Contrast Ultrasound Targeted Treatment of Gliomas in Mice via Drug-Bearing Nanoparticle Delivery and Microvascular Ablation
Summary
Insonation of microbubbles is a promising strategy for tumor ablation at reduced time-averaged acoustic powers, as well as for the targeted delivery of therapeutics. The purpose of the present study is to develop low duty cycle ultrasound pulsing strategies and nanocarriers to maximize non-thermal microvascular ablation and payload delivery to subcutaneous C6 gliomas.
Abstract
Protocol
1. Microbubble Production
- To prepare albumin microbubbles ( MBs), place a 1% solution of serum albumin in normal saline in a flask with a blanket of gas (octafluoropropane) above the aqueous phase. Briefly sonicate the solution (30 sec) with an ultrasound disintegrator equipped with an extended ½” titanium probe. This formulation is similar to Optison (GE Heathcare), which is provided in a concentration range of 0.5- 1.2 x 109 MBs/ml. Determine mean MB diameter with a Multisizer Coulter Counter. The albumin mean MB diameter used in this study was 1.93um ±1.63um.
- To fabricate lipid MBs, prepare an aqueous dispersion of 1 mg/ml polyethyleneglycol-40 stearate (Sigma Chemical Co., St. Louis, MO) and 2 mg/ml distearoyl phosphatidylcholine (Avanti Polar Lipids, Alabaster, AL) and sonciate as previously described (1.1) with decafluorobutane gas. Determine mean MB diameter with a Multisizer Coulter Counter. The mean lipid MB diameter used in this study was 2.01um ±1.29um.
2. Nanoparticle Fabrication
- These methods were adapted from the water-in-oil-in-water emulsion solvent evaporation technique described by Davda (2002) and Chappell (2008).
- Prepare a 2% Poly(vinyl alcohol) PVA solution by dissolving 20g of PVA in 1000 ml of deionized (DI) water. Allow solution to fully dissolve on a stir plate overnight. Centrifuge solution at 1000 rpm for 10 minutes and filter with a 0.22μm sterile filter to remove any residual undissolved PVA.
- To fabricate bovine serum albumin (BSA) loaded poly(lactic-co-glycolic acid) (PLAGA) nanoparticles (NPs), dissolve 180mg of uncapped 85:15 PLAGA in 6ml methylene chloride (MC) in a glass scintillation vial. Vortex the MC/PLAGA solution for 2 minutes.
- Dissolve desired payload (15mg of BSA) in 1.5 ml of PBS. Add the PBS/BSA solution in two parts to the PLAGA/MC solution with intermittent vortexing. Place solution on ice for 5minutes and sonicate at 45W for 120 seconds.
- Add the MC/PLAGA/payload/PBS solution to 24ml of 2% PVA, in two portions with intermediate vortexing.
- Placing the solution on ice for 5minutes and sonicate at 45W for 120.
- Stir the NP emulsion for 12 hours on a stir plate in a fume hood to allow evaporation of MC and NP stabilization.
- Centrifuge the suspension for 25minutes at 20,000 rpm at 4°C twice to isolate NPs and remove residual PVA. Resuspend the pellet in 8ml of DI water and centrifuge for 10 minutes at 1000 rmp at 4°C to isolate the 100nm population of NPs.
- Isolate the supernatant and flash freeze it at -80° C. Lyophilize the frozen sample for 48 hrs. Store the lyophilized particles in a dessicator at -80° C until time of use.
3. Composite Delivery Vehicle Fabrication (Protocol Adapted from VisEn Chemistry Notes)
- Conversion of VivoTag680 to carboxy surface functionality
- Combine one vial of VivoTag680 with 50 μL of 1.0 M HEPES, pH 7 and Succinic anhydride (2.5 mg, 25 ìmol) in 25 μL DMSO. Add 50 μL of 1.0 M NaOH to this solution, mixed thoroughly; allow solution to react for 2 hours at room temperature protected from light. Purify particles using Bio-Rad BioGel (P100, medium) eluting with 0.1 M MES buffer, pH 6.0. Collect the green band.
