Fabricating Complex Culture Substrates Using Robotic Microcontact Printing (R-µCP) and Sequential Nucleophilic Substitution
Summary
Cell culture substrates functionalized with microscale patterns of biological ligands have immense utility in the field of tissue engineering. Here, we demonstrate the versatile and automated manufacture of tissue culture substrates with multiple, micropatterned poly(ethylene glycol) brushes presenting orthogonal chemistries that enable spatially precise and site-specific immobilization of biological ligands.
Abstract
In tissue engineering, it is desirable to exhibit spatial control of tissue morphology and cell fate in culture on the micron scale. Culture substrates presenting grafted poly(ethylene glycol) (PEG) brushes can be used to achieve this task by creating microscale, non-fouling and cell adhesion resistant regions as well as regions where cells participate in biospecific interactions with covalently tethered ligands. To engineer complex tissues using such substrates, it will be necessary to sequentially pattern multiple PEG brushes functionalized to confer differential bioactivities and aligned in microscale orientations that mimic in vivo niches. Microcontact printing (μCP) is a versatile technique to pattern such grafted PEG brushes, but manual μCP cannot be performed with microscale precision. Thus, we combined advanced robotics with soft-lithography techniques and emerging surface chemistry reactions to develop a robotic microcontact printing (R-μCP)-assisted method for fabricating culture substrates with complex, microscale, and highly ordered patterns of PEG brushes presenting orthogonal ‘click’ chemistries. Here, we describe in detail the workflow to manufacture such substrates.
Introduction
The ability of PEG-grafted surfaces to display covalently bound biochemical ligands while simultaneously maintaining inherent non-fouling properties make them an ideal choice for engineering custom microscale environments on culture substrates1,2,3. The biospecific interactions mediated by ligand conjugated PEG brushes enables reductionistic analysis of the effects of biochemical cues found within complex in vivo tissue microenvironments on individual cell phenotypes. Furthermore, bio-orthogonal “click” chemistries can be used to facilitate directional immobilization of ligands so that they are presented in native conformations4-6. Thus, microscale spatial patterning of PEG brushes is a versatile tool to create designer in vitro niches to investigate cell signaling induced by immobilized biochemical cues6,7.
A common method for generating spatial patterns of biochemical cues entails microcontact printing (μCP) gold-coated substrates with patterns of PEG conjugated alkanethiols. Then, the micropatterned self-assembled monolayers (SAMs) of PEG-ylated alkanethiols restricts physical adsorption of biochemical molecules, e.g., proteins, only to non-patterned regions of the substrate8,9. However, the SAMs generated by this technique are sensitive to oxidation in long term cell culture media. Thus, μCP’d alkanethiol SAMs are often further grafted with PEG polymer brushes using surface-initiated atom transfer radical polymerization (SI-ATRP) to increase the region’s non-fouling stability10. Specifically, μCP of the alkanethiol polymerization initiator, ω-meraptoundecyl bromoisobutyrate, on gold-coated surfaces followed by SI-ATRP of poly(ethylene glycol) methyl ether methacrylate (PEGMEMA) monomers generates surfaces with micropatterned long-term, stable, and non-fouling PEG brushes. Moreover, these are capable of being further modified to present diverse chemical moieties11.
Taking advantage of this property, Sha et. al. developed a method to engineer culture substrates with multicomponent PEGMEMA brushes presenting orthogonal “click” chemistries. In this method, they use a series of μCP/SI-ATRP steps interspersed with sequential sodium azide, ethanolamine, and propargylamine nucleophilic substitutions to create culture substrates presenting microscale patterns of multiple immobilized ligands6. While the potential of using such chemistries in conjunction with manual μCP to engineer novel culture substrates is immense, it is limited by the precision and accuracy with which multiple μCP steps can be aligned on a single substrate. A high level of precision and accuracy would be required to reproducibly manufacture complex in vitro niches using these versatile techniques.
