An Enhanced Green Fluorescence Protein-based Assay for Studying Neurite Outgrowth in Primary Neurons
Summary
In this report, we describe a simple protocol for studying neurite outgrowth in embryonic rat cortical neurons by co-transfecting with EGFP and the protein of interest.
Abstract
Neurite outgrowth is a fundamental event in the formation of the neural circuits during nervous system development. Severe neurite damage and synaptic dysfunction occur in various neurodegenerative diseases and age-related degeneration. Investigation of the mechanisms that regulate neurite outgrowth would not only shed valuable light on brain developmental processes but also on such neurological disorders. Due to the low transfection efficiency, it is currently challenging to study the effect of a specific protein on neurite outgrowth in primary mammalian neurons. Here, we describe a simple method for the investigation of neurite outgrowth by the co-transfection of primary rat cortical neurons with EGFP and a protein of interest (POI). This method allows the identification of POI transfected neurons through the EGFP signal, and thus the effect of the POI on neurite outgrowth can be determined precisely. This EGFP-based assay provides a convenient approach for the investigation of pathways regulating neurite outgrowth.
Introduction
Neurites, including both axons and dendrites, are the projections from neurons involved in the establishment of the neural networks. The dynamic outgrowth of neurites is essential for neurodevelopment. However, the underlying regulatory mechanisms underneath remain unclear. In particular, neurite damage is often observed in various neurodegenerative diseases and after brain injuries1. Therefore, investigation of the roles of putative molecules in various neurite outgrowth regulatory pathways would improve our understanding of the process. Moreover, it may reveal novel therapeutic targets for various neurological disorders. Neuronal cell lines are valuable models for studying neuronal processes including neurite outgrowth as they are easy to manipulate and transfect2,3. However, genetic drift has been reported to occur in some commonly used cell lines, which could lead to variations in their physiological responses4. Moreover, differential protein expression has been shown between neuronal cell lines and primary neurons. For instance, PC12, a neuronal cell line derived from rat adrenal gland that is widely used for studying neurite outgrowth2,3, does not express NMDA receptors5. Furthermore, it has been proposed that the reduced responsiveness of the mouse neuroblastoma line neuro-2a to neurotoxins in comparison to primary neurons is due to the lack of expression of certain membrane receptors and ion channels6. Therefore, primary neurons are a more desirable and representative model for the investigation of neurite outgrowth. However, the use of primary neurons is hindered by their low transfection efficiency7.
Here, we describe a method that involves the co-transfection of the protein of interest (POI) and EGFP to primary rat cortical neurons. The EGFP acts as a morphological marker for the identification of successfully transfected neurons and permits the measurement of neurites. We validated this method by using compounds/molecules that have been reported to modulate neurite outgrowth. Moreover, FE65, a neuronal adaptor protein that has been shown to stimulate neurite outgrowth, was used to illustrate this approach8,9. This protocol involves (1) the isolation of primary cortical neurons from embryonic day 18 (E18) rat embryos, (2) the co-transfection of neurons with EGFP and the POI (FE65 in this study) and (3) the imaging and analysis of the neurons by using the image processing software ImageJ with the NeuronJ plugin10,11.
Protocol
All procedures followed were in accordance with the ethical standards of the animal experimentation ethics committee of the Chinese University of Hong Kong.
1. Preparation of Coverslips
- Place a sterile 18 mm circular coverslip into each well of a 12-well tissue culture plate.
- Coat the coverslip with 5 µg/mL poly-D-lysine solution in a humidified 37 °C incubator for at least 1 h.
- Aspirate the poly-D-lysine solution from the tissue culture plate and rinse the coated coverslips once with sterile water.
2. Rat Embryonic Neuron Dissection
- Sacrifice a timed-pregnant Sprague-Dawley rat at a gestational age of 18 days (E18) by either cervical dislocation or CO2 asphyxiation.
