Isolation, Expansion, and Differentiation of Mesenchymal Stem Cells from the Infrapatellar Fat Pad of the Goat Stifle Joint
Summary
Infrapatellar fat pad mesenchymal stem cells (IFP-MSCs) can be isolated easily from the infrapatellar fat pad of the knee joint. They proliferate well in vitro, form CFU-F colonies, and differentiate into adipogenic, chondrogenic, and osteogenic lineages. Herein, the methodology for the isolation, expansion, and differentiation of IFP-MSCs from goat stifle joint is provided.
Abstract
The IFP, present in the knee joint, serves as a promising source of MSCs. The IFP is an easily accessible tissue as it is routinely resected and discarded during arthroscopic procedures and knee replacement surgeries. Additionally, its removal is associated with minimal donor site morbidity. Recent studies have demonstrated that IFP-MSCs do not lose their proliferation capacity during in vitro expansion and have age-independent osteogenic differentiation potential. IFP-MSCs possess superior chondrogenic differentiation potential compared to bone marrow-derived MSCs (BMSCs) and adipose-derived stem cells (ADSCs). Although these cells are easily obtainable from aged and diseased patients, their effectiveness is limited. Hence, using IFP-MSCs from healthy donors is important to determine their efficacy in biomedical applications. As access to a healthy human donor is challenging, animal models could be a better alternative to enable fundamental understanding. Large animals such as dogs, horses, sheep, and goats play a crucial role in translational research. Amongst these, the goat could be a preferred model since the stifle joint of the goat has the closest anatomy to the human knee joint. Moreover, goat-IFP can fulfill the higher MSC numbers needed for tissue regeneration applications. Furthermore, low cost, availability, and compliance with the 3R principles for animal research make them an attractive model. This study demonstrates a simple protocol for isolating IFP-MSCs from the stifle joint of goats and in vitro culture conditions for their expansion and differentiation. The aseptically isolated IFP from the goat was washed, minced, and digested enzymatically. After filtration and centrifugation, the collected cells were cultured. These cells were adherent, had MSCs-like morphology, and demonstrated remarkable clonogenic ability. Further, they differentiated into adipogenic, chondrogenic, and osteogenic lineages, demonstrating their multipotency. In conclusion, the study demonstrates the isolation and expansion of MSCs, which show potential in tissue engineering and regenerative medicine applications.
Introduction
Mesenchymal stem cells (MSCs) are an attractive candidate for cell-based therapies in regenerative medicine1,2. They can be harvested from a variety of tissue sources such as bone marrow, umbilical cord, placenta, dental pulp, and subcutaneous adipose tissue3. However, as the availability of stem cells in adults is limited and their isolation procedure is often invasive (resulting in donor site morbidity), it is desirable to have an alternative stem cell source that could circumvent these challenges.
The knee joint is a depot of various cell types, such as infrapatellar fat pad-derived MSCs, synovial membrane-derived MSCs, synovial fluid-derived MSCs, ligament fibroblasts, articular chondrocytes, etc4,5,6. These cells have the potential to be widely explored in musculoskeletal tissue engineering-based research. Therefore, the knee joint could be a possible and reliable source of multiple types of MSCs. Adipose depot located in the knee joint, known as the infrapatellar fat pad (IFP) or Hoffa's fat pad, is a promising and alternative choice of MSC depot. The IFP is a relatively easily accessible and clinically obtainable source of MSCs, as it is routinely resected and discarded as surgical waste during knee arthroscopy or open knee surgery. Removal of the IFP is associated with minimal donor-site morbidity, which also makes it an attractive tissue source. While having a similar phenotypic profile, MSCs from IFP (IFP-MSCs) have enhanced clonogenic potential when compared with bone marrow-derived mesenchymal stem cells (BM-MSCs)6 and better proliferative capacity compared to subcutaneous adipose-derived stem cells (ADSCs)7. Interestingly, compared to synovial fluid-derived MSCs (SF-MSCs), IFP-MSCs do not lose their proliferative capacity at late passages, nor does doubling time increase at late passages. This suggests that, during cell expansion, IFP-MSCs can achieve a sufficiently large number of cells for in vitro tissue engineering applications without compromising their proliferation rate8. Recent studies have also suggested that IFP-MSCs possess superior chondrogenic differentiation potential compared to bone marrow-derived MSCs (BMSCs) and adipose-derived MSCs (ADSCs), probably due to their anatomical proximity to articular cartilage, indicating their suitability for cartilage tissue engineering6,7,9,10. Moreover, they also possess age-independent osteogenic differentiation potential11. Intra-articular injection of IFP-MSCs has been shown to reduce pain and improve knee joint functions in patients with osteoarthritis (OA)12,13. Further, strong immunosuppressive responses and improved immunomodulatory properties of IFP-MSCs in the presence of inflammatory cytokines during pathological conditions have also been reported6.
IFP-MSCs are a promising and alternate source of MSCs; however, their therapeutic benefit in tissue engineering and regenerative medicine is relatively less explored. The existing studies on IFP-MSCs have majorly utilized cells from human donors. Amongst these, a few recent studies have investigated IFP-MSCs from healthy human donors (non-arthritic patients, aged 17-60 years)6,14, whereas most of the studies have used IFP-MSCs from aged patients undergoing total knee replacement surgery (diseased patients, age 70-80 years). As both age and disease are known to alter the normal functioning of stem cells (reduced number and loss of functional potential), this could potentially lead to inconsistencies in the outcome of the MSC-based studies7,15,16,17. In addition to that, the use of IFP-MSCs from patients with pathophysiological conditions (e.g., arthritis and obesity) also poses difficulty for understanding the basic characteristics of healthy cells in vitro, thereby acting as a limiting factor in the development of MSCs-based therapies. To overcome these issues, the use of IFP-MSCs from healthy donors is vital. As access to a healthy human donor is challenging, animal models could be a better alternative. In this regard, there are a few studies where IFP has been isolated from mice18. However, owing to the small size of the fat pad in normal mice, fat tissues from multiple animals have been combined to get enough tissue to execute elaborate experimental procedures19. Hence, there is a need for a large animal model, which could fulfill the requirement for the higher number of cells and simultaneously comply with the 3R principles (refine, replace, and reduce) in animal research20. The usage of large animals has significant implications in translational research. Specifically, in musculoskeletal tissue engineering, a range of large animals such as dogs, pigs, sheep, goats, and horses have been investigated21. Goat (Capra aegagrus hircus) is an excellent choice of large animal since its stifle joint has the closest anatomy to the human knee joint22,23,24. The subchondral bone trabecular structure and subchondral bone thickness of goats are similar to humans, and the proportion of the cartilage to bone is also reported to be close to humans21. In addition, goats have been widely domesticated throughout the world, making them easily available when they are skeletally mature. Further, low maintenance costs and easy handling have made them an attractive animal model for research22.
