A Protocol to Set Up Needle-Free Connector with Positive Displacement on Central Venous Catheter in Intensive Care Unit
Summary
We present a protocol to show the installation of a needle-free connector with positive displacement on a central venous catheter.
Abstract
Needle-free connectors were initially designed and promoted to avoid blood exposure for healthcare workers. Some recent data suggest that the latest generation of connectors (with positive displacement) may be of interest for reducing central venous line infections. We have been using needle-free connectors for several years in our intensive care unit and here we present a protocol for installing these connectors on central venous catheters. After insertion of the catheter and control of the permeability of the lines, the connectors must be purged with 0.9% NaCl before being connected. The connectors replace all disposable caps used on infusion stopcocks and manifolds. All the connectors are changed every 7 days as recommended by the manufacturer (except when there is macroscopic contamination, which requires an immediate change of the connector). Before each injection, the connector must be disinfected for at least 3 seconds with 70% isopropyl alcohol. The connectors must not be disconnected (unless changed), as the injection is done through the device. Setting up the connectors slightly increases the total time required to place the catheter and there is no formal evidence that these connectors reduce the incidence of infectious or thrombotic complications. However, these devices simplify the management of central venous lines and prevent the catheter circuit from "opening" once it has been sterilely installed.
Introduction
Central venous catheter-related infections (CRI) are a severe complication of central venous catheters in intensive care unit (ICU). The decline in CRI remains an ever-present objective, with a final goal of "zero catheter related infection"1. Needle-free connectors were initially designed and promoted to avoid blood exposure for healthcare workers. There are two main designs of connectors: split septum (no internal moving parts) and closed valve systems (internal moving components) but both designs can be combined in one connector2. Needle-free connectors are categorized according to the type of fluid displacement that occurs after disconnection of a male Luer valve: negative (blood reflux into the catheter), neutral, and positive (with a push of blood out of the catheter lumen)2,3.
Some connectors have been described as a cause of catheter-related infections, in particular in the intensive care unit (ICU)4,5,6. A new generation of needle-free connectors with minimal internal complexity, a reduction or elimination of interstitial or dead space, a visible fluid path to help assess proper flushing technique, and a flat access surface, etc. has been designed to lower the risk of infection. In vitro, these connectors have shown low bacterial colonization7. There are global recommendations from the laboratory manufacturing these connectors; however, there is no practical description of how to install them on catheters8. Hence, it is possible that each team uses them differently. Therefore, we propose a formalized protocol for the installation of these connectors on central venous lines in the ICU.
We present the installation of a positive pressure needleless connector (PPNC) with an internal silicone piston in our ICU but this protocol is applicable with any positive displacement valve. This valve is a mechanical needle-free connector with positive displacement.
Protocol
1. Preparation of connectors and infusion lines
- Sterilely retrieve the connectors.
- On the 3-way extension stopcock, screw 1 connector into each socket of the stopcock and 1 connector into the end of the extension line. Take 0.9% NaCl with a 50 mL syringe to purge the extension and the lines. Purge each 3-way extension with 0.9% NaCl through the 2 connectors of the stopcock.
- Take the infusion manifold. Unscrew each single-use cap. Screw 1 connector into each socket of the manifold to replace the caps. Purge the infusion line through each connector by turning each stopcock sequentially.
NOTE: At the end of this step, there are 3 extensions with 3-way stopcock with purged connectors and an infusion manifold with purged connectors.
2. Placement of the catheter
- Place the central venous catheter sterilely according to usual practice in the unit or a previously described protocol9.
- Check the permeability of each line by aspiration of blood and then reinjection of 0.9% NaCl serum. Clamp the lines.
3. Installation of the connectors
- Screw the extension’s connector of each 3-way extension stopcock into each catheter line. These connectors, directly connected to the lines of the catheter, are the "proximal connectors". Unclamp the lines.
- Connect the infusion manifold line to one of the connectors of the 3-way stopcock of the distal line.
NOTE: There is no need to place a vein guard on the manifold or extensions.
4. Use of connectors and infusion lines
- Before each infusion, disinfect the end of the connector for 3 s with a sterile compress soaked in 70% isopropyl alcohol. Connect the syringe or tubing directly to the connector by screwing and injecting. After injection or infusion, unscrew the device. Do not remove the connector.
