Complete and Partial Aortic Occlusion for the Treatment of Hemorrhagic Shock in Swine
Summary
Here, we present a protocol demonstrating a hemorrhagic shock model in swine that uses aortic occlusion as a bridge to definitive care in trauma. This model has application in testing a wide range of surgical and pharmacological therapeutic strategies.
Abstract
Hemorrhage remains the leading cause of preventable deaths in trauma. Endovascular management of non-compressible torso hemorrhage has been at the forefront of trauma care in recent years. Since complete aortic occlusion presents serious concerns, the concept of partial aortic occlusion has gained a growing attention. Here, we present a large animal model of hemorrhagic shock to investigate the effects of a novel partial aortic balloon occlusion catheter and compare it with a catheter that works on the principles of complete aortic occlusion. Swine are anesthetized and instrumented in order to conduct controlled fixed-volume hemorrhage, and hemodynamic and physiological parameters are monitored. Following hemorrhage, aortic balloon occlusion catheters are inserted and inflated in the supraceliac aorta for 60 min, during which the animals receive whole-blood resuscitation as 20% of the total blood volume (TBV). Following balloon deflation, the animals are monitored in a critical care setting for 4 h, during which they receive fluid resuscitation and vasopressors as needed. The partial aortic balloon occlusion demonstrated improved distal mean arterial pressures (MAPs) during the balloon inflation, decreased markers of ischemia, and decreased fluid resuscitation and vasopressor use. As swine physiology and homeostatic responses following hemorrhage have been well-documented and are like those in humans, a swine hemorrhagic shock model can be used to test various treatment strategies. In addition to treating hemorrhage, aortic balloon occlusion catheters have become popular for their role in cardiac arrest, cardiac and vascular surgery, and other high-risk elective surgical procedures.
Introduction
Hemorrhage continues to be the dominant cause of preventable deaths in patients undergoing traumatic events, accounting for 90% of trauma-related deaths in the military setting and 40% of post-traumatic deaths in the civilian population1,2. Although direct pressure can treat compressible hemorrhage, non-compressible torso hemorrhage remains difficult to treat and can be lethal without prompt hemostatic control. The historical approach of resuscitative thoracotomy or laparotomy with aortic cross-clamping has proved to be extremely invasive3,4. This intervention also requires a complex selection algorithm to determine the candidacy of patients that have undergone traumatic insults5.
In recent years, there has been a resurgence of interest in a previously described approach—resuscitative endovascular balloon occlusion of the aorta (REBOA)6,7,8. Although REBOA has conferred a short-term survival advantages in hemorrhage, a prolonged complete occlusion of the aorta during balloon inflation poses serious concerns that include irreversible end-organ ischemia9,10. In an attempt to overcome this potential morbidity, alternative endovascular strategies to manage hemorrhage are being devised. One such strategy that has seen a growing attention is a partial occlusion of the aorta11,12. The idea of partial aortic balloon occlusion affords the perfusion of vascular beds distal to the site of occlusion, improved physiologic proximal aortic MAPs, and a gradual afterload reduction following the balloon deflation. These changes in parameters are desired modifications to the physiological characteristics of a bleeding animal. Prior to this method's translation to humans, complete and partial aortic balloon occlusion catheters have been heavily tested in swine models of hemorrhagic shock11,12,13.
Swine have been used in studies entailing hemorrhagic shock for many years. Most of the current understanding of the pathophysiology of hemorrhagic shock is derived from studies that have utilized animal models, including swine. Their physiology and homeostatic responses in the setting of pathologic volume depletion following hemorrhage, especially those pertaining to blood clotting and cardiovascular responses, have been well-documented and are like those in humans14. Swine models of hemorrhagic shock also provide opportunities to investigate treatment strategies for hemorrhagic shock and other traumatic injuries.
In this study, we demonstrate a clinically realistic model of hemorrhagic shock in swine to evaluate endovascular treatment strategies, including complete and partial aortic balloon occlusion. We hypothesize that a partial occlusion of the aorta results in a better physiologic and laboratory profile compared to a complete occlusion of the aorta in swine undergoing a controlled fixed-volume hemorrhage.
We aimed to compare the physiologic effects of partial and complete aortic occlusion as a treatment for hemorrhagic shock in a swine model. Partial aortic occlusion was achieved using a selective aortic balloon occlusion in trauma (SABOT) catheter (Figure 1). The SABOT catheter is a two-balloon system that allows intra-luminal blood flow, thereby providing a partial aortic flow to the vascular beds distal to the occlusion. Complete aortic occlusion was achieved using a single-balloon aortic occlusion catheter (e.g.,CODA) (Figure 1). Treatment groups were randomized to undergo resuscitative aortic occlusion either with the complete or with the partial aortic balloon occlusion catheters (n = 2/group).
The major steps of the model include the induction of anesthesia and intubation, the maintenance of anesthesia, instrumentation, 35% TBV hemorrhage (20 min total; half over the first 7 min, and half over the remaining 13 min), aortic balloon occlusion and whole-blood resuscitation (60 min of occlusion; 20% whole-blood resuscitation during the last 20 min of the occlusion), critical care monitoring (240 min) with hemodynamic observation, and euthanasia with tissue harvesting. Figure 2 demonstrates the model utilized in this experiment.
Protocol
In conducting research using animals, the investigators adhered to the Animal Welfare Act Regulations and other Federal statutes relating to animals and experiments involving animals and the principles set forth in the current version of the Guide for Care and Use of Laboratory Animals of the National Research Council. This study protocol was approved by the University of Michigan Institutional Animal Care and Use Committee (IACUC). The experiments were conducted in compliance with all regulations and guidelines regarding animal welfare in research.
1. Animal Selection and Acclimation
- Use female Yorkshire-Landrace crossbred swine (n = 2/group, age range: 5 – 6 months, weight range: 40 – 60 kg) for the experiment.
- After their arrival at the facility, house the animals individually in cages but ensure that there is ample interaction between the animals in neighboring cages.
- House the animals for at least 5 d so that they are completely acclimated before performing the experiment. During this period, feed the animals with a standard diet and observe them to ascertain they are in a good state of health.
- Keep the animals from eating and drinking after midnight before the surgery to prevent the risk of aspiration during the intubation.
2. Anesthesia and Intubation
- Sedate the animal with 5 mg/kg tiletamine/zolazepam given intramuscularly (IM) into the gluteus muscle while the animal is in the housing area. Concurrently, give 0.05 mg/kg atropine IM as an anticholinergic agent to minimize secretions during the intubation.
- Transport the animal from the housing area to the operating room and place it in the supine position on an operating table.
- Use inhaled isoflurane (2% – 5%) to induce anesthesia. Use 21% oxygen (0.4 L/min) mixed with air (4 L/min) as the carrier gases to deliver the inhaled anesthetic gas.
- While the desired depth of anesthesia is being achieved, place a 20 G peripheral intravenous (IV) catheter in the ear vein for the IV administration of fluids, drugs, or anesthesia, as needed.