To address this limitation, several automated and semi-automated μCP systems have been generated. Chakra et. al. developed a μCP system in which custom stamps are placed on a rail system and brought into conformal contact with gold-coated slides using a computer-controlled pneumatic actuator. However, this method requires the precise fabrication of custom stamp designs and reports a 10 μm precision with no report of the accuracy achieved when performing multiple μCP steps12. More recently, a method utilizing an integrated kinematic coupling system reported precision below 1 μm using a single pattern, but were unable to accurately align multiple patterns due to a lack of precise control of stamp features from mold to mold13. Additionally, both of the previous methods require the substrate to remain fixed between patterning steps, thereby significantly limiting the diversity of surface modification chemistries that can be utilized. Here, we describe an automated R-μCP system capable of accurate and precise alignment of multiple μCP steps while allowing maximal flexibility in stamp design and fabrication. Furthermore, the patterned substrates can be repeatedly removed from the system between stampings, thereby permitting the use of diverse substrate modification chemistries, including sequential nucleophilic substitutions. Substrates engineered using such chemistries have been used for cell culture previously by both us6,14 and others7. Thus, we have merged R-μCP and sequential nucleophilic substitution reactions to develop a method for scalable manufacture of culture substrates with complex and micropatterned biochemical cues.
Protocol
1. Generating Elastomeric Stamps
- To generate the PDMS stamp’s silicon masters, design the photomask’s feature patterns using computer-aided design software.
- Design the first pattern as a 20 x 20 array of annuli with 300 μm inner diameter (ID) and 600 μm OD with 1,200 μm center-to-center spacing.
- Design the second pattern as a 20 x 20 array of annuli with 600 μm ID and 900 μm OD with 1,200 μm center-to-center spacing.
- Additionally, place 1 x 1 mm2 square reference marks at all four corners of each array design spaced 1,200 μm center-to-center from the corner pattern at a 45° angle.
- Fabricate the silicon masters for use in this experiment with 1:1 aspect ratios, correlating to a 300 μm feature depth using standard lithography techniques detailed elsewhere15 or in collaboration with a microfluidic foundry.
NOTE: Feature depths lower than 100 μm can lead to abnormal deformation of stamps prior to contact with substrate surfaces.
NOTE: This protocol begins with having already obtained silicon masters with the described patterns of photoresist, which requires specialized equipment and clean rooms to create. It is best to consult with a fabricator or core facility to create these patterned masters.
- Silanize silicon masters O/N by incubation with (tridecafluoro-1,1,2,2-tetrahydroocty) trichlorosilane vapor.
NOTE: Silane vapor is highly toxic and should only be handled in a chemical fume hood. - Create inverse replicas of the silicon masters by curing a 10:1 ratio of PDMS pre-polymer and PDMS curing agent on top of the silanized silicon masters in a petri dish O/N at 60 °C.
- Remove the PDMS stamps from the silicon masters, and bond the stamps to acrylonitrile butadiene styrene (ABS) backings or any other rigid materials using glue.
NOTE: The material of choice need not be transparent or biocompatible. However, it must provide a flat surface for interface with the robotic tooling and higher rigidity than PDMS.
2. Preparing Coverslides
- Rinse microscope coverslides (24 mm x 50 mm #1) in toluene, methanol and sonicate in acetone for 1 min before rinsing with ethanol and drying under a gentle nitrogen stream.
NOTE: Handle toluene and acetone only in a chemical fume hood. - Coat coverslides with ~3.5 nm titanium (Ti) followed by 18.0 nm gold (Au) using a focused electron-beam evaporator.
NOTE: This procedure is compatible with a variety of gold-coated substrates including silicon and polystyrene. While it is possible to generate these on-site, gold-coated substrates are also available via commercial suppliers.
NOTE: Ti layers should be at least 3 nm thick, while Au layers on substrates should be at least 5 nm thick and can range all the way to 1mm in thickness depending on the ideal optical properties of the final substrate16,17. Substrates are not required to be optically clear for fabrication; however, it facilitates microscopic analysis of fabricated substrates. - Rinse gold-coated coverslides with ethanol and dry under gentle nitrogen immediately before use.
3. R-μCP of Outer Annulus
- Before R-μCP with the selectively compliant articulated robot arm (SCARA) system, calibrate the robotic tool effectors and accompanying dual camera systems using the system’s software.