NOTE: Please check local regulations for the sacrifice of pregnant rats. - Open the abdominal cavity of the pregnant rat with dissecting scissors and transfer the uterus to a 10 cm Petri dish.
- Open the uterus and the amniotic sac carefully with small dissecting scissors and remove the placenta from the rat embryo using small dissecting scissors. Transfer the whole embryo to a 10 cm Petri dish with pre-chilled phosphate buffered saline supplemented with glucose (PBS-glucose, 10 mM sodium phosphates, 2.68 mM potassium chloride, 140 mM sodium chloride and 3 g/L glucose) using a pair of small forceps.
- Cut along the sagittal suture of the skull and open it carefully with a pair of small dissecting scissors. Transfer the embryonic brain with a small flat spatula to a 10 cm Petri dish with ice-cold PBS-glucose.
- Separate the two cerebral hemispheres from the cerebellum and brain stem using two #5 tweezers under a dissection microscope.
NOTE: Please see reference12 for the structure of the rat brain. - Remove the meninges using the #5 tweezers.
- Isolate the cortex from the cerebral hemispheres with two straight #5 tweezers.
- Transfer the isolated cortex to ice-cold PBS-glucose in a 15 mL centrifuge tube.
3. Primary Cortical Neuron Culture
NOTE: All procedures in steps 3 and 4 are performed inside a Class II Biosafety cabinet.
- Settle the isolated cortex by gravity at 4 °C for 5 min and aspirate the PBS-glucose.
- Add 1 mL of 0.05% Trypsin-EDTA to the isolated cortex and mix gently by tapping and incubate the tissue in a 37 °C water bath for 10 min to allow enzymatic digestion. Tap the tube gently to mixing every 2 min.
- Add 4 mL of maintenance medium (e.g., Neurobasal Medium) to the tissue/trypsin mixture.
NOTE: All the maintenance medium used in this protocol is supplemented with Penicillin-Streptomycin and B-27 supplement13. - Dissociate the tissue gently by trituration using a 1 mL pipette.
- Pellet the dissociated cells by centrifugation at 200 x g for 5 min. Aspirate the supernatant.
- Repeat steps 3.5 to 3.7 twice.
- Resuspend the cell pellet in 1 mL of maintenance medium.
- Add 10 µL of 0.4% Trypan Blue solution to 10 µL of cell suspension for counting of viable cells by a hemocytometer.
- Plate the neurons at a density of 65,000/cm2 (viable cells) in 1 mL of maintenance medium per well in a 12-well plate.
4. Cell Transfection and Fixation
- At 2 days in vitro (DIV2), transfect 0.5 µg of EGFP construct (pEGFP-C1) to neurons together either with or without of 0.5 µg of POI by using 1 µL of transfection reagent (e.g., Lipofectamine 2000). Use manufacturer's instructions.
NOTE: Mammalian expression constructs were prepared by using an endotoxin free plasmid preparation kit. Treatment with chemicals/molecules (in this manuscript cytochalasin D (Cyto D) and nerve growth factor (NGF) were used) can be done at 6 h after transfection. - Aspirate the culture medium 24 h post-transfection and wash the transfected cells once with 37 °C PBS (10 mM sodium phosphates, 2.68 mM potassium chloride, 140 mM sodium chloride).
- Fix the cells with 4% paraformaldehyde in PBS for 10 min in the dark at room temperature.
- Wash the fixed cells three times with PBS.
- Add a minimal amount of fluorescence mounting medium on a microscope glass slide. Carefully transfer the coverslip from the 12-well plate onto the mounting medium with the sample facing the glass slide.
NOTE: Seal the edge of the coverslip with nail polish if an aqueous mounting medium is used.
5. Measurement of Neurite Outgrowth
- Use a 40x objective for capturing images using an epi-fluorescent microscope.
- Capture images from at least 40 intact neurons with EGFP signal per transfection.
- Open the captured image in ImageJ software with the NeuronJ plugin11 to measure the length of the longest neurite, from the cell body to the tip of the growth cone, of each imaged neuron.