- As the animal acquires the desired depth of anesthesia, gently wash off any waste and debris from the operative sites, including the left and right inguinal regions, left and right sides of the neck, and the abdomen. Use an electric hair clipper to remove all hair from the operative sites. Assess the depth of anesthesia by pinching the hind limb of the animal and observing its (lack of) response.
- Secure the animal to the operating table by using surgical cotton ropes around each extremity, prior to the intubation.
- Use a short piece of surgical cotton rope to lift the mandible along with the tongue to open the oral orifice. Use another piece of cotton rope to pull the maxilla downwards. In case the jaw relaxation is insufficient or the vocal folds are collapsed, increase the level of isoflurane briefly and repeat this step.
- Use the non-dominant hand to hold the handle of a conventional laryngoscope fitted with a 12 in lighted Miller blade. Pass the tip of the Miller blade through the oropharynx. Practice caution to preserve the teeth and oral mucosa while maneuvering the blade in the oral cavity. Slowly advance the blade tip until the epiglottis can be clearly visualized and advance past the laryngeal inlet. Using the wrist, lift the blade, elevating the epiglottis, such that a clear view of the larynx is obtained.
- Place a 6.5 Fr or 7 Fr endotracheal tube (ETT) with a stylet between the vocal folds into the trachea. In case of laryngospasm due to mucosal irritation, apply 2% lidocaine on the vocal folds and the median glosso-epiglottic field and reassess.
- Remove the stylet before fully advancing the tube down the trachea while keeping a firm grip on the tube. Inflate the balloon cuff with 10 – 15 cm3 of air to prevent any air leak around the cuff and aspiration of gastric contents.
- Connect the ETT to the mechanical ventilator through a breathing filter (heat and moisture exchanger). Secure the ETT by tying the cotton tape around the snout (maxilla).
- In the event of a failed intubation, let the animal oxygenate and ventilate briefly before any additional attempts.
- Ensure the appropriate mechanical ventilator settings: a mixture of 21% oxygen and air; a tidal volume of 7 – 10 mL/kg body weight; a respiratory rate of 10 – 15 breaths/min to maintain an end-tidal pCO2 of 40 mmHg ± 2 mmHg.
- Place a pulse oximeter on either the ear, tongue, or tail to measure the blood oxygenation and heart rate. Advance a rectal probe through the anal orifice for continuous temperature monitoring.
Note: The temperature of the animal is to be maintained between 38 °C to 40 °C using a heated temperature-regulated operating table, heat mat, or heating blanket. - Use isoflurane (1% – 3%) for the maintenance of anesthesia throughout the procedure. During this time, assess the depth of anesthesia by testing for pain reflexes using a hind limb pinch. In addition, monitor the respiratory rate on the ventilator to see if it matches the programmed ventilator rate.
Note: Over-breathing indicates an inadequate depth of anesthesia.
3. Surgical Site Sterilization (Preparation and Draping)
- Prepare the incision sites, as well as an extensive area surrounding them for the experimental procedures. The incision sites for the experiment include bilateral inguinal regions, the bilateral neck, and the lower abdomen. Disinfect the operative sites by scrubbing them with a copious amount of povidone-iodine for 5 min.
- Use dry, sterile gauze to remove the povidone-iodine soap from the skin.
- Place sterile surgical towels around the operative sites to preserve the sterile surgical fields. Use towel forceps to secure the towels in place. Cover the animal with a sterile drape to prevent any contamination of the surgical sites.
4. Cannulation
- Femoral artery and vein cannulation
Note: A cannulation of the right femoral artery, left femoral artery, and left femoral vein is performed. The right femoral artery is cannulated using a 14 Fr insertion sheath for the aortic balloon occlusion catheter insertion. The left femoral artery is cannulated using a 5 Fr catheter for distal MAP monitoring. An 8 Fr catheter is placed in the left femoral vein for the fluid administration and blood withdrawal. The open Seldinger technique is employed to gain vascular access for all cannulations15.- Before making the incision, assess the depth of anesthesia by pinching the hind limb of the animal.
- Using a scalpel fitted with a number 10 sterile surgical blade, make a vertical 8-cm incision in the right groin 4 cm above and 4 cm below the right inguinal crease.
- Dissect through the subcutaneous tissue and muscle and use two Weitlaner retractors for gaining proper exposure. Use an Army-Navy retractor for additional retraction if needed.
- Use Mixter Right Angle Forceps and electrocautery to dissect through connective tissues until the neurovascular bundle is clearly exposed.
- Cautiously dissect the artery. Preserve the nerve, which is the lateral-most structure.
- Carefully dissect the vein, which is located most medially, away from the artery.
- Gain proximal and distal control of the artery using 2-0 silk ties.
- Use a 20 G angled introducer needle to puncture the artery. Ensure an appropriate position in the vessel lumen by observing the pulsatile blood flow through the other end of the needle.
- Advance a round-tipped 0.35 in guidewire through the lumen of the angled needle.
- Withdraw the needle over the guidewire. Ensure no migration of the guidewire by holding it in place.
- Pass a 10 Fr dilator over the guidewire to dilate the opening in the artery. Remove the 10 Fr dilator. Insert a 14 Fr insertion sheath over the guidewire.
- Carefully remove the dilator from the insertion sheath along with the guidewire, keeping the cannula inside the artery.
- Flush the insertion sheath to ensure its position inside the vessel lumen.
- Secure the end of the catheter in place with a 3-0 polyglactin suture.
- Use a 3-0 nylon suture to perform a temporary running closure of the overlying skin.
- Repeat the aforementioned steps for the left femoral artery cannulation using a 5 Fr catheter (no initial dilation). Isolate the left femoral vein in a similar fashion and cannulate it using an 8 Fr catheter. Use a 3-0 nylon suture to perform a temporary running closure of the overlying skin.
- Carotid artery and external jugular vein cannulation
Note: A cannulation of the bilateral external jugular veins and left common carotid artery is performed. A 5 Fr catheter is placed in the left common carotid artery for proximal MAP monitoring, and an 8 Fr catheter in the left external jugular vein for additional central venous access for fluid administration and vasopressor infusion. A 9 Fr catheter is placed in the right external jugular vein for pulmonary artery catheterization with a pulmonary artery (e.g., Swan-Ganz) catheter, while a carotid flow probe is placed around the right common carotid artery for carotid flow rate monitoring. The Seldinger approach is used to gain vascular access for all cannulations.- Using a 10-blade scalpel, make a 6 cm vertical incision about 2 cm lateral to the midline on the left side of the neck.
- Use electrocautery to dissect through subcutaneous tissue until sternocleidomastoid (SCM) muscle is exposed.
- Place Weitlaner retractors at the proximal and distal aspects of the incision for a full retraction.
- Dissect along the lateral border of the SCM muscle to expose the left external jugular vein.
