Detection of Abnormal Prion Protein by Immunohistochemistry
Summary
Immunolabeling abnormal prion protein using immunohistochemistry protocols requires specific sample and anti-PrP antibody preparation methodologies. The present protocol describes the key steps in epitope demasking to ensure proper PrP immunolabeling and to minimize non-specific background staining. Also, this approach considers biosafety measures when conducting immunohistochemistry studies with the prion-infected tissues.
Abstract
Abnormal prion proteins (PrPSc) are the disease-associated isoform of cellular prion protein and diagnostic markers of transmissible spongiform encephalopathies (TSEs). These neurodegenerative diseases affect humans and several animal species and include scrapie, zoonotic bovine spongiform encephalopathy (BSE), chronic wasting disease of cervids (CWD), and the newly identified camel prion disease (CPD). Diagnosis of TSEs relies on immunodetection of PrPSc by application of both immunohistochemistry (IHC) and western immunoblot methods (WB) on encephalon tissues, namely, the brainstem (obex level). IHC is a widely used method that uses primary antibodies (monoclonal or polyclonal) against antigens of interest in cells of a tissue section. The antibody-antigen binding can be visualized by a color reaction that remains localized in the area of the tissue or cell where the antibody was targeted. As such, in prion diseases, as in other fields of research, the immunohistochemistry techniques are not solely used for diagnostic purposes but also in pathogenesis studies. Such studies involve detecting the PrPSc patterns and types from those previously described to identify the new prion strains. As BSE can infect humans, it is recommended that biosafety laboratory level-3 (BSL-3) facilities and/or practices are used to handle cattle, small ruminants, and cervid samples included in the TSE surveillance. Additionally, containment and prion-dedicated equipment are recommended, whenever possible, to limit contamination. The PrPSc IHC procedure consists of a formic acid epitope-demasking step also acting as a prion inactivation measure, as formalin-fixed and paraffin-embedded tissues used in this technique remain infectious. When interpreting the results, care must be taken to distinguish non-specific immunolabeling from target labeling. For this purpose, it is important to recognize artifacts of immunolabeling obtained in known TSE-negative control animals to differentiate those from specific PrPSc immunolabeling types, which can vary between TSE strains, host species, and prnp genotype, further described herein.
Introduction
According to the prion hypothesis, the abnormal isoform (PrPSc) is the primary, or sole, component of the infectious agent in transmissible spongiform encephalopathies (TSEs). Confirmation for diagnosis of TSE relies on immunodetection of PrPSc by application of immunohistochemistry (IHC) protocols and/or western immunoblot methods (WB) of encephalon tissues1.
IHC is a method employing monoclonal or, in some cases, polyclonal antibodies (as primary antibodies) as a first step in immunostaining of specifically targeted antigens of interest located in cells of a tissue section. Any effective primary antibody-antigen binding is then visualized using secondary antibodies specific to the primary antibodies. These secondary antibodies are conjugated to enzymes, such as horseradish peroxidase (HRP) or alkaline phosphatase (AP). Visualization is then achieved by adding a substrate to these enzymes, producing insoluble color products localized in areas where the primary antibodies are bound to the targeted antigens. Improved visualization can be achieved by counterstaining, wherein a dye is used to create a contrast between immunolabeled and un-immunolabeled tissue2.
With IHC using formalin-fixed paraffin-embedded tissues (FFPE), formalin fixation can nullify the effectiveness of primary antibodies due to cross-linking by formaldehyde and heating and dehydration during paraffin embedding. These change the conformation of proteins, destroying, denaturing, or masking the epitopes, thus diminishing or abrogating their detection3. As such, this requires antigen retrieval (AR). The AR techniques disrupt formaldehyde-related chemical group cross-linking in the antigenic molecules, thus restoring or unmasking the original antigen-protein conformation. This results in recovering the antibody-antigen (epitope) affinity for immunolabeling. The eventual efficacy of AR depends on the qualities of the targeted antigen and/or the primary antibody2.
Heat-induced antigen (epitope) retrieval (HIER) is one procedure of AR3 and is used routinely for PrPSc IHC detection, as described herein. IHC is essential for diagnosis and used in research laboratories to determine the tissue distribution of a pathology-associated antigen. It is widely used in diagnosing and researching cancer, neuroscience, and infectious diseases4, among others. For TSEs, IHC plays an important role in diagnosis and research for confirming and investigating PrPSc distribution in natural hosts and experimental models. IHC contributes to prion pathogenesis studies and the analysis of PrPSc deposition types and patterns, namely, in neural tissues5, to detect deviations from routinely described infections and identify putative new prion strains.
