A Precise and Quantifiable Method for Collecting Hemolymph from Small Arthropods
Summary
We describe a method to collect quantifiable hemolymph efficiently from small arthropods for subsequent analysis.
Abstract
Arthropods are known to transmit a variety of viruses of medical and agricultural importance through their hemolymph, which is essential for virus transmission. Hemolymph collection is the basic technology for studying virus-vector interactions. Here, we describe a novel and simple method for the quantitative collection of hemolymph from small arthropods using Laodelphax striatellus (the small brown planthopper, SBPH) as a research model, as this arthropod is the main vector of rice stripe virus (RSV). In this protocol, the process begins by gently pinching off one leg of the frozen arthropod with fine-tipped tweezers and pressing the hemolymph out of the wound. Then, a simple micropipette consisting of a capillary and a pipette bulb is used to collect the transudative hemolymph from the wound according to the principle of capillary forces. Finally, the collected hemolymph can be dissolved into a specific buffer for further study. This new method for collecting hemolymph from small arthropods is a useful and efficient tool for further research on arboviruses and vector-virus interactions.
Introduction
Both animal and plant viruses can be transmitted by arthropods, and these viruses pose a severe threat to human health and cause tremendous economic losses in agriculture1,2,3. Importantly, the arthropod hemolymph, which serves as the circulatory system and a vital element of the immune system in arthropods, plays an important role in regulating arboviral transmission. Viruses acquired through the arthropod guts are transported to other tissues only after successfully escaping the adverse hemolymph environment4,5,6,7. The lifecycle of viruses in the arthropod hemolymph involves virus survival in the fluid plasma, entry into the hemocyte, and transport to other tissues, and various virus-vector interaction mechanisms occur in the hemolymph8,9,10,11,12. For example, the vertical transmission of RSV by the SBPH is dependent on a molecular interaction between the SBPH vitellogenin protein and the RSV (rice stripe virus) capsid protein13,14. Some viruses may escape the immune response of the hemolymph by binding specific vector factors15,16,17,18. Therefore, investigating vector-virus interactions in the hemolymph of arthropods is important for developing a better understanding of arbovirus transmission.
The hemolymph of some small insects, such as planthoppers, leafhoppers, and some mosquitoes, is difficult to collect due to their size. To address this issue, several methods have been developed to collect hemolymph, including inserting a syringe needle directly into the insect body to extract a microvolume of the hemolymph, collecting exudate from the wound site with fine-tipped tweezers, and direct centrifugation. These methods have enabled the measurement of relative gene expression levels and viral titers within the hemolymph19,20,21. However, an effective method for quantifying the hemolymph volume, which is necessary for hemocyte counting, protein quantification, and enzyme activity analysis, is currently not available for these small insects.
The SBPH (small brown planthopper) is a type of small insect vector with a body length of about 2-4 mm. The SBPH is capable of transmitting a variety of plant viruses, including RSV, maize rough dwarf virus, and rice black streaked dwarf virus22,23,24. The interaction between the SBPH and RSV has been studied in depth over the past decade. To facilitate working with SBPHs, we developed a novel and simple method of collecting hemolymph. This method, which is based on the principle of capillary forces, uses a capillary with a scale mark to acquire the insect's hemolymph in a precise and quantifiable manner. This allows us to collect a specific volume of hemolymph from small insects efficiently and to study the hemolymph environment of small vectors in more detail.
Protocol
1. Insect rearing
- Raise the SBPHs used in this experiment in rice seedlings (Oryza sativa cv. Nipponbare). Plant 20 rice seedlings in an incubator (65 mm x 200 mm), and grow at 25 °C under a 16 h light/8 h dark photoperiod.
2. Dissection of the SBPHs for hemolymph collection
- Put the SBPHs into a centrifuge tube, and place them in an ice bath for 10-30 min.
NOTE: Do not place the SBPHs in the ice bath for less than 10 min, or the insects may revive. - Place a frozen SBPH on a glass slide (see Table of Materials) under a stereomicroscope (see Table of Materials) with its abdomen facing up. Adjust the focus on the six legs of the insect.