- Vessel cannulation can be achieved by following steps 4.1.8 – 4.1.14. Insert, flush, and secure an 8 Fr catheter in the vein.
- For left common carotid artery exposure, dissect the medial edge of the SCM muscle. Place a Weitlaner retractor to enhance exposure.
- Dissect through the connective tissue longitudinally along the lateral border of the trachea. Preserve the thymus glands if they are encountered.
- Expose the carotid triangle, which contains the carotid artery, the internal jugular vein, and the vagus nerve. Palpate the common carotid artery to determine its trajectory.
- Carefully dissect the common carotid artery away from the vein and the nerve.
- Perform the carotid cannulation by following steps 4.1.8 – 4.1.14. Insert, flush, and secure a 5 Fr catheter in the artery as described previously.
- Repeat steps 4.1. for the dissection and isolation of the right external jugular vein and right common carotid artery.
- Place a 4 mm carotid artery flow probe around the right common carotid artery. Apply transducer gel between the flow probe and the vessel for an optimal flow signaling and capture.
- Following the steps 4.1.8-4.1.14, cannulate the right external jugular vein using a 9 Fr introducer sheath. Flush and secure the catheter in place. Use a 3-0 nylon suture to perform a temporary running closure of the overlying skin.
5. PA Catheter Insertion
- Flush the injection port, the proximal port, and the distal port of the catheter with normal saline (NS) and connect them to the transducer tubing. The distal port is designated as the pulmonary artery (PA) port, while the proximal port is designated as the central venous pressure (CVP) port.
- Check for artifact tracing on the monitor by moving the catheter. This is done to make sure that the catheter is functioning.
- Advance the PA catheter through the sterile sleeve.
- Using a 3 cm3 syringe, inflate the balloon of the PA catheter with no more than 1.5 cm3 of air to test for inflation. Deflate the balloon to insert the catheter in the insertion sheath.
- Insert the PA catheter through the 9 Fr insertion sheath. Once the PA catheter is inserted for at least 18 cm, inflate the balloon with no more than 1.5 cm3 of air.
- Slowly advance the PA catheter and check the monitor for arrhythmias during the insertion. In case of ventricular ectopy, inflate and deflate the balloon in order to prevent progression. If the insertion becomes difficult in any way, the balloon should be deflated and the catheter should be retracted and reinserted.
- Observe the monitor to evaluate the transition from the right atrium (RA) to the right ventricle (RV) to the PA to the pulmonary artery wedge pressure (PWP).
- Deflate the balloon and confirm that a PA trace returns to the monitor.
Note: The appropriate length of the catheter from the insertion site in the right external jugular vein is approximately 45 – 55 cm. - Connect the end of the sterile sleeve to the insertion sheath in order to secure the position of the catheter inside the PA.
- Connect the insertion ports of the PA catheter to the capture unit to obtain the cardiac output (CO) and the saturation of venous oxygen (SVO2).
- Calibrate the PA catheter monitoring system. Using the body length and weight of the animal, conduct an in vivo calibration of the PA catheter monitoring system for the animal. Use a venous blood gas for a further calibration using oxygen saturation and hemoglobin levels.
6. Cystostomy Tube Placement
- Make a 5 cm lower abdominal incision in the midline using a 10-blade scalpel.
- Using electrocautery, dissect through subcutaneous tissue and the linea alba. Visualize each layer during dissection.
- Extracorporealize the urinary bladder.
- With two DeBakey forceps, grasp the urinary bladder on the ventral surface away from the ureteral openings.
- Using electrocautery, make a small opening in the bladder, exposing the inner lumen.
- Use suction to remove any urine from within the bladder lumen. Surgical sponges can be used for any inadvertent spillage of urine outside the bladder.
- Use a 4-0 polypropylene suture to perform a temporary purse-string closure of the bladder.
- Place an 18 Fr Foley catheter inside the bladder lumen and use a 10 cm3 syringe to inflate the balloon. Connect a urinary drainage bag to the Foley catheter and secure it to the side of the operating table.
- Tie the 4-0 polypropylene suture to secure the Foley catheter in the bladder lumen.
- Use a 3-0 nylon suture to perform a temporary running closure of the overlying skin of the abdominal wall.
7. Complete and Partial Aortic Balloon Catheter Insertion
- Randomize animals to receive either the complete or partial aortic balloon occlusion catheters.
- Insert a 0.035 in, 260 cm Amplatz Stiff Guidewire through the 14 Fr insertion sheath in the right femoral artery, and guide it into the supraceliac aorta (Zone 1) using ultrasonography of the abdominal and thoracic aorta.
- Insert the balloon occlusion catheter over the guidewire into Zone 1 of the aorta to perform the occlusion following the hemorrhage.
- Confirm the final placement of the aortic balloon occlusion catheter into Zone 1 of the aorta with ultrasonography.
8. Intraoperative Hemodynamic and Laboratory Monitoring
- Ensure the connection of the invasive catheters to the monitoring system using tubing, transducers, and tubing to the hemodynamic monitors. Invasive monitoring systems should be ‘zeroed’ at the level of the animal’s heart to ensure accurate monitoring.
- Record physiologic data throughout the experiment, including the proximal and the distal MAP, heart rate (HR), the CVP, the CO, the PA pressure, the SVO2, end-tidal CO2, and the core body temperature.
- Record these variables every 5 min during the hemorrhage and the balloon inflation phase, as well as every 15 min during the rest of the experiment.
- Collect blood samples at 4 time-points: baseline (BL), post-shock (PS), post-resuscitation (PR), and at the end (E) of the experiment. 10 mL of blood is collected for both plasma and serum analysis at each time point.
- Perform arterial blood gas (ABG) sampling by collecting up to 1 mL of blood in the ABG syringe. Perform ABG sampling at each of the aforementioned time points, and also when needed during the experiment. Use a blood gas analyzer for the analysis.
Note: Each sample should be analyzed within 10 min of drawing the blood because delays longer than 10 min can decrease the efficiency of the results16.
9. Hemorrhage
- Calculate the TBV.
TBV in mL = weight in g × 0.06 + 0.77 - Hemorrhage 35% of the TBV using an automated pump over a period of 20 min. Remove half over the first 7 min, and the other half over the next 13 min.
- Collect the blood in standard blood collection bags. Store them at 4 °C in anticipation of a future transfusion.
- If the MAP decreases below 30 mmHg, stop the hemorrhage, turn off the isoflurane, and administer 50 – 100 mL boluses of NS.
- Restart the hemorrhage when the MAP returns to 30 mmHg.
10. Aortic Balloon Occlusion Catheter Inflation and Whole-Blood Resuscitation
- Inflate the aortic balloon occlusion catheter with 9 – 12 cm3 of air or until no further decrease in the distal MAP following an additional balloon inflation is noted.
- Remove the wire from the partial aortic balloon occlusion catheter to facilitate distal aortic flow, while leaving it for the complete aortic balloon occlusion catheter. Confirm the placement of the aortic balloon occlusion catheter in Zone 1 using ultrasonography to rule out catheter migration.