NOTE: These implements are pictured in Figure 1.
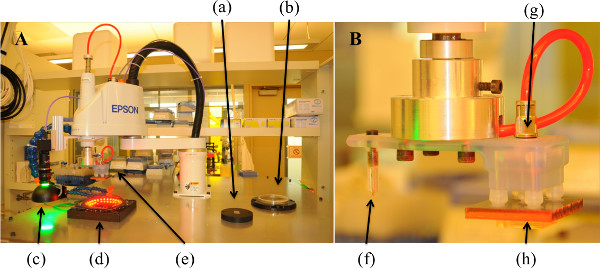
Figure 1. R-μCP System and Robotic Arm Tooling. (A) Large-scale view of R-μCP system with all tooling and fixtures, (a) vacuum chuck, (b) reagent bath, (c) downward facing camera, (d) stamp nesting fixture, (e) robotic tool. (B) Robotic arm tooling depicting (f) diamond-tipped etching tool and (g) pneumatic suction tool holding a (h) ABS-backed PDMS stamp.
- Manually place a PDMS stamp with 600 μm ID and 900 μm OD protruding annuli face down in the stamp nesting fixture. Manually place a freshly cleaned gold-coated coverslide on top of the vacuum chuck, displayed in Figure 1A, and immobilize it using the connected lab vacuum.
- Using the robot controls, align the downward facing camera over the center point of the gold-coated coverslide, as in Figure 2A.
NOTE: This point can initially be defined by visual inspection and does not require great accuracy, as the PDMS stamp is much larger than the gold-coated slide. - Instruct the robotic arm to etch four reference “X” marks at the vertices of a square with dimensions 3.8 mm by 3.8 mm centered on the gold-coated coverslide using the diamond-tipped etching tool attached to the robotic arm. (Depicted in Figure 2B; AUTOMATED PROCESS)
NOTE: This ensures all four reference marks are within one visual frame of the downward facing camera. - Using the robots pneumatic suction tooling, pick-up and hold the PDMS stamp 1 mm above the stamp nesting fixture as in Figure 2C. (AUTOMATED PROCESS)
- Using the upward facing camera and LED illumination ring pictured in Figure 2D and E, visualize the stamp’s square reference marks and identify them with the camera software 10 times in order to determine the stamp’s average X, Y, and angular offset from the center of the robot tooling axis. (AUTOMATED PROCESS)
- As in Figure 2A, use the downward facing camera to visualize and detect the coverslide’s etched reference marks 10 times to determine the coverslide’s average X, Y center locations and angular offset from the center of the robot tooling axis. (AUTOMATED PROCESS).
NOTE: An internal computation is performed by the robotic system’s software to precisely align the stamp’s and coverslip’s X, Y center locations and angular offsets in future R-μCP steps. - 3.8) Place the stamp in a bath of alkanethiol ATRP initiator, ω-mercaptoundecyl bromoisobutyrate (2 mM in ethanol) as depicted in Figure 2F. (AUTOMATED PROCESS)
- Remove the stamp from the alkanethiol solution and place it over nitrogen stream pressurized to 0.48 bar (5 psi) to evaporate the ethanol as in Figure 2G. After 1 min increase the nitrogen stream’s pressure is to 1.03 bar (15 psi) to ensure uniform dryness. (AUTOMATED PROCESS)
NOTE: Incomplete drying will result in total or partial loss of pattern fidelity. - After drying, move the stamp over the calculated center position of the gold-coated slide and lower it at 100 μm increments while monitoring Z-axis motor force as depicted in Figure 2H.
NOTE: This is communicated as torque experienced by the Z-axis motor and displayed in the robot guidance software. (AUTOMATED PROCESS) - Stop lowering the stamp once the predetermined pressure of 79.2 kPa has been achieved, and maintain contact with the gold-coated slide for 15 sec. (AUTOMATED PROCESS)
NOTE: The pressure values here are optimized for the current stamp design and feature height. If the stamp design is changed, specifically for high aspect ratio stamps, tune these accordingly by a trial and error process. - Slowly remove the stamp from the gold-coated slide and place the stamp back in the stamp nesting fixture. (AUTOMATED PROCESS)
4. SI-ATRP of PEGMEMA on Micropatterned Coverslides
- Release vacuum pressure holding the micropatterned coverslide, and transfer it to a 50 ml Schlenk flask. Seal and degas the Schlenk flask using a vacuum pump.