- Prepare two high-precision tweezers (see Table of Materials) with ultra-fine tips. Use one tweezer to press down on the insect's body to keep it in place, and use the other one to carefully pull one of the legs off the insect.
NOTE: For insects with a hard shell, an ophthalmic scalpel can be used for cutting off one of the legs. - Gently press the chest of the insect with a tweezer to make the hemolymph flow out through the wound.
NOTE: The hemolymph presents as transparent droplets without any visible white floccule. Discard the lipid if there is any white floccule, which represents fat body contamination.
3. Hemolymph collection using micropipettes
- Prepare a micropipette. Place a capillary tube (see Table of Materials) with a volume of 1 µL into the pipette bulb (see Table of Materials). For accurate measurements, ensure that the scale line is visible.
NOTE: A small hole present on the top of the pipette bulb is necessary. - When collecting the hemolymph, hold the pipette bulb of the micropipette, and place the capillary tube close to the insect wound. Collect the hemolymph exuding from the wound by simply touching the tip of the capillary to the hemolymph.
- Block the small hole on the top of the pipette bulb slightly with the finger to stop the absorbing process when the liquid inside the capillary tube reaches the desired scale line.
NOTE: Collect one sample of 1 µL of hemolymph incessantly and rapidly to avoid lipid contamination and tube plugging. - Press the pipette bulb to discharge the collected hemolymph into 100 µL of PBS buffer (137 mmol/L NaCI, 2.7 mmol/L KCI, 4.3 mmol/L Na2HPO4, 1.4 mmol/L KH2PO4, pH 7.2-7.4).
NOTE: The capillary tube is inserted into the PBS buffer. - Change to a new capillary tube when collecting another 1 µL sample of hemolymph.
4. Coomassie Blue staining
- Collect 3 hemolymph samples with the same volume (1 µL) from 3rd stage larvae according to the protocols described in step 3, and discharge the collected hemolymph into the PBS buffer to make a total volume of 100 µL.
- Add 25 µL of 5x protein loading buffer (250 mmol/L Tris-HCI [pH 6.8], 10% W/V SDS, 0.05% W/V Bromophenol Blue, 50% W/V glycerol, 5% W/V β-mercaptoethanol) into 100 µL of the diluted hemolymph samples, and heat to denature the proteins.
- Load 20 µL of the boiled samples onto a 10% SDS-PAGE protein gel (see Table of Materials) to separate the proteins.
- Stain the gel in a Coomassie Blue solution (mix 1 g of Coomassie Brilliant Blue R-250 with 250 mL of isopropanol, 100 mL of glacial acetic acid, and 650 mL of ddH2O, and filter out any particles using filter paper) for 1 h at room temperature, and then decolorize the gel for 1 h with decolorizing solution (100 mL of acetic acid, 400 mL of methanol, 500 mL of ddH2O).
5. Protein concentration determination
- Dilute 10 mg/mL bovine serum albumin (BSA) to 5 mg/mL, 2.5 mg/mL, 1.25 mg/mL, 0.625 mg/mL, and 0.3125 mg/mL with PBS buffer using the two-fold dilution method.
- Aspirate 5 µL of PBS buffer and each concentration of BSA solutions in step 5.1, add 1 mL of 1x Bradford dye reagent (see Table of Materials), and incubate for 5 min at room temperature. Use PBS buffer in Bradford dye reagent as the control, and zero at OD = 595 nm. Measure the absorbance values of each of the remaining protein concentrations, and make a standard curve.
- Add 1 µL of hemolymph from larval, female, or male SBPHs into 100 µL of PBS buffer, and then centrifugate the samples at 1,000 x g for 10 min at 4 °C to remove the hemocytes.
- Aspirate 90 µL of supernatant to a new centrifuge tube, and measure the absorbance values of the supernatant according to the instructions in step 5.2.
- Calculate the protein concentrations of the hemolymph samples according to the regression equation of the standard curve. Include three biological replicates with three technical replicates in the experiments.