Because prions of bovine spongiform encephalopathy (BSE) can infect humans, certain laboratory protocols involved in the work with BSE may require the use of BSL-3 facilities and practices6. These include using a sealed secondary container to transport potential BSE-infected tissue samples within the institute and laboratory. It also includes designating containment areas and prion-dedicated equipment for BSE research and analysis whenever possible. This is done to prevent contamination outside the work area and provide a confined space since decontamination procedures become necessary.
Accordingly, the Laboratory of Pathology of INIAV follows recommended biosafety level-3 (BSL-3) facilities and practices6 to manage potential prion-infected samples of tissues from cattle, small ruminants, and cervids associated with the TSE surveillance.
Formalin-fixed and paraffin-embedded tissues included in TSE diagnostic or research procedures, especially in the central nervous system, can be potentially infectious. Hence, these fixed tissues must be treated with formic acid to reduce the infectivity of prions, if present, prior to tissue processing. This is performed by placing fixed, trimmed tissues (approximately 2-4 mm thickness) in a processing cassette. The cassette is then immersed in 98% formic acid (for 1 h). After immersion, the cassettes with tissues are washed in running tap water for 30 min, and returned to the fixative before further processing. If tissue sections are not treated before processing, post-microtomic tissue sections must be immersed in undiluted formic acid for at least 5 min before histological staining7. The IHC protocol for PrPSc includes a routine formic acid epitope-demasking step, also serving to inactivate prions7. After these prion inactivation steps, the resulting fixed tissues can then be processed at BSL-2 using standard BSL-2 practices.
The minimum tissue sampling requirement for TSE diagnosis in any animal included in TSE surveillance is collecting the brainstem (at the obex level). Additionally, for detecting atypical BSE and scrapie, it is advised that part of the cerebellum should also be collected1,8. For CWD diagnosis, both brainstem (obex) and retropharyngeal lymph nodes should be tested as PrPSc could be detected in lymphoid tissues with no detectable PrPSc in obex9, reviewed by Machado et al.10.
The obex portion of the brainstem includes diagnostic TSE target sites, namely, the dorsal vagal nucleus (DVN), solitary tract nucleus (STN) and spinal tract nucleus of the trigeminal nerve (V). These areas consistently present bilateral PrPSc accumulation, even in the early stages of BSE and classical scrapie. In clinical cases of advanced TSE, all the gray-matter areas within the brainstem show widespread PrPSc distribution11.
Before sectioning and processing, the brain samples are evaluated (Figure 1) to ascertain the level of autolysis and the presence of any tissue damage potentially compromising the suitability of the sample for IHC-based confirmatory diagnosis8. To validate the integrity of the preparative protocols and the analytical results, the TSE positive and negative tissue samples are included as controls in conjunction with the preparation of tissues from test cases in each assay.
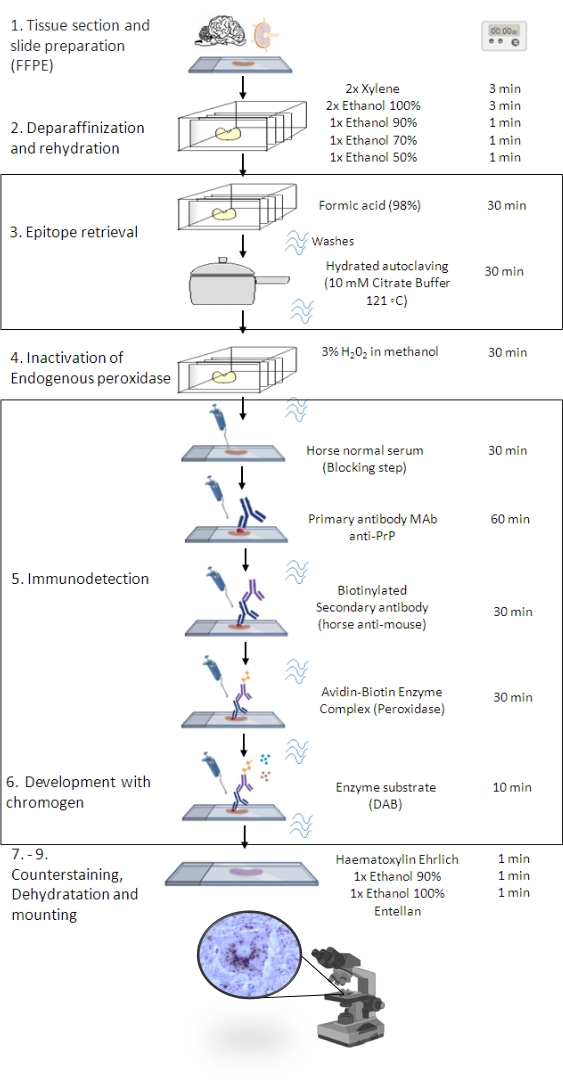
Figure 1: The PrPSc IHC procedure. Representation showing the step-by-step sequence of PrPSc IHC procedure from deparaffinization of tissue sections to eventual immunostaining and detection (FFPE – Formalin-fixed paraffin-embedded; Mab – monoclonal antibody; DAB – 3,3' diaminobenzidine). This figure was created in BioRender.com. Please click here to view a larger version of this figure.
