Flow Cytometric Detection of Newly-formed Breast Cancer Stem Cell-like Cells After Apoptosis Reversal
Summary
Here, we present a protocol to isolate apoptotic breast cancer cells by fluorescence-activated cell sorting and further detect the transition of breast non-stem cancer cells to breast cancer stem cell-like cells after apoptosis reversal by flow cytometry.
Abstract
Cancer recurrence has long been studied by oncologists while the underlying mechanisms remain unclear. Recently, we and others found that a phenomenon named apoptosis reversal leads to increased tumorigenicity in various cell models under different stimuli. Previous studies have been focused on tracking this process in vitro and in vivo; however, the isolation of real reversed cells has yet to be achieved, which limits our understanding on the consequences of apoptosis reversal. Here, we take advantage of a Caspase-3/7 Green Detection dye to label cells with activated caspases after apoptotic induction. Cells with positive signals are further sorted out by fluorescence-activated cell sorting (FACS) for recovery. Morphological examination under confocal microscopy helps confirm the apoptotic status before FACS. An increase in tumorigenicity can often be attributed to the elevation in the percentage of cancer stem cell (CSC)-like cells. Also, given the heterogeneity of breast cancer, identifying the origin of these CSC-like cells would be critical to cancer treatment. Thus, we prepare breast non-stem cancer cells before triggering apoptosis, isolating caspase-activated cells and performing the apoptosis reversal procedure. Flow cytometry analysis reveals that breast CSC-like cells re-appear in the reversed group, indicating breast CSC-like cells are transited from breast non-stem cancer cells during apoptosis reversal. In summary, this protocol includes the isolation of apoptotic breast cancer cells and detection of changes in CSC percentage in reversed cells by flow cytometry.
Introduction
Cancer has been a leading cause of death, causing heavy burden to countries worldwide1. Breast cancer ranks high both in terms of incidence and mortality in female patients among all types of cancer1. Due to the cancer heterogeneity, a combination of drugs is usually used in chemotherapy to achieve cancer cell death2,3,4. However, since common chemotherapeutic drugs often target DNA5,6, protein synthesis7,8 and/or microtubule dynamics9, rapidly growing cells are affected the most while quiescent cells such as cancer stem cell (CSC)s are usually less affected10. CSCs are, therefore, more likely to survive after the treatment, which later leads to drug resistance and cancer relapse10,11. Hence, elimination of CSCs has become an important topic for cancer treatment and study of the origin of CSCs is necessary.
More studies on the phenomenon of apoptosis reversal have been performed in the recent decade12,13,14,15,16,17,18,19. Before the emergence of this concept, it has been widely accepted that cells will irreversibly undergo apoptosis after caspase activation. Caspases are a family of protein enzymes that play key roles in the initiation and execution stages of apoptosis, including the formation of the apoptotic complex and the cleavage of downstream substrates20. Activation of executioner caspases such as caspase 3 or caspase 7 has been considered as the "point of no return" for apoptosis21. However, researchers recently observed that apoptosis reversal occurs both in vitro and in vivo, during which cells can recover from apoptosis even after caspase activation12,13,14,15,16,17,18,19. Moreover, aggressive features such as higher resistance to the original apoptotic inducer and higher invasiveness are detected in the reversed cancer cells15. Hence, it was proposed that the percentage of CSC-like cells would be higher in the reversed population when compared to the untreated cells, eventually contributing to the more malignant features after apoptosis reversal18.
Previously, many efforts have been made to track the apoptosis reversal in vitro and more importantly, in vivo, which greatly help in confirming the universality of this process16,17,19. However, a systemic study on the consequences of reversed cells is lacking due to the unsatisfactory isolation of cells that have genuinely undergone apoptosis reversal. There is a need to acquire pure apoptotic cells and recover them for further study. Thus, we use the traditionally well-accepted marker of executioner caspase activation as the marker of the "point of no return"21 for apoptosis and utilize fluorescence-activated cell sorting (FACS) to discriminate caspase-activated cells stained with Caspase-3/7 Green Detection dye. The dye is covalently linked to a short amino acid sequence, DEVD, which can be recognized and cleaved by active caspases 3/7. The cleavage helps release the dye, which will translocate from the cytosol to the nucleus where it binds to DNA and emits strong fluorescence. This procedure avoids using a bulk cell population in which some cells may not have undergone apoptosis.
CSCs or tumor-initiating cells have been identified in many solid tumors using a single or a combination of several surface marker(s) and very few numbers of these cells are sufficient to form tumors in immunodeficient mice22,23,24,25,26,27. A combination of CD44 and CD24 has been commonly used in breast CSC studies, and CD44+/CD24– cells have been defined as the breast CSCs26,27,28,29,30. Recently, we have performed a series of experiments to confirm the proposed relationship between apoptosis reversal and CSCs and demonstrate that reversed breast cancer cells gained increased tumor-forming ability in vitro and in vivo with an elevated percentage of cells with CSC markers18. Although we could not exclude the possibility that breast CSCs survive better and thus get enriched after apoptosis reversal, importantly, when we isolate non-stem cancer cells and subject them to apoptosis reversal, CSC will emerge in the originally non-stem cancer cell population, suggesting that non-stem cancer cells can contribute to the increase in the percentage of CSCs during apoptosis reversal.
This article aims to demonstrate the transition from breast non-stem cancer cells to breast CSC-like cells after apoptosis reversal and to detect this transition by flow cytometry. The breast non-stem cancer cells are initially prepared by isolating CD44–/CD24+ breast cancer cells by FACS. Then, apoptosis is induced and confirmed by morphological changes under the microscope. Afterwards, apoptotic cells positively-labelled by Caspase 3/7 Green Detection dye are isolated by FACS and further cultured in the absence of apoptotic inducers for apoptosis reversal. The reversed cells are then stained with CSC markers after 7 days of recovery for flow cytometric analysis. Cells with CD44+/CD24– markers re-appear in the reversed population, suggesting that transition from non-stem cancer cells to CSC-like cells has occurred during apoptosis reversal.