- Secure the catheter to the skin using an interrupted 3-0 nylon suture.
- Following 40 min of aortic occlusion, resuscitate the animal with whole blood equal in volume to 20% of the TBV via the left femoral vein catheter over a 20 min period using the automated pump. Use a blood warmer for transfusion, targeting a temperature of 40 °C.
- After the resuscitation, deflate the balloon incrementally over 5 min.
- Remove the aortic balloon occlusion catheter from the 14 Fr insertion sheath.
- Monitor the hemodynamics closely during this time, given the known risk of ischemia-reperfusion injury with the associated hypotension.
11. Critical Care, Observation, and Recovery
- Observe the animals for 4 h with a continuous monitoring of their physiological and laboratory parameters.
- Provide maintenance intravenous fluids (NS infusion at 50 ml/h).
- Target a CVP of 6 mmHg by administering 30 ml/kg boluses of NS followed by 20 ml/kg boluses when the CVP falls below the target. Evaluate the fluid responsiveness hourly.
- Target a MAP of 60 mmHg by using a norepinephrine infusion (0.024 mg/ml).
- Maintain a physiologic temperature of 38 – 40 °C using a heating blanket.
- At the end of the 4 h period, euthanize the animals with a sodium pentobarbital (100 mg/kg) injection.
12. Necropsy
- Remove all essential lines and tubes from the animal.
- Prepare a container with regular ice and place it next to the operating table to freeze the organ tissue immediately after the removal from the body.
- Use a scalpel to make a 6 cm long vertical incision along the left sternal border, dissecting through the skin, subcutaneous tissues, pectoralis muscles, and costal cartilages. Enter the thoracic cavity to expose the heart and the lung.
- Remove the pericardial membrane from the heart by making an incision with the scalpel and grab the apex of the heart using forceps. Use a scalpel to cut a 5 x 5 cm section of the heart at the apex.
- Use scissors to cut a 5 x 5 cm section of the lung from the anterior border of the base of the lung.
- Extend the thoracotomy incision into the anterior midline of the abdomen, exposing the abdominal viscera.
- Use forceps to hold the left lobe of the liver and cut a 5 x 5 cm section.
- Repeat the same process for getting samples from the spleen.
- Use the right hand to create a window to enter the left retroperitoneum to grab the kidney. After the renal parenchyma is mobilized, transect the renal artery, vein, and ureter. Remove the renal capsule.
- Gently squeeze stool away from a 5 cm long section of small bowel and use scissors to cut a 5 cm long section of bowel from the mesentery.
- Use the scalpel to cut a 5 x 2 cm long section of quadriceps muscle from the thigh.
- Process these samples further by cutting them into small sections to be placed in microcentrifuge tubes. Flash-freeze these tissue samples by placing the tubes into dry ice or liquid nitrogen.
- Use 50 ml conical tubes containing a formalin solution to fix 3 cm long, finely cut sections of tissue for a histological evaluation.
Representative Results
Hemodynamic and Physiological Parameters:
The MAP decreased immediately after the hemorrhage (Figures 3A – 3D). During the balloon inflation phase, animals in the complete occlusion group experienced a higher proximal MAP compared to the animals in the partial occlusion group (Figures 3A and 3B). The average distal MAP during the balloon inflation was higher in the partial occlusion group compared to the complete occlusion group (average distal MAP, balloon inflation phase; partial: 31 ± 2.9 mmHg, complete: 16.5 ± 1.14 mmHg; p > 0.05), reflecting the partial distal aortic flow (Figures 3C and 3D). Following resuscitation, the proximal and distal MAPs increased in both groups and returned to the baseline following the balloon deflation for the remainder of the critical care phase (Figures 3A – 3D).
All animals experienced reflex tachycardia immediately following the hemorrhage, which underwent an incremental increase during the balloon inflation phase in both groups (Figure 4A). Following the balloon deflation, the HRs were significantly higher for the remainder of the critical care phase in the complete occlusion group compared to the partial occlusion group, although this difference in HR was not statistically significant.
Following the hemorrhage, the CVP decreased in both groups (Figure 4B). It underwent a rising trend following the balloon inflation. Following the balloon deflation, the complete occlusion group demonstrated a greater decrease in CVP compared to the partial occlusion group, although not statistically different. Following the additional resuscitation in the critical care phase, the CVP recovered toward the baseline in both groups. Similarly, the CO decreased following the hemorrhage, increased during the balloon inflation, and returned to the baseline following the balloon deflation and resuscitation for both groups (Figure 4C).
The carotid flow decreased in both groups immediately following the hemorrhage (Figure 4D). Following the balloon inflation, the complete occlusion group demonstrated higher carotid flow rates compared to the partial occlusion group. Following the resuscitation and balloon deflation, the carotid flow rate recovered toward the baseline in both groups. However, this carotid flow was lower in the complete occlusion group as compared to the partial occlusion group.
Laboratory Parameters:
No appreciable differences in the baseline pH and lactate level were noted between the groups. Following the balloon inflation, the animals in both groups experienced a decrease in pH (Figure 5A). The pH nadir in the complete occlusion group was notably lower than that in the partial occlusion group (complete: 7.14 ± 0.01, partial: 7.32 ± 0.02, p = 0.1). The lactate level was significantly higher throughout balloon inflation and the remainder of the critical care phase in the complete occlusion group (complete: 17.5 ± 0.71 mmol, partial: 6.1 ± 0.28 mmol, p = 0.03) (Figure 5B). This difference in lactate levels decreased slowly until the levels were similar at the end of the critical care phase.
Resuscitation Requirements:
The total fluid requirement for animals in the complete occlusion group was significantly higher than for the animals in the partial occlusion group (total additional fluid resuscitation for the animals in the complete occlusion group: 47.5 ± 3.4 cm3/kg, total additional fluid resuscitation for the animals in the partial occlusion group: 3.7 ± 0.4 cm3/kg, p = 0.003) (Figure 6A). Similarly, the norepinephrine requirement in the complete occlusion group was significantly higher than in the partial occlusion group (complete: 289.7 ± 25.4 µg/kg, partial: 32 ± 13.8 µg/kg, p = 0.006) (Figure 6B).

Figure 1: Aortic balloon occlusion catheters. (A) Partial aortic occlusion is achieved using a selective aortic balloon occlusion in trauma (SABOT) catheter, while complete aortic occlusion is achieved using the complete aortic balloon occlusion catheter. (B) The partial aortic balloon occlusion catheter is a two-balloon system that allows an intra-luminal blood flow providing a distal aortic flow. Complete aortic occlusion is provided using a single-balloon system. Please click here to view a larger version of this figure.