Protocol
The IHC procedure described herein is a component of the research project FAIRJ-CT98-7021, "The establishment of a European network for the surveillance of ruminant TSE and the standardization and harmonization of the process and criteria for the identification of suspect cases". It follows the diagnostic criteria defined by the World Organization for Animal Health, WOAH (previously named the Office International des Épizooties, OIE)1 and European Reference Laboratory for animal TSEs8,11. The IHC procedure described is also accredited according to NPISOIEC17025 (general requirements for the competence of testing and calibration laboratories) since 2008. Such accreditation implies a high-quality management system incorporating standard operative procedures, qualified personnel, accurately calibrated equipment, rigorous control of data and reagents, participation in inter-laboratory proficiency assays, non-conforming work, and risk assessment to increase the effectiveness of the management system, achieving improved results, and preventing laboratory hazards. Prion-infected tissue controls with BSE, classical scrapie, atypical scrapie, and CWD were collected (formalin-fixed, paraffin-embedded, FFPE) from various sources. The bovine and sheep sections are from the cases diagnosed under an animal TSE surveillance program. The CWD sections are from the control samples kindly provided by Professor Stefanie Czub (Canadian Food Inspection Agency, National Center for Animal Diseases) in 2003 and from the CWD Proficiency testing 2008 organized by the European Reference Laboratory for TSEs (APHA, Weybridge).
1. Tissue sectioning and slide preparation
- Cut sections of formalin-fixed, paraffin-embedded (FFPE) tissues at 3-5 µm thickness using a microtome.
- Float sections onto purified water with a temperature approximately 10 ˚C below the melting point of the paraffin used. Lift the sections from the water onto specially treated microscope slides (see Table of Materials). Allow the water to drain thoroughly from the slides.
- Incubate the slides overnight at 50 ˚C to enhance the slide adhesion of tissues.
NOTE: It is preferable to prepare freshly sectioned tissues for IHC protocols, although TSE positive and negative control sections can be prepared in advance and stored. Steps 2 to 4 must be performed in a chemical fume hood.
2. Deparaffinization and rehydration
- Place the slides with the tissue sections into a stainless-steel staining basket (see Table of Materials). Immerse the basket into a xylene bath (stainless-steel staining container) for 3 min, remove and immerse once again.
- Remove xylene and rehydrate sections by immersing them in an absolute ethanol bath for 3 min. Remove and immerse once again.
- Air dry the sections and place in a 90% ethanol bath for 1 min. Transfer to a 70% ethanol bath for another minute, followed by a final transfer to a 50% ethanol bath for 1 min. For each ethanol bath treatment, gently agitate the slide basket twice and drain the basket prior to the next transfer.
3. Epitope retrieval
- Carefully immerse the tissue sections in 98% formic acid at room temperature for 30 min. Rinse the sections in tap water for 5 min, followed by rinsing twice in distilled water.
CAUTION: Formic acid is highly corrosive. Protective goggles and gloves must be worn. - Perform Heat-Induced Antigen Retrieval (HIER) following the steps below.
NOTE: This step is performed by hydrated autoclaving at 121 °C for 30 min in a specific pressure chamber (see Table of Materials).- Pre-heat the citrate buffer (10 mM, pH 6.1, see Table of Materials) at 98 °C for about 20 min in a stainless-steel staining container placed inside the pressure chamber filled with 500 mL of distilled water.
NOTE: For the present study, the program Set Point was 1 (SP1) for the specific equipment used. - After the alarm indicates that the equipment attained the programmed time and temperature, immerse the basket with slides and initiate the program to Set Point 2 (121° C for 30 min). For quality control purposes, place a section of adhesive autoclave indicator tape on the basket to monitor the temperature and pressure, and record the initial and final pressure of this program.
- If the number of slides to be immunostained does not reach the basket's capacity, use clean, blank slides to occupy any empty positions. After the program has terminated, allow the passive return of the chamber to ambient pressure (at least 30 min).
NOTE: Longer durations may reduce non-specific background staining. - Carefully transfer the slide basket to a stainless-steel staining container filled with distilled water for 5 min.