In the present study, a simple protocol for the isolation of IFP-MSCs from the stifle joint of Capra aegagrus hircus (goat) and in vitro culture conditions for their expansion and differentiation are demonstrated. The isolated cells are adherent, have MSC-like morphology, form CFU-F (colony-forming unit-fibroblast) colonies, and possess adipogenic, chondrogenic, and osteogenic differentiation potential. Therefore, IFP-MSCs show potential as an alternative source of MSCs for biomedical applications.
Protocol
The protocol is based on the isolation of IFP-MSCs from goats. Goat IFP and blood were collected from a local abattoir. Since such tissue collections are outside the purview of an Institutional Animal Ethics Committee, ethical approval was not required.
1. Isolation of IFP-MSCs from the femorotibial joint of goat knee
- Collect goat femorotibial joint (sample) encompassing ~15 cm each of the femoral and tibial regions of the hind limbs. Immediately place the sample in a sterilized sample collection box and store it at 4 °C for transportation to the laboratory for further processing. Process the samples aseptically in a biosafety cabinet, using sterilized surgical tools throughout the procedure (Figure 1, Step 1).
- Place the sample on a 150 mm Petri dish and rinse it thoroughly with autoclaved phosphate-buffered saline (PBS) supplemented with antibiotics (5 µg/mL amphotericin B, 200 U/mL penicillin-streptomycin, and 50 µg/mL ciprofloxacin) to avoid contamination. Always ensure that the sample is kept hydrated using PBS.
- Carefully examine the anatomical regions of the sample. For ease of access to the joint capsule, ensure that the extra tissues from both the long bones are completely removed (Figure 1, Step 2).
- To achieve this, first, hold the end surface of the femur bone with one hand and, with sharp scissors, cut longitudinally towards the joint. Remove the surrounding muscles and adipose tissue from the bone during the process. Be careful that the tissue surrounding the immediate joint capsule remains undisturbed initially.
- Similarly, make another incision at the tibial end of the sample and remove the muscles and adipose tissue from the tibial bone. Ensure that both the long bones are completely exposed and the joint capsule is clearly visible after this step (Figure 1, Step 3).
- Next, make an incision from either side of the synovium membrane to cut open the articulating joint (Figure 1, Step 4). Cut free the patella from both the synovium membrane and patellar ligaments using a fresh batch of sterilized scissors and forceps. Immediately keep the separated patella in a Petri dish containing PBS (Figure 1, Step 5).
- Remove the entire fat pad present on the inner surface of the patella using scissors and forceps and collect the fat pad in another fresh Petri dish (Figure 1, Step 6). Mince the collected fat pad using a scalpel. Include other surgical tools (e.g., scissors or surgical blades) as required to obtain the smallest possible fat pieces/segments of ~2-3 mm.
NOTE: Good mincing is critical to result in a good yield of cells. Always keep the tissue hydrated and do not let it dry at any stage of the isolation procedure. - Transfer the minced tissue using a spatula to a 50 mL centrifuge tube and wash it with PBS supplemented with antibiotics at room temperature (RT) for 15 min in a test tube mixer. Repeat the washing step 3x, 15 min each.
- For enzymatic digestion, incubate the minced tissue (~5 g) in Dulbecco's Modified Eagle's Media (DMEM low glucose) containing 1.5 mg/mL type II collagenase (20 mL) and supplemented with 2% FBS at 37 °C for 12-16 h.
NOTE: Perform the digestion in a test tube mixer at ~15 rotations per minute (RPM). - Carefully aspirate the digested tissue and filter it through a cell strainer (70 µm) to remove any undigested tissue. Centrifuge the filtrate in a 15 mL centrifuge tube at 150 x g for 5 min. Remove the supernatant and wash the pellet with DMEM low glucose at least 2x.
NOTE: Pour the digested tissue slowly into the strainer to avoid spillage. Use a low volume centrifuge tube (15 mL) to get a good pellet. - Resuspend the obtained cell pellet in complete DMEM media (DMEM low glucose supplemented with 10% FBS and antibiotics [2.5 µg/mL amphotericin, 100 U/mL penicillin-streptomycin, and 25 µg/mL ciprofloxacin]), referred to as expansion media in the protocol in the subsequent steps.
- Seed the cells on 150 mm Petri dishes and culture them in the expansion media supplemented with 5 ng/mL basic fibroblast growth factor (bFGF) and 50 µg/mL 2-Phospho-L-ascorbic acid trisodium salt. Incubate the cells at 37 °C in a 5% CO2 incubator to allow the cells to adhere and proliferate. Monitor the attachment and growth of the cells on a daily basis.
2. Maintenance and expansion of isolated cells
- Change the media every 3 days until the cells become confluent. Add fresh 5 ng/mL bFGF and 50 µg/mL 2-Phospho-L-ascorbic acid trisodium salt directly to the Petri dish every time the media is changed. After the cells become 80%-90% confluent, subculture the cells.
NOTE: Remove the non-adherent entities during each media change. In case oil droplets are seen after 3 days of incubation, wash the cells with PBS 1x and then add fresh media to the Petri dish. Continue with PBS wash before each media change until the oil droplets are completely removed. - Sub-culturing of cells: Remove the expansion media from the Petri dish and wash the cells 1x with PBS. Add 4 mL of 0.25% trypsin/EDTA to the dish and incubate the cells at 37 °C in a 5% CO2 incubator for 4 min.
NOTE: Confluency (80%-90%) is achieved within 7 days. Uniformly distribute the trypsin/EDTA solution over the entire surface of the Petri dish during trypsinization. - Dislodge the cells by tapping the plate on the side and observing under a microscope to confirm that all the cells are detached. Neutralize the trypsin after adding an equal volume (4 mL) of expansion media.
- Collect the dissociated cells using a pipette in a fresh 15 mL polypropylene tube and centrifuge at 150 x g for 5 min at RT. Discard the supernatant and resuspend the pellet in the desired volume of expansion media.
- Count the cells using the standard cell counting method and subculture in 150 mm Petri dishes at a seeding density of 1 x 104 cells/cm2 for further expansion. For all assays, use a confluent monolayer of cells of passage number P2-P5.
NOTE: Cryopreserve the excess cells.
3. Evaluation of the clonogenic ability of IFP-MSCs using colony forming assay (CFU-F)
- For CFU-F assay, seed the cells in expansion media in a 6-well tissue culture plate at a seeding density of 50-100 cells/cm2. Add media to obtain a final volume of 3 mL.
NOTE: Optimize the cell concentration for assays and treatment. - Culture the cells for 12 days at 37 °C in a 5% CO2 incubator under standard culture conditions to allow the cells to form colonies. Change the media every 3 days. Be gentle while changing the media.
- After 12 days, rinse the colonies 1x with PBS and fix them using 20% methanol for 20 min at RT. Stain the colonies with crystal violet dye (0.5% [w/v] in 20% methanol) for 20 min at RT. Count the individual colonies using an inverted microscope.
