Estimation of Urinary Nanocrystals in Humans using Calcium Fluorophore Labeling and Nanoparticle Tracking Analysis
Summary
The objective of this study was to determine whether nanoparticle tracking analysis (NTA) could detect and quantify urinary calcium containing nanocrystals from healthy adults. The findings from the current study suggest NTA could be a potential tool to estimate urinary nanocrystals during kidney stone disease.
Abstract
Kidney stones are becoming more prevalent worldwide in adults and children. The most common type of kidney stone is comprised of calcium oxalate (CaOx) crystals. Crystalluria occurs when urine becomes supersaturated with minerals (e.g., calcium, oxalate, phosphate) and precedes kidney stone formation. Standard methods to assess crystalluria in stone formers include microscopy, filtration, and centrifugation. However, these methods primarily detect microcrystals and not nanocrystals. Nanocrystals have been suggested to be more harmful to kidney epithelial cells than microcrystals in vitro. Here, we describe the ability of Nanoparticle Tracking analysis (NTA) to detect human urinary nanocrystals. Healthy adults were fed a controlled oxalate diet prior to drinking an oxalate load to stimulate urinary nanocrystals. Urine was collected for 24 hours before and after the oxalate load. Samples were processed and washed with ethanol to purify samples. Urinary nanocrystals were stained with the calcium binding fluorophore, Fluo-4 AM. After staining, the size and count of nanocrystals were determined using NTA. The findings from this study show NTA can efficiently detect nanocrystalluria in healthy adults. These findings suggest NTA could be a valuable early detection method of nanocrystalluria in patients with kidney stone disease.
Introduction
Urinary crystals form when urine becomes supersaturated with minerals. This can occur in healthy individuals but is more common in individuals with kidney stones1. The presence and accumulation of urinary crystals can increase one's risk of developing a kidney stone. Specifically, this occurs when crystals bind to Randall's plaque, nucleate, accumulate, and grow over time2,3,4. Crystalluria precedes kidney stone formation and assessment of crystalluria may have predictive value in kidney stone formers3,5. Specifically, crystalluria has been suggested to be useful to predict the risk of stone recurrence in patients with a history of calcium oxalate containing stones6,7.
Crystals have been reported to negatively impact renal epithelial and circulating immune cell function8,9,10,11,12,13. It has been previously reported that circulating monocytes from calcium oxalate (CaOx) kidney stone formers have suppressed cellular bioenergetics compared to healthy individuals14. In addition, CaOx crystals reduce cellular bioenergetics and disrupt redox homeostasis in monocytes8. Consumption of meals rich in oxalate may cause crystalluria which could lead to renal tubule damage and alter the production and function of urinary macromolecules that are protective against kidney stone formation15,16. Several studies have demonstrated that urinary crystals can vary in shape and size depending on the pH and temperature of the urine17,18,19. Further, urinary proteins have been shown to modulate crystal behavior20. Daudon et al.19, proposed that crystalluria analysis could be helpful in the management of patients with kidney stone disease and in assessing their response to therapies. A few conventional methods currently available to evaluate the presence of crystals include polarized microscopy21,22, electron microscopy23, particle counters3, urine filtration24, evaporation3,5 or centrifugation21. These studies have provided valuable insight to the kidney stone field regarding crystalluria. However, a limitation of these methods has been the inability to visualize and quantify crystals less than 1 µm in size. Crystals of this size may influence the growth of CaOx stones by attaching to Randall's plaque.
Nanocrystals have been shown to cause extensive injury to renal cells compared to larger microcrystals25. The presence of nanocrystals has been reported in urine using a nanoparticle analyzer26,27. Recent studies have used fluorescently labeled bisphosphate probes (alendronate-fluorescein/alendronate-Cy5) to examine nanocrystals using nanoscale flow cytometry28. The limitation of this dye is that it is not specific and will bind to almost all types of stones except cysteine. Thus, accurately assessing the presence of nanocrystals in individuals may be an effective tool to diagnose crystalluria and/or predict stone risk. The purpose of this study was to detect and quantify calcium containing nanocrystals (<1 µm in size) using nanoparticle tracking analysis (NTA). To achieve this, NTA technology was used in combination with a calcium binding fluorophore, Fluo-4 AM to detect and quantify calcium containing nanocrystals in the urine of healthy adults.