- Pre-heat the citrate buffer (10 mM, pH 6.1, see Table of Materials) at 98 °C for about 20 min in a stainless-steel staining container placed inside the pressure chamber filled with 500 mL of distilled water.
4. Inactivation of endogenous peroxidase
- Immerse the slide basket containing the sample sections in a bath of 3% hydrogen peroxide (H2O2) in methanol for 30 min. Rinse the sections under running water (5 min). Drain and immerse the sections in 1x Tris-buffered saline (TBS) for an additional 5 min.
5. Immunodetection
NOTE: For the present study, immunodetection was executed using the capillary gap format7 using a commercially available slide clip assembly system (see Table of Materials). Other immunohistochemistry slide systems are also applicable.
- Place each slide onto a commercially available cover-plate holder (see Table of Materials) pre-moistened with TBS, avoiding bubbles, with the tissue side facing the holder and slide edges coinciding with the two lower points of the holder.
- Hold the slide-holder assembly between the thumb and the forefinger, with one finger on top of the sample slide and the other on the bottom of the holder. Then, place the assembly in the gallery of the system.
- To ensure the set is well assembled, fill the well between the sample slide and the holder with TBS, which must not immediately overflow. From this point on, ensure that approximately 80 µL of TBS must be retained between the holder and the slide. Do not allow the sections to dry.
- Once in the system, to decrease background staining prior to the treatment with primary antibody (anti-PrP, see Table of Materials), pre-incubate the sample slides with 20% normal serum from the same species as the secondary antibody host being used for immunostaining (in the present case, horse serum) for 30 min in TBS.
- Thaw the number of aliquots of antibody to be used according to the animal species under analysis, the number of sample sections to be examined, and the amount of working dilution.
NOTE: For best results, use 10% normal serum from the same species as the source of the secondary antibody and primary antibody solutions. If possible, for best results, the host source of the normal serum and secondary antibody should be from the same species. - Without washing the sections, apply the primary antibody solution directly (200 µL) in each well of the slide-holder set and incubate for 60 min at room temperature.
- For washing, fill the wells of the slide-holder sets with TBS and wait for 5 min. Repeat twice.
- Dilute the biotinylated secondary antibody (monoclonal anti-murine Horse Ab, see Table of Materials) at 1/200 in TBS with 10% horse serum. Prepare the volume required depending on the number of sections to be treated.
- Apply the secondary antibody solution (200 µL) to each well of the slide-holder set. Incubate for 30 min at room temperature.
- Wash as described in step 5.5.
- For incubation with avidin-biotin complex peroxidase (ABC/HRP complex, see Table of Materials), prepare the reagent 30 min before use. Apply the ABC/HRP complex solution (200 µL) in each well of the slide-plate holder set. Incubate for 30 min at room temperature.
- Wash as described in step 5.5.
6. Development with DAB chromogen
CAUTION: DAB is a potential carcinogen. As a result, appropriate care is necessary while working with this reagent, including eye protection, lab coats, gloves, and good laboratory procedures. Dispose of following local regulations.
- Dilute chromogen according to the manufacturer's instructions (see Table of Materials) immediately before use. Apply the chromogen solution (400 µL) to each well of the slide-plate set.
- Incubate for up to 30 min at room temperature. In PrPSc positive sections, a 10 min incubation period is usually sufficient.
- Remove the residual chromogen solution by washing the slides in distilled water. Remove the slides from the coverplate holders and place them in a plastic container with distilled water.
7. Hematoxylin counterstaining
- Immerse the stainless-steel staining basket with the sections into the hematoxylin solution bath (see Table of Materials) for 1 min. Rinse gently in cold running water for 10 min or tepid water for 5 min.
- Dehydrate the sections by placing them in 90% ethanol for 1 min, followed by one bath in absolute ethanol for 1 min. Then, place the slides in a xylene bath for 1 min.
- Mount the sections with a commercially available mounting medium (see Table of Materials).
NOTE: These steps are carried out in a chemical fume hood at room temperature (19 ˚C-24 ˚C). This procedure includes a test record (Supplementary File 1) for recording the completion of all the steps, the equipment used, and the lab technician. In addition, a worksheet (Supplementary File 2) has been designed to easily determine the volumes of the reagents according to the number of sections in each assay, the working dilutions, and the manufacturer's instructions as well as to record the room temperature, equipment, and reagent batches.