NOTE: A colony is defined as a cluster of more than 50 cells.
4. Differentiation potential of IFP-MSCs
- Inducing adipogenic differentiation of IFP-MSCs
- To induce adipogenesis, seed the cells at a density of 25 x 103 cells/cm2 in a 24-well tissue culture plate. Add media to obtain a final volume of 500 µL. Culture the cells at 37 °C in a 5% CO2 incubator.
- One day post-confluency, replace the expansion medium with 500 µL of adipogenic differentiation media. It requires approximately 2 days for the cells to reach confluency.
- Prepare the adipogenic differentiation media by supplementing expansion media (DMEM + 10% FBS + 2.5 µg/mL amphotericin, 100 U/mL penicillin-streptomycin, and 25 µg/mL ciprofloxacin) with 25 mM D (+)-glucose, 10 µg/mL insulin, 1 µM dexamethasone, and 100 µM indomethacin.
NOTE: Differentiation media is unstable at 4 °C after 1 month; hence, prepare the media as and when required. - Consider the cells cultured in the expansion media supplemented with 25 mM D (+) glucose as uninduced controls. Change the media every 3 days for 14 days. At the end of the 14 days, wash the cells 1x with PBS. Fix the cells using neutral buffered formalin (NBF; 4 % formaldehyde in PBS) for 15 min at 4 °C.
- Following fixation, wash the wells 1x with distilled water and then incubate the cells with 60% isopropanol for 5 min at RT. After removing isopropanol, stain the lipid droplets by incubating the cells with 500 µL of the working solution of Oil Red O dye for 5 min at RT. Wash the wells 1x with distilled water to remove the unbound dye and image the cells using an inverted microscope.
NOTE: Prepare the stock solution of Oil Red O (ORO) by dissolving 300 mg of ORO in 100 mL of 99% isopropanol. To prepare the working solution, mix three parts of stock ORO and two parts of distilled water and allow it to mix at RT for 10 min. Filter the mixture using 0.2 µm filter paper. The working solution is ready to use. The stock solution can be prepared and stored at RT for 1 month in the dark, whereas the working solution needs to be freshly prepared prior to each use/staining.
- Inducing chondrogenic differentiation of IFP MSCs
- Isolation of goat plasma:
- Prepare 3.4% (w/v) sodium-citrate in distilled water. Add 5 mL of the prepared sodium citrate solution to a 50 mL centrifuge tube.
- Collect 45 mL of the freshly isolated goat blood in the same tube. Immediately tilt the tube to mix the blood with sodium citrate thoroughly to prevent clotting and transport it to the laboratory at 4 °C.
- Centrifuge the collected goat blood at 1000 x g for 20 min at 4 °C. Transfer the supernatant (plasma) to a fresh tube inside a biosafety cabinet. Filter the plasma through 0.2 µm filters, aliquot, and store at -20 °C for further use.
NOTE: Maintain the temperature of 2-8 °C while handling the samples. It is important to minimize the freeze-thaw cycles of plasma.
- Culture the expanded cells to 80%-90% confluency, dissociate the cells using trypsin/EDTA solution, and pellet down the cells at 150 x g for 5 min in a 15 mL centrifuge tube. Resuspend the pellet in goat plasma at a density of 2 x 106 cells per 50 µL of plasma solution.
- Add a drop of plasma-containing cells (50 µL) onto a sterilized 90 mm Petri dish and then add calcium chloride at a final concentration of 0.3% (w/v). Mix well to fabricate cross-linked plasma hydrogel.
- Incubate the fabricated hydrogel at 37 °C for 40 min. After incubation, transfer the hydrogels to 24-well tissue culture plates containing chondrogenic media.
- Prepare chondrogenic media by supplementing DMEM low glucose with 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 1.25 mg/mL bovine serum albumin (BSA), 1 mM proline, 50 µg/mL 2-Phospho-L-ascorbic acid trisodium salt, 100 nM dexamethasone, 1x insulin, transferrin, sodium selenite + linoleic-BSA (ITS+1), antibiotics (2.5 µg/mL amphotericin, 100 U/mL penicillin-streptomycin and 25 µg/mL ciprofloxacin), and 10 ng/mL transforming growth factor- β1 (TGF-β1).
- The hydrogels cultured in complete DMEM media (DMEM low glucose with 10% FBS and antibiotics [2.5 µg/mL amphotericin, 100 U/mL penicillin-streptomycin, and 25 µg/mL ciprofloxacin]) without TGF-β1 are considered as uninduced controls. Change the media every 3 days for 14 days.
- After 14 days of chondrogenic differentiation of cells in plasma hydrogels, wash the hydrogels with PBS 3x for 5 min each and then fix the samples in NBF for 3 h.
CAUTION: Formaldehyde is a potential carcinogen and can cause irritation to the nose, throat, and lungs upon inhalation. Perform all procedures related to formaldehyde in a fume hood. - Remove NBF, wash the hydrogels with PBS 3x for 5 min each, and then incubate in 35% (w/v) sucrose at RT overnight to allow the solution to be completely absorbed into the samples. Acclimatize the hydrogels in optimal cutting temperature (OCT) compound for 3 h. Embed the samples in OCT compound using silicone molds under liquid nitrogen.
- Cut the samples at a thickness of 10-12 µm on gelatin-coated glass slides using a cryotome and store them at -20 °C for future use. To examine the presence or deposition of the extracellular matrix after chondrogenic differentiation, stain the sections with Alcian Blue and Safranin-O dye, which bind to sulfated glycosaminoglycans.
NOTE: To prepare Alcian Blue dye, dissolve 1 g of Alcian Blue dye in 100 mL of 0.1 N HCl and mix thoroughly in a test tube mixer overnight. The solution can be stored at RT for 1 month in the dark. To prepare Safranin-O dye, dissolve 0.1 g of Safranin-O dye in 100 mL of distilled water and mix thoroughly in a test tube mixer overnight. - For Alcian Blue staining, wash the hydrogel sections 1x with distilled water for 5 min. To fix the sections, add 4% NBF to the slides and incubate for 30 min.
- Wash the sections 1x with distilled water for 5 min and then 2x with 0.1 N HCl for 30 s each. Remove the HCl and gently dry the slides with tissue paper. Add the prepared Alcian Blue stain to the sections and incubate for 30 min at 37 °C in a humidified chamber.
- Wash the unbound dye with 0.1 N HCl and then distilled water for 3 min. Dehydrate the sections in absolute ethanol, clear them in xylene, and finally mount the stained sections in a resinous medium for imaging.
- For Safranin-O staining, wash the hydrogel sections with distilled water 1x for 5 min. To fix the sections, add 4% NBF to the slides and incubate for 30 min. Wash the sections 1x with distilled water for 5 min, and then dip the slides in 1% acetic acid for 10-15 s. Dry the slides gently with tissue paper.