- Activation of carboxy-modified VivoTag680
- Combine carboxy-modified VivoTag680 obtained in 0.1 M MES buffer, pH 6, with 1 mg of 1-Ethyl-3-(3-dimethylaminopropyl)-carbodiimide (EDC) and 2.2 mg of N-hydroxysulfosuccinimide (Sulfo-NHS). Allow to react for 2 h at room temperature.
- Purify activated particles from excess activating agent (EDC) by gel filtration using Bio-Rad BioGel (P100, medium) eluting with 0.1 M MES buffer, pH 6.0 and collect the green band. Immediately conjugate the activated particles to amine-containing molecules (e.g. BSA-loaded Nanoparticle). Allow the solution to react for 2 hours at room temperature. Amine conjugated NPs were purified from excess EDC by centrifuging at 20,000 rpm for 60min at 4 °C, pouring off the supernatant after centrifugation and resuspending in 0.1 MES buffer.
- Stop here to fabricate VivoTag680 tagged NPs for drug delivery studies.
- Conjugation of carboxy-modified PLGA-BSA-VivoTag680 Nanoparticles (NP680) to Albumin Microbubbles
- Combine NP680 obtained in 0.1 M MES buffer, pH 6 with 1 mg of 1-Ethyl-3-(3 dimethylaminopropyl)-carbodiimide (EDC) and 2.2 mg of N-hydroxy-sulfosuccinimide (Sulfo-NHS). Allow the solution to react for 1 hour at room temperature.
- Purify activated nanoparticles from excess EDC by centrifuging at 20,000 rpm for 60min at 4 °C, pouring off the supernatant after centrifugation. Resuspend nanoparticles in 0.1 MES buffer.
- Wash albumin MBs three times in degassed PBS to remove excess BSA from solution.
- Immediately conjugate the activated particles to amine-containing molecules (e.g. Albumin Microbubbles). Allow NP680 solution to react for 2 hour at room temperature. Purify NP680 conjugated microbubbles (MNCA) from unbound NP680s by washing three times with degassed PBS.
- Determine the concentration of MNCAs using a Coulter Counter.
4 .Tumor Model
All animal experiments were in compliance with an animal protocol approved by the University of Virginia Animal Care and Use Committee.
- C6 Giloma rat tumor cell line was provided by Dr. Jason Sheehan of UVA (Charlottesville, VA).
- Cells line was tested to ensure cells were mycoplasma free.
- Maintain the cell line in F-12K Nutrient Mixture supplemented with 16% horse serum, 3% fetal bovine serum (FBS) and 1% Pen-Strep, (Gibco, USA) at 37°C and 5% CO2.
- Inoculate C57BLJ6/Rag1 mice (Jackson) with 3 x 106 C6 Giloma tumor cells suspended in 300 μl of PBS subcutaneously in the left hindlimb. Allow tumor to grow for 12 days to reach a maximum diameter of 8-10mm.
5. In Vivo Ultrasound Application
- Anesthetize mice with an intraperitoneal (IP) combination injection of ketamine hydrochloride (60 mg/kg body weight) and xylazine (0.1 mg/kg body weight) prior to treatment.
- Prepare a maintenance anesthetic of ketamine hydrochloride (20 mg/kg body weight) and xylazine (0.3 mg/kg body weight). Administer as needed.
- Cannulate the tail vein of each animal for the intravenous (I.V.) administration of a MB, MB/NP or MNCA solution.
- Tumor perfusion measurements
- I.V. infuse a lipid MB solution (1×108 MBs/g body weight in 0.3 ml of 0.9% saline) at a rate of 15μl/min with a continuous infusion pump (Harvard Apparatus PHD 2000; Harvard Apparatus, Holliston, MA).