- Analyze the data obtained with the software to determine the effect of the targeted proteins in neurite outgrowth.
Representative Results
To test this methodology, we used Cyto D and nerve growth factor NGF, which have been shown to inhibit and stimulate neurite outgrowth respectively14,15,16. The neurite length of neurons transfected with EGFP were measured after treatment with Cyto D or NGF. The transfection efficiency of EGFP to the neurons was 2.7% (1,068 neurons counted). As shown in Figure 1A, Cyto D suppressed neurite extension in a dose-dependent manner. Conversely, neurite outgrowth was potentiated in the neurons treated with NGF (Figure 1B).
Next, we investigated the utility of this system by transfecting the neuronal adaptor FE65, which has been shown to promote neurite outgrowth. Primary rat cortical neurons were co-transfected with FE65 and EGFP. Despite the low transfection efficiency, immuno-fluorescence analysis revealed that over 80% of the neurons were successfully co-transfected with EGFP and FE65 (Figure 2A). Similar to previous reports8,9, FE65 significantly stimulated the neurite outgrowth by 2x (Figure 2B). We also analyzed the expression of EGFP and FE65 at different time points by Western blot analysis. As shown in Figure 2C, EGFP and FE65 were detected 6 h and 12 h post-transfection, respectively. Similar expression levels of the proteins were observed in 1 d to 7 d post-transfection neurons. This indicates that the analysis of neurite outgrowth could be done as early as 6 h post-transfection or in more mature neurons. Together, this data suggest that the mentioned protocol is suitable for determining the role of putative neurite outgrowth regulatory proteins by classical transfection.
We also monitored the effect of gene dosage on neurite outgrowth by transfecting primary rat cortical neurons with different amounts of FE65 plasmid DNA. As shown in Figure 2D, a dose-dependent increase in neurite extension was observed from 0 – 0.5 µg of FE65 plasmid DNA. However, there was no significant difference between neurons transfected with either 0.5 µg or 1 µg of plasmid DNA (Figure 2D).
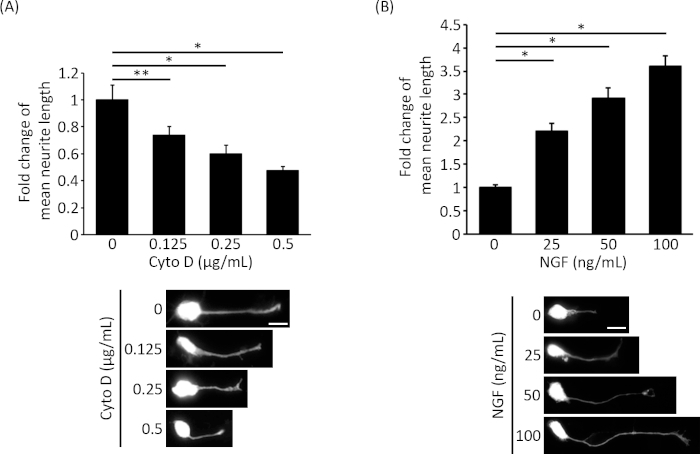
Figure 1: Neurite outgrowth is modulated by Cyto D and NGF. E18 rat cortical neurons were transfected on DIV2 with an EGFP expression vector. 6 h post-transfection, the cells were treated with (A) 0-0.5 µg/mL Cyto D or (B) 0-100 ng/mL NGF for 24 h. Then the neurons were fixed and imaged accordingly. Images were captured with 40x objective using an epi-fluorescence microscope and the length of the longest neurite from the cell body to the tip of the growth cone was measured by using ImageJ with the NeuronJ plugin. Three independent experiments were performed and at least 40 neurons were measured in each group. The bar chart showed the fold change in mean neurite length. Unpaired t-test was adopted for the statistical analysis. *p < 0.001, **p < 0.05. Error bars = S.E.M. Please click here to view a larger version of this figure.