- Add prepared Safranin-O dye to the sections and incubate for 10 min at RT. Wash the unbound dye with 100% ethanol. Dip the slides in 100% ethanol for 5 min. Air-dry the slides, clear them in xylene, and finally mount the stained sections in a resinous medium for imaging.
- Isolation of goat plasma:
- Inducing osteogenic differentiation of IFP MSCs
- To induce osteogenic differentiation, trypsinize the cells, as stated before (Step 2.2.). Seed the cells at a density of 3 x 103 cells/cm2 in a 24-well tissue culture plate. Add media to obtain a final volume of 500 µL. Culture the cells at 37 °C in a 5% CO2 incubator. 1 day post-seeding, replace the expansion media with 500 µL osteogenic differentiation media.
- Prepare the osteogenic media by supplementing expansion media (DMEM + 10% FBS +2.5 µg/mL amphotericin, 100 U/mL penicillin-streptomycin, and 25 µg/mL ciprofloxacin) with 25 mM D (+)-glucose, 10 nM dexamethasone, 10 mM β-glycerophosphate, 50 µg/mL 2-Phospho-L-ascorbic acid trisodium salt, and 10 nM vitamin D3.
NOTE: The cells cultured in expansion media supplemented with 25 mM D (+) glucose are considered uninduced controls. - Change the osteogenic media every 3 days for 14 days or 28 days. At the end of 14 days, measure the extent of osteogenesis by alkaline phosphatase (ALP) staining.
NOTE: To prepare the substrate for ALP staining, dissolve one 5-Bromo-4- Chloro-3-Indolyl Phosphate (BCIP)/Nitroblue Tetrazolium (NBT) tablet in 10 mL of distilled water in a 15 mL centrifuge tube. Let the tablet dissolve completely. The substrate solution is ready to use. Since the substrate solution cannot be stored for a longer duration, based on need, break the tablet in half and dissolve it in 5 mL of distilled water. To prepare the permeabilization buffer, add Tris buffer (100 mM) and magnesium chloride (5 mM) to distilled water. Add Triton X100 (0.1%) to the solution and let it dissolve. Adjust the pH of the solution to 9.25-9.75. - For ALP staining, after 14 days of induction, remove the media and wash the cells with PBS. Fix the cells using NBF (4%) for 1 min. Wash the cells with PBS and permeate using the prepared lysis buffer for 2-3 min.
- Add 500 µL of the prepared substrate solution to the cells and let it incubate for 15 min to 1 h, depending upon the expression level. Check the development of purple/blue color in between.
- Remove the substrate solution and wash with distilled water. Image the stained cells using an inverted microscope.
- At the end of 28 days, measure the extent of calcification after osteogenic induction by Alizarin Red-S staining.
NOTE: To prepare the Alizarin Red-S staining solution (40 mM), dissolve 2 g of Alizarin Red-S in 100 mL of distilled water and adjust the pH to 4.1-4.3 with HCl or NH4OH. Filter the solution through a 0.22 µm membrane and store at 4 °C in the dark. The pH of the solution is important; hence it is recommended either to make a fresh solution or to re-measure the pH of the solution if it is more than 1 month old. - For Alizarin Red-S staining, after 28 days of induction, remove the media and wash the cells 1x with cold PBS. Fix the cells using 70% ethanol for 1 h at 4 °C.
- Remove the fixative and wash the cells 2x with distilled water. Remove the water completely and add 1 mL of Alizarin Red-S solution to each well.
- Incubate the cells with the solution for 1 h at RT and image the cells using an inverted microscope.
Representative Results
Isolation of IFP-MSCs from the femorotibial joint of goat
The steps involved in the isolation of IFP-MSCs from the stifle joint of a goat are depicted in Figure 1. The fat pad present in the inner non-articulating surface of the patella was removed, minced, and enzymatically digested. The IFP-MSCs were successfully isolated and cultured in vitro (Figure 2A).
Expansion and clonogenic ability of IFP-MSCs
The isolated cells were cultured in vitro in expansion media. The cells started adhering to the tissue culture plate within 12 h of seeding and were mostly round in shape at day 0. They were homogenously adherent to the plate and attained elongated morphology within 24 h. This morphology was maintained throughout the duration of the culture. The cells proliferated efficiently in culture and became 80%-90% confluent within 6 days of expansion (Figure 2A). Additionally, the isolated cells displayed clonogenic capacity, assessed by colony-forming unit-fibroblast (CFU-F) assay (Figure 2B). These results indicate that the isolated cells have efficient proliferative and self-renewal capabilities in vitro.
Differentiation potential of IFP-MSCs
To characterize whether the isolated cells were multipotent and possessed characteristic features of MSCs, they were induced to differentiate into multiple lineages. The isolated cells, when induced towards adipogenic lineage, produced lipid droplets, as can be observed from the brightfield images (Figure 3A). This was further confirmed by Oil Red O staining on day 14 and day 21, suggesting the ability of these cells to differentiate into adipocytes. On the other hand, no visible oil droplets were observed in the cells cultured in the absence of adipogenic-inducing factors (Figure 3B,C).
To determine the chondrogenic potential of the isolated cells, the cells were encapsulated in plasma hydrogel and were allowed to differentiate into the chondrogenic lineage. As depicted from the gross images of the plasma hydrogel (Figure 4A), the neotissue formed after 14 days in the induced group appeared white and glossy (similar to articular cartilage), whereas in the uninduced group, it was pale white, and reduced glossiness was observed. Additionally, the cells in the induced group were able to secrete one of the major components of the cartilage matrix (i.e., sulfated glycosaminoglycans), evidenced by positive histological staining for Alcian Blue (Figure 4B) and Safranin O (Figure 4C). In contrast, the hydrogel sections from the uninduced groups were negative for Safranin O and Alcian Blue staining. These results collectively indicate the chondrogenic differentiation potential of the MSCs in the presence of chondrogenic stimuli only.
The isolated cells also demonstrated osteogenic differentiation potential. After 14 days and 21 days in culture, no spontaneous mineralization was observed (uninduced group). However, when induced with osteogenic media, mineralization was evident by the positive staining for alkaline phosphatase (ALP) at the end of 14 days (Figure 5A). Moreover, after 28 days of culture in osteogenic media, calcified depositions were visualized by positive Alizarin Red-S staining (Figure 5B). These results demonstrate that the isolated cells from goat infrapatellar fat pad, when induced, have the potential to differentiate into adipogenic, chondrogenic, and osteogenic lineages.