- Add 5.5 ml of ATRP reaction mixture containing the macromonomer PEGMEMA (208.75 mmol), water (34.4 ml), methanol (43.8 ml), copper(II) bromide (1 mmol), and 2’,2-bipyridine (3 mmol) to the Schlenk flask.
- Add 0.5 ml of L-sodium ascorbate (454.3 mM) in water to initiate the reaction, and allow it to continue for 16 hr at RT under inert gas.
NOTE: Following addition of L-sodium ascorbate, reaction color will shift from light green to dark brown as shown in Figure 3A-B.
NOTE: The reaction can continue beyond 16 hr, but it will not significantly increase the length of the PEG brushes.

Figure 3. Initiation of SI-ATRP. (A) Initiation of reaction and (B) subsequent color change following addition of L-sodium ascorbate. (C) Microscope image of micropatterned coverslide surface following SI-ATRP procedure. Scale bar 1 mm.
- Remove the micropatterned coverslide from the Schenk flask and rinse with ethanol, water, and ethanol and dry under a gentle nitrogen stream.
NOTE: Following SI-ATRP, surface modifications should be visible to the eye and can be imaged and analyzed under a microscope as in Figure 3C.
NOTE: This is the simplest method to test the precision of the trial as it obviates the need to immunostain the substrates.
5. Azide Functionalization of Micropatterned PEGMEMA Chains
- Place the micropatterned coverslide in a 20 ml glass reaction vial.
- Add 6 ml of N,N-dimethylformamide (DMF) containing 100 mM sodium azide to the reaction vial. Maintain this reaction at 37 °C for 24 hr.
NOTE: Handle DMF only in a chemical fume hood. Exercise caution when weighing out sodium azide. - Upon completion, remove the micropatterned coverslide from the reaction vial and rinse with ethanol then water and dry under a nitrogen stream.
6. Passivation of Bromine Functionalized PEGMEMA Chains
- Place micropatterned coverslide in 20 ml glass reaction vials.
- Add 6 ml of dimethyl sulfoxide (DMSO) containing 100 mM ethanolamine and 300 mM triethylamine to the reaction vial. Hold this reaction at 40 °C for 24 hr.
NOTE: Handle DMSO, ethanolamine, and triethylamine only in a chemical fume hood. - Upon completion remove the micropatterned coverslide from the reaction vial, rinse with ethanol then water and dry under a nitrogen stream.
7. R-μCP of Inner Annulus and SI-ATRP of PEGMEMA
- Carry out R-μCP steps as previously described in Steps 3.2 and 3.6 – 3.12, substituting a PDMS stamp with 300 μm ID and 600 μm OD protruding annuli, such that the annuli with smaller features are placed inside the previously micropatterned annuli.
NOTE: The pressure threshold for this stamp is 132.0 kPa. As previously mentioned, these pressure settings are optimized for the stamp features being used in this experiment. - Carry out SI-ATRP steps as previously described in steps 4.1 – 4.4.
8. Acetylene Functionalization of Micropatterned PEGMEMA Chains
- Place micropatterned coverslide in a 20 ml glass reaction vial.
- Add 6 ml of DMSO containing 100 mM propargylamine to the reaction vial. Maintain this reaction at RT for 24 hr.
NOTE: Handle DMSO and propargylamine only in a chemical fume hood. - Upon completion remove the micropatterned coverslide from the reaction vial and rinse with ethanol then water and dry under a nitrogen stream.
9. Copper-catalyzed “Click” Biotinylation of Acetylene Terminated PEGMEMA Chains
- Place micropatterned coverslide in a 20 ml glass reaction vial.