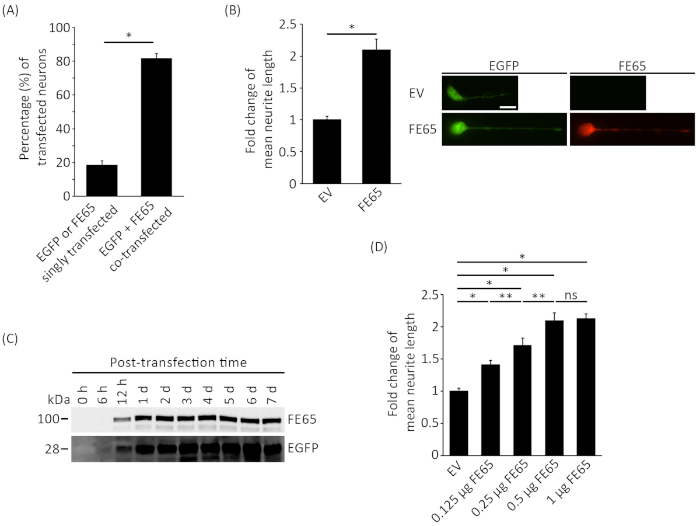
Figure 2: FE65 stimulates neurite outgrowth. E18 rat cortical neurons were transfected on DIV2 with either empty vector control (EV) or FE65 together with an EGFP expression vector. Cells were fixed and imaged 24 h after transfection. (A) The transfected neurons were counterstained with anti-FE65 antibody and the number of cells with EGFP or FE65 singly transfected and EGFP + FE65 co-transfected were counted. Three independent experiments were performed and at least 100 cells were counted in each experiment. Data were expressed as the percentage of cells with EGFP and EGFP + FE65 signals. *p < 0.001. Error bars = S.D. (B) The length of the longest neurite from the cell body to the tip of the growth cone was measured by using ImageJ with the NeuronJ plugin. Representative neuron images were shown in the right panel. FE65 was stained by a goat anti-FE65 antibody as described8,17. Three independent experiments were performed and at least 40 neurons were measured per transfection. The bar chart showed the fold change in mean neurite length. The data were analyzed by unpaired t-test. *p < 0.001. Error bars = S.E.M. Scale bar = 10 µm. (C) Western blot analysis of the expression of levels of EGFP and FE65 at the post-transfection time points as indicated. EGFP and FE65 were detected by mouse anti-GFP and anti-myc (to FE65 C-terminal myc tag), respectively. (D) The average neurite length of neurons transfected with various amounts of FE65 plasmid DNA as indicated. Statistical analyses were performed using one-way ANOVA tests with Bonferroni post-hoc test. *p < 0.001, **p < 0.05. Error bars = S.E.M. Scale bar = 10 µm. Please click here to view a larger version of this figure.
Discussion
As stated before, PC12 and its subclones are widely employed for studying neurite extension because they have excellent transfection efficiency2,3. In contrast, primary neurons have a low transfection rate, which is a major obstacle for studying neurite outgrowth regulators by transfection7. Here, we describe a convenient protocol for quantifying neurite outgrowth in primary neurons. Despite the low overall transfection efficiency, more than 80% of the transfected neurons were successfully co-transfected with the two proteins: FE65 and EGFP (Figure 2A). Therefore, by analyzing the EGFP labelled neurons, the effect of FE65 on neurite outgrowth could be precisely determined (Figure 2B).
Another advantage of the EGFP-based approach described is that immunofluorescence staining can be omitted. Immunofluorescence staining of Tuj 1, a neuron-specific class III β-tubulin, is widely employed as a morphological marker when studying neurite elongation18,19,20,21. In addition to Tuj 1, staining of the POI is required for the identification of transfected neurons19,22,23. It is known that the consistency of immunofluorescence staining, which is crucial for neurite outgrowth measurement, is affected by many factors including sample preparation and antibodies24. Hence, the overall simplicity of the EGFP-based method could improve the accuracy of the assay.