6. Microscopic detections
- Use micropipettes to collect hemolymph from 10-15 SBPHs at different developmental stages. Add the collected hemolymph into a tube containing 20 µL of 4% paraformaldehyde solution (see Table of Materials). Incubate the mixture at room temperature for 30 min to fix the hemolymph.
- Pipette 20 µL of fixed hemocytes onto the center of a slide coated with silane (see Table of Materials).
- Dry up the droplet in a slide drying rack.
NOTE: Avoid excessive drying. - Cover the sample with gold antifade reagent 4',6-diamidino-2-phenylindole (DAPI) (see Table of Materials), and inspect the slide under an inverted microscope (see Table of Materials).
7. Cell quantification
- Collect 1 µL of hemolymph from the SBPHs with a micropipette, and dissolve it into 20 µL of PBS buffer.
- Dilute the hemolymph samples five-fold with PBS buffer.
- Pipette 20 µL of hemolymph solution into the center of the cell counting chamber (see Table of Materials). Cover the droplet with a coverslip (see Table of Materials).
- Count the cells in the five squares on the cell counting chamber (Supplementary Figure 1), under an inverted microscope (see Table of Materials). Include three biological replicates with three technical replicates in each of the experiments.
Cells/µL = total count of five middle squares × 5 × dilution factor × 104
8. Statistical analyses
- Perform statistical analyses with the corresponding software (see Table of Materials). Create a new file, and select the column, import the data, and generate a scatter plot with a bar.
- Calculate the mean and SD for each group of data by using column statistics, and use a one-way ANOVA tool to evaluate the significance of the differences among the groups. Determine the mean and SD from three biological replicates with three independent experiments.
Representative Results
Micropipette model and hemolymph collection
We have developed a simple micropipette whose action is based on the capillary forces of the capillary tube. The micropipette is composed of a capillary tube and a pipette bulb (Figure 1A). Capillary tubes are available in different volume sizes ranging from 1 µL to 20 µL, and the capillary tube volumes are selected according to the requirements. Capillary tubes with smaller volumes are not suggested because the extra fine apertures of smaller-volume tubes can make it difficult to absorb liquid such as hemolymph. The pipette bulb contains a hole on the top that cannot be plugged during the hemolymph collection. This pipette bulb is convenient for holding the micropipette during the liquid collection (Figure 1B) and also assists in transferring the collected liquid from the capillary tube into the collection buffer.
In order to collect hemolymph easily, in this work, the SBPHs were firstly frozen in ice or in the refrigerator. These frozen SBPHs were then localized on a slide under a stereomicroscope, and one of the legs of each SBPH was pulled off with fine-tipped tweezers (Figure 1C). To ensure a large wound and optimal hemolymph collection, it is best to pull off the leg at the root (Figure 1Ca, b). In order to minimize the risk of contamination by the fat body, only clear liquid drops without any white floccule were collected (Figure 1Cc). The micropipette was used to absorb the desired volumes of hemolymph (Figure 1Cd). To collect 1 µL of hemolymph, approximately 30-40 larval SBPHs or 8-15 adult SPBHs had to be dissected.
Analysis of the collected hemolymph
To assess the accuracy of the micropipette as a method for evaluating the volume of collected hemolymph, we tested the protein concentrations of different samples. Hemolymph from larvae was collected using a micropipette with a capillary volume of 1 µL, and three protein samples were collected separately and tested by running an SDS-PAGE gel. The results showed that the amount of protein in the three lanes was nearly equal (Figure 2A). For larvae, the total protein content was 3.707 mg/mL ± 1.382 mg/mL. We also collected the same volume of hemolymph from adult female and male SBPHs, and these showed protein concentrations of 3.515 mg/mL ± 1.400 mg/mL and 3.621 mg/mL ± 0.860 mg/mL, respectively (Table 1). There were no significant differences in the protein among the three samples (Figure 2B).