- Add 6 ml of copper sulfate (15 mM) / Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA, 30 mM) (1:1 v/v water/DMF) containing 562 μM azide-PEG4-Biotin conjugate to the reaction vial. Add 1.2 ml of L-ascorbic acid (0.15 mM) in water to the mixture to initialize the reaction.
- Bubble nitrogen through the reaction solution for 10 sec, seal the vial with Parafilm, and react for 24 hr at RT.
- Upon completion remove micropatterned coverslide from the reaction vial, rinse with water and place it in a 12-well polystyrene dish.
10. Immunofluorescent Detection of Biotinylated Acetylene Groups
- Block micropatterned coverslide in DPBS (3% Donkey Serum) for 1 hr at RT. Stain for biotinylated acetylene groups with Streptavidin-546 conjugate (2 μg/ml) in DBPS (3% Donkey Serum in PBS) for 2 hr at RT.
- Rinse micropatterned coverslide 5 times with DPBS for 10 min with gentle agitation.
NOTE: Figure 4B depicts an image of the slide after this step is completed.
11. Copper-free “Click” Biotinylation of Azide Terminated PEGMEMA Chains
NOTE: If desired, this substrate modification step can be performed in situ during cell culture.
- Leave micropatterned coverslide in 12-well polystyrene dish following DBPS rinses.
- Add 2 ml of DBPS (or cell culture media) containing 20 μM DBCO-PEG4-Biotin, and allow this reaction to continue for 24 hr at RT (or at 37 °C in an incubator).
- Upon completion, rinse micropatterned coverslide 5 times with DPBS (or simply perform a media change).
12. Immunofluorescent Detection of Biotinylated Azide Groups
- Stain for biotinylated azide groups with Streptavidin-488 conjugate (2 μg/ml) in 2 ml DPBS (3% Donkey Serum) for 2 hr at RT.
- Rinse micropatterned coverslide 5 times in DPBS for 10 min with gentle agitation.
- Image micropatterned coverslide using confocal fluorescence microscope. Figure 4A-C depicts images of the slide after this step is completed.
NOTE: When using substrates for tissue culture assays, skip sections 10 and 12 of this protocol. After the desired degree of functionalization, sterilize the engineered coverslide by rinsing both sides with 100% ethanol and drying under a nitrogen stream. Transfer the slide to a sterile biological safety cabinet and rinse the slide with PBS five times before proceeding with cell seeding and culture.
Representative Results
The use of manual alignment μCP techniques to engineer culture substrates with arrays of PEG-grafted brushes functionalized with orthogonal “click” chemistries has been demonstrated in previous work6. However, this offers minimal control of pattern orientation and often results in overlap of functionalized areas. Here, a novel R-μCP system is used to overcome this limitation, and its ability to accurately pattern an array of PEG brush annuli with 300 μm ID and 600 μm OD presenting terminal alkyne groups within a separate array of PEG brush annuli with 600 μm ID and 900 μm OD presenting terminal azide groups is demonstrated14. Following the reaction of alkyne presenting PEG brushes with Azide-PEG3-Biotin and the reaction of azide presenting PEG brushes with DBCO-PEG4-Biotin, the substrate was immunostained with fluorescent probes and imaged using a confocal microscope (Figure 4). Analysis of these images using a custom MATLAB program calculated that the two PEG brush arrays were aligned with sub-10 μm accuracy, i.e., X ~ 6.4 μm, Y ~ 1.7 μm, and θ ~ 0.02°, which is at the manufacturer’s cited limit of our SCARA model (i.e., ~10 μm) and indicative of the R-μCP system’s prior performance14. Thus, we believe that the use of higher-end SCARAs in this system would provide even lower, sub-micron resolution. These results demonstrate the versatile substrate engineering capabilities enabled by the combined use of the R-μCP system and sequential nucleophilic substitution reactions. The minimal cross-reactivity evidenced by fluorescent labeling of the orthogonal surface chemistries serves to illustrate the potential of this system for precise immobilization of biochemical cues to generate complex culture substrates for tissue engineering applications.

