Blind Endotracheal Intubation in Neonatal Rabbits
Summary
We describe a technique of endotracheal intubation in newborn rabbits after esophageal catheterization with a gastric tube.
Abstract
The newborn rabbit is a useful animal model for various pathologies and procedures. Airway management of the rabbit is complex due to its anatomical characteristics, which is further complicated in the case of the newborn. Of the different methods of advanced airway management, endotracheal intubation is less aggressive than tracheostomy, and is more feasible than supraglottic management given the lack of supraglottic devices of such a small size. As direct glottis visualization is very difficult in animals this size, this blind intubation model is presented as an effective alternative, especially for experiments requiring prolonged anesthesia. Using this method, we performed blind intubations with a 90% success rate.
Introduction
The peculiar anatomy of the rabbit's respiratory system makes endotracheal intubation complicated. The oral cavity is deep and narrow with a limited range of mouth opening and a relatively large tongue, making direct visualization of the larynx difficult even with the use of a laryngoscope1,2. Furthermore, intubation of the newborn rabbit, which is 10-20 times smaller than the adult, is even more of a challenging procedure. In addition to these anatomical characteristics, rabbits are prone to laryngospasm secondary to manipulation of the airway.
Several intubation techniques have been described in the literature, such as laryngoscope tracheal intubation, blind intubation, otoscope intubation, retrograde intubation, endoscope intubation, tracheostomy and supraglottic device ventilation3,4,5,6,7,8,9. All these techniques have been carried out successfully in adult rabbits; however, there are few publications regarding advanced airway management in the newborn rabbit10. One study describes performing tracheostomies in animals of this age to assess breathing mechanics and lung volumes, though this is an aggressive and irreversible technique11. This method for endotracheal intubation has proven to be an effective and low-cost method of airway management in newborn rabbits.
Protocol
The intubation procedure outlined below was approved by the Barcelona University Institutional Animal Care and Use Committee (CEEA 180/19)
1. Experimental preparation
- Obtain the following materials: 2 mm diameter polyvinyl chloride (PVC) endotracheal tube, 6 French (Fr) polyurethane feeding tube, flat and wide tip dissection forceps and sterile lubricant (Figure 1).
- During the intubation and surgery processes, maintain sterile conditions.
2. Anesthesia
- Anesthetize the animal with 5 mg/kg xylazine and 25 mg/kg ketamine applied together intramuscularly in the vastus lateralis of the femoral quadriceps. Alpha-2 agonists such as dexmedetomidine can also be used instead of xylazine.
- Allow 3-5 minutes for the medication to reach effect.
- Readminister the same dose until deep anesthesia is achieved (negative pedal, palpebral and corneal reflexes). Ideally, the rabbit will maintain spontaneous breathing until intubation.
3. Endotracheal intubation
- Place the rabbit on a thermal blanket at 37 °C. Place the animal in dorsal recumbency with the head in hyperextension. Align the head as straight and midline as possible. A four-centimeter roll made of sterile gauze placed in the dorsal region of the neck may be helpful to achieve at least 45° of extension (Figure 2).
- Use flat and wide tip dissection forceps to open the oral cavity approximately one centimeter wide. Use forceps with a blunt and wide end to avoid mouth trauma.
- Measure the distance between mouth and the region of stomach (first rib). Introduce a lubricated 6 French (Fr) polyurethane gastric tube to the pre-measured mark (Figure 3-4).
- Move the feeding tube to the left and fix with adhesive tape to the surgical field.
- Introduce the 2 mm diameter PVC endotracheal tube, covered with sterile lubricant to the 3 cm mark.
- If there are any difficulties in advancing the tube, move it completely and reattempt until a smooth glide is achieved. The risk of trauma to the larynx and epiglottis is significant with multiple intubation attempts. Fix the tube with adhesive tape to the surgical field (Figure 5-6).
- Connect the endotracheal tube to the ventilator with following settings: Tidal volume – 10 mL/kg; respiratory rate: 40 breaths/min; FiO2 100%, PEEP 6.
- Confirm the correct position of the tube by the detection of CO2 in exhaled air (ETCO2), direct visualization of thoracic expansion and fogging of the tube. Monitoring of the animal by pulse oximetry and lung auscultation can also be helpful (Figure 7).
- At the conclusion of the experiment, use a standardized euthanasia protocol that consists of intravenous administration of pentobarbital 120-240 mg/kg. Repeat doses until breath cessation, reflex absence and asystole are acheived.
NOTE: In the surgical experiments, the animal is alive during the ventilation process and finally is euthanized.