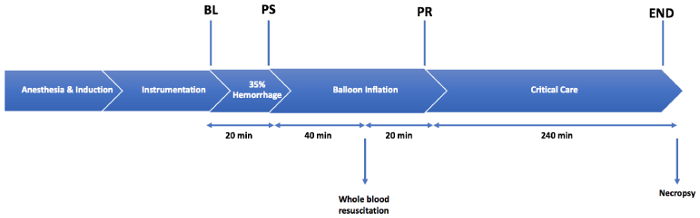
Figure 2: Injury protocol. An injury consisting of a 35% total blood volume hemorrhage is followed by a 1 h period of aortic balloon occlusion. A resuscitation is performed with 20% whole blood over 20 min, after 40 min of balloon occlusion. The animals are monitored in the critical care phase for 4 h following the balloon deflation. BL = the baseline; PS = post-shock; PR = post-resuscitation period. Please click here to view a larger version of this figure.
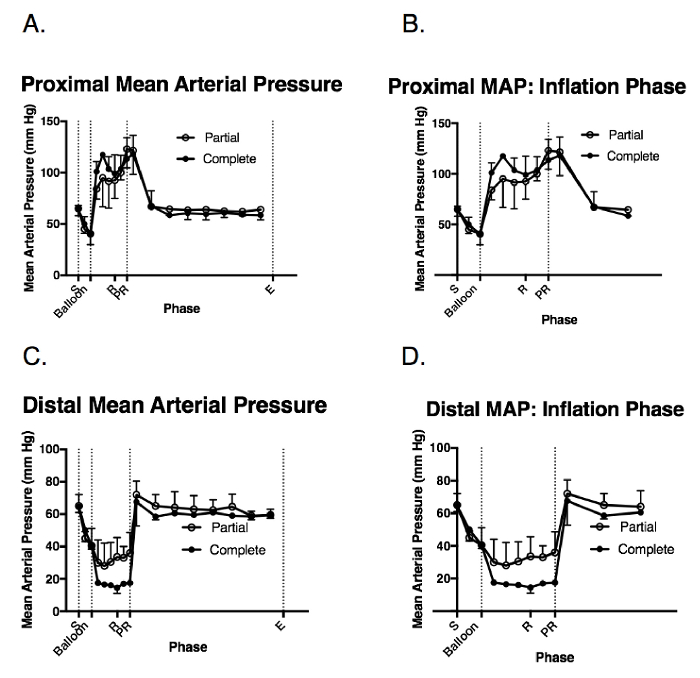
Figure 3: Hemodynamic response to the injury and balloon inflation. These panels show the intraoperative measurements of (A) the proximal pean arterial pressure (MAP), (B) the proximal MAP during the balloon inflation, (C) the distal MAP, and (D) the distal MAP during the balloon inflation. The data are presented as the group mean ± the standard error (SE). S = the shock period (20 min); Balloon = the balloon inflation (60 min); R = the resuscitation (20 min); PR = the post-resuscitation period/balloon deflation; E = the end of the injury phase (5 h following the shock period completion); Complete = the complete aortic balloon occlusion catheter; Partial = the partial aortic balloon occlusion catheter. Please click here to view a larger version of this figure.
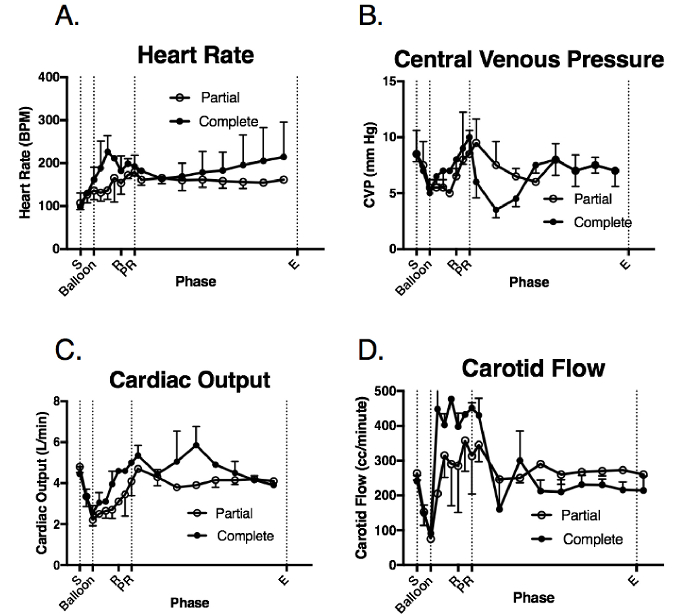
Figure 4: Systemic and physiologic response to the injury and balloon deployment. These panels show the intraoperative measurements of (A) the heart rate (HR), (B) the central venous pressure (CVP), (C) the cardiac output (CO), and (D) the carotid flow (CF). The data are presented as group mean ± SE. S = the shock period (20 min); Balloon = the balloon inflation (60 min); R = the resuscitation (20 min); PR = the post-resuscitation period/balloon deflation; E = the end of the injury phase (5 h following the shock period completion); Complete = the complete aortic balloon occlusion catheter; Partial = the partial aortic balloon occlusion catheter. Please click here to view a larger version of this figure.
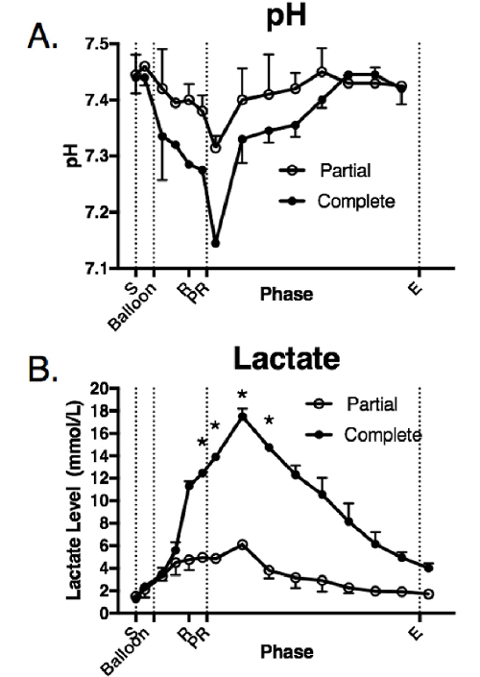
Figure 5: Laboratory parameters in response to the injury and balloon inflation. These panels show the intraoperative measurements of (A) pH and (B) lactate. The data are presented as group mean ± SE. The asterisks indicate the time points that were significantly different (p < 0.05). S = the shock period (20 min); Balloon = the balloon inflation (60 min); R = the resuscitation (20 min); PR = the post-resuscitation period/balloon deflation; E = the end of the injury phase (5 h following the shock period completion). Complete = the complete aortic balloon occlusion catheter; Partial = the partial aortic balloon occlusion catheter. Please click here to view a larger version of this figure.