Apoptosis reversal has been observed in multiple cancer cell lines as well as normal primary cells treated with different apoptotic stimuli in vitro12,13. This process has also been traced in Drosophila model in vivo16,17,19. Much information regarding the underlying mechanism of cancer relapse in different cancer disease models and the origin of CSCs can be obtained through the use of the technique as described in this manuscript.
Protocol
1. Preparation of Breast Non-stem Cancer Cells
- Culture MCF-7 and MDA-MB-231 cells in 10 mL of phenol red-free Roswell Park Memorial Institute (RPMI) 1640 medium supplemented with 10% heat-inactivated fetal bovine serum (FBS) in a 100 mm dish. Culture T47D in 10 mL of phenol red-free RPMI 1640 medium supplemented with 2 mM L-glutamine, 10% heat-inactivated FBS, and 1% v/v Penicillin-Streptomycin (PS) in a 100 mm dish. Culture cells at 37 °C in a 5% CO2/95% air cell culture incubator.
NOTE: Phenol-red affects fluorescence-based detection but also has a potential influence on the growth of breast cancer cells due to its estrogen-like effects31. For example, phenol red could stimulate progesterone receptor and growth of MCF-7 cells31. It could also stimulate the growth of T47D cells31. - Wash cells with 2 mL of phosphate buffered saline (PBS) twice. Add 2 mL of 0.05% trypsin-EDTA to MCF-7 and MDA-MB-231 or 2 mL of 0.25% trypsin-EDTA to T47D cells.
NOTE: The concentration of trypsin-EDTA needed for separation of cells among different breast cancer cell types vary while an inappropriate concentration may affect the expression pattern of some cell surface markers such as CD44 and CD2432. - Culture the dishes for 5 min at 37 °C under an atmosphere of 5% CO2/95% air. Frequently check the detachment of cells under the microscope to prevent cells from over-digestion by trypsin-EDTA.
- When over 90% cells detach, add 5 mL of completed RPMI medium to the dish and pipette it over the cell layer surface several times.
- Transfer the cells to a 15 mL conical tube and centrifuge at 300 x g and 4 °C for 5 min.
NOTE: Usually, around 6 x 106 cells can be obtained from a 100 mm dish when the confluency reaches 70%. - Discard the supernatant and resuspend the pellets at 106 cells/100 μL of FACS buffer (PBS containing 0.5% BSA and 0.1% sodium azide) in a 1.5 mL microcentrifuge tube.
- Divide the cells into several tubes. For MCF-7 cells, divide the cells into 4 tubes: two for isotype controls, one for CD24 single staining as CD24 positive control and one for dual staining.
- For T47D cells, divide the cells into 3 tubes: two for isotype controls and one for dual staining.
- For MDA-MB-231 cells, divide the cells into 4 tubes: two for isotype controls, one for CD44 single staining as CD44 positive control and one for dual staining.
- Centrifuge the tubes again at 300 x g and 4 °C for 5 min. Discard the supernatant, and then add Fc block diluted 1:50 in FACS buffer.
- Incubate the samples on ice for 20 min in the dark before centrifugation at 300 x g and 4 °C for 5 min. Discard the supernatant.
- Add fluorochrome-conjugated monoclonal antibodies against human CD44 (PerCP-Cy5.5) and CD24 (PE) at 1:40 and 1:10 dilutions (in FACS buffer) in the dual staining groups. For positive controls, add CD44 (PerCP-Cy5.5) to MDA-MB-231 and add CD24 (PE) to MCF-7 cells at the same concentration, respectively.
NOTE: The combination of CD44 and C24 has been commonly used in breast CSC study26,27,28,29,30. - For the isotype control groups, add PerCP-Cy5.5 Mouse IgG2b,κ at a 1:40 dilution as the isotype control for CD44 antibodies and PE Mouse IgG2a,κ at a 1:10 dilution as the isotype control for CD24 antibodies at 106 cells/100 μL.
NOTE: Isotype controls for the antibodies of interest are recommended as negative controls33. - Incubate the samples (from steps 1.12 and 1.13) at 4 °C in the dark for 30 min. Centrifuge at 300 x g and 4 °C for 5 min. Discard the supernatant.
- Wash the pellet twice with 500 μL of PBS and centrifuge at 300 x g and 4 °C for 5 min.
- Resuspend the pellet in 0.5 mL of PBS and filter through a 40 µm nylon mesh before running on a fluorescence-activated cell sorter.
- For gating and compensation purposes, stain MCF-7 cells with PE-conjugated anti-CD24 antibodies and stain MDA-MB-231 cells with PerCP-Cy5.5-conjugated anti-CD44 antibodies as positive controls. Use cells stained with isotype controls as negative controls (Figure 1).
- Collect cells with CD44–/CD24+ markers in round-bottom polystyrene 12 x 75 mm tubes containing 1 mL of collection medium (phenol red-free RPMI 1640 medium supplemented with 20% heat-inactivated FBS and 2% v/v PS). Centrifuge the tubes at 300 x g and RT for 5 min and discard the supernatant.
NOTE: CSC-like cells in breast cancer cells are defined as CD44+/CD24– cells26,27,28,29,30, and breast non-stem cancer cells are defined as CD44–/CD24+ cells18 (Figure 1). - Plate the sorted breast non-stem cancer cells in the culture dish containing fresh collection medium for further culture at 37 °C in 5% CO2 cell culture incubator.
2. Apoptotic Induction and Detection
- Prepare 1 mM staurosporine12,34 in DMSO. For the treated MCF-7 group, add 25 μL of 1 mM staurosporine to the completed medium of MCF-7 cells to make up to 10 mL of final volume (2.5 μM staurosporine) in a 15 mL conical tube. Pipette to mix the content evenly.
- Remove the culture medium from the cells, wash the cells with 2 mL of PBS once. Then add the 10 mL of medium with 2.5 μM staurosporine to MCF-7 cells for 6 h to induce apoptosis when cell density reaches 70% confluency.