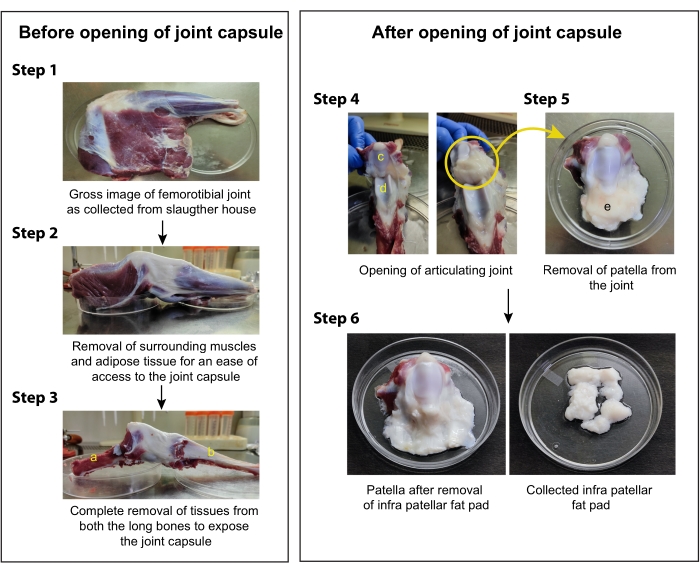
Figure 1: Schematic for the isolation of IFP-MSC from the stifle joint of goat knee. Step 1. Collect the goat knee sample from the slaughterhouse and proceed with the dissection in a biosafety cabinet; Step 2. Carefully examine the anatomy of the tissue and remove the surrounding muscles and adipose tissue; Step 3. Without disturbing the joint capsule, remove the muscles and adipose tissues to completely expose both the long bones (femur and tibia); Step 4. Make an incision in the synovium membrane and cut open the articulating joint to expose the patella and trochlear cartilage; Step 5. Remove the patella from the articulating joint carefully, without disturbing the infrapatellar fat pad (IFP), and keep it on a petri dish containing PBS; Step 6. Slowly remove the entire fat pad from the patella and keep it in a fresh petri dish for mincing and enzymatic digestion. (a. Femur, b. Tibia, c. Patella, d. Trochlear cartilage, e. Infrapatellar fat pad). Please click here to view a larger version of this figure.
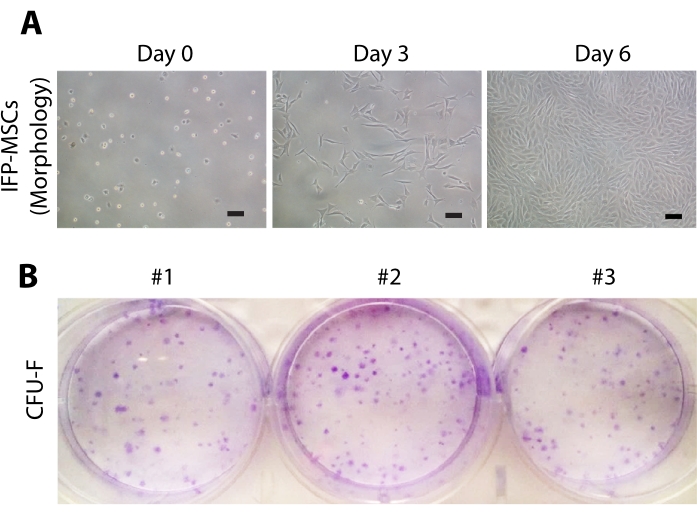
Figure 2: Morphology and clonogenic potential of IFP-MSCs. (A) Micrographs were taken at passage 3 using an inverted brightfield microscope at 10x magnification. Immediately after seeding (day 0), the cells remained round in shape, at day 3 most of the cells attained elongated morphology, and on day 6 the IFP-MSCs became 80%-90% confluent (Scale bar = 100 µm). (B) Representative pictures (n = 3) of CFU-F colonies stained with crystal violet dye after 12 days of seeding. Please click here to view a larger version of this figure.
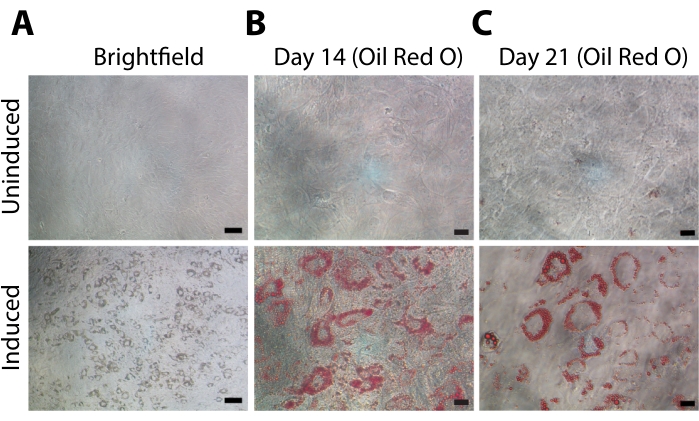
Figure 3: Adipogenic differentiation of IFP-MSCs. The upper panel indicates micrographs of uninduced IFP-MSCs, and the lower panel represents IFP-MSCs induced into the adipogenic lineage. (A) Representative brightfield images showing the formation of lipid vacuoles during adipogenesis (Scale bar = 100 µm). Representative images of Oil Red O staining of lipid droplets at (B) day 14 and (C) day 21 of differentiation (Scale bar = 20 µm). Please click here to view a larger version of this figure.
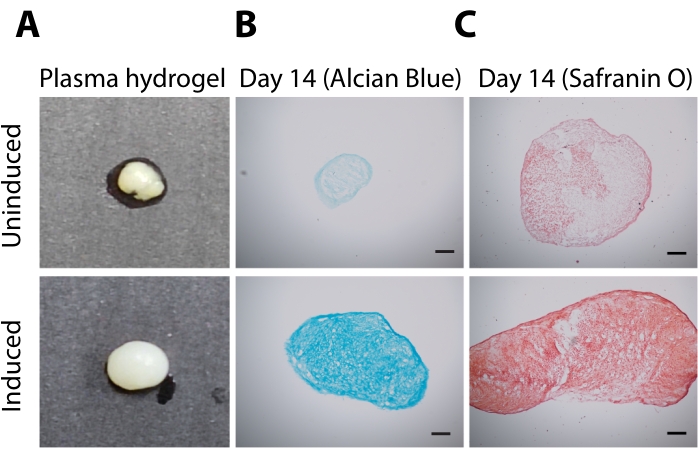
Figure 4: Chondrogenic differentiation of IFP-MSCs. The upper panel indicates hydrogel constructs/sections from uninduced hydrogel, and the bottom panel represents hydrogel subjected to chondrogenesis. (A) Gross images of plasma hydrogel constructs after 14 days in culture in the absence (uninduced) or presence (induced) of chondrogenic media supplemented with TGF-β1. Representative images of 10 µm sections of hydrogel stained with sGAG binding dye (B) Alcian Blue (blue) and (C) Safranin O (red) (Scale bar = 100 µm). Please click here to view a larger version of this figure.