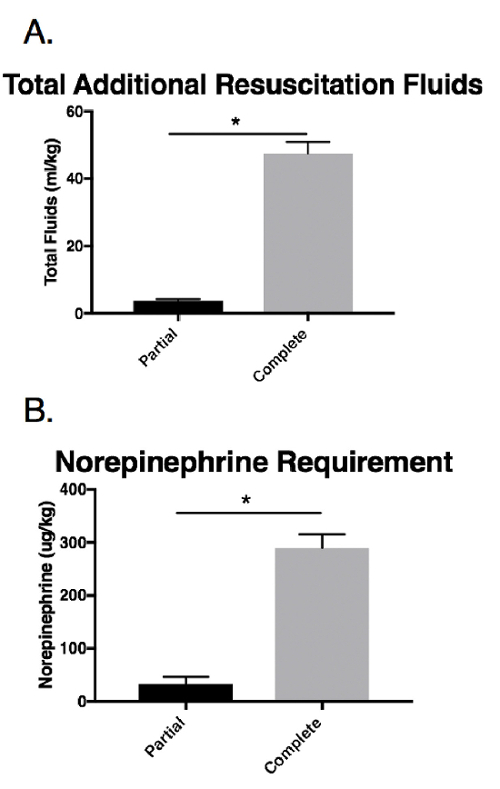
Figure 6: Resuscitation requirements in response to the injury and balloon inflation. These panels show the intraoperative measurements of (A) total additional resuscitation fluids and (B) the norepinephrine use. The data are presented as group mean ± SE. The asterisks indicate significant differences (p < 0.05). Complete = the complete aortic balloon occlusion catheter; Partial = the partial aortic balloon occlusion catheter. Please click here to view a larger version of this figure.
Discussion
In this protocol, we highlighted a hemorrhagic shock model in swine. This model has been shown to be both reliable and reproducible16,17,18,19. Models similar to this have been employed in several scientific studies investigating the effects of hemorrhagic shock on animal physiology16,20. Furthermore, this model has also been used for testing both pharmacologic and surgical treatment interventions in hemorrhagic shock with marked success12,13,16,19,21.
This model comprises several steps that require great attention to detail. The intubation of a swine is a complex procedure since the animal has a long, beak-like snout and a narrow, long oropharyngeal cavity. Additionally, swine generally have a high tendency to undergo laryngospasm, making orotracheal intubation even more challenging22. An appropriate induction of anesthesia, promoting good muscular relaxation, should be achieved before attempting the intubation. In our experience, having an assistant to use surgical cotton ropes to lift the mandible and tongue and depress the maxilla helps facilitate a wider opening of the oral cavity for easier intubation. If the intubation becomes difficult due to laryngospasm, topical xylocaine spray should be used. Although not utilized in this protocol, a neuromuscular blocker can be administered to promote laryngeal relaxation. Using a neuromuscular blocker, however, requires close supervision by a trained professional. We have also found it helpful to apply lubricating gel to the end of the endotracheal tube, as well as rotating the tube while advancing it through the laryngeal opening. Following the intubation, the endotracheal tube placement should be confirmed with end-tidal CO2. Nevertheless, although pigs can be intubated in various positions, we find intubation in the supine position to be the easiest, especially if the individual performing the intubation has experience with human intubation.
Femoral artery and vein cannulation can be technically challenging. The use of good retraction is important and can be achieved by using self-retaining retractors. An additional retractor, such as an Army-Navy, may be used if needed. Care should be taken while dissecting the neurovascular bundle, since the femoral nerve, which is the lateral-most structure in the bundle, has to be preserved. This is particularly important if the animal needs to survive the experiment. In addition, the cannulation of the right femoral artery is a critical step to the experiment. Following the guidewire cannulation into the vessel, a 14 Fr insertion sheath is inserted. A successful execution of this step requires an initial dilatation with a 10 Fr dilator to upsize the vessel. Also of prime importance is compressing the femoral artery at the site of arteriotomy, following the removal of the 10 Fr dilator, to minimize blood loss. Although not routinely performed in animal studies, gaining proximal and distal control before performing the arteriotomy and venotomy, as demonstrated in this study, can help minimize the bleeding and allow for troubleshooting, should problems arise during the cannulation.
An appropriate positioning and deployment of the aortic balloon occlusion catheters are critical. Caution should be practiced while advancing the catheter inside the aorta, as aggressive manipulation can result in an iatrogenic injury to the femoral artery or the aorta. Although several locations may be targeted for the deployment of the catheter, we chose to position the occlusion balloons in aortic Zone 1, which is the supraceliac aorta. The balloon positioning can be confirmed by manual palpation or fluoroscopy; however, ultrasonography can also be used to easily confirm the balloon placement, which was used for this study. Following the appropriate positioning, the balloon inflation should be conducted with care. In general, balloons should be inflated slowly until no further decrease in the distal MAP is noted. Overinflation of the balloon can potentially cause balloon rupture, which may precipitate an aortic injury. Close attention to the proximal and distal MAP helps to achieve the desired degree of aortic balloon occlusion, whether partial or complete.
Insertion sheaths and aortic balloon occlusion catheters have become smaller in profile in recent years. In this study, we used a 14 Fr insertion sheath before advancing the partial aortic balloon occlusion catheter (i.e., SABOT) into the femoral artery. Currently, this catheter is in phase I of its development, with plans for a future revision involving adjustable balloons and the distal aortic flow, as well as a smaller, low-profile system. Smaller 7 Fr catheters, however, have gained popularity in recent years, as they are associated with fewer ischemic complications. Smaller, low-profile sheaths and aortic balloon occlusion catheters may also be used for deployment in this hemorrhagic shock model, with excellent results.
Several models of hemorrhage are used to test hemorrhagic shock in large animals23,24,25. We employ a fixed-volume model of hemorrhage. In this model, a predetermined hemorrhage volume, which is based on a calculated TBV, is withdrawn from the body over a set period of time. We used a 35% TBV hemorrhage over 20 min, which is commonly used in fixed-volume hemorrhagic shock models26,27,28,29. This model is widely used to investigate shock-induced physiologic changes and compensatory mechanisms, as well as pathophysiologic responses, in hemorrhagic shock. Although this method is highly popular, the degree of shock that is induced as a result of the fixed-volume hemorrhage varies from animal to animal. Furthermore, as the blood-volume-to-body-weight ratio varies, it is important to control for weight in this model in order to achieve reproducible results. Other model types in practice include a fixed-pressure hemorrhage model, an uncontrolled hemorrhage model, and a hemorrhage model with ischemic markers as endpoints. Each of these models, however, has its own set of limitations.
Controlled hemorrhage models have been used to test aortic balloon occlusion catheters with success12. In this study, we used a closed hemorrhage system because this type of hemorrhage model can be employed in a wide variety of experiments. Our goal was to provide readers with the foundation to replicate a hemorrhagic shock model and to deploy aortic balloon occlusion catheters. However, to create the most clinically relevant and meaningful comparison of partial versus complete aortic occlusion, these catheters should ultimately be tested in the setting of an ongoing distal hemorrhage. In combination with other traumatic insults, this model of hemorrhagic shock can be extrapolated to a more clinically realistic model of traumatic injuries16,18.