- Prepare 1 mM paclitaxel35,36 in DMSO. For the treated T47D group, add 12.5 μL of 1 mM paclitaxel to the completed medium of T47D cells to make up to a final volume of 10 mL (5 μM paclitaxel) in a 15 mL conical tube. Pipette to mix the content evenly.
- Remove the culture medium from the cells, and wash the cells with 2 mL of PBS once. Then add the 10 mL of medium with 5 μM paclitaxel to the T47D cells for 10 h to induce apoptosis when cell density reaches 70% confluency.
NOTE: The induction time and the concentration of the inducers that induce apoptosis should be optimized whenever a new cell line is used for the first time. - For the solvent-treated MCF-7 group, add 25 μL of sterile dimethyl sulfoxide (DMSO) to the completed medium of MCF-7 cells to make up to a final volume of 10 mL (i.e., 0.25% v/v DMSO) in a 15 mL conical tube. Pipette to mix the content evenly.
- Remove the culture medium from the solvent treated MCF-7 cells, wash with 2 mL of PBS once. Then add the 10 mL of medium with 0.25% v/v DMSO for 6 h as the solvent control for STS.
- For the solvent-treated T47D group, add 5 μL of sterile DMSO to the completed medium of T47D cells to make up to 10 mL final volume (i.e., 0.05% v/v DMSO) in a 15 mL conical tube. Pipette to mix the content evenly.
- Remove the culture medium from the solvent treated T47D cells, wash with 2 mL of PBS once. Then, add the 10 mL of medium with 0.05% v/v DMSO for 10 h as the solvent control for paclitaxel.
- To observe the morphological changes of treated cells, stain cells with 50 nM Mitotracker Red CMXRos and 250 ng/mL Hoechst 33342, and incubate for another 20 min at 37 °C under an atmosphere of 5% CO2/95% air. Observe the typical apoptotic cell morphology under a 60x confocal laser scanning microscope (Figure 2).
NOTE: Typical apoptosis morphology includes cell shrinkage, membrane blebbing, mitochondria fragmentation and nuclear condensation but with intact cellular membrane12,13,14,15,18,37.
3. Isolation of Apoptotic Cells and Apoptosis Reversal Procedure
- Stain both the apoptotic inducer- and solvent-treated MCF-7 or T47D cells with 3 μM Caspase-3/7 Green Detection dye at the cell concentration of 106/mL in the dark for 30 min at 37 °C under an atmosphere of 5% CO2/95% air.
- Filter the cells through a 40 µm nylon mesh before running on the sorter for sorting.
- For gating purposes, first gate the major population (R1) and exclude the debris by plotting the graph in dots with forward scatter and side scatter (Figure 3).
- Use the untreated cells without adding the Caspase-3/7 Green Detection dye as a negative control to gate the negative region (R2) (Figure 3A).
- Use cells treated with staurosporine12,18,34 for 24 h and stained with the dye as a positive control to gate the positive region (R3) (Figure 3B).
- Collect positive cells (R3) from the inducer-treated groups in round-bottom polystyrene 12 x 75 mm tubes containing 1 mL of collection medium (same as the collection medium in step 1.18) (Figures 3C-3D). Centrifuge the tubes at 300 x g and RT for 5 min and discard the supernatant.
- Collect negative cells (R2) from solvent-treated groups in round-bottom polystyrene 12 × 75 mm tubes with 1 mL of collection medium as in step 3.6 (Figures 3E-3F).
NOTE: A small portion of cells (such as 10,000 cells) can be collected and re-run on the sorter to check the pattern of active caspases marker. If over 90% of sorted solvent-treated cells are shown in the caspase-negative region or over 90% of sorted inducer-treated cells are shown in the caspase-positive region, these collected cells are believed to be relatively pure (Figures 3C-3F). Otherwise, the sorting region and/or the sorter should be reset. - Resuspend the sorted cells in fresh collection medium and seed them in 12-well tissue culture plates and culture at 37 °C under an atmosphere of 5% CO2/95% air for 7 days for apoptosis reversal.
4. Confirmation of Apoptosis in Caspase-activated Cells
- Mix 10 mM HEPES, 140 mM NaCl, and 2.5 mM CaCl2 to prepare annexin-binding buffer (pH 7.4). Store the buffer at 4 °C and avoid the buffer to expose to light.
- Prepare a 100 µg/mL working solution of propidium iodide (PI) by diluting 5 µL of the 1 mg/mL PI stock solution in 45 µL of annexin-binding buffer. Store the solution at 4 °C and avoid the solution to expose to light.
NOTE: PI is a potential mutagen and should be handled with care. - Prepare the inducer-treated cells as in steps 2.1 to 2.5.
- Collect the inducer-treated MCF-7 or T47D cells by pipetting the medium over the cell layer 3-5x to detach the cells. Transfer the cells to a 15 mL conical tube.
- Centrifuge at 300 x g and RT for 5 min. Discard the supernatant.
- Resuspend the cells with completed medium at the cell concentration of 106/mL in a 1.5 mL microcentrifuge tube and stain cells with 3 μM Caspase 3/7 Green Detection dye in the dark for 30 min at 37 °C.
- Centrifuge at 300 x g and RT for 5 min. Discard the supernatants.
- Wash the cells with 1 mL of ice-cold PBS and centrifuge at 300 x g and 4 °C for 5 min. Discard the supernatants.
- Resuspend the cells in 100 μL of annexin-binding buffer with the add 5 μL of the annexin V conjugate and 1 µL of 100 µg/mL PI working solution to each 100 μL of cell suspension and incubate the cells for 15 min at room temperature.
NOTE: Annexin V and PI are commonly used together to label apoptotic cells in flow cytometry38,39,40. - Add 400 µL of annexin-binding buffer, mix gently. Keep the samples on ice before running on a flow cytometer.