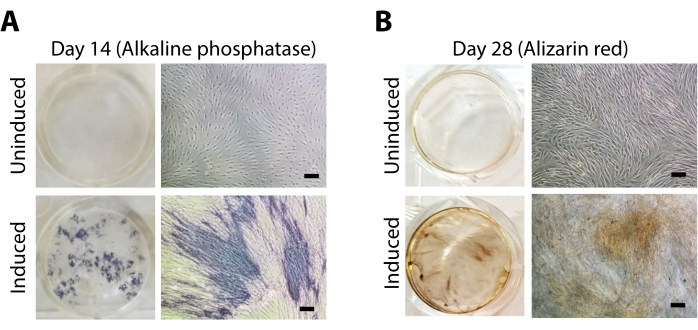
Figure 5: Osteogenic differentiation of IFP-MSCs. The upper panel indicates uninduced cells, and the bottom panel indicates cells induced with osteogenic media. Micrographs and representative images of IFP-MSCs stained with (A) BCIP-NBT (alkaline phosphatase) after 14 days and (B) Alizarin Red-S (calcium deposition) after 28 days of differentiation (Scale bar = 100 µm). Please click here to view a larger version of this figure.
Discussion
In the present protocol, a simple, reliable, and reproducible method for the isolation of MSCs from goat IFP has been provided. Cells isolated using this method have been successfully used in our previous studies for in vitro tissue regeneration. It was observed that the isolated cells were proliferative, were responsive to various growth factors, and retained their biological activity when seeded on electrospun fibers and scaffolds25,26. Moreover, it was observed that a large number of high-quality MSCs can be obtained and co-cultured with chondrocytes in injectable hydrogels to engineer articular cartilage in vitro27,28,29. While the procedure for the dissection and isolation of cells is simple, there are a few steps that need to be carefully followed for the successful isolation and expansion of MSCs from IFP. Firstly, since it is not possible to maintain truly aseptic conditions while bringing the sample from the slaughterhouse, it is advised to wipe the outer tissue with 70% ethanol to sterilize the tissue before starting the isolation procedure. The next important step is dissecting the fat pad from the knee joint. The IFP is located below the patella in the knee joint. The proximal end of the IFP is attached to the distal end of the patella30,31. Hence, it becomes very important to identify the correct location of the patella while performing the isolation procedure. Next, as IFP articulates with the trochlear cartilage in the femur and is covered with the synovium membrane30,31, making an incision to the synovium membrane is necessary to cut open the knee joint. It is also crucial to observe the overall health of the collected infrapatellar fat pad. As the IFP is composed of adipocytes (cells) and adipose connective tissue, such as collagen and glycosaminoglycans, the tissue is minced and digested using collagenase enzyme to release the cells9,32. The floating adipocytes are separated by centrifugation at a slow speed to have a homogenous culture of MSCs. The remaining adipocytes in the stromal vascular fraction must be removed from the tissue culture plate at each media change by washing the cell monolayer with PBS before adding fresh media. This step needs to be continued until all the visible adipocytes are removed. It is suggested to reseed the cells in a new plate after P0 cells reach confluency to get rid of the remaining adipocytes, which might have adhered to the plate wall and are difficult to remove. Finally, as the goat samples are mostly obtained from a slaughterhouse, it is difficult to trace the exact age of the goat, and, therefore, while isolating the cells, variation between samples needs to be carefully observed and documented. It is suggested to characterize the cells for self-renewal (CFU-F), proliferation, and differentiation potential after each isolation before they are used for various applications and mechanistic studies. Due to the requirement of a large number of cells in cell-based tissue engineering applications, the culture conditions that facilitate proliferation, delay senescence, and maintain the stemness of cells become centrally important. It was observed that MSCs cultured in 2-Phospho-L-ascorbic acid trisodium salt (PAA) and basic fibroblast growth factor (bFGF) showed a ~42-fold increase in their numbers compared to MSCs cultured in bFGF alone. In addition, the co-treatment of PAA and bFGF reduced the production of reactive oxygen species (due to their antioxidant properties) and senescence in stem cells. Therefore, the use of PAA during in vitro expansion of MSCs is desirable33.
While enzymatic digestion of tissues is universally employed and is an efficient method for isolating MSCs, the use of enzymes such as collagenases is time-consuming and expensive34. In the present study, the complete digestion of the tissue using collagenase took 12-16 h (overnight), which might lead to a decreased viability or altered surface phenotype of cells35. Hence, the method can further be optimized to reduce the duration of collagenase treatment for tissue digestion. Alternatively, an enzyme-free explant culture method that is used for the isolation of adipose-derived MSCs from lipoaspirates or inguinal fat pads can also be explored for the isolation of IFP-MSCs34,36,37.
IFP-MSCs, due to their ability to differentiate into chondrogenic, adipogenic, and osteogenic lineages possess wide therapeutic potential. Previous reports have demonstrated that IFP-MSCs exhibit better chondrogenic potential than other stem cells6,7,9,10. Hence, IFP-MSCs have gained interest and are considered a better candidate for cell-based therapy for the repair of cartilage defects and alleviating cartilage degradation in osteoarthritis. In a preclinical study, co-delivery of IFP-MSCs with cartilage extracellular matrix components through intraarticular injection resulted in enhanced cartilage regeneration in osteochondral defects38. Similarly, intra-articular injection of IFP-MSCs in the rabbit osteoarthritis (OA) model led to a decrease in the following disease parameters: cartilage degradation, osteophyte formation, subchondral bone thickening, and synovitis39. Moreover, results from a previously reported randomized clinical trial13 and a recently published open level phase 1 clinical trial40 have demonstrated that intra-articular injections of IFP-MSCs in patients with knee osteoarthritis are safe and result in the reduction of pain and improvement of joint function, indicating their promising therapeutic potential in ameliorating knee OA-related complications. IFP-MSCs also have been shown to develop augmented immunomodulatory properties in the presence of proinflammatory cues by facilitating the degradation of an inflammatory regulator, Substance P (SP), leading to a reduction in proinflammatory and catabolic molecules and, as a consequence, improved treatment of inflammation and fibrosis of synovium and IFP6. More importantly, IFP-MSCs processed in regulatory-compliant conditions showed better proliferation, differentiation capacity, and immunomodulatory properties compared to standard in vitro culture conditions, which demonstrates their potential for clinical success when used for mesenchymal stem cell (MSC)-based therapy41.
In conclusion, the present protocol describes the isolation of infrapatellar fat pad (IFP)-derived mesenchymal stem cells (MSCs) from goat stifle joint by enzymatic digestion and their maintenance in culture. The isolated cells have proliferative and self-renewal potential and can differentiate into adipogenic, osteogenic, and chondrogenic lineages. IFP-MSCs are a promising source of MSCs and have translational potential for regenerative medicine applications.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
SH acknowledges support from the Institute Post-Doctoral Fellowship of IIT Kanpur and SYST grant from DST (SEED Division) (SP/YO/618/2018). AM acknowledges the Indian Institute of Technology-Kanpur (IIT-Kanpur) for an Institute fellowship. DSK acknowledges Gireesh Jankinath Chair Professorship and Department of Biotechnology, India, for funding (BT/PR22445/MED/32/571/2016). AM, SH, and DSK thank The Mehta Family Centre for Engineering in Medicine at IIT-Kanpur for their generous support.
