Optimizing the Rearing Procedure of Germ-Free Wasps
Summary
Nasonia wasp embryos were dissected from Lucillia sericata pupae after parasitization for 12-24 h and washed with alcohol and 10% sodium hypochlorite solution to obtain germ-free embryos. After rearing the germ-free embryos and supplying them with Nasonia rearing medium to grow and develop in vitro, germ-free Nasonia adults were obtained.
Abstract
Aseptic rearing technology is a method of culturing insects under sterile or almost sterile conditions, which can effectively eliminate the influence of external microorganisms on insect microbiota and thus promote the rapid development of insect microbiota research. Nasonia (wasp genus) is a parasitic wasp insect that has many advantages, such as a short lifespan, high genetic variation, easy operation, etc., and is widely used as an insect model system. Unlike antibiotic treatment, which can only reduce the number of microorganisms in animals, aseptic rearing techniques can control both the composition and quantity of microorganisms in animals, further facilitating the study of host-microbe interactions. However, previous versions of Nasonia rearing medium (NRM) have some defects and problems, such as a complex and time-consuming preparation process, easy contamination by bacteria or fungi, and short storage time. Therefore, this study solves these problems by optimizing the tools used in the NRM preparation process, storage conditions, and component ratios. The optimized medium could allow storage at -20 °C for at least 3 months and eliminate the possibility of NRM contamination during feeding sterile wasps. This further improves the survival rate and health level of aseptic Nasonia, which is important for using Nasonia as a model for microbial research.
Introduction
Germ-free animals are animals that have no detectable living microorganisms and parasites1. Germ-free embryos can be obtained by dissecting the mother under aseptic conditions and subsequently raised in barrier systems2. Such animals can be used to study the effects of microorganisms on animals, such as on the intestinal microbiota, immune system, and metabolism1. With certain technical means, many insects and even mammals can be rendered sterile3,4. Germ-free animals have a unique role and have been widely used in various aspects of microbiology research5. For example, the use of germ-free Nasonia wasps has revealed that microorganisms can help hosts adapt to new environments under long-term exogenous environmental stress6,7.
Nasonia parasitoids are small parasitic wasps that inject their eggs into the pupae of flies4. There are four known species of Nasonia, including Nasonia vitripennis, Nasonia longicornis, Nasonia giraulti, and Nasonia oneida8. N. vitripennis can be found worldwide, while the other three species have limited ranges in North America4. Nasonia parasitoid wasps are regarded as ideal model insects because of their characteristics, such as easy cultivation, short reproduction cycle, sequenced genome, and long term diapause8,9. They can be used to study various aspects of insect evolution, genetics, development, behavior, and symbiosis10. Moreover, Nasonia parasitoid wasps can also help control harmful flies in agriculture and disease11. The successful establishment of a sterile insect system involves two major steps: (1) sterilization of the embryos and (2) provision of sterile food to the larvae in vitro. In order to obtain sterile food, Brucker and Bordenstein12 developed Nasonia rearing medium (NRMv1) in 2012 by using chemicals such as antibiotics, bleach, and fetal bovine serum to kill bacteria12. However, the chemical sterilization method resulted in low survival and eclosion rates of N. vitripennis13. Then, in 2016, Shropshire et al. developed NRMv2 by using a filter sterilization method instead of a chemical sterilization method to eliminate the hazards of antibiotics and other substances, and optimized the breeding process13. Unfortunately, this method still has some disadvantages, such as the challenges associated with preparing and using the medium, as well as the risks of drowning, underfeeding, or dehydration for the embryos, larvae, and closed pupae14. Wang and Brucker14 recently improved the Nasonia rearing media version 3 (NRMv3) and the germ-free rearing version 2 (GFRv2) protocols. These improvements reduced the cost and media consumption. However, the NRMv3 has a very short storage time and is highly susceptible to contamination.
Building on NRMv3, the NRM preparation tool storage method and nutrient ratio were optimized in this study. This methodological refinement facilitates using N. vitripennis as a model for microbiome studies. Compared with the NRMv3 developed by Wang et al.14, the improved tool for squeezing Sarcophaga bullata pupa, one of the NRM raw materials, greatly enhances the production efficiency of S. bullata pupa tissue fluid compared with the 60 mL syringe with a bottom hole used by Wang et al.14. We adjusted the nutrient ratio of NRM, which led to a certain increase in the survival rate of germ-free Nasonia wasps without affecting their development time. In addition, the NRM was packed into small-capacity centrifuge tubes (1.5 mL) and frozen in a -20 °C refrigerator to extend the storage time. It is worth noting that while we used the housefly Lucilia sericata as the host and source for NRM preparation, this protocol can likely be adapted for other Nasonia hosts that are available in the laboratory.