In our protocol, neurons are fixed for neurite measurement 24 h post-transfection. Thus, neurons are exposed to the transfection reagent for 24 h. It is long known that transfection reagents are toxic to cells25. If the effect of the POI on neurite extension needs to be determined beyond DIV3, fresh medium replenishment may help to minimize the toxicity. Moreover, alternative gene delivery methods may be considered such as Ca2+ phosphate co-precipitation and nucleofection, both of which are shown to have a less toxic effect to cells26. Additionally, we show here that the highest effect of FE65 on neurite outgrowth is observed in the neurons transfected with that 0.5 µg of FE65 and 0.5 µg of EGFP plasmids by using 1 µL of transfection reagent. For other POIs, the optimal amounts of plasmid DNA and transfection reagents should be determined as proteins have widely different turnover rates.
In addition to gene delivery method, cell density is another critical parameter to be considered. If the neuron density is too high, it becomes difficult to identify the origins of neurites as they would overlap with each other. Although plating cells at a low density may resolve the issue, the survival of primary neurons would be significantly reduced at a low cell density27. Primary rat hippocampal neurons have been shown to grow healthily at the density between 40,000 to 100,000/cm2 28. In this protocol, a density of 65,000/cm2 of rat cortical neurons is used. Nevertheless, it is important to determine the appropriate cell density for different types of neurons under different experimental conditions.
The neurite measurement of developing rat primary neurons (i.e. DIV3 neurons) is described here. However, more mature neuronal phenotypes can be observed in neurons beyond DIV329. As the effects of POIs or drugs on neurite extension in mature neurons may be different from the developing neurons, investigation by using neurons from different stages would provide a more comprehensive perspective. It is worth noting that we were still able to observe EGFP expression in the neurons 7 d post-transfection. This may facilitate the study of POIs or drugs on neurite outgrowth in mature neurons.
Human induced pluripotent stem cell (hiPSC)-derived neurons are valuable tools in identification of novel therapeutic approaches. For instance, investigation of neurite outgrowth in these cells could reveal novel targets in neuroregeneration as the use of hiPSC-derived neurons avoids the species differences. Similar to primary rodent neurons, hiPSC-derived neurons are difficult to transfect30, which may reduce the co-transfection efficiency of EGFP and the POI. Hence, the use of polycistronic mammalian expression vectors could ensure that all the transfected cells express the entire set of transfected genes including EGFP and POIs. Additionally, alternative gene delivery methods, such as electroporation, may improve transfection efficiency. Again, cell viability is an issue which needs to be considered when using such gene delivery approaches.
It is known that the rat embryonic brain cortex contains different types of neurons including spiny stellate neurons, bipolar neurons and long-projecting neurons31. While this protocol stated here can reveal the general effect of POIs on neurite outgrowth, it is possible that the same POI may exhibit different responses in different types of neurons. For example, myostatin increases the number of sensory axons but not that of motor axons32. In such a scenario, modification of the protocol is necessary, such as prior isolation of specific types of neurons by flow cytometry. Alternatively, specific neuronal marker antibodies may be used for the identification of the required types of neurons during imaging.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This work was supported by funds from the Research Grants Council Hong Kong, Health and Medical Research Fund (Hong Kong), CUHK direct grant scheme, the United College endowment fund and the TUYF Charitable Trust.