In addition, we also observed the hemocytes inside the collected larval hemolymph to assess the quality and purity of the hemolymph samples. The hemocytes varied in size between 2-20 nm, and no fat body was detected (Figure 2C). The majority of the cells identified were plasmatocytes, ranging from 5-15 nm in diameter and often appeared in aggregates25. We then counted the cell concentrations of the hemolymph from larvae, adult females, and adult males, and the cell concentrations identified were 1.794 x 105/µL ± 0.614 x 105/µL, 1.256 x 105/µL ± 0.603 x 105/µL, and 1.553 x 105/µL ± 0.474 x 105/µL, respectively (Table 2). The hemocyte cell count in the larvae, adult females, and adult males showed no significant differences (Figure 2D). These results indicate that the micropipette collection method is a reliable and accurate way to collect hemolymph from SBPHs.
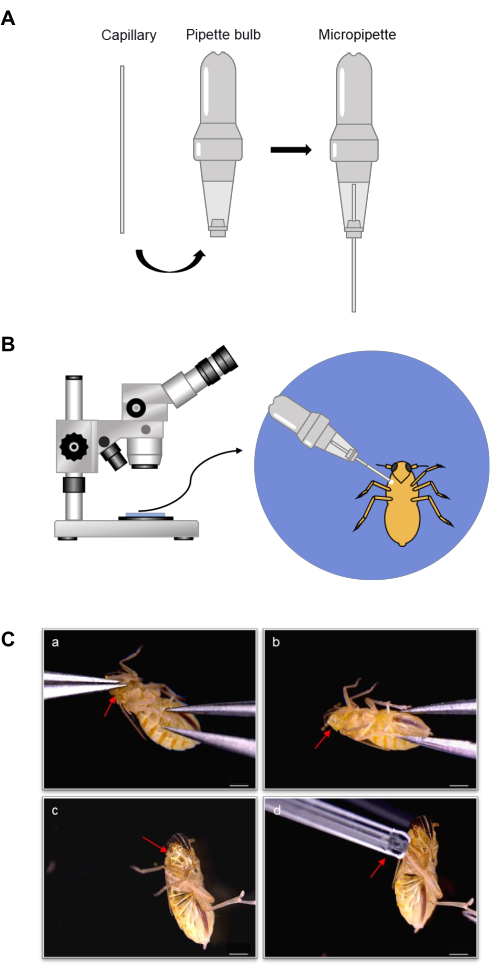
Figure 1: Schematic of the micropipette model and hemolymph collection. (A) Micropipette composition. The micropipette consists of a capillary and a pipette bulb. The capillary volumes range from 1-20 µL. The pipette bulb has a small hole on the top and the bottom. The capillary is inserted into the bottom hole, and the scale is visible. (B) Overview diagram of hemolymph collection with a micropipette. The insect abdomen is kept facing up while a leg is detached, hemolymph outflow is induced, and the hemolymph is collected into the pipette under a stereomicroscope. (C) Process of hemolymph collection with a micropipette. One of the legs is pulled off at the root with fine-tipped tweezers (a), and the insect's body is pressed to make hemolymph flow out (b,c). The hemolymph from the wound is collected into the capillary (d). Scale bar = 500 µm. Please click here to view a larger version of this figure.