Representative Results
Given that non-specific target immunolabeling is possible, it is important to ascertain the level of non-specific immunolabeling in known TSE-negative control animals. This is an important step to properly interpret specific PrPSc immunolabeling7 (Figure 2). Non-target labeling by anti-PrP antibodies has been noted to occur as discrete intraneuronal fine-particulate staining (more evident in the hypoglossal nucleus and accessory cuneate nucleus), irregular fine-linear threads of stain in the neuropil (STN, V, olivary nucleus, accessory cuneate nucleus, lateral fascicular nucleus, reticular formation dorsal to the olivary nuclei, and area postrema)12, diffuse nonparticulate labeling in some gray and white matter areas, and diffuse cytoplasmic labeling of neurons13 (Figure 3).
Based on several studies, specific, targeted PrPSc immunolabeling can be found in many different types depending on the TSE strain, host species, and prnp genotype. The following PrPSc types have been described (reviewed by Orge et al.5): Intraneuronal; Intraglial; Stellate; Perivascular; Subpial; Subependimal; Linear; Fine granular; Aggregates (or coarse granular, coarse particulate to coalescing and moss-like); Perineuronal; Plaque-Like (or plaques, vascular plaques); Punctate; Globular.
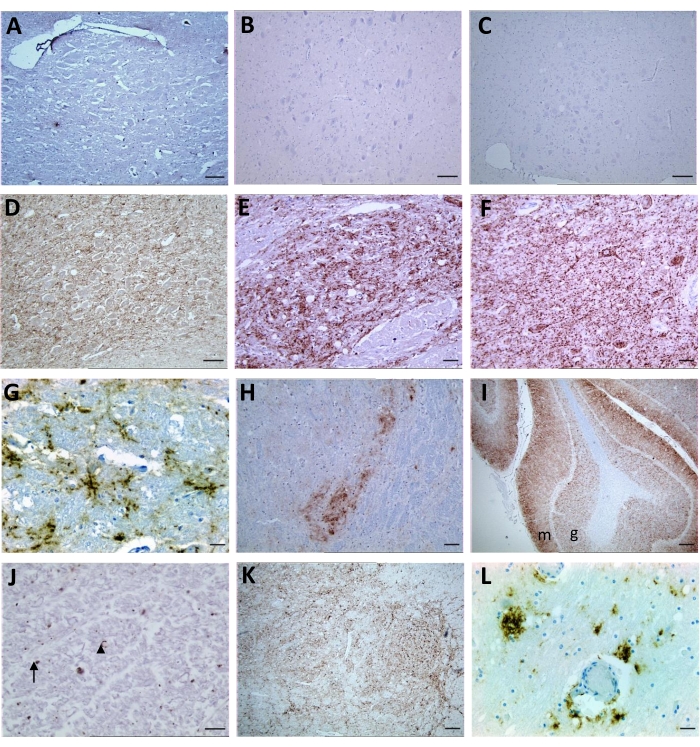
Figure 2: Representative images following immunolabeling for PrPSc of tissue sections from non-infected controls and prion-infected animals with BSE, classical scrapie, atypical scrapie, and CWD. (A,B,C) Sections from negative controls of bovine, sheep, and cervid, respectively; (D) BSE positive: granular PrPSc deposits in the neuropil of DVN and (E) STN; (F) Sheep classical scrapie positive: granular PrPSc in the neuropil and intraneuronal in DVN and (G) stellate PrPsc in the reticular formation; (H) Sheep atypical scrapie: granular PrPSc in the neuropil of V at the obex region, granular at molecular (m) and (I) granular (g) layers of the cerebellum and (J) punctate (arrow) and globular (arrowhead) PrPSc at white matter of the brainstem; (K) CWD: granular PrPSc at V and (L) reticular formation and perivascular at reticular formation. For A, B, C, and D- Scale bar = 50 µm; E, F, G, H, and L- Scale bar = 20 µm; I and K- Scale bar = 1 mm; J- Scale bar = 10 µm. Please click here to view a larger version of this figure.