Materials
| #5 tweezers | Regine | 5-COB | |
| 18 mm Circle Cover Slips | Thermo Scientific | CB00180RA | Sterilize before use. |
| B27 Supplement | Gibco | 17504044 | |
| Cytochalasin D | Invitrogen | PHZ1063 | Dissolved in DMSO. |
| D-(+)-Glucose | Sigma-Aldrich | G8270 | |
| Dimethyl Sulfoxide | Sigma-Aldrich | D2650 | |
| Dissecting Scissors, 10 cm | World Precision Instruments | 14393 | |
| Dissecting Scissors, 12.5 cm | World Precision Instruments | 15922 | |
| EndoFree Plasmid Maxi Kit | QIAGEN | 12362 | |
| Fluorescence Mounting Medium | Dako | S302380 | |
| Lipofectamine 2000 Transfection Reagent | Invitrogen | 11668019 | |
| Neurobasal Medium | Gibco | 21103049 | |
| NGF 2.5S Native Mouse Protein | Gibco | 13257019 | |
| Nugent Utility Forceps, 10mm, Straight Tip | World Precision Instruments | 504489 | |
| Paraformaldehyde | Sigma-Aldrich | P6148 | |
| pEGFP-C1 | Clontech | #6084-1 | |
| pCI FE65 | Please see references 8 and 15 | ||
| PBS Tablets | Gibco | 18912014 | |
| Penicillin-Streptomycin | Gibco | 15140122 | |
| Poly-D-lysine hydrobromide | Sigma-Aldrich | P7280 | |
| Spatula | Sigma-Aldrich | S4147 | |
| Trypsin-EDTA (0.05%), phenol red | Gibco | 25300062 | |
| Trypan Blue Solution, 0.4% | Gibco | 15250061 |
Referencias
- Kaplan, A., Bueno, M., Hua, L., Fournier, A. E. Maximizing functional axon repair in the injured central nervous system: Lessons from neuronal development. Developmental Dynamics. 247 (1), 18-23 (2018).
- Harrill, J. A., Mundy, W. R. Quantitative assessment of neurite outgrowth in PC12 cells. Methods in Molecular Biology. 758, 331-348 (2011).
- Yeyeodu, S. T., Witherspoon, S. M., Gilyazova, N., Ibeanu, G. C. A rapid, inexpensive high throughput screen method for neurite outgrowth. Current Chemical Genomics. 4, 74-83 (2010).
- Ben-David, U., et al. Genetic and transcriptional evolution alters cancer cell line drug response. Nature. 560 (7718), 325-330 (2018).
- Edwards, M. A., Loxley, R. A., Williams, A. J., Connor, M., Phillips, J. K. Lack of functional expression of NMDA receptors in PC12 cells. Neurotoxicology. 28 (4), 876-885 (2007).
- LePage, K. T., Dickey, R. W., Gerwick, W. H., Jester, E. L., Murray, T. F. On the use of neuro-2a neuroblastoma cells versus intact neurons in primary culture for neurotoxicity studies. Critical Reviews in Neurobiology. 17 (1), 27-50 (2005).
- Karra, D., Dahm, R. Transfection techniques for neuronal cells. Journal of Neuroscience. 30 (18), 6171-6177 (2010).
- Cheung, H. N., et al. FE65 interacts with ADP-ribosylation factor 6 to promote neurite outgrowth. The FASEB Journal. 28 (1), 337-349 (2014).
- Li, W., et al. Neuronal adaptor FE65 stimulates Rac1-mediated neurite outgrowth by recruiting and activating ELMO1. Journal of Biological Chemistry. 293 (20), 7674-7688 (2018).
- Schneider, C. A., Rasband, W. S., Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nature Methods. 9 (7), 671-675 (2012).
- Meijering, E., et al. Design and validation of a tool for neurite tracing and analysis in fluorescence microscopy images. Cytometry. Part A: the Journal of the International Society for Analytical Cytology. 58 (2), 167-176 (2004).
- Swanson, L. W. Brain maps 4.0-Structure of the rat brain: An open access atlas with global nervous system nomenclature ontology and flatmaps. The Journal of Comparative Neurology. 526 (6), 935-943 (2018).
- Brewer, G. J. Serum-free B27/neurobasal medium supports differentiated growth of neurons from the striatum, substantia nigra, septum, cerebral cortex, cerebellum, and dentate gyrus. Journal of Neuroscience Research. 42 (5), 674-683 (1995).