Protocol
All experiments outlined in this work were approved by the University of Alabama at Birmingham (UAB) Institutional Review Board. Healthy adults (33.6 ± 3.3 years old; n=10) were enrolled in the study if they had a normal blood comprehensive metabolic panel, non-tobacco users, non-pregnant, a BMI between 20-30 kg/m2, and free of chronic medical conditions or acute illnesses. Healthy participants signed a written informed consent form prior to the start of the study.
1. Clinical protocol and urine collection
- Have participants consume a low oxalate diet prepared by the UAB Center for Clinical and Translational Sciences Bionutrition Core for 3 days and fast overnight before collecting their urine (24-hour sample).
- The following day, have participants return their 24-hour urine sample (pre-oxalate) before consuming an oxalate load (smoothie containing fruits and vegetables, ~8 mM oxalate). Have participants subsequently collect their urine for 24 hours (post-oxalate sample) and return their urine the following day.
- Maintain all urine samples at room temperature (RT) prior to processing as described below and shown in Figure 1.
2. Urine Processing
NOTE: All materials and equipment used are listed in Table of Materials.
CAUTION: Wear personal protective equipment at all times while handling clinical samples and reagents. Specifically, gloves, face and eye shields, respiratory protection, and protective clothing.
- Measure and record urine pH and volume. Mix thoroughly prior to adding 50 mL of urine into a labeled sterile 50 mL conical tube.
- Centrifuge sample at 1200 x g for 10 min at RT using a benchtop centrifuge.
NOTE: Keep the sample at RT to prevent further crystal formation as cooler temperatures can promote crystallization. - Discard the supernatant and wash and resuspend the pellet again with 5 mL of 100% ethanol. Centrifuge the sample at 1200 x g for 10 minutes at RT using a benchtop centrifuge.
- Discard the supernatant and resuspend the pellet in 1 mL of 100% ethanol. Store the sample at -20 °C for later processing OR stain the sample as described below.
NOTE: There is no significant difference in data points (i.e., particle size/concentration) between stored or freshly stained samples.
3. Nanoparticle Tracking Analysis (NTA)
- Sample Preparation
- Gold nanoparticles: Use gold nanoparticles to optimize settings on the instrument. Dilute 100 nm sized gold nanoparticles 1:1000 in ultrapure water.
- Human Urine: Dilute urine samples 20 times in water prior to staining with 5 mM Fluo-4 AM (a calcium fluorescence dye) for 30 min in the dark. Analyze the samples using NTA.
- Prepare Calcium Oxalate (CaOx) crystals as previously described29. Dilute 10 mM stock solution (14.6 mg in 10 mL of water) to 50 µM in water and stain the diluted samples using 5 mM Fluo-4 AM for 30 min in dark prior to analysis.
- Calcium Phosphate (CaP) crystals: Dilute 10 mM stock solution (50.4 mg in 10 mL of water) to 50 µM in water and stain the diluted samples using 5 mM Fluo-4 AM for 30 min in dark prior to NTA analysis.
- Instrument Set-up, Camera Settings, and Data Collection
NOTE: The computer and instrument setup used for this method are shown in Figure 2.- Turn on the computer and then the instrument. Open the software and turn on the camera.
- Once the software window is open, click the capture icon in the top left corner on the window to start the capture mode. The camera initialization takes a few seconds.
- Clean the platform by first pumping air into it using a 1 mL syringe until the platform appears clean. Gently add water to the apparatus 2-3 times using another 1 mL syringe to remove any air bubbles.
NOTE: Look for any air bubbles in the platform as well as in the tubing. It is important to not have bubbles throughout the apparatus prior to and while running samples. If bubbles are present, clean the platform again with air and water. - Once the platform is clean, add water to check for any contamination on the surface by viewing the camera. Next, add gold nanoparticles as a control to the sample loading pump injector to set up the instrument.