5. Measurement of Breast CSC-like Cells by Flow Cytometry
- Harvest the reversed MCF-7 in both inducer- or solvent-treated groups with 0.05% trypsin-EDTA. Harvest T47D cells in both inducer- or solvent-treated groups or with 0.25% trypsin-EDTA as in steps 1.2 to 1.5.
- Stain the cells with fluorochrome-conjugated monoclonal antibodies against human CD44 (PerCP-Cy5.5) and CD24 (PE) as in step 1.12. Meanwhile, prepare the isotype controls as in step 1.13.
- Run the cells on a flow cytometer and detect the percentage of cells with CD44+/CD24– markers.
Representative Results
In order to observe the transition from breast non-stem cancer cells to breast CSC-like cells, a first sorting of CD44–/CD24+ breast cancer cells were needed. For the MCF-7 cell line, which has around 0.15% cells with CSC markers in the original population (Figure 1), this step helped exclude the possibility of CSC enrichment during apoptosis reversal. On the contrary, if there were no cells with CSC markers in the original population, such as for T47D cells (Figure 1), this sorting procedure could be omitted. Indeed, gating affected the definition of the positive and negative of each marker, which would eventually influence the percentage of CSC determined. Therefore, appropriate controls including isotype controls for antibodies of interest, single stained controls for each marker should be carefully chosen and prepared for gate adjustment (Figure 1).
With breast non-stem cancer cells, the apoptosis reversal model could thereafter be established. Typical morphological changes could be observed after adding apoptotic inducers and cells should recover from apoptosis with similar morphology after drug withdrawal (Figure 2). Caspase-3/7 recognizes the amino acid sequence DEVD in the Caspase-3/7 Green Detection dye and the active forms of caspase-3/7 are able to cleave this site41. Originally, the fluorescence of the Caspase-3/7 Green Detection dye in cytosol is weak while if the dye is cleaved and translocated to the nucleus, the fluorescence signal would be amplified after its binding to DNA in the nucleus. This obvious difference could be distinguished by flow cytometry. Hence, those caspase-activated cells could be labelled and sorted out based on their higher fluorescence intensity comparing to those without caspase activation (Figures 3A-3D). During the apoptotic induction process, an appropriate solvent control must be included: solvent-treated cells without caspase activation were collected (Figures 3E and 3F) to exclude the possibility that the FACS procedure or the solvent itself was the cause for transition, if any.
In order to show that those FACS-sorted caspase-activated cells were indeed apoptotic, we co-stained these cells with Annexin V and PI. One of the early-stage changes in apoptotic cells is the translocation of phosphatidylserine from the inner side of plasma membrane to the external surface of the cell13,38,39,40; this externalization of phosphatidylserine could be detected by Annexin V. While this change is not unique to apoptosis, PI can be used for distinguishing apoptosis from necrosis based on the integrity of the cell membrane. Thus, cells with Annexin V binding but without PI staining are regarded as apoptotic cells. Caspase-activated cells in the apoptotic inducer treatment groups were found to be Annexin V positive and PI negative, suggesting that they were apoptotic cells (Figure 4).
After the second sorting based on caspase activation in apoptotic cells, these apoptotic cells were collected and subsequently cultured for recovery. The reversed cells that were alive were able to attach the culture container bottom and continue to proliferate. After 7 days, staining of CD44 and CD24 was performed in the reversed cells while controls were prepared at the same time as before. Compared with the solvent-treated groups (Figure 1), flow cytometric analysis showed that there were events (cells) appearing in the CD44+/CD24– quadrant in the reversed breast non-stem cancer cell population (Figure 1). Since we had already excluded cells with CD44+/CD24– in advance of apoptosis induction and only CD44–/CD24+ breast non-stem cancer cells were chosen, these CD44+/CD24– CSC-like cells could only be transited from breast non-stem cancer cells during apoptosis reversal.
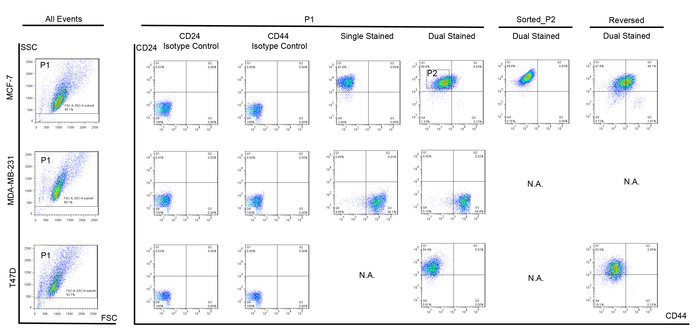
Figure 1: Representative breast CSC marker staining on breast cancer cells in flow cytometry.
MCF-7, MDA-MB-231 and T47D were stained with fluorochrome-conjugated monoclonal antibodies against human CD44 (PerCP-Cy5.5) and CD24 (PE). Cells were first gated by forward scatter (FSC) and side scatter (SSC) (P1) to exclude debris. Isotype controls of CD24 and CD44 were used as negative controls for CD24 and CD44 respectively. MCF-7 cells stained with CD24 was used as positive control for CD24 and MDA-MB-231 cells stained with CD44 was used as positive control for CD44. Non-stem MCF-7 breast cancer cells (P2) were sorted. These sorted cells and T47D cells were subjected to apoptosis reversal procedure. CSC-like (CD44+CD24–) cells appeared after apoptosis reversal. Please click here to view a larger version of this figure.