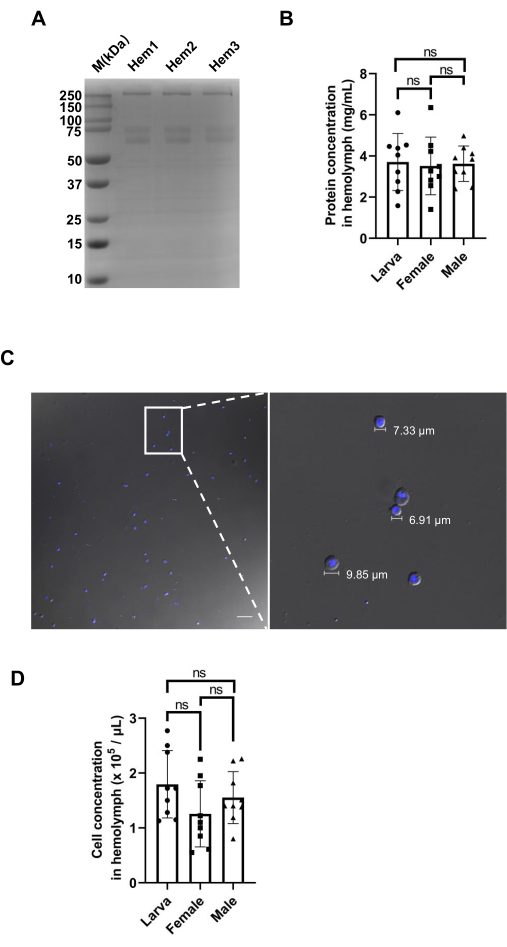
Figure 2: Analysis of the SBPH hemolymph. (A) Coomassie Blue staining showing three replicates of collected larval hemolymph. Hem indicates hemolymph. (B) Total protein concentrations of the hemolymph of larval, female, and male SBPHs. (C) Microscopic images showing the cells present in the hemolymph in SBPHs at 20x and 60x magnification, respectively. The nucleus was stained with DAPI (blue). Scale bar = 50 µm. (D) Hemocyte density of larval, female, and male SBPHs. The mean and SD were calculated from three independent experiments. Please click here to view a larger version of this figure.
| Hemolymph | Protein concentration (mg/mL) |
| Larva | 3.707±1.382 |
| Female | 3.515±1.400 |
| Male | 3.621±0.860 |
Table 1: Protein concentrations of hemolymph from different SBPHs. The data were obtained from three biological replicates.
| Hemolymph | Cell concentration (105 / μL) |
| Larva | 1.794±0.614 |
| Female | 1.256±0.603 |
| Male | 1.553±0.474 |
Table 2: The total concentrations of hemocytes in the hemolymph from different SBPHs. The data were obtained from three biological replicates.
Supplementary Figure 1: Determining the number of cells. The four corner squares (1, 2, 3, and 4) and the central square (5) are counted on the cell counting chamber. For border cells, only the two boundaries (top and left) are counted. Please click here to download this File.
Discussion
Hemolymph is the medium of the circulatory system in arthropods, and arboviruses can only invade other arthropod tissues if they are able to survive the hostile hemolymph environment. Collecting a high-quality sample of hemolymph is the first step in studying the vector-virus interactions that occur in the hemolymph. It has been reported that insect hemolymph can be obtained from several sites on the insect's body, including a wound on the front leg, a minor incision in the head area, or a tear wound at the abdomen26,27,28,29. Different collection methods may work for certain acquisitions. The method involving creating a small incision at the head-neck joint is more suitable for hemolymph collection from large insects such as honeybees26. Creating a wound in the head area of the SBPH is challenging as it may damage other organs and introduce contamination from other tissues. After making a tear wound at the abdomen, the insects are immersed in the buffer, and then the buffer is centrifuged to extract the hemolymph. This is a convenient method to collect lipids from small insects21. However, as there is a high amount of fat body within the hemocoel of SBPHs, a wound on the body part may cause fat body leakage from the wound and lead to the contamination of the hemolymph. This method is more suitable for collecting hemolymph from adult SBPHs, which have less fat body than larvae. To effectively prevent fat body contamination, it is recommended to make a wound at the leg site rather than directly on the body. In previous studies of SBPH hemolymph, fine-tipped tweezers were used to facilitate the collection of small drops of liquid from the wound13. Although the collected hemolymph was clear from impurities, the volume of hemolymph was not measured. In this study, we developed a simple micropipette that can be used to accurately and quantitatively collect hemolymph from very small insect vectors.
For successful hemolymph collection, several critical steps must be taken into account. Firstly, it is essential to anesthetize the insects at 4 °C for 15 min in order to produce hemolymph that is free of fat body. Secondly, it is simpler to obtain pure hemolymph by pulling off the first leg. Thirdly, the collection of the hemolymph should be done in a continuous and swift manner, as otherwise the hemolymph may coagulate at the bottom and block the capillary.