Resuscitation strategies following traumatic injuries in animal models vary widely. While some are proponents of 'fluid responsiveness'-guiding requirements for ongoing resuscitation28, others propose objective thresholds for administering fluid boluses and vasopressors21,26. In this study, we employed thresholds to determine the fluid bolus administration and vasopressors use for their ease of reproducibility. Although 'fluid responsiveness' replicates clinical practice, objective thresholds for fluid administration and vasopressors may limit a wide variability and the subjectivity of resuscitation requirements in hemorrhagic shock models.
For years, swine have been used in various models of hemorrhagic shock that have provided opportunities to test a wide range of treatment strategies11,12,13,16,17,19,20,21,30. However, it is important to realize that swine are not the perfect animal model and physiologic changes do not exactly translate to humans. For example, some researchers may recommend splenectomy prior to the hemorrhagic shock to better mimic the human physiology, although this is controversial topic31.
In conclusion, this protocol demonstrates the basic foundation for replicating a hemorrhagic shock model in swine and for the deployment of aortic balloon occlusion catheters. The findings of a study that used a similar model of hemorrhagic shock are currently being used in Phase II clinical trials investigating the role of valproic acid (VPA) in traumatic injuries16,19,32,33,34. Also, to be noted is the importance of the role of aortic balloon occlusion catheters in the present era. Aortic balloon occlusion catheters have not only found an application in hemorrhagic shock, they are also being used in cardiac and vascular surgeries, as well as in high-risk elective surgical procedures where a control of the aortic flow is useful in an otherwise devastating circumstance. Overall, we feel that the swine model of hemorrhagic shock described and the aortic balloon occlusion are highly relevant and can be employed in a multitude of experimental studies.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We would like to acknowledge Rachel O'Connell, and Jessica Lee for their assistance with the animal studies. We would also like to acknowledge Maj. General Harold Timboe, MD, MPH, U.S. Army (Ret.), who has been an advisor and mentor for this project.
Materials
| Yorkshire-Landrace Swine | Michigan State University Veterinary Farm | ||
| Anesthesia: Telazol | Pfizer | Dose: 2-8 mg/kg; IM | |
| Anti-cholinergic: Atropine | Pfizer | Dose: 1mg, IM | |
| Anesthesia: Isoflurane | Baxter | Dose: 1-5%, INH | |
| Betadine | Humco | ||
| Alcohol 70% | Humco | NDC 0395-4202-28 | |
| Datex-Aespire Anesthesia Machine | GE Healthcare | 7900 | |
| Endotracheal tube | DEE Veterinary | 20170518 | Appropriate size for animal (6.5 or 7.0F) |
| Laryngoscope | Miller | 85-0045 | |
| Stylet | Hudson RCI | 5-151–1 | |
| Jelco 20G IV Catheter | Smiths Medical | 4054 | |
| Operating Room Monitor (Vital Signs Monitor) | SurgiVet Advisor | V9201 | May require at least 2 |
| Surgical Gowns | Kimberly Clark | 90142 | Use appropriate size for surgeon. |
| Sterile surgical gloves | Cardinal Health (Allegiance) | 22537-570 | Use appropriate size for surgeon. |
| Cautery Pencil | Medline | ESPB 2000 | |
| Suction tubing | Medline | DYND50251 | |
| Sunction tip: Yankauer | Medline | DYND50130 | |
| Bovie Aaron 1250 Electrocautery Unit | Bovie Medical Co. FL | BOV-A1250U | |
| Salpel Blade – Size #10 | Cardinal Health (Allegiance) | 32295-010 | |
| Scalpel Handle | Martin | 10-295-11 | |
| Debakey Forceps | Roboz | RS-7562 | |
| Weitlander Retractor | Roboz | RS-8612 | |
| Mayo Scissors | Roboz | RS-76870SC | |
| Army-navy Retractor | Teleflex | 164715 | |
| Mixter Right-angle Forceps | Teleflex | 175073 | |
| 5F (1.7 mm) 11 cm Insertion Sheath with 0.35" Guidewire | Boston Scientific | 16035-05B | |
| 8F (2.7 mm) 11 cm Insertion Sheath with 0.35'' Guidewire | Boston Scientific | 16035-08B | |
| 20G angled Introducer Needle | Arrow | AK-09903-S | |
| 14F (4.78 mm) 13 cm Insertion Sheath with 10F dilator | Cook Medical | G08024 | |
| 2-0 Silk 18'' 45 cm | Ethicon | A185H | |
| 3-0 Vicryl 36'' 90 cm | Ethicon | J344H | |
| 3-0 Nylon 18'' 45 cm | Ethicon | 663G | |
| 4-0 Prolene 30'' 75 cm | Ethicon | 8831H | |
| 20 ml syringe | Metronic/Covidien | 8881512878 | |
| 3 mL syringe | Metronic/Covidien | 1180300555 | |
| 6 mL syringe | Metronic/Covidien | 1180600777 | |
| 1000ml 0.9% Saline | Baxter | 2B1324X | |
| Foley Catheter (18F 30 cc) | Bard | 0166V18S | |
| Urinary Drainage Bag | Bard | 154002 | |
| 9F 10 cm Insertion Sheath | Arrow | AK-09903-S | |
| Swan-Ganz pulmonary artery catheter (8F) | Edwards Lifesciences co. CA | 746F8 | |
| Carotid Flow Probe System | Transonic, Ithaca, NY | 3, 4, or 6 mm probes | |
| SABOT catheter | Hayes Inc. | ||
| CODA balloon catheter | Cook Medical | 8379144 | |
| Ultrasound, M-Turbo | SonoSite | ||
| Amplatz Stiff Guidewire (0.035 inch, 260 cm) | Cook Medical | G03460 | |
| Arterial Blood Gas Syringes | Smiths Medical | 4041-2 | |
| Arterial Blood Gas Analyzer | Nova Biochemical | ABL800 | |
| Masterflex Pump | Cole Palmer | HV-77921-75 | |
| Blood Collection Bags | Terumo | 1BBD606A | |
| Macro IV drip set | Hospira | 12672-28 | |
| Pentobarbital | Pfizer | Dose: 100 mg/kg; IV | |
| Eppendorf Tubes | Sorenson | 11590 | |
| 50 cc conical tubes | Falcon | 352097 | |
| Formalin | Fisherbrand | 431121 | |
| Bair Hugger Normothermia System | Arizant Healthcare, Inc. |
Riferimenti
- Kauvar, D. S., Lefering, R., Wade, C. E. Impact of hemorrhage on trauma outcome: an overview of epidemiology, clinical presentations, and therapeutic considerations. The Journal of Trauma: Injury, Infection and Critical. 60, S3-S11 (2006).
- Kauvar, D. S., Wade, C. E. The epidemiology and modern management of traumatic hemorrhage: US and international perspectives. Critical Care. 9, S1-S9 (2005).