Representative Results
In the laboratory, this technique is carried out by medical personnel with experience in experimentation with New Zealand rabbits and expertise in pediatric airway management. The procedure has been performed by one investigator in 10 animals with a global success rate of 90%. Previously, another researcher had practiced optimizing both the position of the animal and the correct tube fixation length. None of the animals were intubated in esophagus. Only one attempt (10%) failed due to the extremely small size of the animal (Table 1). Once blind intubation has been practiced, success is achieved on the first attempt in most cases, even if the procedure is performed by personnel not previously trained in rabbit airway management. We achieved a 100% intubation success rate in one or two attempts in animals weighing equal to or greater than 120 g.
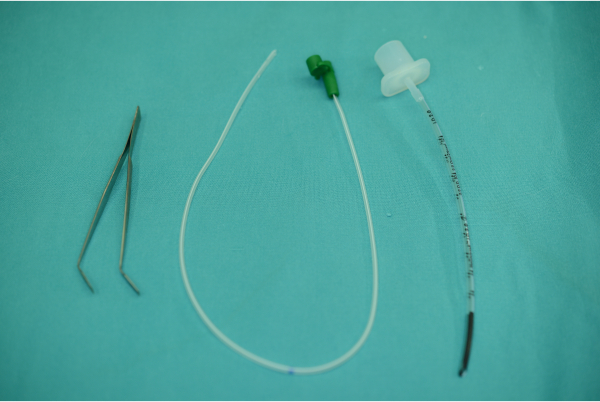
Figure 1: Materials needed for the experiment: Dissection forceps, feeding tube, endotracheal tube, sterile lubricant. Please click here to view a larger version of this figure.

Figure 2: Position of the animal before intubation: Dorsal recumbency and mild hyperextension. Please click here to view a larger version of this figure.
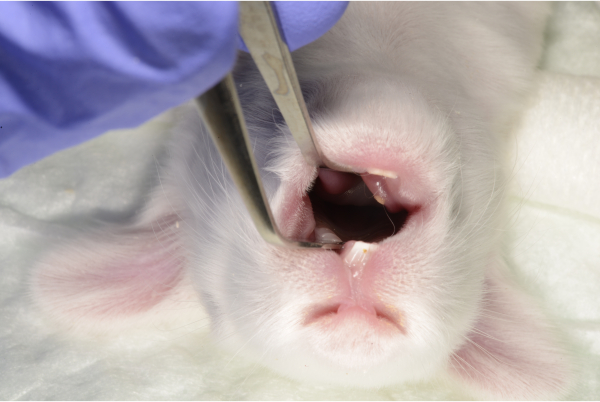
Figure 3: Mouth opening with dissection forceps. Please click here to view a larger version of this figure.
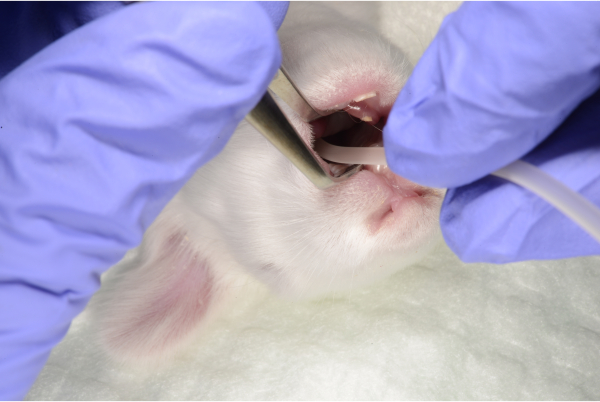
Figure 4: The feeding tube is introduced into the stomach. Please click here to view a larger version of this figure.
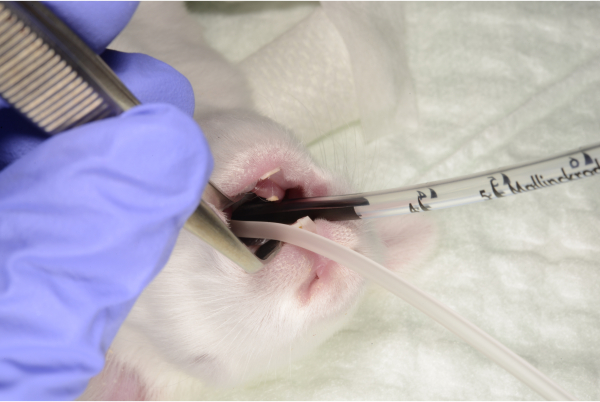
Figure 5-6: The endotracheal tube is introduced 3 cm blindly and fixed, along with the feeding tube, to the surgical field. Please click here to view a larger version of this figure.