- Yamada, K. M., Spooner, B. S., Wessells, N. K. Axon growth: roles of microfilaments and microtubules. Proceedings of the National Academy of Sciences of the United States of America. 66 (4), 1206-1212 (1970).
- Casella, J. F., Flanagan, M. D., Lin, S. Cytochalasin D inhibits actin polymerization and induces depolymerization of actin filaments formed during platelet shape change. Nature. 293 (5830), 302-305 (1981).
- Calabrese, E. J. Enhancing and regulating neurite outgrowth. Critical Reviews in Toxicology. 38 (4), 391-418 (2008).
- Lau, K. F., et al. Dexras1 Interacts with FE65 to Regulate FE65-Amyloid Precursor Protein-dependent Transcription. Journal of Biological Chemistry. 283 (50), 34728-34737 (2008).
- Cui, X., et al. Niacin treatment of stroke increases synaptic plasticity and axon growth in rats. Stroke. 41 (9), 2044-2049 (2010).
- Khodosevich, K., Monyer, H. Signaling involved in neurite outgrowth of postnatally born subventricular zone neurons in vitro. BMC Neuroscience. 11, 18 (2010).
- Tang, F., et al. Resveratrol Enhances Neurite Outgrowth and Synaptogenesis Via Sonic Hedgehog Signaling Following Oxygen-Glucose Deprivation/Reoxygenation Injury. Cellular Physiology and Biochemistry: International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology. 43 (2), 852-869 (2017).
- He, W., Liu, Y., Tian, X. Rosuvastatin Improves Neurite Outgrowth of Cortical Neurons against Oxygen-Glucose Deprivation via Notch1-mediated Mitochondrial Biogenesis and Functional Improvement. Frontiers in Cellular Neuroscience. 12, 6 (2018).
- Tesarova, P., et al. Receptor for advanced glycation end products (RAGE)–soluble form (sRAGE) and gene polymorphisms in patients with breast cancer. Cancer Investigation. 25 (8), 720-725 (2007).
- Park, S. Y., et al. Hippocalcin Promotes Neuronal Differentiation and Inhibits Astrocytic Differentiation in Neural Stem Cells. Stem Cell Reports. 8 (1), 95-111 (2017).
- Radbruch, A. Immunofluorescence: Basic Considerations. Flow Cytometry and Cell Sorting. , 38-52 (2000).
- Wang, T., Larcher, L. M., Ma, L., Veedu, R. N. Systematic Screening of Commonly Used Commercial Transfection Reagents towards Efficient Transfection of Single-Stranded Oligonucleotides. Molecules. 23 (10), (2018).
- Sariyer, I. K. Transfection of neuronal cultures. Methods in Molecular Biology. 1078, 133-139 (2013).
- Banker, G. A., Cowan, W. M. Rat hippocampal neurons in dispersed cell culture. Brain Research. 126 (3), 397-425 (1977).
- Banker, G. A., Cowan, W. M. Further observations on hippocampal neurons in dispersed cell culture. The Journal of Comparative Neurology. 187 (3), 469-493 (1979).
- Biffi, E., Regalia, G., Menegon, A., Ferrigno, G., Pedrocchi, A. The influence of neuronal density and maturation on network activity of hippocampal cell cultures: a methodological study. PLoS One. 8 (12), e83899 (2013).
- Hiragi, T., et al. Differentiation of Human Induced Pluripotent Stem Cell (hiPSC)-Derived Neurons in Mouse Hippocampal Slice Cultures. Frontiers in Cellular Neuroscience. 11, 143 (2017).
- Zeng, H., Sanes, J. R. Neuronal cell-type classification: challenges, opportunities and the path forward. Nature Reviews. Neuroscience. 18 (9), 530-546 (2017).
- Jones, M. R., Villalon, E., Northcutt, A. J., Calcutt, N. A., Garcia, M. L. Differential effects of myostatin deficiency on motor and sensory axons. Muscle & Nerve. 56 (6), E100-E107 (2017).