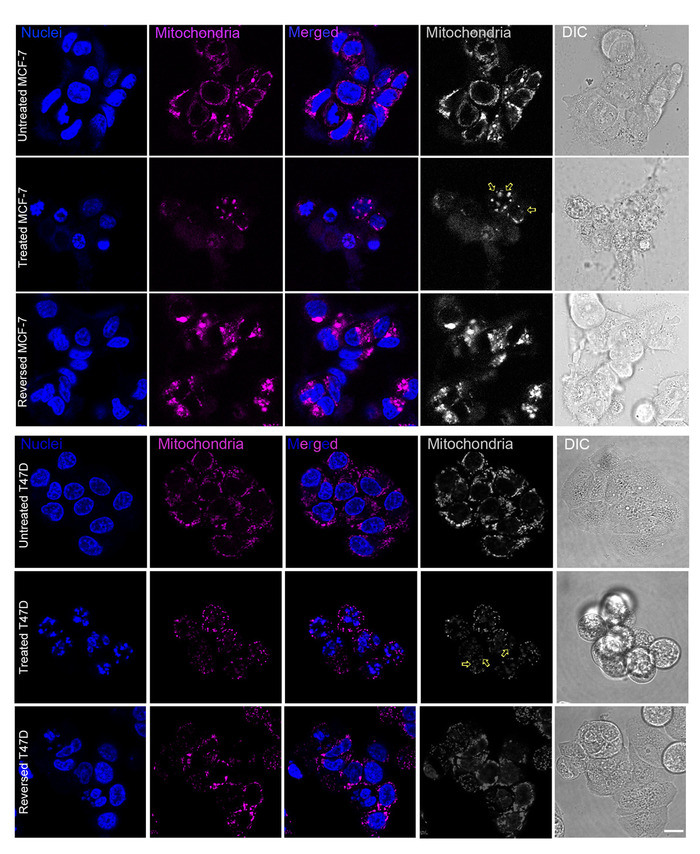
Figure 2: Morphological changes of breast cancer cells under apoptotic stimulus induction.
Breast cancer cells showed typical apoptotic morphological changes including cell shrinkage, membrane blebbing, mitochondria fragmentation (yellow arrows in the monochrome figures) and nuclear condensation. Nuclei (in blue): nuclei of cells stained with Hoechst 33342 were shown in blue color. Mitochondria (in pink): mitochondria of cells stained with Mitotracker Red CMXRos were shown in pink color. Merged: figures merged in dual colors showing both mitochondria and nuclei. Mitochondria (in gray): mitochondria of cells stained with Mitotracker Red CMXRos were shown in monochrome. Differential Interference Contrast (DIC): whole cells. Upper: MCF-7 cells were treated with staurosporine (STS) for 6 h then reversed for 24 h. Lower: T47D cells were treated with paclitaxel for 10 h with reversal for 24 h. Scale bars = 20 μm. Please click here to view a larger version of this figure.
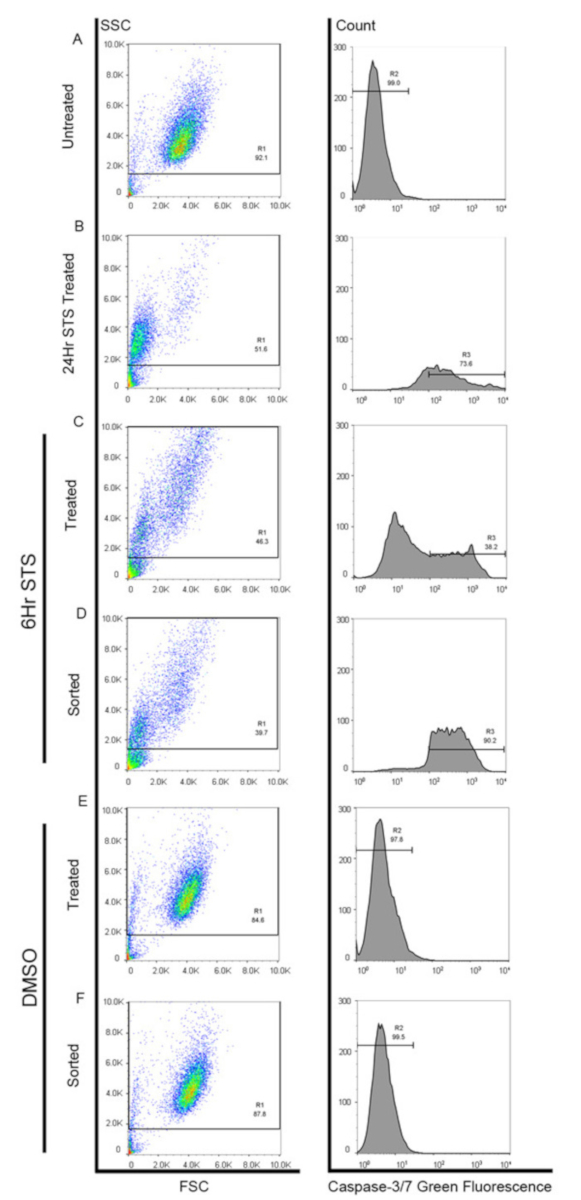
Figure 3: Caspase activation analysis by sorter. (A) Unstained MCF-7 cells without staurosporine (STS) treatment were used as negative control (R2). (B) MCF-7 cells treated with staurosporine (STS) for 24 h. Cells in R3 region were regarded as the caspase-activated cells. (C) MCF-7 cells treated with staurosporine (STS) for 6 h. Cells in R3 region were FACS-sorted. (D) FACS-sorted caspase-activated MCF-7 cells after staurosporine (STS) treatment for 6 h were re-run. (E) DMSO-treated MCF-7 cells. (F) FACS-sorted cells without caspase activation after DMSO treatment for 6 h were re-run. Please click here to view a larger version of this figure.