Figure 2. Schematic of R-μCP Process. (A) Downward facing camera visualizing immobilized gold-coated slide, (B) etching reference marks on gold-coated coverslide, (C) removing PDMS stamp from stamp nesting fixture, (D–E) visualizing PDMS stamp reference marks with upward facing camera, (F) placing PDMS stamp in alkanethiol initiator bath, (G) drying alkanethiol initiator solvent over nitrogen streams, and (H) stamping alkanethiol coated PDMS stamp on gold-coated coverslide.
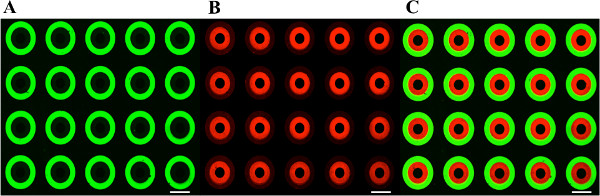
Figure 4. Immunostained Images of MicroPatterned Slide. R-μCP micropatterned coverslide orthogonally functionalized to present terminal (A) azide and (B) alkyne groups, biotinylated using copper-free and copper-mediated click chemistry, and detected with Streptavidin-488 and -546 respectively. (C) Overlay of both fluorescent channels. All scale bars 500 μm.
Discussion
Ideal substrates for tissue engineering would be bioinspired and thereby recapitulate the spatial distribution of critical bioactive ligands found within the native tissues. They would also possess dynamic properties that enable temporal adjustments of the ligands and the spatial patterns in which they are presented to permit directed tissue morphogenesis and spatially restricted induction of cell fate. Fabrication of such substrates requires the immobilization of multiple biochemical cues in complex and highly ordered orientations on substrates. While simulating all the endogenous factors of a cell’s niche in vitro is unreasonable, the R-μCP system described here allows for immobilization of multiple regions of orthogonal bioactive chemistries with microscale control of spatial orientation.
The utility of these substrates increases further when considering that while PEG brush arrays bound with immobilized ligands can elicit biospecific interactions, unbound PEG brushes remain resistant to protein adsorption and thus can serve to control cell adhesion and migration. Although passive, micropatterned adsorption of extracellular matrix (ECM) proteins or polymer-PEG conjugates is a simpler method for controlling cell adhesion and migration, these technique only provide transient control since adsorption is a reversible process11. Also, molecules adsorb to surfaces in unpredictable orientations and at random concentrations thereby limiting the fidelity and control of biospecific interactions. Moreover, compounds typically used to create non-fouling regions, such as bovine serum albumin or Pluronic F127, do not possess designated reactive sites for further functionalization limiting post-adsorption in situ modifications. SAMs of alkanethiols can be generated with such reactive groups for additional surface modification, but they too have limited durability in tissue culture conditions due to the lack of oxidative shielding afforded by grafted PEG brushes10,11. Conversely, the merger of our R-μCP platforms with PEG-brush grafting and sequential nucleophilic substitution reactions provides the ability to engineer substrates with durable, microscale cytophobic regions that can be modified a priori or in situ with bio-orthogonal functionalities. This will permit greater control of biospecific interactions that can regulate in vitro tissue morphology and growth.
A key strength of R-μCP compared to other systems for aligning sequential microcontact printings is the ability to remove the substrate from the system between repeated stampings while still maintaining high alignment accuracy12,13. This enables greater diversity in the types of surface chemistries that can be applied, and the duration of surface modification reactions can be varied to tune the densities of subsequently immobilized ligands6. Thus, R-μCP can be used to establish focal concentration gradients in the biochemical cues that mediate biospecific interactions14. With the capacity to precisely control both the location and degree of biospecific interactions, the R-μCP system offers a unique method for investigating the roles of bioactive ligands in tissue morphogenesis and establishes a robust system for generating culture substrates simulating the presentation of biological cues in the in vivo niche.