- If needed, rinse an unused line with 3 mL of saline. There is no need to clamp or infuse an unused line with a vein guard. Leave the proximal valve in place even if the line is not perfused.
5. Replacement and maintenance of connectors and infusion lines
- Always change the connectors every 7 days except for the proximal connectors. Thus, every week, the nurse prepares the extensions with 3-way stopcock and the infusion manifold in sterile conditions (see step 1).
- Unscrew the used extensions at the proximal valve and screw the new sterile and purged extensions to the catheter on the proximal connector (see step 3).
- Only change a proximal connector if it is soiled (to maintain the catheter in a "closed" system).
- In case of macroscopic contamination, rinse the connector with 10 mL of 0.9% NaCl. If there is still contamination, replace the connector. In the event of transfusion, infusion of lipid solution (e.g. propofol) or parenteral nutrition, change the tubing and connectors of the line concerned every 24 hours.
- Change other infusion lines and tubing according to the practices and protocols of each unit.
Representative Results
Once all the elements are in place, the catheter has connectors on almost all the junctions between two infusion lines (Figure 1). Thus, it has proximal connectors on each line and two connectors on the sockets of the 3-way extensions (Figure 2). Each infusion line has connectors at all its female sockets (Figure 3). Once the assembly is in place, any injection or infusion (continuous or discontinuous) must be made, after disinfection, through one of the connectors of the infusion lines or extensions (Figure 4).
Maintaining the connectors in place keeps the infusion system closed as well as sterile mounted and minimizes the risk of bacterial contamination. One of our previous works, the first study that prospectively analyzed these devices over several years, showed a significant decrease in CRI incidence during the six-year period framing the introduction of the connectors (Figure 5)10. In this work, the incidence of CRI before using connectors was 6.2 CRI/1000 catheter-day vs. 2.7 CRI/1000 catheter-days after using connectors10. Moreover, we did not find any increase of CRI after the beginning of connector use (Figure 5) and there was no significant difference concerning the kind of bacteria species involved in CRI10. We have not identified any specific complications associated with the use of these connectors during these years of use.
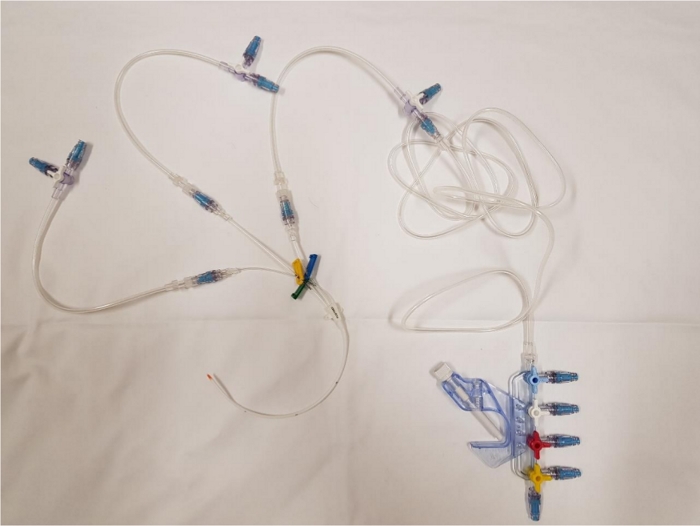
Figure 1: Global view of the catheter once the connectors have been placed. Please click here to view a larger version of this figure.
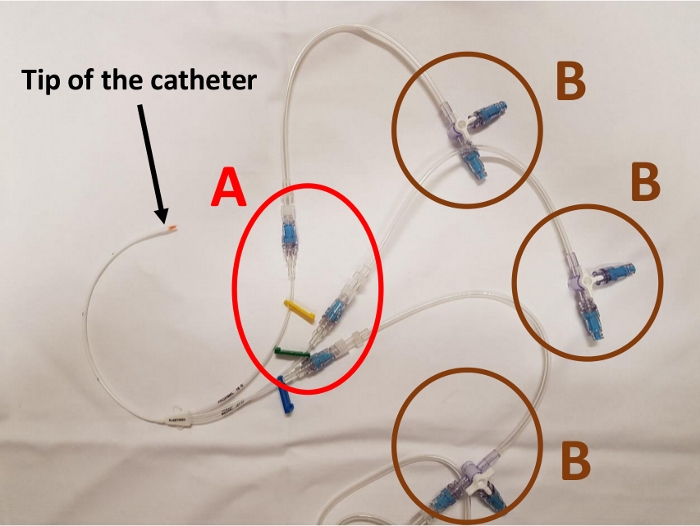
Figure 2: Proximal part of the catheter with proximal connectors (A) and connectors on the 3-way extension stopcock (B). Please click here to view a larger version of this figure.