- Mattox, K. L., Allen, M. K., Feliciano, D. V. Laparotomy in the emergency department. Journal of the American College of Emergency Physicians. 8 (5), 180-183 (1979).
- Pust, G. D., Namias, N. Resuscitative thoracotomy. International Journal of Surgery. 33 (Pt B), 202-208 (2016).
- Burlew, C. C., et al. Trauma Association critical decisions in trauma: resuscitative thoracotomy. Journal of Trauma and Acute Care Surgery. 73 (6), 1359-1363 (2012).
- DuBose, J. J., et al. The AAST prospective Aortic Occlusion for Resuscitation in Trauma and Acute Care Surgery (AORTA) registry: Data on contemporary utilization and outcomes of aortic occlusion and resuscitative balloon occlusion of the aorta (REBOA). Journal of Trauma and Acute Care Surgery. 81 (3), 409-419 (2016).
- Biffl, W. L., Fox, C. J., Moore, E. E. The role of REBOA in the control of exsanguinating torso hemorrhage. Journal of Trauma and Acute Care Surgery. 78 (5), 1054-1058 (2015).
- Manzano Nunez, R., et al. A meta-analysis of resuscitative endovascular balloon occlusion of the aorta (REBOA) or open aortic cross-clamping by resuscitative thoracotomy in non-compressible torso hemorrhage patients. World Journal of Emergency Surgery. 12, 30 (2017).
- Gupta, B. K., et al. The role of intra-aortic balloon occlusion in penetrating abdominal trauma. Journal of Trauma. 29 (6), 861-865 (1989).
- Inoue, J., et al. Resuscitative endovascular balloon occlusion of the aorta might be dangerous in patients with severe torso trauma: A propensity score analysis. Journal of Trauma and Acute Care Surgery. 80 (4), 559-566 (2016).
- Russo, R. M., et al. Extending the golden hour: Partial resuscitative endovascular balloon occlusion of the aorta in a highly lethal swine liver injury model. Journal of Trauma and Acute Care Surgery. 80 (3), 378-380 (2016).
- Russo, R. M., et al. Partial Resuscitative Endovascular Balloon Occlusion of the Aorta in Swine Model of Hemorrhagic Shock. Journal of the American College of Surgeons. 223 (2), 359-368 (2016).
- Williams, T. K., et al. Extending resuscitative endovascular balloon occlusion of the aorta: Endovascular variable aortic control in a lethal model of hemorrhagic shock. The Journal of Trauma and Acute Care Surgery. 81 (2), 294-301 (2016).
- Hannon, J. P., Swindle, M. M. Hemorrhage and hemorrhagic-shock in swine: A review. Swine as Models in Biomedical Research. , 197-245 (1992).
- Garry, B. P., Bivens, H. E. The Seldinger technique. Journal of Cardiothorac Anesthesia. 2 (3), 403 (1988).
- Halaweish, I., et al. Addition of low-dose valproic acid to saline resuscitation provides neuroprotection and improves long-term outcomes in a large animal model of combined traumatic brain injury and hemorrhagic shock. The Journal of Trauma and Acute Care Surgery. 79 (6), 911-919 (2015).
- Alam, H. B., et al. Surviving blood loss without blood transfusion in a swine poly-trauma model. Surgery. 146 (2), 325-333 (2009).
- Jin, G., et al. Traumatic brain injury and hemorrhagic shock: evaluation of different resuscitation strategies in a large animal model of combined insults. Shock. 38 (1), 49-56 (2012).
- Nikolian, V. C., et al. Valproic acid decreases brain lesion size and improves neurologic recovery in swine subjected to traumatic brain injury, hemorrhagic shock, and polytrauma. The Journal of Trauma and Acute Care Surgery. 83 (6), 1066-1073 (2017).
- Langeland, H., Lyng, O., Aadahl, P., Skjaervold, N. K. The coherence of macrocirculation, microcirculation, and tissue metabolic response during nontraumatic hemorrhagic shock in swine. Physiological Reports. 5 (7), (2017).
- Johnson, M. A., et al. The effect of resuscitative endovascular balloon occlusion of the aorta, partial aortic occlusion and aggressive blood transfusion on traumatic brain injury in a swine multiple injuries model. Journal of Trauma Acute Care Surgery. 83 (1), 61-70 (2017).
- Theisen, M. M., et al. Ventral recumbency is crucial for fast and safe orotracheal intubation in laboratory swine. Laboratory Animals. 43 (1), 96-101 (2009).
- Li, Y., Alam, H. B. Modulation of acetylation: creating a pro-survival and anti-inflammatory phenotype in lethal hemorrhagic and septic shock. Journal of Biomedicine and Biotechnology. 2011, 523481 (2011).
- Nikolian, V. C., et al. Valproic acid decreases brain lesion size and improves neurologic recovery in swine subjected to traumatic brain injury, hemorrhagic shock, and polytrauma. The Journal of Trauma and Acute Care Surgery. 83 (6), 1066-1073 (2017).
- Dekker, S. E., et al. Normal saline influences coagulation and endothelial function after traumatic brain injury and hemorrhagic shock in pigs. Surgery. 156 (3), 556-563 (2014).
- Causey, M. W., McVay, D. P., Miller, S., Beekley, A., Martin, M. The efficacy of Combat Gauze in extreme physiologic conditions. The Journal of Surgical Research. 177 (2), 301-305 (2012).
- Frankel, D. A., et al. Physiologic response to hemorrhagic shock depends on rate and means of hemorrhage. The Journal of Surgical Research. 143 (2), 276-280 (2007).
- Morrison, J. J., et al. The inflammatory sequelae of aortic balloon occlusion in hemorrhagic shock. The Journal of Surgical Research. 191 (2), 423-431 (2014).
- White, J. M., et al. A porcine model for evaluating the management of noncompressible torso hemorrhage. Journal of Trauma. 71, S131-S138 (2011).
- Alam, H. B., et al. Putting life on hold-for how long? Profound hypothermic cardiopulmonary bypass in a Swine model of complex vascular injuries. Journal of Trauma. 64 (4), 912-922 (2008).
- Bebarta, V. S., Daheshia, M., Ross, J. D. The significance of splenectomy in experimental swine models of controlled hemorrhagic shock. The Journal of Trauma and Acute Care Surgery. 75 (5), 920 (2013).
- Georgoff, P. E., et al. Alterations in the human proteome following administration of valproic acid. Journal of Trauma and Acute Care Surgery. 81 (6), 1020-1027 (2016).
- Dekker, S. E., et al. Different resuscitation strategies and novel pharmacologic treatment with valproic acid in traumatic brain injury. Journal of Neuroscience Research. 96 (4), 711-719 (2017).
- Georgoff, P. E., et al. Safety and Tolerability of Intravenous Valproic Acid in Healthy Subjects: A Phase I Dose-Escalation Trial. Clinical Pharmacokinetics. 57 (2), 209-219 (2017).