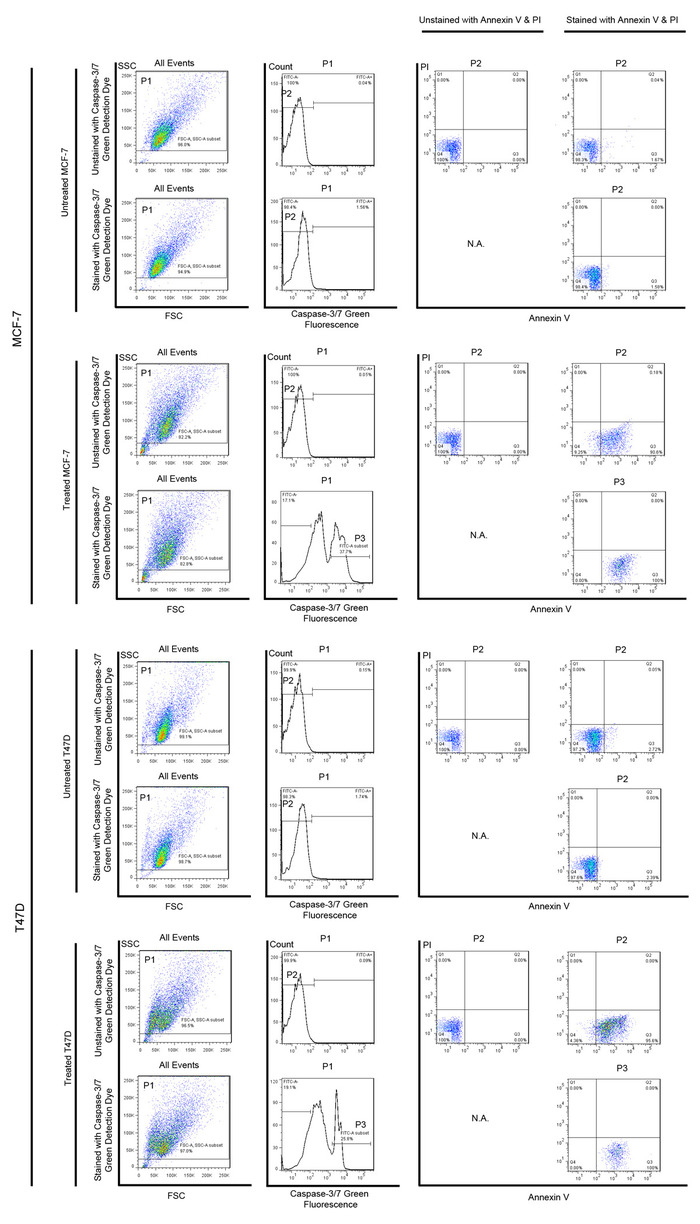
Figure 4: Annexin V and PI staining of caspase-activated cells in flow cytometry. MCF-7 and T47D cells were firstly gated by forward scatter (FSC) and side scatter (SSC) (P1) to exclude debris. Cells that were untreated with apoptotic inducers and without any staining were used as negative controls. For cells treated with apoptotic inducers, caspase-activated cells [i.e., Caspase-3/7 Green positive cells] were selected (P3) to show the fluorescence intensity of Annexin V and PI staining. All Caspase-3/7 Green positive cells were Annexin V positive and PI negative, suggesting that they were apoptotic. Please click here to view a larger version of this figure.
Discussion
This protocol describes a direct and clear way for detecting the transition of breast non-stem cancer cells into breast CSC-like cells as a result of apoptosis reversal. Confirmation of the CSC properties of these reversed cells could be assisted by using in vitro mammosphere formation assay and in vivo xenograft transplantation in immunodeficient mice18,24,26,27,42,43,44,45. Here, we use two breast cancer cell lines, but this protocol can be further applied in other breast cancer cell lines. Since this phenomenon is apparent in the breast cancer cell line in which fewer number of CD44+/CD24– cells exist originally, it is suggested not to choose cell lines such as MDA-MB-231 in which there are more than 90% of CD44+ cells.
Since gating is important in flow analysis, proper and sufficient controls should be prepared in advance. For sorting, the purity should also be determined before the actual collection of cells. For example, to know the purity of the first sorting, a small portion of cells (such as 10,000 cells) can be collected and re-run on the sorter to check the pattern of CSC markers. If over 90% of these sorted cells are CD44–/CD24+ and are not shown in the CD44+/CD24– region, the collected cells are believed to be relatively pure (Figure 1). If cells appear in the CD44+/CD24– region, the sorting region and/or the sorter should be reset. Sorting should be conducted as sterile as possible either by using a sorter inside culture hood or adding antibiotics in the collection and culture medium for sorted cells.
Cancer cell types other than breast cancer can also be chosen and induced with various apoptotic stimuli to establish the apoptosis reversal model. While caspase activation could be detected as early as 2 h, the sorting time should be carefully selected. Since cells in the population are not synchronized, at one specific time point, each cell may have reached different stages of apoptosis. Meanwhile, cells keep going on to an apoptotic death after caspase activation. Therefore, if the inducers are removed only when all the cells reach caspase activation, some cells may have already reached the stage of DNA fragmentation, making them unable to recover even after inducer removal. It is not suggested to induce apoptosis for too long time to achieve a higher percentage of caspase-activated cells but with the cost of leaving a large number of cells dying after collection. Besides, the incubation time and the dye concentration that labels caspase-activated cells should be optimized whenever a new cell line is used for the first time.
Another concern as of successfully inducing apoptosis reversal in the apoptotic cells is the low recovery rate of the cells (usually less than 10%). The cells have gone through apoptosis plus the sorting procedure; therefore, they require very gentle handling during centrifugation and transfer at the collection and resuspension steps. Also, the choice of plate or dish used for subsequent culture should not be dependent on the number of cells collected but on the expected number of recoveries. Otherwise, cells could be allocated too sparsely to reverse and re-grow. Given that non-stem cancer cells grow at a higher speed and may dominate in the re-constituted cell population46, the detection time should not be too far away from the first recovery day.
This method first isolates breast non-stem cancer cell then applies them for apoptotic induction where a second sorting is done based on caspase activation before cells get recovered. The re-appearance of breast CSC-like cells in the reversed population is detected by flow cytometry. Since this in vitro apoptosis reversal procedure isolates pure apoptotic cancer cells, a number of experiments now can be conducted to understand the consequences of these real reversed cells. Apart from breast cancer cells, other solid tumor types as well as hematologic malignancy that are with known CSC markers can be studied and various apoptotic stimulus can be used. Hence, this method is extendable and applicable to a boarder scope of cancer investigation.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Innovative Technology Fund of Innovation Technology Commission: Funding Support from the State Key Laboratory of Agrobiotechnology (CUHK), the Lo Kwee-Seong Biomedical Research Fund and the Lee Hysan Foundation. Y.X. was supported by the postgraduate studentship from the CUHK.