- An Acuson Sequoia 512 ultrasonography system (Siemens Medical Solutions, Mountain View, CA) equipped with an 8 13 MHz linear 15L8 probe was used in this study to quantify contrast enhanced tumor perfusion. Couple the 15L8 probe to the tumor with a water-based ultrasound gel (Parker Laboratories Aquasonic 100; Parker Laboratories, Inc., Fairfield, NJ). Scan the tumor in B-Mode to obtain the best imaging plane.
- In contrast pulse sequence (CPS) mode, acquire a continuous video capturing microbubbles signal intensity 5 seconds prior, and 20 following, the high-amplitude “burst” pulse at a frame rate of 13Hz. Repeat measurements in four imaging planes.
- Wait ten minutes to infuse albumin MBs and initiate therapeutic low frequency treatment to allow lipid MBs to clear from the circulation.
- Therapeutic Ultrasound Treatment
- Ten minutes following tumor perfusion measurements (5.3) couple a 0.75” diameter 1 MHz unfocused transducer (A314S; Panametrics, Waltham, MA) to the skin above the flank tumor. I.V. infuse an albumin MB (1 x105 MBs/g body weight in 0.3 ml of 0.9% saline), MB/NP (1 x105 MBs/g and 0.2 ug NPs/g body weight in 0.3 ml of 0.9% saline) or MNCA solution (1 x105 MNCAs/g body weight in 0.3 ml of 0.9% saline).
- Drug Delivery Treatment
- Insonate for sixty minutes with the “1-Burst” pulse sequence (5.4.2.2) during a continuous infusion of NPs, a co-injection of MBs and NPs, or MNCAs (5.4.1).
- The “1-Burst” pulsing sequence consists of 100 consecutive 1 MHz sinusoids (duty cycle= 0.00002) each of 1V peak to- peak amplitude from a waveform generator (AFG-310; Tektronix, Inc., Beaverton, OR). Amplify the waveform signal by a 55 dB RF with a power amplifier (ENI 3100LA; Electronic Navigation Industries, Richardson, TX).
- Ablative Treatment
- Identical to 5.4.2.1, however insonate with the “5-Burst-Medium” or “5-Burst – Extended” pulsing sequence
- The “5-Burst-Medium” pulsing sequence consists of 5000 1 MHz sinusoids (duty cycle= 0.005) each of 1V peak to- peak amplitude . The “5-Burst-Exteneded” pulsing sequence consists of 10000 1 MHz sinusoids (duty cycle= 0.01) each of 1V peak to- peak amplitude.
- During the time course of the experiment monitor tumor temperature by inserting a needle thermocouple probe (Omega T-type, HYP1-30-1/2-T-G-60-SMPW-M) 2cm into the tumor. Record temperature measurements every five minutes.
- Repeat tumor perfusion measurements as described in 5.3 ten minutes following therapeutic treatment.
6. Tumor Perfusion Quantification
- Using the CPS program on the Sequoia, enter ACQ mode. Select the region of interest encompassing the complete tumor volume. Perfusion recovery curves will be generated of the form y = A (1 – e -βt) + C (Sadlowski 2002; Chromas 2001; Yeh 2004) that are fit to the CPS data. Evaluate β, a relative measure of red blood cell velocity, pre-treatment and post-treatment.
- Export CPS files for off-line analysis. Convert data into avi files. Break avi files into frame-by-frame image sequences. Using Compaq Visual Fortan, or a similar software package, generate a time averaged image of all frames from the destructive pulse to 8 seconds following the destructive pulse.
- To determine perfused tumor area, using ImageJ software, apply a threshold of 247 to the 8-bite time averaged CPS image and quantify the percent enhanced pixels within the tumor volume. Overlay the corresponding B-mode image with the time averaged thresholded image to accurately define tumor area.
- Average the percent perfused area for a minimum of four CPS clips to obtain a final perfused area at a given time point for a particular tumor.
- Take the product of percent perfused area (6.4) and β (.1) to determine relative tumor blood flow.
7. Nanoparticle Biodistribution in Tumor
- Twelve days prior to treatment with fluorescently labeled NPs place mice on a low alfasprot diet (Harlan, Indianapolis IN) to reduce autofluorescence caused by normal mouse chow when imaging.