Materials
| β-glycerophosphate | Sigma-Aldrich | G9422-10G | 10 mM |
| 0.25% Trypsin- 0.02% EDTA | Hi-Media | TCL049 | |
| 15-mL centrifuge tube | Corning | ||
| 2-Phospho-L-ascorbic acid trisodium salt | Sigma | 49752-10G | 50 µg/mL |
| 2-Propanol | Sigma-Aldrich | I9516 | |
| 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) | HiMedia | TCL021-50ml | 10 mM |
| 50-mL centrifuge tube | Corning | ||
| Alcian Blue | Hi-Media | RM471 | For sufated gycosaminoglycans staining |
| Alizarin Red S | S D Fine-Chem Limited | 26048-25G | For calcium deposition |
| Amphotericin B | HiMedia | A011 | 2.5 µg/mL |
| Basic fibroblast growth factor (bFGF) | Sino Biologicals | 10014-HNAE | 5 ng/mL |
| BCIP/NBT ALP Substrate | Sigma | B5655-5TAB | For ALP staining |
| Biological safety cabinet | |||
| BSA | HiMedia | MB-083 | Long name: Bovine Serum Albumin (1.25 mg/mL ) |
| Cell strainer | HiMedia | TCP-182 | 70 µm |
| Centrifuge | REMI | ||
| Ciprofloxacin | RANBAXY LAB. Limited | B17407T1 | 2.5 µg/mL |
| Crystal Violet | S D Fine-Chem Limited | 42555 | |
| D(+)-glucose | Merck | 1.94925.0521 | 25 mM |
| Dexamethasone | Sigma-Aldrich | D2915 | 1 µM |
| DMEM LG | SIGMA | D5523 | Long name: Dulbecco’s Modified Eagle’s Media Low Glucose |
| Ethanol | Merck | 100983 | |
| FBS | Gibco | 10270 | Long name: Fetal Bovine Serum |
| Formaldehyde solution 37%-41% | Merck | 61780805001730 | |
| Indomethacin | Sigma-Aldrich | I7378 | 100 µM |
| Insulin | Sigma-Aldrich | I9278 | 10 µg/mL |
| Inverted microscope | Nikon Eclipse TS 100 | ||
| ITS + 1 | Sigma-Aldrich | I2521-5mL | Long name: insulin, transferrin, sodium selenite + linoleic-BSA |
| L-Proline | HiMedia | TO-109-25G | 1 mM |
| Magnesium chloride | Merck | 61751605001730 | For lysis buffer |
| Methanol | Meck | 1.07018.2521 | |
| Micropipettes and sterile tips (20 µL, 200 µL, 1000 µL) | Thermoscientific | ||
| MUSE Cell analyser | Merck Millipore | For cell counting | |
| OCT compound | Tissue-Tek | 4583 | Long name: Optimal Cutting Temperature |
| Oil Red O dye | S D Fine-Chem Limited | 54304 | For lipid vacuole staining |
| Penicillin-Streptomycin | HiMedia | A007 | 100 U/mL |
| Petri dishes (150 mm and 90 mm) | NEST | ||
| Safranin O | S D Fine-Chem Limited | 50240 | For sufated gycosaminoglycans staining |
| Sodium citrate | Sigma-Aldrich | C3434 | 3.4 % (w/v) |
| Sterile scissors, forceps and scalpels | For isolation of IFP-MSC | ||
| Sucrose | Merck | 1.94953.0521 | 35 % (w/v) |
| TGF-β1 | Sino Biologicals | Long name: Transforming growth factor- β1 (10 ng/mL) | |
| Tissue culture incubator 37 °C, 5% CO2 | Thermoscientific | ||
| Tris buffer | Merck | 61771405001730 | For lysis buffer |
| Triton X100 | S D Fine-Chem Limited | 40632 | For lysis buffer |
| Type II collagenase | Gibco | 17101015 | 1.5 mg/mL |
| Vitamin D3 | Sigma | C9756-1G | 10 nM |
| Well plates (6 -WP and 24-WP) | NEST |
Riferimenti
- Han, Y., et al. Mesenchymal stem cells for regenerative medicine. Cells. 8 (8), (2019).
- Pittenger, M. F., et al. Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regenerative Medicine. 4 (1), 22 (2019).
- Guo, Y., Yu, Y., Hu, S., Chen, Y., Shen, Z. The therapeutic potential of mesenchymal stem cells for cardiovascular diseases. Cell Death & Disease. 11 (5), 349 (2020).
- Ge, Z., Goh, J. C. H., Lee, E. H. Selection of cell source for ligament tissue engineering. Cell Transplantation. 14 (8), 573-583 (2005).
- Harvanová, D., Tóthová, T., Sarissky, M., Amrichová, J., Rosocha, J. Isolation and characterization of synovial mesenchymal stem cells. Folia Biologica. 57 (3), 119-124 (2011).
- Kouroupis, D., et al. Infrapatellar fat pad-derived MSC response to inflammation and fibrosis induces an immunomodulatory phenotype involving CD10-mediated Substance P degradation. Scientific Reports. 9 (1), 10864 (2019).
- Lopa, S., et al. Donor-matched mesenchymal stem cells from knee infrapatellar and subcutaneous adipose tissue of osteoarthritic donors display differential chondrogenic and osteogenic commitment. European Cells & Materials. 27, 298-311 (2014).
- Garcia, J., et al. Characterisation of synovial fluid and infrapatellar fat pad derived mesenchymal stromal cells: The influence of tissue source and inflammatory stimulus. Scientific Reports. 6 (1), 24295 (2016).
- Tangchitphisut, P., et al. Infrapatellar fat pad: an alternative source of adipose-derived mesenchymal stem cells. Arthritis. 2016, 4019873 (2016).
- Ding, D. -. C., et al. Human infrapatellar fat pad-derived stromal cells have more potent differentiation capacity than other mesenchymal cells and can be enhanced by hyaluronan. Cell Transplantation. 24 (7), 1221-1232 (2015).
- Khan, W., Adesida, A., Tew, S., Andrew, J., Hardingham, T. The epitope characterisation and the osteogenic differentiation potential of human fat pad-derived stem cells is maintained with ageing in later life. Injury. 40 (2), 150-157 (2009).
- Koh, Y. G., et al. Mesenchymal stem cell injections improve symptoms of knee osteoarthritis. Arthroscopy. 29 (4), 748-755 (2013).