Materials
| MCF-7 | American Type Culture Collection (ATCC) | HTB-22 | |
| MDA-MB-231 | American Type Culture Collection (ATCC) | HTB-26 | |
| T47D | American Type Culture Collection (ATCC) | HTB-133 | |
| Reagent | |||
| 0.05% trypsin-EDTA | Invitrogen | 25300054 | |
| 0.25% trypsin-EDTA | Invitrogen | 25200072 | |
| Alexa Fluor 680 annexin V conjugate | Invitrogen | A35109 | |
| bovine serum albumin | USB | 9048-46-8 | |
| CaCl2 · 2H2O | Sigma-Aldrich | C-5080 | |
| CellEvent caspase-3/7 green fluorescent dye | Invitrogen | C10423 | |
| dimethyl sulfoxide | Sigma-Aldrich | D2650 | |
| Fc block | Miltenyi Biotec | 130-059-901 | |
| fetal bovine serum | Invitrogen | 16000044 | heat-inactivated |
| HEPES | USB | 16926 | |
| Hoechst 33342 | Invitrogen | H3570 | |
| L-glutamine | Invitrogen | 25030081 | |
| Mitotracker Red CMXRos | Invitrogen | M7512 | |
| monoclonal antibodies against human CD24 | BD Biosciences | 555428 | PE Clone:ML5 Lot:5049759 RRID:AB_395822 |
| monoclonal antibodies against human CD44 | BD Biosciences | 560531 | PERCP-CY5.5 Clone:G44-26 Lot:7230770 RRID:AB_1727485 |
| NaCl | Sigma-Aldrich | 31434 | |
| paclitaxel | Sigma-Aldrich | T7402 | |
| PE Mouse IgG2a, κ Isotype Control | BD Biosciences | 554648 | Clone:G155-178 (RUO) RRID:AB_395491 |
| Penicillin-Streptomycin | Invitrogen | 15070-063 | |
| PerCP-Cy5.5 Mouse IgG2b, κ Isotype Control | BD Biosciences | 558304 | Clone:27-35 RRID:AB_647257 |
| phosphate buffered saline | Thermo Fisher Scientific | 21600010 | |
| propidium iodide | Invitrogen | P1304MP | |
| Roswell Park Memorial Institute 1640 medium | Invitrogen | 11835055 | phenol red-free |
| sodium azide | Sigma-Aldrich | S2002 | |
| staurosporine | Sigma-Aldrich | S4400 | |
| Equipment | |||
| 100 mm culture dish | Greiner Bio-One | 664160 | |
| 12-well tissue culture plates | Thermo Fisher Scientific | 150628 | |
| Cell Strainer 40-μm nylon mesh | BD Biosciences | 08-771-1 | |
| FACSuite software bundle v1.0 | BD Biosciences | 651360 | |
| FACSVerse | BD Biosciences | 651155 | |
| FluoView FV1000 confocal microscope | Olympus | IX81 | 60X objective |
| FV10-ASW Viewer software Ver.4.2b | Olympus | – | |
| round-bottom polystyrene 12 × 75 mm tubes | BD Biosciences | 352003 | |
| S3e sorter | Bio-Rad | 1451006 |
Referencias
- McGuire, S. World Cancer Report 2014. Geneva, Switzerland: World Health Organization, International Agency for Research on Cancer, WHO Press, 2015. Advances in Nutrition. 7 (2), 418-419 (2015).
- Slamon, D. J., et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. New England Journal of Medicine. 344 (11), 783-792 (2001).
- Geyer, C. E., et al. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. New England Journal of Medicine. 355 (26), 2733-2743 (2006).
- Cameron, D., et al. A phase III randomized comparison of lapatinib plus capecitabine versus capecitabine alone in women with advanced breast cancer that has progressedon trastuzumab: updatedefficacy andbiomarker analyses. Breast Cancer Research and Treatment. 112 (3), 533-543 (2008).
- Liu, L. F. DNA topoisomerase poisons as antitumor drugs. Annual Review of Biochemistry. 58, 351-375 (1989).
- Hertel, L. W., et al. Evaluation of the antitumor activity of gemcitabine (2′,2′-difluoro-2′-deoxycytidine). Investigación sobre el cáncer. 50 (14), 4417-4422 (1990).
- Ni, H., et al. Analysis of expression of nuclear factor kappa B (NF-kappa B) in multiple myeloma: downregulation of NF-kappa B induces apoptosis. British Journal of Haematology. 115 (2), 279-286 (2001).
- Richardson, P. G., Mitsiades, C., Hideshima, T., Anderson, K. C. Bortezomib: proteasome inhibition as an effective anticancer therapy. Annual Review of Medicine. 57, 33-47 (2006).
- Dumontet, C., Jordan, M. A. Microtubule-binding agents: a dynamic field of cancer therapeutics. Nature Review Drug Discovery. 9 (10), 790-803 (2010).
- Dean, M., Fojo, T., Bates, S. Tumour stem cells and drug resistance. Nature Review Cancer. 5 (4), 275-284 (2005).
- Pirozzi, G., et al. Prognostic value of cancer stem cells, epithelial-mesenchymal transition and circulating tumor cells in lung cancer. Oncology Reports. 29 (5), 1763-1768 (2013).
- Tang, H. L., Yuen, K. L., Tang, H. M., Fung, M. C. Reversibility of apoptosis in cancer cells. British Journal of Cancer. 100 (1), 118-122 (2009).
- Tang, H. L., et al. Cell survival, DNA damage, and oncogenic transformation after a transient and reversible apoptotic response. Molecular Biology of the Cell. 23 (12), 2240-2252 (2012).