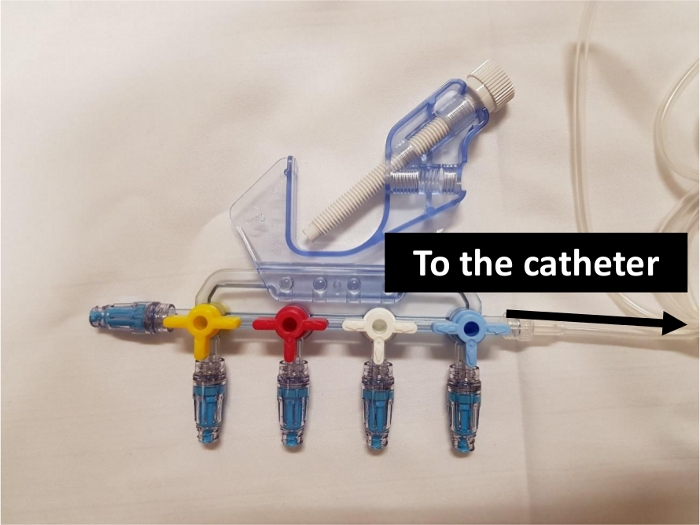
Figure 3: 4-port manifold with connectors. Please click here to view a larger version of this figure.
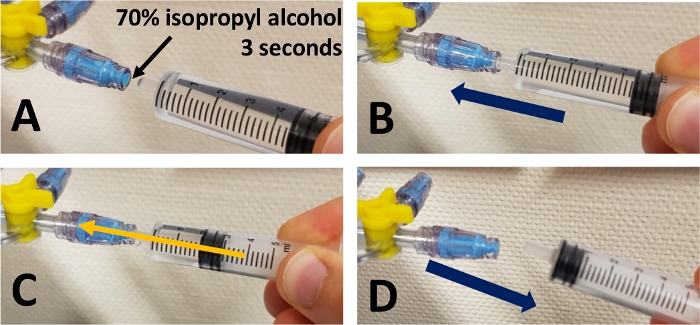
Figure 4: Steps of injection through a connector. (A) Disinfect the surface of the connector for 3 seconds with 70% isopropyl alcohol. Wait until the connector is dry. (B) Insert the tip of the syringe or infuser into the connector. (C) Inject or infuse the medication. (D) Remove the syringe or infuser. Please click here to view a larger version of this figure.
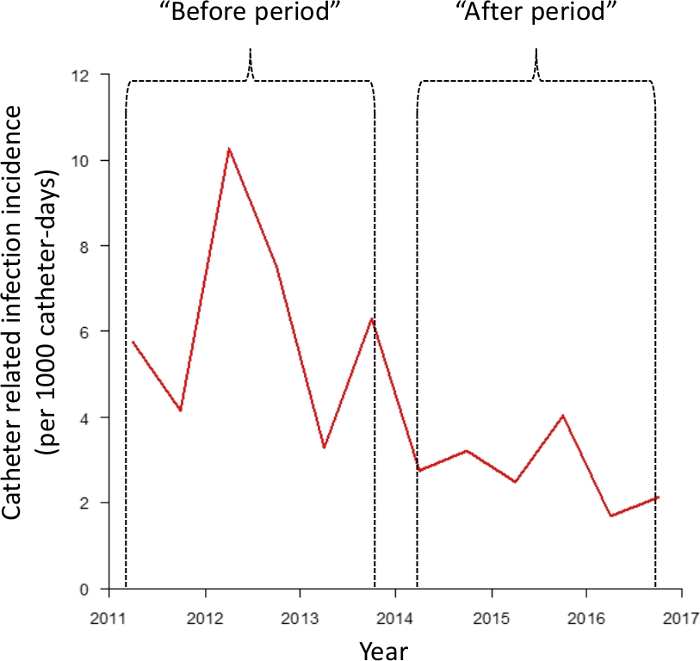
Figure 5: Evolution of catheter-related infection incidence before and after the use of connectors. Figure taken from Clavier et al.10 Please click here to view a larger version of this figure.