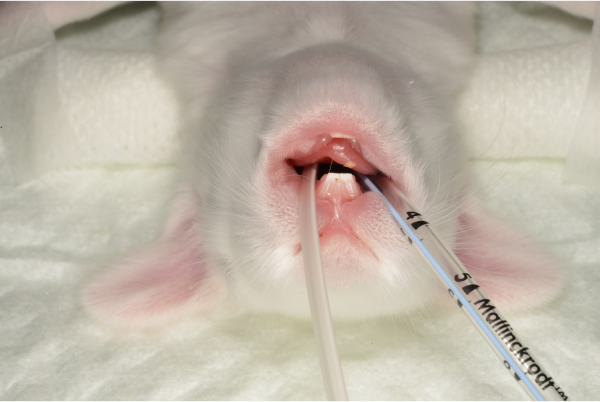
Figure 6: The fixed endotracheal tube and feeding tube. Please click here to view a larger version of this figure.

Figure 7: The animal is connected to the ventilator. Please click here to view a larger version of this figure.
| ANIMAL NUMBER | WEIGHT (g) | SUCCESS | ATTEMPTS | DEATH |
| 1 | 150 | + | 1 | – |
| 2 | 125 | + | 2 | – |
| 3 | 180 | + | 1 | – |
| 4 | 210 | + | 1 | – |
| 5 | 120 | + | 2 | – |
| 6 | 140 | + | 2 | – |
| 7 | 200 | + | 1 | – |
| 8 | 190 | + | 1 | – |
| 9 | 180 | + | 1 | – |
| 10 | 110 | – | 3 | + |
| TOTAL | 160.5 | 90% | 1.5 | 10% |
Table 1: Animal sample features and endotracheal intubation attempts.
Discussion
Rabbits are widely used in animal experimentation due to their availability, high reproduction rate with short intergenerational times, and easy handling2. In our center, we use 5-7-day old New Zealand rabbits (Oryctolagus cuniculus) as a neonatal model to study the neurologic repercussions of extracorporeal surgery on the developing brain. As a result, we utilize procedures that require deep anesthesia and must use endotracheal intubation for periods of time up to an hour.
In the adult rabbit, several airway management techniques have been described, usually in a surgical context. When we tried to extrapolate these techniques to the rabbit kits, several problems arose. The rabbits weigh between 110 and 210 grams and have a maximum mouth opening range of 1 cm. These characteristics make direct visualization of the larynx difficult and it is complicated to find endoscopic material small enough. Another option described in the literature is to place an otoscope in the oral cavity and use it as a guide to introduce the PVC tube7. In newborn rabbits, both the presence of the tongue and the small size of the mouth make direct visualization of the glottis difficult. In addition, there are no supraglottic devices of adequate dimensions for newborns.
Due to the circumstances mentioned above, there are two fundamental mechanisms to secure the airway of the very small rabbit: tracheostomy and blind intubation. Tracheostomy is an invasive approach that has been used in both adult and newborn rabbits10, 12. It has the advantage of direct tracheal visualization, allowing the proceduralist to identify the exact location of the endotracheal tube. However, there is a high risk of mechanical complications and infection of the incision area, in addition to a decreased rate of survival of the animals once the procedures are completed13. Endoscopic/fiberoptic intubation is a method that would allow for direct visualization without surgery5. Another option to prevent laryngeal trauma is the use of a tight-fitting face mask, which has some additional drawbacks such as suboptimal ventilation and gastric distension.
In the laboratory, blind intubation is used to manage newborn rabbit airways. This method has been described in the adult rabbit as a feasible technique with high success index6. In the initial experiments, we tried to perform this technique without occlusion of the digestive tract. The result was a high rate of introduction of the endotracheal tube into the esophagus. With the insertion of the gastric tube prior to intubation, however, successful intubation was achieved in 90% of cases. Regarding the size of the endotracheal tube, we initially tried to use peripheral venous catheters up to 14 gauge in diameter, observing difficulties in both insertion (greater rigidity) and ventilation (diameter too small and difficulty connecting to the ventilator). In our experience, the use of 2 mm internal diameter endotracheal tubes introduced to a depth no more than 3 cm has been the most effective in ventilating small rabbits, shown by correct gasometric and ETCO2 values. While intubation in adult rabbits generally requires hyperextension of the head to essentially a 90° angle to the horizontal surface of the table, we were able to perform the procedure with only a mild head extension.