- Shave the imaging site (flank to liver) to remove all hair.
- Remove residual hair with a chemical hair removal agent, such as Nair.
- Rinse of all residual chemical hair remover or a chemical burn may occur.
- Image mice on the FMT system (VisEn Medical) before treatment and 0, 1, 4, and 24 hours after treatment as described in 5.4.2.
- Prior to imaging anesthetize mice with an IP combination injection of ketamine hydrochloride (60 mg/kg body weight) and xylazine (0.1 mg/kg body weight). Place mouse on the imaging cartridge. Acquire reflectance images in white light and fluorescent modes. Carry out fluorescent tomographic imaging in the VT680 channel.
- Employ the FMT software to generate 3D reconstructions of the imaging data utilizing a normalized Born equation. Following the reconstruction, select volumes of interest (VOI’s) by drawing a region of interest (ROI’s) in all 3 imaging planes (X, Y, Z). A mean fluorescent value and total VOI volume and fluorochrome concentration, due to fluorescently tagged BSA NPs, will be generated for VOI’s encompassing the feet and liver.
8. Tumor Growth Rate
- Evaluate tumor volume by taking daily measurements using digital calibers. Calculate tumor volumes using an ellipsoid approximation; V= 1/6 π abc. Where a, b, and c are the maximum diameters of the tumor measured in three orthogonal planes.
9. Tumor Processing and Analysis
- Seven days following treatment euthanize animals. Cannulate the left ventricle and exsanguinate blood with 10ml of 2% Heparinized Tris CaCl2 Buffer (.68mM) perfusion, followed by 10ml Tris CaCl2 Buffer (.68mM), each for 10 minutes at 100 mmHg.
- Perfusion-fix tissue with an infusion of 4% paraformaldehyde in PBS (4°C) for 10minutes at 100 mmHg. Allow sample to fix for 60 minutes.
- Excise specimens, embed in paraffin, and cut into 5-micron sections.
- Perform standard hematoxylin-eosin staining to evaluate histological changes that may have occurred as a result of the ultrasound exposures.
- To detect apoptotic cells, use a terminal deoxynucleotidyl transferase mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay (ApoptTag kit, Intergen Co., Norcross, GA, USA).
10. Representative Results
1. Nanoparticle Fabrication (2.0)
- If this protocol is preformed properly NPs will be spherical in shape, as determined by scanning electron microcopy (SEM), and have a Gaussian distribution around 100nm, as determined by light scattering techniques. Representative results are shown in Figure 1.
2. Targeted Drug Delivery (5.4.1)
- Based on published studies performed in our laboratory (Song 2002, Chappell 2008) and unpublished results present in Figure 2, ultrasonic microbubble destruction results in enhanced delivery of intravascular nanoparticles to tumor tissue. We have also shown that our MNCA delivery technique results in heightened nanoparticle delivery immediately following treatment.
3. Tumor Ablation (5.4.3)
- We have shown that prolonged ultrasonic microbubble destruction, at relatively low duty cycles (0.005-0.01), results in mechanical ablation of the tumor microvasculature and regression of tumor growth. If this protocol is performed properly one should expect changes in (i) rate of tumor growth (Figure 3); (ii) tumor hemodynamics (Figure 4) ; (iii) necrotic area ; and (iv) apoptosis .

Figure 1. Characterization of PLAGA nanoparticles bearing BSA. (A) SEM image of properly fabricated nanoparticles. (B) SEM image of improperly fabricated nanoparticles.
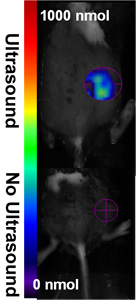
Figure 2. (A) Fluorescence-mediated tomography (FMT) images of ultrasound treated (top) and control treated (bottom) subcutaneous gliomas immediately following treatment with a MNCAs, where nanoparticles (NPs) are bearing 680 fluorescence signals are superimposed on grayscale planar excitation light image.