- Adjust the camera level on the screen or on the knob to the right side of the instrument until the image starts to display colored pixels and then reduce the camera level.
- Then adjust the screen to optimize the image. Left-click the mouse button on the video image. Hold the left mouse button and drag image up and down to get the entire view.
NOTE: The normal camera lens and filter is used to assess gold nanoparticles and unstained samples. - Set up the infusion speed and focus the camera so that the gold nanoparticles are visible on the camera screen. Set the infusion speed to high (i.e., 500 µL/min) for initial set up to ensure the gold nanoparticles are detected. Once detected, reduce the speed to 50 µL/min.
- Adjust the camera level to visualize the particles. For unstained samples, adjust the screen gain at level 5 to achieve the camera focus, and set the camera level at 8. Once the focus is set, record the sample (i.e., 1 measurement for 60 seconds only).
NOTE: The focus and continuous flow speed are important for obtaining clear and sharp images of the particles for counting. - After optimization, clean the apparatus again with water before assessing samples. View the camera to ensure that the tubing is clean and particles are not present.
NOTE: Wash the chamber between each sample until no particles are detected by the camera. - To analyze stained samples, adjust the camera to the filter position containing the suitable fluorescent filter. Load diluted and stained samples onto the sample loading pump injector and reduce the speed to 20 µL/min for analysis of the sample.
- Next adjust the screen gain and camera level as these are important parameters. For stained (fluorescent) samples, set the screen gain to 5 and the camera level at level 13.
NOTE: These parameters will vary based on the sample type and every sample will need to be optimized to gain focus. - Use standard measurement to measure the samples for 5 captures per sample where one capture-duration is for 60 seconds.
- Save and store data after each measurement. The software will save image and video files for each measurement. The software provides output data (e.g., crystal size: 10 nM – 1000 nM and concentration) in both excel and pdf formats.
- Calculate the average number of nanoparticles for all 5 readings for each individual sample. Analyze the data using standard deviation or standard error of the mean and use t-tests for paired analysis.
Representative Results
The findings from this study show NTA can efficiently detect the mean size and concentration of calcium containing urinary nanocrystals in human urine. This was achieved by using the fluorophore, Fluo-4 AM, and nanoparticle tracking analysis. Fluo-4 AM was able to bind to both CaOx and CaP crystals. As shown in Figure 3A, CaOx crystals were determined to be between 50-270 nm in size and have a mean concentration of 1.26 x 109 particles/mL. CaP crystals were between 30-225 nm in size and had a mean concentration of 2.22 x 109 particles/mL (Figure 3B). To determine if NTA could assess nanocrystals in human urine, healthy adults were asked to consume a controlled oxalate diet followed by a high oxalate load. Twenty-four hour urine samples before and after the load were collected to assess urinary nanocrystal size and concentration. Pre-oxalate urine samples contained some urinary nanocrystals (1.65 x 108 ± 3.29 x 107 particles/mL) between 110-300 nm (Figure 4). In contrast, there was a significant increase (p<0.0001) in urinary nanocrystals present in post-oxalate samples (7.05 x 108 ± 1.08 x 108 particles/mL; 100-320 nm) (Figure 4). To confirm the reproducibility of the method, samples were measured three times and there was no significant variation in technical replicates (Figure 5).

Figure 1: Protocol for isolating and staining human urinary nanocrystals. Please click here to view a larger version of this figure.

Figure 2: Description of Nanoparticle Tracking Analysis (NTA). (A) Computer and instrument set up used for these studies. (B) Samples are injected into an inlet tubing using a syringe pump at a continuous rate prior to filling the optical surface. Samples are then observed by the objective lens and captured by the camera as samples flow through the platform before exiting through the outlet tubing to be discarded. Please click here to view a larger version of this figure.