Protocol
1. Preparation of germ-free Nasonia rearing medium
- Place the commercially available L. sericata pupae (see Table of Materials) on a surface that can accommodate all the pupae, such as a tray or a sheet of paper. Discard any underdeveloped larvae, dark old pupae, empty pupal shells, sawdust, or other impurities. Keep only young pupae that are brownish-red in color and transfer them to a beaker (approximately 3,000-4,000 pupae).
NOTE: After conducting many experiments, it was found that the medium made from the dark old pupae easily turned dark and stopped the development of germ-free wasps. This phenomenon did not occur when using the young brownish-red pupae to produce medium. The reason for this is still unclear and requires further investigation to verify it. - Add enough deionized water to the beaker to cover the entire surface of the pupae. Wrap the mouth of the beaker with tinfoil or gauze, shake the beaker to clean the impurities on the surface of the pupae, and then pour out the water. Repeat three to five times.
- Place the cleaned L. sericata pupae in the filter tank of a garlic press, squeeze hard, and collect the tissue fluid of the L. sericata pupae in a 50 mL sterile centrifuge tube. Then, pour out the pupa dregs and put new pupae in the filter tank of the garlic press until all are squeezed.
NOTE: Compared to the needle extrusion device used by Wang et al.14, the garlic press is more convenient and can separate the interstitial fluid more thoroughly. This advancement lays the foundation for large-scale production and long-term storage of NRM. - Centrifuge the mixture at 4 °C (25,000 x g) for 10 min. After centrifugation, the mixture will be separated into three layers from bottom to top: sediment layer, protein layer, and fat layer (Figure 1).
- To prevent clogging during filtration, aspirate the protein layer using a 18 G sterile needle and transfer it to a new 50 mL conical sterile polypropylene centrifuge tube.
- Add commercial liquid Drosophila medium (see Table of Materials) to the protein extract at a 1:1 ratio.
NOTE: More convenient commercialized L. sericata was used as the host and NRM raw material, compared with NRMv3. Therefore, the nutritional ratio was adjusted from 1:2 to 1:1 based on NRMv3, which was more suitable for the growth and development of sterile Nasonia (Figure 2). - Filter the mixed medium (Drosophila medium and protein extract of L. sericata pupa) using a vacuum filtration system with different pore sizes (8, 1.2, 0.8, and 0.45 µm; see Table of Materials) to remove particles of different sizes. To prevent clogging, change the filter paper when the flow slows down.
NOTE: The use of a new 50 mL sterile centrifuge tube is required for each filtration. - Centrifuge the liquid again at 4 °C (15,000 x g) for 10 min and sterilize the supernatant medium through a 0.22 µm syringe filter. Repeat the above steps once.
- Store the culture medium at -20 °C after aliquoting (Figure 3A) for long-term storage.
NOTE: To prevent contamination and repeated freezing and thawing, it is recommended to open each tube of NRM only once when using the culture medium after packaging (Figure 3A).
2. Germ-free egg collection
- To ensure a stable male-to-female ratio in offspring, place the pupae in a Drosophila vial during the pupal stage at a ratio of 50 females to 15 males because of their haploid genetic characteristics (males develop from haploid cells, while females develop from diploid cells that result from fertilized eggs)4,15.
- After emerging to adults, allow the males and females to mate for 1.5 days, and then put about 40 L. sericata pupae into the vial. Within 12-24 h after parasitization, use a sterile dissecting needle to carefully open one end of the pupal shell under a stereomicroscope (Figure 4).
- Hold the other end by hand and find the wasp embryos. Use a dissecting needle to transfer embryos from the surface of the L. sericata pupa tissue to a sterile cell strainer with phosphate-buffered saline (PBS)14.
NOTE: Older L. sericata pupae should be used for parasitism because the dry L. sericata pupa tissue is easier to transfer embryos. In order to ensure the smooth transfer of eggs from the L. sericata pupa tissue surface, try to be careful not to puncture the tissue when the dissecting needle opens the pupal case so that the interstitial fluid does not flow out.