Discussion
Setting up the connectors slightly increases the total time required to place the catheter. However, their use has several advantages: no need to maintain a continuous saline perfusion for non-infused lines, rapid stopping of infusion of the lines if necessary (in case of urgent transport of the patient for example), no need to use single-use caps several times a day to close the infusion lines. The use of connectors simplifies the daily use of catheter infusion lines without any particular complications. It should be noted that during our four year experience of these PPNC connectors there have been no complications with rapid fluid administration while this has been described with other types of connectors11. It is essential to maintain complete asepsis when installing the connectors in order to keep the infusion system closed. In addition, regular examination of the connectors for contamination is essential to be able to rinse or change the implicated connectors and avoid bacterial growth. A recent work reports the in vitro efficacy of a pulsative flushing technique to prevent bacterial colonization of vascular access devices but this technique has not been evaluated directly on needleless connectors12. Due to the technical aspect of this kind of connector, nurses must be trained before using these connectors in everyday practice. In this context, the use of Aseptic Non Touch Technique, which is a safe method for managing intravascular devices, is very relevant13. Thus, even after the connectors are set up, direct contact with the connectors should be minimized as much as possible to minimize the risk of contamination. In this context, the use of a disinfectant cap on needleless connectors can help to limit the contact with connectors and is effective in reducing central line-associated bloodstream infections14.
The inclusion of extension tubing, stopcocks and manifolds increase the complexity of the system and expose it to a risk of contamination of stopcocks and manifolds15. Our choice of extensions and infusion lines is the result of a reflection on the balance between patient safety and infectious risk. In the ICU, continuous uninterrupted infusion of certain drugs is essential (e.g., catecholamine, sedation). In our protocol, the use of a 3-way extension stopcock allows medication relays to be easily made without ever interrupting the infusion, which is a guarantee of safety for the patient. The use of a unique 4-port manifold with a perfusion line allows the infusion of several drugs simultaneously (e.g., antibiotics, analgesics, insulin) but the number of these manifolds should be limited to prevent the risk of infection.
Some studies have shown interest in a closed system to prevent colonization and catheter infections16,17. Additionally, several studies have reported a decrease in catheter infection after using PPNCs10,18,19. It is also possible that these connectors may decrease thrombotic catheter occlusions, but there is insufficient evidence to conclude a direct causal link between needleless connectors and catheter occlusions20,21. Moreover, data in the literature support the safe and prolonged use of these devices10,16,19. However, different department may sometimes use different methods to mount connectors on infusion lines and most studies do not describe the method used to install connectors on catheters. It could appear interesting that studies on connectors report their installation protocol to better assess potential differences of practice between teams.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The authors are grateful to Nikki Sabourin-Gibbs, Rouen University Hospital, for her help in editing the manuscript. The authors would like to thank the communication department of Rouen University Hospital and the teams of the Medical Training Center for their participation in the video shoot.
Materials
| BD MaxZero™ needle-free connector | Becton Dickinson | MZ1000-07 | we present the installation of the connector with MaxZero but this protocol is applicable with any positive displacement valve. |
| 4-port mannifold with PE/PVC extension | Cair-LGL | RPB4310A | |
| PE/PVC extension line with 3-way stopcock | Cair-LGL | PE3302M | |
| NaCl 0.9% 250 ml | Baxter | 2B1322 | |
| BD Plastipak™ 50mL Luer-Lock Syringe | BECTON DICKINSON MEDICAL | 613-3925 |
Riferimenti
- Worth, L. J., McLaws, M. L. Is it possible to achieve a target of zero central line associated bloodstream infections. Current Opinion in Infectious Diseases. 25 (6), 650-657 (2012).
- Kelly, L. J., Jones, T., Kirkham, S. Needle-free devices: keeping the system closed. British Journal of Nursing (Mark Allen Publishing). 26 (2), 14-19 (2017).
- Casey, A. L., Karpanen, T. J., Nightingale, P., Elliott, T. S. The risk of microbial contamination associated with six different needle-free connectors. British Journal of Nursing (Mark Allen Publishing). 27 (2), 18-26 (2018).