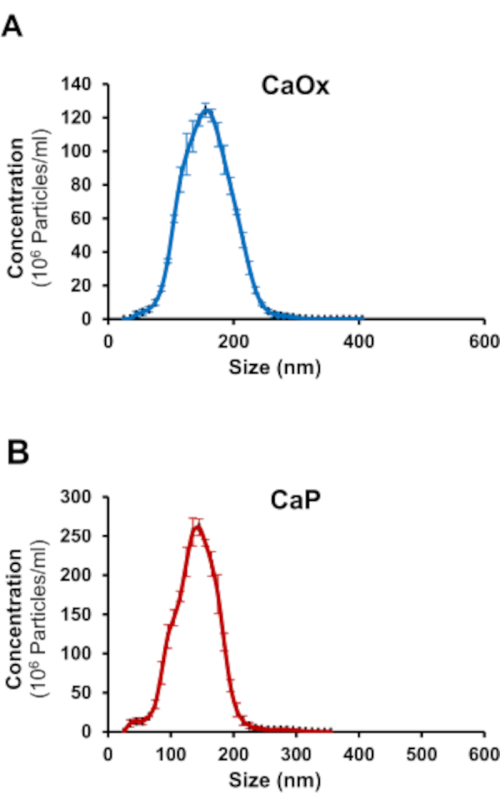
Figure 3: NTA detects Fluo-4 AM labeled calcium oxalate (CaOx) and calcium phosphate (CaP) crystals. Representative graphs of (A) CaOx and (B) CaP crystals showing size distribution and concentration. Please click here to view a larger version of this figure.
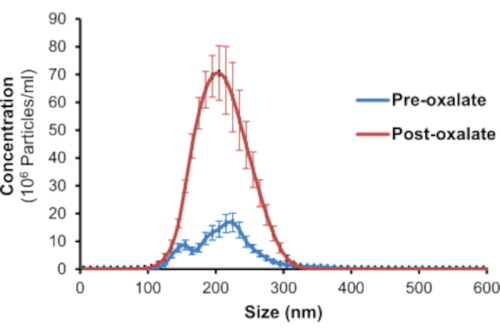
Figure 4: NTA detects Fluo-4 AM labeled 24-hour human urinary nanocrystals. Representative graph of Fluo-4 AM labeled urinary nanocrystals in 24-hour pre-oxalate and post-oxalate samples from a healthy adult on a controlled oxalate diet. Please click here to view a larger version of this figure.
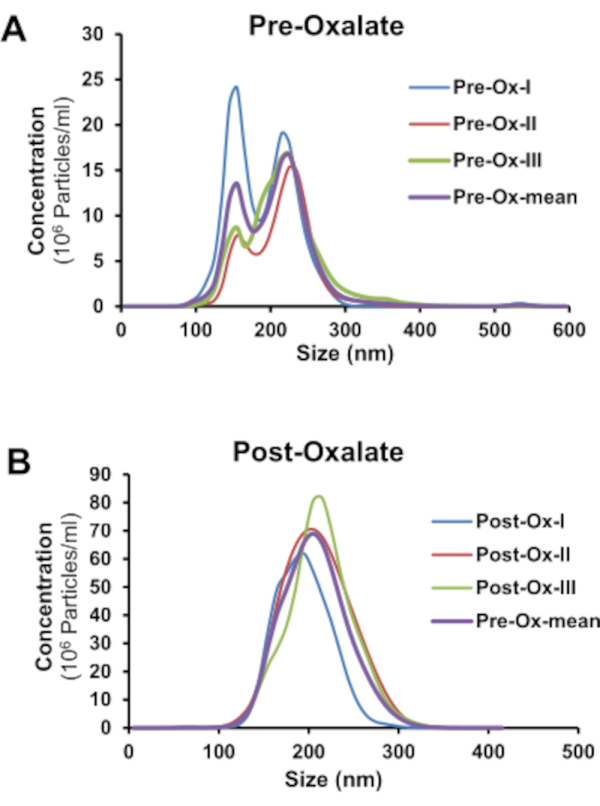
Figure 5: Technical replicates of human nanocrystals in 24-hour urine collections using NTA. Technical replicates of Fluo-4 AM labeled urinary nanocrystals in 24-hour (A) pre-oxalate and (B) post-oxalate samples from a healthy adult on a controlled oxalate diet. Please click here to view a larger version of this figure.