The capillary collection technique has been widely used for animal blood collection30,31. We modified the technique to suit the distinct characteristics of insect hemolymph. As SBPHs possess a small body size, a capillary with a minimal volume of 1 µL was chosen. Using a capillary with a volume lower than 1 µL is not recommended. It is noteworthy that hemolymph contains a high density of proteins and a considerable number of cells (Figure 2), which elevate its liquid density and make it difficult to absorb hemolymph via capillaries with smaller volumes.
The method of collecting hemolymph by capillary is a simple, cost-effective, and viable technique that enables the reliable and precise quantification of the hemolymph. This newly developed method could also be applied for collection of other trace fluids from small vectors, such as honeydew. It is vital to highlight that the insects remain alive after the extraction of hemolymph using this method. This is beneficial for studies involving other insect organs or for repeated hemolymph collection. This work presents a valuable technology for the study of entomology and virus-vector interactions.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Key R&D Program of China (No. 2022YFD1401700) and by the National Science Foundation of China (No. 32090013 and No. 32072385).
Materials
| 10% SDS-PAGE protein gel | Bio-rad | 4561035 | Protein separation and detection |
| 4% paraformaldehyde | Solarbio | P1110 | For fixation of the cells or tissues |
| Bradford dye reagent | Bio-rad | 5000205 | Protein concentration detection |
| Capillary | Hirschmann | 9000101 | For collecting hemolymph |
| Cell counting chamber | ACMEC | AYA0810 | Hemocytes counting |
| Glass slide | Gitoglas | 10127105A | For holding insects |
| Glass slide coated with silane | Sigma | S4651-72EA | For holding microscope samples |
| Gold antifade reagent with DAPI | Invitrogen | P36935 | Nucleus staining |
| Microscope cover glass | Gitoglas | 10212424C | For microscopic observation |
| Pipette bulb | Hirschmann | 9000101 | For collecting hemolymph |
| Prism 8.0 software | GraphPad Software | / | Statistical analyses |
| Stereomicroscope | Motic | SMZ-168 | For insect dissection |
| Tweezers | Tianld | P5622 | For insect dissection |
| Zeiss inverted microscope | Zeiss | Observer Z1 | Hemocytes observation |
References
- Hogenhout, S. A., Ammar el, D., Whitfield, A. E., Redinbaugh, M. G. Insect vector interactions with persistently transmitted viruses. Annual Review of Phytopathology. 46, 327-359 (2008).
- Ray, S., Casteel, C. L. Effector-mediated plant-virus-vector interactions. Plant Cell. 34 (5), 1514-1531 (2022).
- Islam, W., et al. Plant-insect vector-virus interactions under environmental change. Science of the Total Environment. 701, 135044 (2020).
- Cory, J. S. Insect virus transmission: Different routes to persistence. Current Opinion in Insect Science. 8, 130-135 (2015).
- Wang, X. W., Blanc, S. Insect transmission of plant single-stranded DNA viruses. Annual Review of Entomology. 66, 389-405 (2021).
- Yi, H. Y., Chowdhury, M., Huang, Y. D., Yu, X. Q. Insect antimicrobial peptides and their applications. Applied Microbiology and Biotechnology. 98 (13), 5807-5822 (2014).
- Liu, W. W., et al. Proteomic analysis of interaction between a plant virus and its vector insect reveals new functions of hemipteran cuticular protein. Molecular & Cellular Proteomics. 14 (8), 2229-2242 (2015).
- Wang, L., Van Meulebroek, L., Vanhaecke, L., Smagghe, G., Meeus, I. The bee hemolymph metabolome: A window into the impact of viruses on bumble bees. Viruses. 13 (4), 600 (2021).
- Jia, D., et al. Vector mediated transmission of persistently transmitted plant viruses. Current Opinion in Virology. 28, 127-132 (2018).
- Anderson, J. F., Main, A. J., Ferrandino, F. J. Horizontal and vertical transmission of West Nile Virus by Aedes vexans (Diptera: Culicidae). Journal of Medical Entomology. 57 (5), 1614-1618 (2020).