One of the important elements to be considered when performing this procedure is the friable nature of the newborn rabbit trachea. Keeping this is mind, we introduce the pre-lubricated tube slowly and carefully to avoid tracheal perforation. We recommend not inserting the tube further than 3 cm, as this was found to be the optimal length during practice attempts, and halting the intubation if the proceduralist encounters any resistance; in this situation, it is preferable to remove the tube and start over. We experienced one case of likely tracheal perforation in the smallest animal in the cohort.
The main limitation of this technique is the difficulty in detecting the correct position of the endotracheal tube once introduced. In our experience, the advancement of the device without resistance, the detection of appropriate ETCO2, the fogging of the tube and visualization of thoracic expansion during ventilation, as well as the maintenance of vital signs have been sufficient to corroborate the suitability of the procedure in all cases. In some of the experimental subjects, arterial gasometries were performed during ventilation showing adequate levels of carbon dioxide and oxygen concentration. In addition, though histopathology analysis was not performed, previously published literature has described mucosal injury to the trachea in the absence of clinical changes secondary to intubation14.
In conclusion, the model of intubation in newborn rabbits we have described is a rapid technique that uses basic supplies and allows for effective ventilation. We believe this technique can be incorporated in the armamentarium of airway management of neonatal rabbits.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the Instituto de Salud Carlos III [PFIS2017-0224], "Beca Ruza" from the Sociedad Española de Cuidados Intensivos Pediátricos and the Vanderbilt Medical Scholars Program.
The sponsors were not involved in the collection, analysis, or interpretation of the data.
We would like to thank Isabel Salas for the help provided within the animal laboratory and the Mr. Carles Fábrega and Ms. Gemma Fernandez-Asensio from the Audio-Visual Techincal Team from Sant Joan de Déu Hospital for their technical support with the video recording and production.
Materials
| 2 mm diameter Polyvinyl Chloride (PVC) endotracheal tube | Vygon | 520.20 | |
| 6 French polyurethane feeding tube | Vygon | 310.06 | |
| Anesthesia (Ketamine and Xylazine) | |||
| Multiparameter monitor Intellivue MP5 | Phillips | ||
| Plain and wide tip dissection forceps | LensforVision | AK005 | |
| Sterile lubricant (Silkospray) | Rush | ||
| Ventilator Servo 900c | Maquet |
References
- Gilroy, B. A. Endotracheal intubation of rabbits and rodents. Journal of the American Veterinary Medical Association. 179 (11), 1295 (1981).
- Graham, J. Common procedures in rabbits. The Veterinary Clinics of North America. Exotic Animal Practice. 9 (2), 367-388 (2006).
- Fick, T. E., Schalm, S. W. A simple technique for endotracheal intubation in rabbits. Laboratory Animals. 21 (3), 265-266 (1987).
- Conlon, K. C., et al. Atraumatic endotracheal intubation in small rabbits. Laboratory Animal Science. 40 (2), 221-222 (1990).
- Johnson, D. H. Endoscopic intubation of exotic companion mammals. The Veterinary Clinics of North America. Exotic Animal Practice. 13 (2), 273-289 (2010).
- Falcão, S. C., et al. Technique of blind tracheal intubation in rabbits (Oryctolagus cuniculi) supported by previous maneuver of esophageal cannulization. Acta Cirurgica Brasileira. 26 (5), 352-356 (2011).
- Miura, H., et al. Endotracheal intubation in rabbits. The Japanese Journal of Veterinary Anesthesiology. 19 (1), 25-29 (2012).
- Thompson, K. L., et al. Endotracheal Intubation of Rabbits Using a Polypropylene Guide Catheter. Journal of Visualized Experiments. (129), (2017).
- Comolli, J., et al. Comparison of endoscopic endotracheal intubation and the v-gel supraglottic airway device for spontaneously ventilating New Zealand white rabbits undergoing ovariohysterectomy. Veterinary Record. , 1-8 (2020).
- Hua, S., et al. Effects of different ventilation strategies on lung injury in newborn rabbits. Pediatric Pulmonology. 47 (11), 1103-1112 (2012).
- Kitchen, M. J., et al. The role of lung inflation and sodium transport in airway liquid clearance during lung aeration in newborn rabbits. Pediatric Research. 73 (4-1), 443-449 (2012).
- Boon, M. S., et al. Tubeless tracheotomy for survival airway surgery in the leporine model. The Laryngoscope. 125 (3), 680-684 (2015).
- Dion, G. R., et al. Morbidity and mortality associated with preclinical tracheostomy models. The Laryngoscope. 128 (2), 68-71 (2018).
- Phaneuf, L. R., et al. Tracheal injury after endotracheal intubation and anesthesia in rabbits. Journal of the American Association for Laboratory Animal Science. 45 (6), 67-72 (2006).