Discussion
NTA has been used in the present study to assess nanocrystals in human urine using a calcium binding probe, Fluo-4 AM. There is no standard method available to detect nanocrystals in the urine. Some research groups have detected nanocrystals in the urine and relied on the use of extensive protocols or methods that are limited in their ability to quantify the samples27,28. This study shows a specific and sensitive method for detecting calcium containing nanocrystals in the urine of humans who participated in a dietary feeding study which consisted of ingesting a high oxalate load. The amount of oxalate consumed was equivalent to real world consumption of oxalate (e.g., ½ spinach salad).
NTA is a well characterized high resolution tool that uses Brownian motion to measure particles in solution30. It has been used to assess biological nanoparticles in a variety of biological samples31,32,33. In addition, NTA can accurately predict the size as well as concentration of particles in any type of biological sample. This method does not require any labeling; however, labeling may be used to detect specific particles. Fluo-4 AM was used in this study to efficiently and specifically detect calcium containing nanocrystals in urine samples. Calcium fluorescent probes were initially used to measure free cytosolic calcium34. Fluo-4 is an analogue of Fluo-3 whose fluorescence increases >100-fold upon binding to free calcium35. In addition, Fluo-4 has been shown to assess calcium particles in the synovial fluid of patients with arthritis using flow cytometry36. Thus, we used Fluo-4 AM for these studies.
All samples were continuously injected into the platform for accurate detection. Determining the concentration and particle size depends on the flow rate, as a high flow rate (i.e., 50 µL/min) can affect accurate assessment of the concentration, as well as the particle size compared to a static setting and a lower flow rate (i.e. 20 µL/min)37. Thus, a steady slow flow rate provides accurate measurement of the number of particles present in samples. Other important parameters that might affect the particle count and size include the camera level, detection threshold, and focus38,39,40. A consistent particle measurement in samples (CV approx. 20%) was observed in the current study, which was consistent with findings from another study39. Lastly, the presence of nanocrystals in human urine has been confirmed using electron microscopy29. This research demonstrates NTA can successfully measure urinary nanocrystals from humans.
One advantage of this protocol is the use of Fluo-4 AM to evaluate calcium containing particles in solution. Another advantage is the minimal variability observed in detecting nanocrystals within samples. One limitation of NTA in this setting, is the inability to distinguish the morphology of nanocrystals. However, this method could be beneficial to detect crystalluria for predicting stone risk in individuals with a history of calcium containing kidney stones. This protocol cannot replace current methodologies but may provide new insight about urinary nanocrystals. The use of NTA to assess urinary calcium containing crystals is a novel approach that should highlight the importance of nanocrystalluria beyond standard microscopy and methods mentioned above. Additional investigations are warranted to explore the reliability of this method in the kidney stone population.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
The authors thank all study participants and the UAB CCTS Bionutrition Core and UAB High Resolution Imaging Service Center for their contributions. This work was supported by NIH grants DK106284 and DK123542 (TM), and UL1TR003096 (National Center for Advancing Translational Sciences).
Materials
| Benchtop Centrifuge | Jouan Centrifuge | CR3-12 | |
| Calcium Oxalate monohydrate | Synthesized in the lab as previously described29. | Store at RT; Stock 10 mM | |
| Calcium Phosphate crystals (hydroxyapatite nanopowder) | Sigma | 677418 | Store at RT; Stock 10 mM |
| Ethanol | Fischer Scientific | AC615095000 | Store at RT; Stock 100% |
| Fluo-4 AM* | AAT Bioquest, Inc. | 20550 | Store at Freezer (-20°C); Stock 5 mM |
| Gold Nanoparticles | Sigma | 742031 | Store at 2-8°C |
| NanoSight Instrument | Malvern Instruments, UK | NS300 | |
| Syringe pump | Harvard Apparatus | 98-4730 | |
| Virkon Disinfectant | LanXESS Energizing Company, Germany | LSP | |
| *Fluorescence dyes are light sensitive; stock and aliquots should be stored in the dark at -20°C. | |||
Referencias
- Fogazzi, G. B. Crystalluria: a neglected aspect of urinary sediment analysis. Nephrology, Dialysis, Transplantation. 11 (2), 379-387 (1996).