- Xie, X., Wang, S. S., Wong, T. C. S., Fung, M. C. Genistein promotes cell death of ethanol-stressed HeLa cells through the continuation of apoptosis or secondary necrosis. Cancer Cell International. 13 (1), 63 (2013).
- Wang, S. S., Xie, X., Wong, C. S., Choi, Y., Fung, M. C. HepG2 cells recovered from apoptosis show altered drug responses and invasiveness. Hepatobiliary & Pancreatic Diseases International. 13 (3), 293-300 (2014).
- Tang, H. L., Tang, H. M., Fung, M. C., Harwick, J. M. In vivo CaspaseTracker biosensor system for detecting anastasis and non-apoptotic caspase activity. Scientific Reports. 5, 9015 (2015).
- Tang, H. L., Tang, H. M., Hardwick, J. M., Fung, M. C. Strategies for tracking anastasis, a cell survival phenomenon that reverses apoptosis. Journal of Visualized Experiments. (96), (2015).
- Xu, Y., So, C., Lam, H. M., Fung, M. C., Tsang, S. Y. Apoptosis reversal promotes cancer stem cell-like cell formation. Neoplasia. 20 (3), 295-303 (2018).
- Tang, H. M., Fung, M. C., Tang, H. L. Detecting anastasis in vivo by CaspaseTracker biosensor. Journal of Visualized Experiments. (132), (2018).
- Li, J., Yuan, J. Caspases in apoptosis and beyond. Oncogene. 27 (48), 6194-6206 (2008).
- Green, D. R., Amarante-Mendes, G. P. The point of no return: mitochondria, caspases, and the commitment to cell death. Results and Problems in Cell Differentiation. 24, 45-61 (1998).
- Singh, S. K., et al. Identification of a cancer stem cell in human brain tumors. Investigación sobre el cáncer. 63 (18), 5821-5828 (2003).
- Ricci-Vitiani, L., et al. Identification and expansion of human colon-cancer-initiating cells. Nature. 445 (7123), 111-115 (2007).
- O’Brien, C. A., Pollett, A., Gallinger, S., Dick, J. E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 445 (7123), 106-110 (2007).
- Cioffi, M., et al. Identification of a distinct population of CD133(+) CXCR4(+) cancer stem cells in ovarian cancer. Scientific Reports. 5, 10357 (2015).
- Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J., Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America. 100 (7), 3983-3988 (2003).
- Ponti, D., et al. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Investigación sobre el cáncer. 65 (13), 5506-5511 (2005).
- Liu, S., et al. Breast cancer stem cells transition between epithelial and mesenchymal states reflective of their normal counterparts. Stem Cell Reports. 2 (1), 78-91 (2013).
- Sheridan, C., et al. CD44+/CD24- breast cancer cells exhibit enhanced invasive properties: an early step necessary for metastasis. Breast Cancer Research. 8 (5), R59 (2006).
- Phillips, T. M., McBride, W. H., Pajonk, F. The response of CD24(-/low)/CD44+ breast cancer-initiating cells to radiation. Journal of the National Cancer Institute. 98 (24), 1777-1785 (2006).
- Welshons, W. V., Wolf, M. F., Murphy, C. S., Jordan, V. C. Estrogenic activity of phenol red. Molecular and Cellular Endocrinology. 57 (3), 169-178 (1998).
- Tsuji, K., et al. Effects of Different Cell-Detaching Methods on the Viability and Cell Surface Antigen Expression of Synovial Mesenchymal Stem Cells. Cell Transplantation. 26 (6), 1089-1102 (2017).
- Herzenberg, L. A., Tung, J., Moore, W. A., Herzenberg, L. A., Parks, D. R. Interpreting flow cytometry data: a guide for the perplexed. Nature Immunology. 7 (7), 681-685 (2006).
- Belmokhtar, C. A., Hillion, J., Ségal-Bendirdjian, E. Staurosporine induces apoptosis through both caspase-dependent and caspase-independent mechanisms. Oncogene. 20 (26), 3354-3362 (2001).
- Saunders, D. E., et al. Paclitaxel-induced apoptosis in MCF-7 breast-cancer cells. International Journal of Cancer. 70 (2), 214-220 (1997).
- Miller, A. V., et al. Paclitaxel-induced apoptosis is BAK-dependent, but BAX and BIM-independent in breast tumor. PLoS One. 8 (4), e60685 (2013).
- Elmore, S. Apoptosis: a review of programmed cell death. Toxicologic Pathology. 35 (4), 495-516 (2007).
- Vermes, I., Haanen, C., Steffens-Nakken, H., Reutelingsperger, C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. Journal of Immunological Methods. 184 (1), 39-51 (1995).
- Ng, S. Y., et al. Role of voltage-gated potassium channels in the fate determination of embryonic stem cells. Journal of Cellular Physiology. 224 (1), 165-177 (2010).
- Lo, I. C., et al. TRPV3 channel negatively regulates cell cycle progression and safeguards the pluripotency of embryonic stem cells. Journal of Cellular Physiology. 231 (1), 403-413 (2016).
- Talanian, R. V., et al. Substrate specificities of caspase family proteases. Journal of Biological Chemistry. 272 (15), 9677-9682 (1997).
- Dontu, G., et al. In vitro propagation and transcriptional profiling of human mammary stem/progenitor cells. Genes & Development. 17 (10), 1253-1270 (2003).
- Vermeulen, L., et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nature Cell Biology. 12 (5), 468-476 (2010).
- Shaw, F. L., et al. A detailed mammosphere assay protocol for the quantification of breast stem cell activity. Journal of Mammary Gland Biology and Neoplasia. 17 (2), 111-117 (2012).
- Desiderio, V., et al. Increased fucosylation has a pivotal role in invasive and metastatic properties of head and neck cancer stem cells. Oncotarget. 6 (1), 71-84 (2015).
- Gupta, P. B., et al. Stochastic State Transitions Give Rise to Phenotypic Equilibrium in Populations of Cancer Cells. Cell. 146 (4), 633-644 (2011).