- Koh, Y. G., Choi, Y. J. Infrapatellar fat pad-derived mesenchymal stem cell therapy for knee osteoarthritis. Knee. 19 (6), 902-907 (2012).
- Kouroupis, D., Willman, M. A., Best, T. M., Kaplan, L. D., Correa, D. Infrapatellar fat pad-derived mesenchymal stem cell-based spheroids enhance their therapeutic efficacy to reverse synovitis and fat pad fibrosis. Stem Cell Research & Therapy. 12 (1), 44 (2021).
- Stocco, E., et al. Infrapatellar fat pad stem cells responsiveness to microenvironment in osteoarthritis: from morphology to function. Frontiers in Cell and Developmental Biology. 7, 323 (2019).
- Hindle, P., Khan, N., Biant, L., Péault, B. The infrapatellar fat pad as a source of perivascular stem cells with increased chondrogenic potential for regenerative medicine. Stem Cells Translational Medicine. 6 (1), 77-87 (2017).
- Stolzing, A., Jones, E., McGonagle, D., Scutt, A. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mechanisms of Ageing and Development. 129 (3), 163-173 (2008).
- Wu, C. L., Diekman, B. O., Jain, D., Guilak, F. Diet-induced obesity alters the differentiation potential of stem cells isolated from bone marrow, adipose tissue and infrapatellar fat pad: the effects of free fatty acids. International Journal of Obesity (2005). 37 (8), 1079-1087 (2013).
- Barboza, E., et al. Profibrotic infrapatellar fat pad remodeling without M1 macrophage polarization precedes knee osteoarthritis in mice with diet-induced obesity. Arthritis & Rheumatology. 69 (6), 1221-1232 (2017).
- Allen, M. J., Hankenson, K. D., Goodrich, L., Boivin, G. P., von Rechenberg, B. Ethical use of animal models in musculoskeletal research. Journal of Orthopaedic Research. 35 (4), 740-751 (2017).
- Moran, C. J., et al. The benefits and limitations of animal models for translational research in cartilage repair. Journal of Experimental Orthopaedics. 3 (1), (2016).
- Kuyinu, E. L., Narayanan, G., Nair, L. S., Laurencin, C. T. Animal models of osteoarthritis: classification, update, and measurement of outcomes. Journal of Orthopaedic Surgery and Research. 11 (1), 19 (2016).
- Chu, C. R., Szczodry, M., Bruno, S. Animal models for cartilage regeneration and repair. Tissue Engineering Part B: Reviews. 16 (1), 105-115 (2010).
- Proffen, B. L., McElfresh, M., Fleming, B. C., Murray, M. M. A comparative anatomical study of the human knee and six animal species. The Knee. 19 (4), 493-499 (2012).
- Bhutada, S. S., Sriram, M., Katti, D. S. Sulfated carboxymethylcellulose conjugated electrospun fibers as a growth factor presenting system for tissue engineering. Carbohydrate Polymers. 268, 118256 (2021).
- Waghmare, N. A., Arora, A., Bhattacharjee, A., Katti, D. S. Sulfated polysaccharide mediated TGF-β1 presentation in pre-formed injectable scaffolds for cartilage tissue engineering. Carbohydrate Polymers. 193, 62-72 (2018).
- Arora, A., Mahajan, A., Katti, D. S. TGF-β1 presenting enzymatically cross-linked injectable hydrogels for improved chondrogenesis. Colloids & Surfaces B: Biointerfaces. 159, 838-848 (2017).
- Arora, A., Sriram, M., Kothari, A., Katti, D. S. Co-culture of infrapatellar fat pad-derived mesenchymal stromal cells and articular chondrocytes in plasma clot for cartilage tissue engineering. Cytotherapy. 19 (7), 881-894 (2017).
- Mahajan, A., Singh, A., Datta, D., Katti, D. S. Bioinspired injectable hydrogels dynamically stiffen and contract to promote mechanosensing-mediated chondrogenic commitment of stem cells. ACS Applied Materials & Interfaces. 14 (6), 7531-7550 (2022).
- Nakano, T., Wang, Y. W., Ozimek, L., Sim, J. S. Chemical composition of the infrapatellar fat pad of swine. Journal of Anatomy. 204 (4), 301-306 (2004).
- Sun, Y., Chen, S., Pei, M. Comparative advantages of infrapatellar fat pad: an emerging stem cell source for regenerative medicine. Rheumatology. 57 (12), 2072-2086 (2018).
- Han, W., et al. Infrapatellar fat pad in the knee: is local fat good or bad for knee osteoarthritis. Arthritis Research & Therapy. 16 (4), 1-8 (2014).
- Bae, S. H., et al. L-ascorbic acid 2-phosphate and fibroblast growth factor-2 treatment maintains differentiation potential in bone marrow-derived mesenchymal stem cells through expression of hepatocyte growth factor. Growth Factors. 33 (2), 71-78 (2015).
- Priya, N., Sarcar, S., Majumdar, A. S., SundarRaj, S. Explant culture: a simple, reproducible, efficient and economic technique for isolation of mesenchymal stromal cells from human adipose tissue and lipoaspirate. Journal of Tissue Engineering and Regenerative Medicine. 8 (9), 706-716 (2014).
- Tsuji, K., et al. Effects of different cell-detaching methods on the viability and cell surface antigen expression of synovial mesenchymal stem cells. Cell Transplantation. 26 (6), 1089-1102 (2017).
- Jing, W., et al. Explant culture: an efficient method to isolate adipose-derived stromal cells for tissue engineering. Artificial Organs. 35 (2), 105-112 (2011).
- Sherman, L. S., Condé-Green, A., Naaldijk, Y., Lee, E. S., Rameshwar, P. An enzyme-free method for isolation and expansion of human adipose-derived mesenchymal stem cells. Journal of Visualized Experiments. (154), e59419 (2019).
- Muttigi, M. S., et al. Matrilin-3 codelivery with adipose-derived mesenchymal stem cells promotes articular cartilage regeneration in a rat osteochondral defect model. Journal of Tissue Engineering and Regenerative Medicine. 12 (3), 667-675 (2018).
- Toghraie, F., et al. Treatment of osteoarthritis with infrapatellar fat pad derived mesenchymal stem cells in Rabbit. The Knee. 18 (2), 71-75 (2011).
- Chen, H. -. H., et al. Infrapatellar fat pad-derived mesenchymal stromal cell product for treatment of knee osteoarthritis: a first-in-human study with evaluation of the potency marker. Cytotherapy. 24 (1), 72-85 (2022).
- Kouroupis, D., Bowles, A. C., Best, T. M., Kaplan, L. D., Correa, D. CD10/Neprilysin enrichment in infrapatellar fat pad-derived mesenchymal stem cells under regulatory-compliant conditions: implications for efficient synovitis and fat pad fibrosis reversal. The American Journal of Sports Medicine. 48 (8), 2013 (2020).