- Place 20-30 embryos on a cell strainer and wash them evenly with 1,000 µL of 10% commercial sodium hypochlorite solution (see Table of Materials), followed by another wash with 1,000 µL of 1x sterile PBS. Then, wash once with 1,000 µL of 70% ethanol solution and wash three times with 1,000 µL of 1x sterile PBS.
- First, place a 5 mm diameter polypropylene mesh sheet (see Table of Materials) that has been pre-wetted with 1x PBS into a 24-well plate. Then, using a sterilized small brush, gently brush the wasp embryos on the cell strainer onto the polypropylene mesh sheet. This can be done for all four wells in a vertical column of the 24-well plate.
3. Germ-free wasp rearing
- Add 50 µL of NRM to each well in a laminar flow hood. To maintain a humid environment for growth, add 1 mL of sterile water between each well in the 24-well plate. A small beaker containing 30 mL of sterile water can also be placed in the 5 L sterile plastic box with the 24-well plate.
- Throughout the entire experiment, keep the sterile plastic box and the 24-well plate in a climate chamber at a constant temperature of 25 ± 2 °C and under constant light.
- Before transferring and adding NRM every day, thaw the frozen NRM on a laminar flow bench. In a sterile environment, use alcohol-disinfected tweezers to transfer the polypropylene mesh with larvae on it from one well to another. Lastly, add 50 µL of NRM that has been equilibrated to room temperature (Figure 3B). Repeat the above operations every day until pupae are observed.
NOTE: With the development of wasps, the amount of NRM should be increased appropriately to ensure that the germ-free wasps have sufficient nutrition. For example, the fourth instar larvae were supplied with 60 to 70 µL of NRM. Since different developmental stages may coexist in the same well, use a sterilized brush to transfer out the pupae. Add a small amount of NRM to the remaining larvae. Use aseptic techniques to avoid microbial invasion and pollution. - After feeding for 9 to 11 days, more than 80% of the larvae develop into white or yellow pupae. At this time, move the filter to a clean well plate and stop adding culture medium to wait for eclosion.
Representative Results
The preparation efficiency of NRM was greatly improved by improving the preparation tools. In addition, the problem of NRM pollution in the feeding process was eliminated by optimizing the strategy and preservation method. At the same time, the adjusted NRM had a more suitable nutritional ratio for the growth and development of germ-free wasps with L. sericata as hosts. The survival rate of germ-free wasps from larvae to pupae was significantly improved compared to germ-free wasps reared with NRMv3 using GFRv2 (Figure 2). Further, the generation period was not different between the aseptic wasps and the conventionally reared wasps (about 14 days a generation) (Figure 5). These improvements are highly significant for developing the Nasonia model organism and studying the host-microorganism interaction mechanism.

Figure 1: Mixture separated by centrifugation. Please click here to view a larger version of this figure.
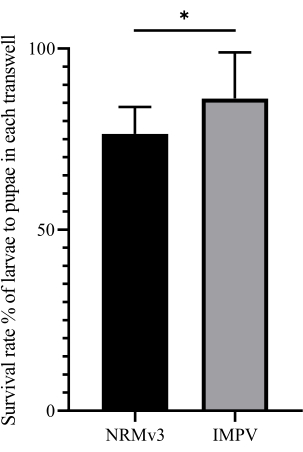
Figure 2: The survival rate of larvae to pupae in each transwell. The survival rate improved significantly compared to germ-free females reared on NRMv3 with GFRv2 (two independent-sample T-tests, *p < 0.05). Please click here to view a larger version of this figure.
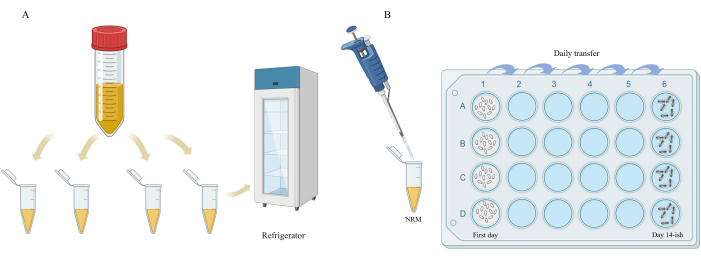
Figure 3: Schematic diagram of NRM dispensing and germ-free wasp rearing. (A) The filtered NRM from the 50 mL centrifuge tube was aliquoted into sterile 1.5 mL centrifuge tubes and frozen. Each tube of NRM was only opened once. (B) There was a polypropylene mesh sheet underneath the embryos of the Nasonia wasps. The centrifuge tube contained the packaged NRM. "Day 14-ish" means adult emergence occurs on about the 14th day. Please click here to view a larger version of this figure.