- Kuo, R. L. Urine calcium and volume predict coverage of renal papilla by Randall’s plaque. Kidney International. 64 (6), 2150-2154 (2003).
- Robertson, W. G., Peacock, M., Nordin, B. E. Calcium crystalluria in recurrent renal-stone formers. Lancet. 2 (7610), 21-24 (1969).
- Robertson, W. G., Peacock, M. Calcium oxalate crystalluria and inhibitors of crystallization in recurrent renal stone-formers. Clinical Science. 43 (4), 499-506 (1972).
- Hallson, P. C., Rose, G. A. A new urinary test for stone “activity”. British Journal of Urology. 50 (7), 442-448 (1978).
- Daudon, M., Hennequin, C., Boujelben, G., Lacour, B., Jungers, P. Serial crystalluria determination and the risk of recurrence in calcium stone formers. Kidney International. 67 (5), 1934-1943 (2005).
- Baumann, J. M., Affolter, B. From crystalluria to kidney stones, some physicochemical aspects of calcium nephrolithiasis. World Journal of Nephrology. 3 (4), 256-267 (2014).
- Patel, M., et al. Oxalate induces mitochondrial dysfunction and disrupts redox homeostasis in a human monocyte derived cell line. Redox Biology. 15, 207-215 (2018).
- Khan, S. R. Role of renal epithelial cells in the initiation of calcium oxalate stones. Nephron Experimental Nephrology. 98 (2), 55-60 (2004).
- Mulay, S. R., et al. Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1beta secretion. Journal of Clinical Investigation. 123 (1), 236-246 (2013).
- Umekawa, T., Chegini, N., Khan, S. R. Oxalate ions and calcium oxalate crystals stimulate MCP-1 expression by renal epithelial cells. Kidney International. 61 (1), 105-112 (2002).
- Huang, M. Y., Chaturvedi, L. S., Koul, S., Koul, H. K. Oxalate stimulates IL-6 production in HK-2 cells, a line of human renal proximal tubular epithelial cells. Kidney International. 68 (2), 497-503 (2005).
- Lu, X. Renal tubular epithelial cell injury, apoptosis and inflammation are involved in melamine-related kidney stone formation. Urological Research. 40 (6), 717-723 (2012).
- Williams, J., Holmes, R. P., Assimos, D. G., Mitchell, T. Monocyte Mitochondrial Function in Calcium Oxalate Stone Formers. Urology. 93, 221-226 (2016).
- Balcke, P., et al. Transient hyperoxaluria after ingestion of chocolate as a high risk factor for calcium oxalate calculi. Nephron. 51 (1), 32-34 (1989).
- Khan, S. R., Kok, D. J. Modulators of urinary stone formation. Frontiers in Bioscience. 9, 1450-1482 (2004).
- Rodgers, A., Allie-Hamdulay, S., Jackson, G. Therapeutic action of citrate in urolithiasis explained by chemical speciation: increase in pH is the determinant factor. Nephrology, Dialysis, Transplantation. 21 (2), 361-369 (2006).
- Verplaetse, H., Verbeeck, R. M., Minnaert, H., Oosterlinck, W. Solubility of inorganic kidney stone components in the presence of acid-base sensitive complexing agents. European Urology. 11 (1), 44-51 (1985).
- Frochot, V., Daudon, M. Clinical value of crystalluria and quantitative morphoconstitutional analysis of urinary calculi. International Journal of Surgery. 36, 624-632 (2016).
- Grover, P. K., Thurgood, L. A., Wang, T., Ryall, R. L. The effects of intracrystalline and surface-bound proteins on the attachment of calcium oxalate monohydrate crystals to renal cells in undiluted human urine. BJU International. 105, 708-715 (2010).
- Bader, C. A., Chevalier, A., Hennequin, C., Jungers, P., Daudon, M. Methodological aspects of spontaneous crystalluria studies in calcium stone formers. Scanning Microscopy. 8 (2), 215-231 (1994).