- Salgado, C. D., Chinnes, L., Paczesny, T. H., Cantey, J. R. Increased rate of catheter-related bloodstream infection associated with use of a needleless mechanical valve device at a long-term acute care hospital. Infection Control and Hospital Epidemiology. 28 (6), 684-688 (2007).
- Jarvis, W. R. Health care-associated bloodstream infections associated with negative- or positive-pressure or displacement mechanical valve needleless connectors. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America. 49 (12), 1821-1827 (2009).
- Btaiche, I. F., Kovacevich, D. S., Khalidi, N., Papke, L. F. The effects of needleless connectors on catheter-related bloodstream infections. American Journal of Infection Control. 39 (4), 277-283 (2011).
- Chernecky, C., Waller, J. Comparative evaluation of five needleless intravenous connectors. Journal of Advanced Nursing. 67 (7), 1601-1613 (2011).
- . Infusion Resource Library – BD Available from: https://www.bd.com/en-us/offerings/capabilities/infusion/infusion-resource-library?contenttype=22_productline=115 (2019)
- Kim, S. C., Klebach, C., Heinze, I., Hoeft, A., Baumgarten, G., Weber, S. The supraclavicular fossa ultrasound view for central venous catheter placement and catheter change over guidewire. Journal of Visualized Experiments. (94), 52160 (2014).
- Clavier, T., et al. Impact of MaxZeroTM needle-free connector on the incidence of central venous catheter-related infections in surgical intensive care unit. Australian Critical Care: Official Journal of the Confederation of Australian Critical Care Nurses. , (2018).
- Lehn, R. A., Gross, J. B., McIsaac, J. H., Gipson, K. E. Needleless connectors substantially reduce flow of crystalloid and red blood cells during rapid infusion. Anesthesia and Analgesia. 120 (4), 801-804 (2015).
- Ferroni, A., et al. Pulsative flushing as a strategy to prevent bacterial colonization of vascular access devices. Medical Devices (Auckland, N.Z). 7, 379-383 (2014).
- Flynn, J. M., Keogh, S. J., Gavin, N. C. Sterile v aseptic non-touch technique for needle-less connector care on central venous access devices in a bone marrow transplant population: A comparative study. European Journal of Oncology Nursing: The Official Journal of European Oncology Nursing Society. 19 (6), 694-700 (2015).
- Merrill, K. C., Sumner, S., Linford, L., Taylor, C., Macintosh, C. Impact of universal disinfectant cap implementation on central line-associated bloodstream infections. American Journal of Infection Control. 42 (12), 1274-1277 (2014).
- Mermel, L. A., Bert, A., Chapin, K. C., LeBlanc, L. Intraoperative stopcock and manifold colonization of newly inserted peripheral intravenous catheters. Infection Control and Hospital Epidemiology. 35 (9), 1187-1189 (2014).
- Rosenthal, V. D., Maki, D. G. Prospective study of the impact of open and closed infusion systems on rates of central venous catheter-associated bacteremia. American Journal of Infection Control. 32 (3), 135-141 (2004).
- Bouza, E., et al. A needleless closed system device (CLAVE) protects from intravascular catheter tip and hub colonization: a prospective randomized study. The Journal of Hospital Infection. 54 (4), 279-287 (2003).
- Tabak, Y. P., Jarvis, W. R., Sun, X., Crosby, C. T., Johannes, R. S. Meta-analysis on central line-associated bloodstream infections associated with a needleless intravenous connector with a new engineering design. American Journal of Infection Control. 42 (12), 1278-1284 (2014).
- Wallace, M. C., Macy, D. L. Reduction of Central Line-Associated Bloodstream Infection Rates in Patients in the Adult Intensive Care Unit. Journal of Infusion Nursing: The Official Publication of the Infusion Nurses Society. 39 (1), 47-55 (2016).
- Btaiche, I. F., Kovacevich, D. S., Khalidi, N., Papke, L. F. The effects of needleless connectors on catheter-related thrombotic occlusions. Journal of Infusion Nursing: The Official Publication of the Infusion Nurses Society. 34 (2), 89-96 (2011).
- Williams, A. Catheter Occlusion in Home Infusion: The Influence of Needleless Connector Design on Central Catheter Occlusion. Journal of Infusion Nursing: The Official Publication of the Infusion Nurses Society. 41 (1), 52-57 (2018).