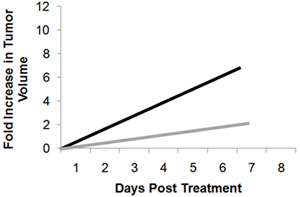
Figure 3. Representative fold change in tumor growth following microbubble insonation with the five-burst extended pulsing protocol.
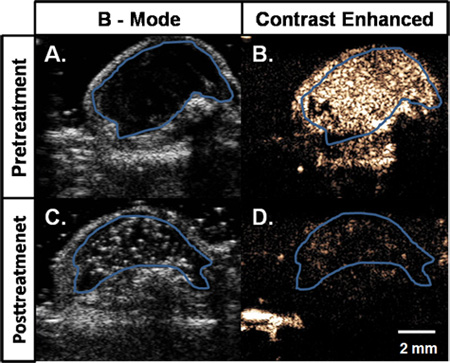
Figure 4. B-Mode (A,C) and contrast-enhanced ultrasonography (B,D) images of a subcutaneous C6 glioma tumor in a mouse. In the initial pretreatment image (A) the boundary of mostly hypoechoic tumor has been traced in blue. Time average enhancement after intravenous injection of a contrast agent is shown in (B) pretreatment. Posttreatment, before contrast injection, tumor is again mostly hypoechoic (D). After contrast injection, there is significantly less enhancement in the tumor.
Discussion
Critical Steps
Cannulation of mouse tail vein:
Intravenous injection into the mouse tail vein can be a challenging procedure. However, a tail vein catheter can greatly improve the likelihood of a successful injection. To make the catheter, repeatedly bend back and forth a 25 gauge needle until it breaks from the hub. Insert the blunt end into the end of PE 20 tubing and seal the connection with silicon glue. To prepare the catheter for cannulation, attach a syringe loaded with 1% heparinized saline to the cather and infuse liquid into the dead space of the catheter. Position an anesthetized mouse on its side so the lateral tail vein is in view. Dilate the tail with warm water (105° – 110°F). Insert the needle into the vein. If successful blood will usually enter the catheter. Verify that the needle is in the vein by clearing the vein with a small amount of saline. Secure the catheter in place with tape before attaching the syringe to the infusion pump.
Nanoparticle Resuspension:
Nanoparticles may aggregate following lyophilization. Resuspend particles in PBS, repeatedly vortex and sonicate briefly (10 sec) in a sonic water bath. It is critical to properly resuspend the lyophilized sample. Characterize the suspension with SEM microscopy and light scattering techniques following lyophilization and resuspension.
Possible Modifications:
In terms of adjusting the nanoparticle fabrication protocol, BSA serves as a surrogate drug and can be interchanged for a multitude of therapeutic agents. Depending on the solubility of the therapeutic agent, nanoparticles can be fabricated as an oil-in-water emulsion or as an oil-in-water-in-oil emulsion. The loading efficiency and release of the therapeutic agent from PLAGA NPs, may also be adjusted considerably if desired. To increase the loading efficiency, increase the wt/wt loading of drug in polymer carriers via the optimization of NP fabrication parameters. To tailor the release rate of the therapeutic agent, vary the hydrolysis rate constant via changes in molecular weight and the lactic/glycolic ratio of PLAGA. By talioring both the loading efficiency and release rate of therapeutic agent, in and from PLAGA NPs, the desired local concentration can be delivered to tissue. In terms of adjusting the delivery protocol, the most important factors are the degree of ultrasonic MNCA destruction and microvascular permeabilization. It has been reported that albumin shelled microbubbles have a half-life of 1.3 ± 0.69 minutes (mean ± SD) (Optison 2008). We hypothesize that continuous agent infusion and prolonged ultrasound application will increase the extent of microvessel permeabilization. By increasing treatment time we aim to transiently heighten permeabilization of the microvasculature.