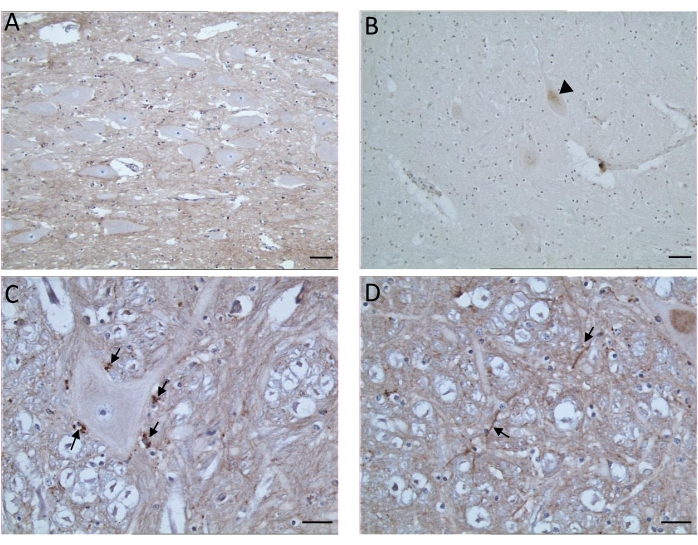
Figure 3: Representative images showing examples of immunostaining artifacts observed following the PrPSc IHC procedure. (A) Diffuse neuropil background (DVN). (B) Diffuse cytoplasmic (head arrow) labeling of neurons (reticular formation). (C) Artifactual chromogen deposits (arrows) (reticular formation). (D) Irregular fine lines (arrows) of staining in the neuropil (reticular formation). Samples of PrPSc negative bovine sections from immunohistochemistry technical proficiency testing organized by the European Reference Laboratory for TSEs. A and B- Scale bar = 20 µm; C and D- Scale bar = 10 µm. Please click here to view a larger version of this figure.
Table 1 summarizes the description of immunolabeled PrPSc types observed in BSE, classical and atypical scrapie, and CWD for a positive diagnosis and established criteria for negative, inconclusive, and unsuitable results11 and reviewed by Orge et al.5.
Table 1: Summary of PrPSc types observed in BSE, classical and atypical scrapie, and CWD for a positive diagnosis, and criteria for negative, inconclusive, and unsuitable results. Please click here to download this Table.
Supplementary File 1: Form serving as the INIAV "TSE Immunohistochemical test record". The task is completed as each step of the IHC protocol is completed and certified by the technician performing the respective task. Please click here to download this File.
Supplementary File 2: Reagent preparation worksheet for TSE immunohistochemical protocol. Please click here to download this File.
Discussion
TSEs are potential zoonotic diseases. After the emergence of BSE in 1986 in the United Kingdom, Portugal became one of the European Union Member States with a higher incidence of this disease14,15. In order to control this disease, other TSEs that have emerged (classical and atypical scrapie, BSE variants, and currently the surveillance of chronic wasting disease in cervids), surveillance mechanisms were developed by the Directorate General of Food and Veterinary (DGAV) and the National Institute for Agricultural and Veterinary Research, IP (INIAV) that conducts the TSE diagnosis as the TSE National Veterinary Laboratory. This laboratory follows the recommended biosafety BSL-3 facilities and practices6 in managing the potential prion-infected samples of tissues from cattle, small ruminants, and cervids associated with TSE surveillance. Immunodetection of PrPSc by immunohistochemistry (IHC) and/or western immunoblot methods (WB) of encephalon and lymphoid tissues are the reference methods for confirming the diagnosis of TSE1.
The IHC technique is a useful complementary tool for diagnosis and anatomopathological investigation in veterinary and human medicine. Several critical points are identified, and, in practice, it is necessary to adjust in every laboratory for each animal species, the antibody used, and also the different tissues tested16,17. The fixation, antigen retrieval, background, and artifacts, in addition to the type of antibody used (monoclonal versus polyclonal, the clone used, animal origin) and animal species tested, are examples of factors that need to be properly controlled and standardized in each laboratory so that the final results obtained with immunohistochemistry would be properly interpreted as specific immunolabeling2,17.
The current work presented the protocol followed in the TSE National Reference Laboratory in Portugal, with the critical points and all reference methodology made to ensure the correct diagnosis of TSE diseases. The IHC results depend on the quality of the sample, which must include the target sites for TSE. The lack of these TSE target sites together with the absence of specific PrPSc immunolabeling, is an important limitation of this methodology to assure a diagnosis. Even in cases where freezing or autolysis promotes artifacts, it is possible to make a conclusive diagnosis of whether or not a TSE is present if those target sites are present in the sample. Furthermore, the identification of immunostaining in non-specific targets in different neuronal tissues, based on daily observation of negative and positive controls that have always been introduced whenever this technique is performed, indicates the quality of the protocol produced and presented here and the reason for its certification5,12,13.
As possible incidents, the loss of tissue sections during the hydrated autoclaving and/or removal of the cover-plate holders, the presence of bubbles trapped between the slides and the cover- plates, and the inadequate dilution of the reagents are highlighted. To overcome these events, it is recommended to use positively charged microscope slides within the expiration date and that are well stored, clip the tissue sections carefully with the cover plates, use calibrated equipment, standardize the reagent preparation worksheet (Supplementary File 2), and test immunolabeling batch reagents before use.