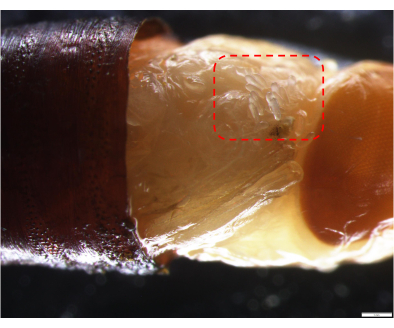
Figure 4: Images of transferred wasp eggs under the stereomicroscope. The eggs laid by the Nasonia wasp are within the red dashed box. Please click here to view a larger version of this figure.
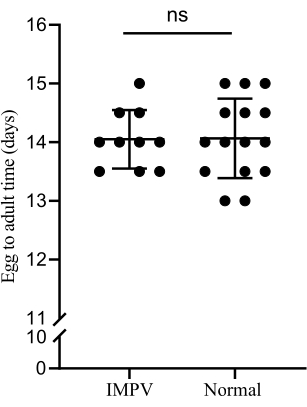
Figure 5: Development time comparison. The development time of germ-free females (about 14 days) was not significantly different from that of the conventionally reared wasps when using the improved NRM of this study (two independent-sample T-tests, ns, not significant, p > 0.05). Please click here to view a larger version of this figure.
Discussion
With the application of high-throughput detection technologies such as genomics and metabolomics, researchers have gradually realized that there is huge genetic diversity and metabolic complexity in the gut microbiota16. These symbiotic bacteria are closely related to various physiological or pathological states, such as host nutritional metabolism, tumors, immunity, and aging through complex interactions with the host17. However, the research related to the network of the composition and function of the microbial population in the host is very difficult because the composition of microorganisms in each natural individual is not exactly the same16. Therefore, an ideal model is needed to explore the interaction and related mechanisms between microorganisms and hosts. Nasonia wasps can be used as an ideal model in the microbiome field because of its many biological advantages and the characteristics of germ-free wasps6. Compared to antibiotic treatment, using sterile animals allows for control over the composition and quantity of microorganisms, which is crucial for studying microbial function. Additionally, antibiotic treatment may lead to several side effects, including antibiotic resistance, intestinal dysfunction, and intestinal barrier damage18. Therefore, developing a more efficient method to obtain germ-free wasps is of great significance for studying host-microbiome interactions.
The acquisition of germ-free Nasonia mainly includes two steps – obtaining sterile embryos and providing sterile food. The embryos were obtained by dissecting L. sericata pupae 12-24 h after N. vitripennis parasitization under a stereoscope13. This step required enough skill to move the embryos faster and without damaging them to the cell strainer. Based on experience, it is more convenient to dissect the old L. sericata pupae that are black. However, young red L. sericata pupae are preferred for preparing NRM.
Moreover, the normal development of sterile embryos depended on the production of sterile food and the operation process of changing the medium every day12. We found that the main source of contamination for rearing germ-free wasps by Wang et al.'s14 GFRv2 was NRM. Therefore, the culture was packed and stored in the refrigerator at -20 °C in the present study, ensuring that each medium is only opened once. This not only had no effect on the growth and development of Nasonia wasps, but also greatly prolonged the culture medium's storage time and eliminated the chance of NRM contamination. In total, this method further increased the survival rate of aseptic wasps.
However, the present method still has some drawbacks. For example, the daily transfer of sterile embryos in a 24-well plate may increase the risk of pollution. In addition, the process of squeezing a mass of fly pupae is also time-consuming and labor-intensive. If the specific nutrients that play a role in the fly pupae can be identified, they could be purchased and added directly to the medium, which would greatly improve the efficiency of NRM preparation. It is expected that this method will be further optimized in future research.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Funding: this work was supported by the National Science Foundation of China (32270538), the National Key R&D Program of China (2022YFF0710603), the Natural Science Foundation of Beijing (6222046), and the CAS strategic funding via the CAS-CSIRO funding scheme (152111KYSB20210011) awarded to G.H.W. Author contributions: all authors developed the scope and focus of the review and contributed to the writing of the manuscript.