The non-thermal mechanical ablation protocol may be adjusted by changing MB concentration or the ultrasound peak pressure. It is expected that increasing MB concentration and/or ultrasound peak pressure will increase the degree of damage to the tumor vasculature.
Applications:
We are developing techniques to lower the acoustic power required by transcranial high-intensity focused ultrasound (HIFU) to reach the desired therapeutic effect.
In part, due to complications associated with treatment through the skull and importance of surrounding tissue, thermal tumor tissue ablation with high intensity focused ultrasound (HIFU) has not yet achieved widespread use as a therapeutic option for brain cancer. Preliminary conclusions from the first three patients of a clinical train have indicated deep brain HIFU treatment at sub-ablative focal temperatures results in cranial heating (McDannold 2009). The key to success of HIFU as a clinical treatment for transcranial treatment of brain tumors is the capacity to localize the delivery of acoustic energy to a well-delineated region. The ability to deliver acoustic energy is complicated by bone or air interfaces, such as the skull and previous surgical resections, which generate phase and power aberrations as the ultrasound energy is attenuated along the propagation path (Tanter 1998). These aberrations often contribute to pre-focal heating (McDonnald 2009) and cavitations in healthy tissues.
Over the past several years, the use of microbubbles to lower the acoustic power for targeted, reversible BBB disruption and tumor ablation has attracted much research interest (Hynynen 2001, Sheikov 2004, McDannold 2006a; 2006b, Meairs 2005). McDannold et al. (2006a) have demonstrated that the intravascular injection of microbubbles at the time of HIFU treatment resulted in a 91% reduction in the time-averaged acoustic power threshold for mechanical damage compared to controls in which no microbubbles were present, showing the potential for generating lesions with reduced temperature elevation. Reducing the thermal threshold for lesion formation in turn lowers the probability of heat accumulation in off target tissue or bone. Furthermore, it has been shown that the intravenous administration of microbubbles at the time of ultrasound treatment results in transient, localized, BBB opening with powers approximately two orders of magnitudes lower than those required for lesion formation.
It is the goal of this work to develop ultrasound-microbubbles techniques to both lower the acoustic power required by transcranial HIFU and control the degree of microvascular permeabilization. The two specific therapeutic applications of this work in the brain are targeted drug delivery and non-thermal ablation. The relative levels of increased permeability and permanent ablation can be controlled by adjusting acoustic power levels depending on whether the emphasis is on improved drug delivery, permanent microvascular ablation, or a combination of drug delivery and permanent microvascular ablation. This potential ability to control how the microvessels respond creates an opportunity to develop different permutations of our treatment strategy for specific transcranial therapeutic applications. We believe this approach has the potential to drastically transform how brain tumors are treated.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Supported by NIH R01 HL74082, the Hartwell Foundation, and the Focused Ultrasound Surgery Foundation.
Materials
| Material Name | Type | Company | Catalogue Number | Comment |
|---|---|---|---|---|
| ApoptTag kit | Intergen Co. | S7110 | ||
| un-capped 85:15 poly(lactic-co-glycolic acid) (PLAGA) | Lakeshore Biomaterials | Custom | ||