The description of new TSE variants, namely, atypical scrapie, also validates the importance of a standard IHC technique in this diagnosis. With this technique, it is possible to observe the existence of different patterns for PrPSc immunostaining, depending on the prion strain, host species, and prnp genotype.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This article was funded by the Project POCI-01-0145-FEDER-029947 "Chronic wasting disease risk assessment in Portugal" supported by FCT (Fundação para a Ciência e a Tecnologia) – FEDER -Balcão2020. Also, the authors of the research unit CECAV received funding from the FCT, under the project UIDB/CVT/0772/2020.We thank Bruce C. Campbell, Research Director (retired), Western Regional Research Center, USDA, for his assistance.
Materials
| Absolute ethanol | Labchem | LB0507-9010 | Undituled |
| Diluted 90%, 70% and 50% in distilled water | |||
| Avidin-biotin complex and peroxidase Vectastain Elite ABC kit Peroxidase |
Vector Laboratories | PK-6100 | Prepare and gently mix 30 min before use according to kit instructions. Do not mix after standing. |
| Biotinylated secondary antibody (Horse anti-mouse IgG H+L) | Vector Laboratories | BA-2000-1.5 | Dilute at 1/200 in TBS with 10% horse normal serum. Prepare the volume required depending on the number of sections. |
| Chromogen Diaminobenzidine- DAB, substrate kit, Peroxidase | Vector Laboratories | SK-4100 | Prepare before use according to kit instructions. Use 400 µL of solution per section. |
| DakoCytomation Pascal pressure chamber | DAKO | S2800 | |
| Ehrlich’s Hematoxylin: | |||
| Hematoxylin | Merck | 115938 | Dissolve 2 g of hematoxylin in 100 mL of absolute ethanol. Add 100 mL of distilled water, 10 mL of glacial acetic acid and 15 g of potassium alum with constant stirring. Add 100 mL of glycerin. The natural oxidation process takes 2 months, before use. |
| Absolute ethanol | Labchem | LB0507-9010 | |
| Glacial acetic acid | Merck | 101830 | |
| Potassium alum | Merck | 1.01047.1000 | |
| Glycerin | Merck | 1.04091.1000 | |
| Endogenous Peroxidase Block solution (3% concentration H2O2): | 40 mL Hydrogen peroxide (30% w/w) in 360 mL Methanol. Prepare before use |
||
| Hydrogen peroxide (30% w/w) | Scharlau | HI0136 | |
| Methanol | Sigma Aldrich | 322415-2L | |
| Formic acid 98% | Merck | 1.00264.1000 | Undiluted |
| Microtome | Shandon-AS325 | Microtome | Shandon-AS325 |
| Mounting medium Entellan | Merck | 107960 | Ready- to- use. |
| Normal serum (20% ) block solution in TBS: Horse normal serum |
Gibco |
16050-122 |
Prepare final volume according to the number of sections in the assay (200 µL of solution per section). |
| Primary antibody anti-PrP Mouse MAb 2G11 | BIORAD | MCA2460 | PrP 146-R154R171182 Ovine including atypical scrapie, cervine, feline. Not suitable for bovine. According to the number of sections in the assay (200 µL of solution per section) and antibody dilution, prepare final volume in TBS supplemented with 10% of normal serum from the species the secondary antibody was raised in (horse normal serum) Usual antibody dilution: MAb 2G11 1/100 but working dilution should be established in every new batch to get the concentration to give the strongest labelling with lowest background. For storage, freeze aliquot volumes of a minimum of 10 μL into sterile microtubes. Defrost and use one aliquot at a time. |
| Primary antibody anti-PrP Mouse MAb 12F10 | Cayman Chemical Company | 189710 | PrP142-160 Bovine, not suitable for ovine Usual antibody dilution: 1/200 but working dilution should also be established. Prepare as MAb 2G11 |
| Shandon CoverplateTM chamber | Thermo Scientific | 72110017 | |
| Shandon Sequenza® Immunstaining center | Thermo Scientific | 73300001 | |
| Shandon Sequenza® Immunstaining slide rack | Thermo Scientific | 73310017 | |
| Solution Citrate Buffer (10 mM pH 6.1): | 2.55 g Tri-sodium citrate dihydrate and 0.255 g Citric acid in one litre purified water. Adjust pH of working solution to 6.1 using 10 mM citric acid solution (1.05 g citric acid in 500 mL purified water) Prepare on assay day. |
||
| Tri-sodium citrate dihydrate | Sigma-aldrich | S4641-500G | |
| Citric acid | Sigma Aldrich | C0759 | |
| Staining jar and basket | Deltalab | 19360 | |
| 19361 | |||
| Superfrost Plus microscope slides | VWR | 631-0108 | |
| Tris-Buffered Saline solution (TBS) (50 mM TRIZMA BASE; 0.8% NaCI; pH 7.6): | 10xTBS (stock solution 0.5 M TRIZMA BASE; 8% NaCI; pH 7.6): TRIZMA BASE 60,57 g and NaCl 80 g in 800 mL purified water. Adjust pH of stock solution using Hydrochloric acid 37% and final volume to one litre with purified water (keep 5± 3 °C until 2 months) Dilute TBS stock solution 1/10 on assay day. |
||
| TRIZMA BASE | Sigma Aldrich | T6066-1KG | |
| Sodium Chloride (NaCl) | Merck | 106404 | |
| Xylene | Panreac Applied Chem ITW reagents | 251769 | Undiluted |
References
- . WOAH, Manual of Diagnostic Tests and Vaccines for Terrestrial Animals Online Access. Chapter 3.4.5.-Bovine Spongiform Encephalopathy (Version May 2021) and Chapter 3.8.11. – Scrapie (Version May 2022) Available from: https://www.woah.org/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (2022)
- Ramos-Vara, J. A. Principles and methods of immunohistochemistry. Methods in Molecular Biology. 1641, 115-128 (2017).