- Daudon, M., Cohen-Solal, F., Jungers, P. . Eurolithiasis. 9th European Symposium on Urolithiasis. , 261-263 (2001).
- Werness, P. G., Bergert, J. H., Smith, L. H. Crystalluria. Journal of Crystal Growth. 53 (1), 166-181 (1981).
- Fan, J., Chandhoke, P. S. Examination of crystalluria in freshly voided urines of recurrent calcium stone formers and normal individuals using a new filter technique. Journal of Urology. 161 (5), 1685-1688 (1999).
- Sun, X. Y., Ouyang, J. M., Yu, K. Shape-dependent cellular toxicity on renal epithelial cells and stone risk of calcium oxalate dihydrate crystals. Scientific Reports. 7 (1), 7250 (2017).
- He, J. Y., Deng, S. P., Ouyang, J. M. Morphology, particle size distribution, aggregation, and crystal phase of nanocrystallites in the urine of healthy persons and lithogenic patients. IEEE Trans Nanobioscience. 9 (2), 156-163 (2010).
- Gao, J., et al. Comparison of Physicochemical Properties of Nano- and Microsized Crystals in the Urine of Calcium Oxalate Stone Patients and Control Subjects. Journal of Nanomaterials. 2014, 9 (2014).
- Gavin, C. T., et al. Novel Methods of Determining Urinary Calculi Composition: Petrographic Thin Sectioning of Calculi and Nanoscale Flow Cytometry Urinalysis. Scientific Reports. 6, 19328 (2016).
- Kumar, P., et al. Dietary Oxalate Induces Urinary Nanocrystals in Humans. Kidney International Reports. 5 (7), 1040-1051 (2020).
- Carr, B., Hole, P., Malloy, A., Nelson, P., Smith, J. Applications of nanoparticle tracking analysis in nanoparticle research–A mini-review. European Journal of Parenteral Sciences and Pharmaceutical Sciences. 14 (2), 45 (2009).
- Dragovic, R. A., et al. Sizing and phenotyping of cellular vesicles using Nanoparticle Tracking Analysis. Nanomedicine: Nanotechnology, Biology, and Medicine. 7 (6), 780-788 (2011).
- Dragovic, R. A., et al. Isolation of syncytiotrophoblast microvesicles and exosomes and their characterisation by multicolour flow cytometry and fluorescence Nanoparticle Tracking Analysis. Methods. 87, 64-74 (2015).
- Gercel-Taylor, C., Atay, S., Tullis, R. H., Kesimer, M., Taylor, D. D. Nanoparticle analysis of circulating cell-derived vesicles in ovarian cancer patients. Analytical Biochemistry. 428 (1), 44-53 (2012).
- Minta, A., Kao, J. P., Tsien, R. Y. Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. Journal of Biological Chemistry. 264 (14), 8171-8178 (1989).
- Harkins, A. B., Kurebayashi, N., Baylor, S. M. Resting myoplasmic free calcium in frog skeletal muscle fibers estimated with fluo-3. Biophysical Journal. 65 (2), 865-881 (1993).
- Hernandez-Santana, A., Yavorskyy, A., Loughran, S. T., McCarthy, G. M., McMahon, G. P. New approaches in the detection of calcium-containing microcrystals in synovial fluid. Bioanalysis. 3 (10), 1085-1091 (2011).
- Tong, M., Brown, O. S., Stone, P. R., Cree, L. M., Chamley, L. W. Flow speed alters the apparent size and concentration of particles measured using NanoSight nanoparticle tracking analysis. Placenta. 38, 29-32 (2016).
- Maas, S. L., et al. Possibilities and limitations of current technologies for quantification of biological extracellular vesicles and synthetic mimics. Journal of Controlled Release. 200, 87-96 (2015).
- Hole, P., et al. Interlaboratory comparison of size measurements on nanoparticles using nanoparticle tracking analysis (NTA). Journal of Nanoparticle Research. 15, 2101 (2013).
- Tomlinson, P. R., et al. Identification of distinct circulating exosomes in Parkinson’s disease. Annals of Clinical and Translational Neurology. 2 (4), 353-361 (2015).