Materials
| 0.22 Sterile vacuum filter | NEST | 331011 | |
| 10% SodiumHypochlorite | LIRCON | XB-84BS-1 | |
| 1x PBS solution | Solarbio | P1020 | |
| 200 mesh nylon net | BIOBYING | BY-378Z | |
| 24 well-plate | NEST | 702001 | |
| 8, 1.2, 0.8, and 0.45 µm filters | Shanghai Xingya Purification Material Factory | HN-AA-JT-10079 | |
| Absolute ethyl alcohol | Macklin | E809057-500ml | |
| Cell Strainer | BIOLOGIX | 15-1100 | |
| Commercial Drosophila Medium | Boer | B645446-500ml | |
| Dissecting needle | Bioroyee | 17-9140 | |
| Garlic press | Taobao | No Catalog numbers | Purchase on Taobao |
| Lucillia sericata pupae | Hefei Dayuan Biotechnology Co., Ltd. | No Catalog numbers | Purchase on Taobao |
| Small writing brush | Cestidur | BL0508 | |
| Stereoscope | SOPTOP | RX50 | |
| Tweezers | SALMART | A109001-56 |
References
- Diviccaro, S., et al. Exploring the impact of the microbiome on neuroactive steroid levels in germ-free animals. International Journal of Molecular Sciences. 22 (22), 12551 (2021).
- Pang, X., et al. Inter-species transplantation of gut microbiota from human to pigs. The ISME Journal. 1 (2), 156-162 (2007).
- Uzbay, T. Germ-free animal experiments in the gut microbiota studies. Current Opinion in Pharmacology. 49, 6-10 (2019).
- Zhu, Z., Liu, Y., Hu, H., Wang, G. -. H. Nasonia-microbiome associations: a model for evolutionary hologenomics research. Trends in Parasitology. 39 (2), 101-112 (2022).
- Li, J., Wei, H. Establishment of an efficient germ-free animal system to support functional microbiome research. Science China Life Sciences. 62 (10), 1400-1403 (2019).
- Wang, G. H., et al. Changes in microbiome confer multigenerational host resistance after sub-toxic pesticide exposure. Cell Host Microbe. 27 (2), 213-224 (2020).
- Wang, G. H., Dittmer, J., Douglas, B., Huang, L., Brucker, R. M. Coadaptation between host genome and microbiome under long-term xenobiotic-induced selection. Science Advances. 7 (19), (2021).
- Dittmer, J., Brucker, R. M. When your host shuts down: larval diapause impacts host-microbiome interactions in Nasonia vitripennis. Microbiome. 9 (1), 85 (2021).
- Dittmer, J., et al. Disentangling a holobiont-recent advances and perspectives in Nasonia wasps. Frontiers in Microbiology. 7, 1478 (2016).
- Brooks, A. W., Kohl, K. D., Brucker, R. M., van Opstal, E. J., Bordenstein, S. R. Phylosymbiosis: relationships and functional effects of microbial communities across host evolutionary history. PLoS Biology. 14 (11), e2000225 (2016).
- Heavner, M. E., et al. Partial venom gland transcriptome of a Drosophila parasitoid wasp, Leptopilina heterotoma, reveals novel and shared bioactive profiles with stinging Hymenoptera. Gene. 526 (2), 195-204 (2013).
- Brucker, R. M., Bordenstein, S. R. In vitro cultivation of the hymenoptera genetic model, Nasonia. PLoS One. 7 (12), e51269 (2012).
- Shropshire, J. D., van Opstal, E. J., Bordenstein, S. R. An optimized approach to germ-free rearing in the jewel wasp Nasonia. PeerJ. 4, e2316 (2016).
- Wang, G. H., Brucker, R. M. An optimized method for Nasonia germ-free rearing. Scientific Reports. 12 (1), 219 (2022).
- Brucker, R. M., Bordenstein, S. R. The hologenomic basis of speciation: gut bacteria cause hybrid lethality in the genus Nasonia. Science. 341 (6146), 667-669 (2013).
- Fontaine, C. A., et al. How free of germs is germ-free? Detection of bacterial contamination in a germ free mouse unit. Gut Microbes. 6 (4), 225-233 (2015).
- Mazmanian, S. K., Liu, C. H., Tzianabos, A. O., Kasper, D. L. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell. 122 (1), 107-118 (2005).
- Weersma, R. K., Zhernakova, A., Fu, J. Interaction between drugs and the gut microbiome. Gut. 69 (8), 1510-1519 (2020).