- Krenacs, L., Krenacs, T., Stelkovics, E., Raffeld, M., Oliver, C., Jamur, M. Heat-Induced antigen retrieval for immunohistochemical reactions in routinely processed paraffin sections. Immunocytochemical Methods and Protocols. Methods in Molecular Biology. 588, 103-119 (2010).
- Duraiyan, J., Govindarajan, R., Kaliyappan, K., Palanisamy, M. Applications of immunohistochemistry). Journal Pharmacy Bioallied Sciences. 4, 307-309 (2012).
- Orge, L., et al. Neuropathology of Animal Prion Diseases. Biomolecules. 11 (3), 466 (2021).
- . Biosafety in Microbiological and Biomedical Laboratories. 6th Edition Revised June 2020 Available from: https://www.cdc.gov/labs/pdf/SF_19_308133-A_BMBL6_00-BOOK-WEB-final-3.pdf (2022)
- APHA. Fixation, tissue processing, histology and immunohistochemistry procedures for diagnosis of animal TSE (BSE, scrapie, atypical scrapie, CWD). Histo & IHC protocols for TSE diagnosis_Rev_Jan2019.pdf. TSEglobalNet – International Reference Laboratory for TSE. , (2022).
- Sample requirements for TSE testing and confirmation. Version 1.0. TSE EURL Available from: https://www.izsplv.it/it/istituto/212-centri-eccellenza/laboratori-internazionali-riferimento/422-eurl_tses.html (2019)
- TSE EU Reference Laboratory Guidelines for the detection of Chronic Wasting Disease in cervids. Version 1.0. TSE EURL Available from: https://www.izsplv.it/it/istituto/212-centri-eccellenza/laboratori-internazionali-riferimento/422-eurl_tses.html (2019)
- Machado, C. N., et al. TSE Monitoring in Wildlife Epidemiology, Transmission, Diagnosis, Genetics and Control. Wildlife Population Monitoring. IntechOpen. , (2019).
- APHA. Neuropathology: Confirmatory diagnosis of transmissible spongiform encephalopathies (TSEs) in cattle and small ruminants. Confirmatory (Histo & IHC) diagnostic criteria Rev_Jan2019.pdf. TSEglobalNet – International Reference Laboratory for TSE. , (2019).
- Ryder, S. J., Spencer, Y. I., Bellerby, P. J., March, S. A. Immunohistochemical detection of PrP in the medulla oblongata of sheep: The spectrum of staining in normal and scrapie-affected sheep. The Veterinary Record. 148 (1), 7-13 (2001).
- Simmons, M. M., et al. Experimental classical bovine spongiform encephalopathy: definition and progression of neural PrP immunolabeling in relation to diagnosis and disease controls. Veterinary Pathology. 48 (5), 948-963 (2011).
- Orge, L., et al. Identification of H-type BSE in Portugal. Prion. 9 (1), 22-28 (2015).
- Orge, L., Simas, J. P., Fernandes, A. C., Ramos, M., Galo, A. Similarity of the lesion profile of BSE in Portuguese cattle to that described in British cattle. Veterinary Record. 147 (17), 486-488 (2000).
- Pires, M. A., Travassos, F. S., Gärtner, F. Atlas of veterinary pathology. Biopathology. Lidel VII. 195 (6), 179-180 (2004).
- Pires, M. A., et al. Immunology protocols, didactic series. Applied Sciences. , 357 (2010).