- Gadhave, K. R., et al. Low frequency of horizontal and vertical transmission of cucurbit leaf crumple virus in whitefly Bemisia tabaci Gennadius. Phytopathology. 110 (6), 1235-1241 (2020).
- Logan, R. A. E., et al. Vertical and horizontal transmission of cell fusing agent virus in Aedes aegypti. Applied and Environmental Microbiology. 88 (18), e0106222 (2022).
- Huo, Y., et al. Transovarial transmission of a plant virus is mediated by vitellogenin of its insect vector. PLoS Pathogens. 10 (3), e1003949 (2014).
- Wei, J., et al. Vector development and vitellogenin determine the transovarial transmission of begomoviruses. Proceedings of the National Academy of Sciences of the United States of America. 114 (26), 6746-6751 (2017).
- Medzhitov, R. Toll-like receptors and innate immunity. Nature Reviews Immunology. 1 (2), 135-145 (2001).
- Kingsolver, M. B., Huang, Z., Hardy, R. W. Insect antiviral innate immunity: Pathways, effectors, and connections. Journal of Molecular Biology. 425 (24), 4921-4936 (2013).
- Pei, R. J., Chen, X. W., Lu, M. J. Control of hepatitis B virus replication by interferons and Toll-like receptor signaling pathways. World Journal of Gastroenterology. 20 (33), 11618-11629 (2014).
- Kao, Y. T., Lai, M. M. C., Yu, C. Y. How dengue virus circumvents innate immunity. Frontiers in Immunology. 9, 2860 (2018).
- Gilliam, M., Shimanuki, H. Coagulation of hemolymph of the larval honey bee (Apis mellifera L). Experientia. 26 (8), 908-909 (1970).
- Huo, Y., et al. Insect tissue-specific vitellogenin facilitates transmission of plant virus. PLoS Pathogens. 14 (2), e1006909 (2018).
- Chen, X., et al. A plant virus ensures viral stability in the hemolymph of vector insects through suppressing prophenoloxidase activation. mBio. 11 (4), e01453 (2020).
- Vidano, C. Phases of maize rough dwarf virus multiplication in the vector Laodelphax striatellus (Fallén). Virology. 41 (2), 218-232 (1970).
- Yu, Y. L., et al. Laodelphax striatellus Atg8 facilitates Rice stripe virus infection in an autophagy-independent manner. Journal of Insect Science. 28 (2), 315-329 (2021).
- Zhang, J. H., et al. Cytochrome P450 monooxygenases CYP6AY3 and CYP6CW1 regulate Rice black-streaked dwarf virus replication in Laodelphax striatellus (Fallen). Viruses. 13 (8), 1576 (2021).
- Ribeiro, C., Brehelin, M. Insect haemocytes: What type of cell is that. Journal of Insect Physiology. 52 (5), 417-429 (2006).
- Butolo, N. P., et al. A high quality method for hemolymph collection from honeybee larvae. PLoS One. 15 (6), e0234637 (2020).
- Nesa, J., et al. Antimicrobial potential of a ponericin-like peptide isolated from Bombyx mori L. hemolymph in response to Pseudomonas aeruginosa infection. Scientific Reports. 12 (1), 15493 (2022).
- Mahmoud, S., et al. Curcumin-injected Musca domestica larval hemolymph: Cecropin upregulation and potential anticancer effect. Molecules. 27 (5), 1570 (2022).
- Patton, T. G., et al. salivary gland, and hemolymph collection from Ixodes scapularis ticks. Journal of Visualized Experiments. (60), e3894 (2012).
- Piyankarage, S. C., Augustin, H., Featherstone, D. E., Shippy, S. A. Hemolymph amino acid variations following behavioral and genetic changes in individual Drosophila larvae. Amino Acids. 38 (3), 779-788 (2010).
- Fiorotti, J., et al. Disclosing hemolymph collection and inoculation of metarhizium blastospores into Rhipicephalus microplus ticks towards invertebrate pathology studies. Journal of Visualized Experiments. (148), e59899 (2019).