| Vivo Tag 680 | VisEn Medical | 10120 | Used to Tag BSA | |
| Poly(vinyl alcohol) | Sigma-Aldrich | 363136 | ||
| MicroTip Sonicator | Misonix | S-4000 | ||
| Sequoia | Simons Medical | P.O.A | Equipped with CPS | |
| FreeZone 2.5 | Labconco | 7670020 | Equipped with Nitrogen Trap | |
| Methylene chloride (CH2Cl2) | Fisher Scientific | D37-500 | ||
| FMT 250 | VisEn Medical | P.O.A | ||
| F-12K Nutrient Mixture | Gibco | 21127-022 | ||
| polyethyleneglycol-40 stearate | Sigma Chemical | 9004-99-3 | ||
| distearoyl phosphatidylcholine | Avanti Polar Lipids | 770365 | ||
| Multisizer Coulter Counter | Beckman Coulter | P.O.A | ||
| Waveform Generator | Tektronix, Inc. | AFG-310 | ||
| water-based ultrasound gel | Parker Laboratories | Aquasonic 100 | ||
| Infusion pump | Harvard Apparatus | Harvard Apparatus PHD 2000 | ||
| 1-Ethyl-3-(3-dimethylaminopropyl)-carbodiimide (EDC) | Pierce Biotechnology | 25952-53-8 | ||
| N-hydroxysulfosuccinimide (Sulfo-NHS) | Pierce Biotechnology | 106627-54-7 | ||
| Succinic anhydride | Sigma Aldrich | 603902 | ||
| Power Amplifier | Electronic Navigation Industries | ENI 3100LA | ||
| Needle Thermocouple Probe | Omega | HYP1-30-1/2-T-G-60-SMPW-M | ||
| BioGel (P100, medium) | Bio-Rad | 150-4170 | ||
| .75’’ diameter 1 MHz unfocused transducer | Panametrics | A314S |
References
- Chappell, J., Song, J., Burke, C., Klibanov, A., Price, R. Targeted delivery of nanoparticles bearing fibroblast growth factor-2 by ultrasonic microbubble destruction for therapeutic arteriogenesis. Small. 10, 1769-1777 (2008).
- Chomas, J. E., Pollard, R., Wisner, E., Ferrara, K. Subharmonic Phase-Inversion for Tumor Perfusion Estimation. IEEE. 2, 1713-1716 (2001).
- Davda, J., Labhasetwar, V. Characterization of nanoparticle uptake by endothelial cells. Int J Pharm. 233, 51-51 (2002).
- Hynynen, K., McDannolod, N. Noninvasive MR imaging-guided focal opening of the blood-brain barrier in rabbits. Radiology. 220, 640-646 (2001).
- McDannold, N., Vykhodtseva, N., Hynynen, K. Microbubble contrast agent with focused ultrasound to create brain lesions at low power levesl: MR imaging and histological study in rabbits. Radiology. 241, 95-106 .
- McDannold, N., Vykhodtseva, N., Hynynen, K. Targeted disruption of the blood-brain barrier with focused ultrasound: Association with cavitation activity. Phys Med Biol. 51, 793-807 .
- McDannold, N., Clement, G. T., Black, P., Jolesz, F., Hynynen, K. Transcranial magnetic resonance imaging- guided focused ultrasound surgery of brain tumors: initial findings in 3 patients. Neurosurgery. 66, 323-332 (2009).
- Meairs, S., Alonso, A. Ultrasound microbubblesand the blood brain barrier. Progr Biophys Mol Biol. 93, 354-362 (2007).
- . . Optison, products insert. , (2009).
- Sadlowskie, A., Chromas, J., Pollard, R., Bloch, S., Griffey, S., Wisner, W., Ferrara, K. W. Mean Flow Rate and Intergrated Perfusion Estimates Obtained with Contrast-Assisted Ultrasound. IEEE Ultrasonics Symposium. , 1977-1980 (2002).
- Song, J., Chappell, J. C., Qi, M., VanGieson, E. J., Kaul, S., Price, R. J. Influence of injection site, microvascular pressure and ultrasound variables on microbubble-mediated delivery of microspheres to muscle. J Am Coll. Cardiol. 39, 726-731 (2002).
- Sheikov, N., McDannold, N., Vykhodtseva, N., Jolesz, F., Hynynen, K. Cellular mechanisms of blood-brain barrier opeing induced by ultrasound in the presences of microbubbles. Ultrasound Med. Biol. 30, 979-989 (2004).
- Tanter, J., Fink, M. Focusing and steering through absorbing and aberrating layers: Application to ultrasonic propagation through the skull. J Acoust Soc Am. 103, 2403-2410 (1998).
- Yeh, C. K., Kruse, D. E., Lim, M. C., Redline, D. E., Ferrara, K. W. A New High Frequency Destruction/Reperfusion System. IEEE. 1, 433-436 (2003).