A critical aspect of cellular niches, specifically within development, is spatiotemporal modulation of signaling factors18. Though soluble biochemical cues can be added to culture media in a temporally defined manner, there is limited spatial control over which cells encounter these cues. With the R-μCP system described here, it is possible to construct micropatterned culture substrates with terminally functionalized PEG brushes presenting azide functional groups that have no effect on cell fate in culture. While azide groups are capable of undergoing copper-catalyzed “click” reactions with alkyne presenting molecules, this cannot be performed in the presence of cells in culture given the toxicity of copper. However, high-strain molecules like dibenzocyclooctyne (DBCO) undergo a highly selective and biocompatible copper-free azide-alkyne cycloaddition reaction19,20. Through conjugation of DBCO-containing linkers, micropatterned culture substrates can be modified in situ with new biological ligands21. With the ability to render cytophobic regions cytophilic or add different biochemical cues for use in multi-step signaling procedures, culture substrates generated with this method have novel adaptive capabilities and thus provide a more robust system for instructing tissue morphogenesis in vitro.
Despite the immense potential and utility of the R-μCP platform, it does have certain drawbacks. The use of numerous SI-ATRP steps to generate PEG-grafted substrates greatly increases the fabrication time compared to methods involving inkjet printing or microdroplet deposition of ECM proteins. While the precision of inkjet printing techniques rivals that of the current R-μCP system, the adsorption of ECM proteins is reversible thereby providing less control of cell-protein interactions. Also, substrates generated via inkjet printing techniques cannot be removed during fabrication, thereby impeding the use of diverse substrate modification chemistries that permit, for example, spatially restricted in situ substrate modifications22. Due to these limitations inkjet techniques are better suited for rapid generation of culture substrates primarily concerned with short-duration, static arrangements of biomolecules and cell types23 whereas the R-μCP system is much better suited toward generating multi-faceted substrates designed to exhibit long-term, dynamic control over both spatial and temporal cellular interactions in 2D with diverse biological ligands.
Microcontact printing enables rapid deposition of nano-to-microscale ‘ink’ features over large surface areas, but there has always been significant heterogeneity in the concentration of deposited ‘ink’ substances. This is also a current limitation of the R-μCP platform as can be observed in Figure 4. We hypothesize that the heterogeneity in fluorescent modification of the PEG brushes is due to the robot’s application of an uneven normal force over the entire PDMS stamp, which is also likely not perfectly flat. This results in non-uniform contact pressure between all regions of the stamp and the substrate thereby causing uneven deposition of the alkanethiol initiator and subsequent thickness of the grafted PEG brush. In order to manufacture PEG-grafted substrates with even deposition of alkanethiols and thereby surface functionality, the stamping tool design will need to be optimized to apply a distributed and uniform normal force over the entire PDMS stamp. This will facilitate uniform contact with the gold-coated slides across the entire interface independent of stamp imperfections. Also, in Figure 4B, one can observe slight cross-contamination of acetylene groups on the azide terminated PEG brush. While the surface density of immobilized ligands due to cross-contamination is minimal compared to that on the intended PEG-brush, this can be reduced to near zero by increasing the duration of the ethanolamine passivation reaction (see Section 6) as demonstrated previously6.
The primary application of the R-μCP system described here is for manufacturing complex tissue culture substrates that could be used to exert spatial and temporal control of cell fate. However, the high precision and accuracy of the R-μCP platform makes it an attractive method for other applications as well. The ability to pattern cytophilic areas with differential ligand chemistries followed by in situ modification of previously inert, cytophobic regions allows for co-culture of multiple cell lines with tight control over their spatial orientation. This coupled with the inherent high-throughput nature of micropatterning present an alternative to current methods for high-throughput screening of both single cells and multi-cell combinations24. While the R-μCP system has great potential in the realm of biology, it could also be applied to the field of microelectromechanical systems (MEMS). In MEMS fabrication, it is desirable to transfer MEMS components with high precision and accuracy for mass production. With novel kinetic stamping techniques, the R-μCP system described here could be adapted to effectively print components of silicon or gallium nitride on silicon wafers for use in generating MEMS25. Thus, the R-μCP platform could be used for a wide range of potential applications.
In conclusion, use of the R-μCP system to generate PEG-grafted culture substrates orthogonally functionalized using sequential nucleophilic substitution reactions presents not only an ideal platform for potentially controlling tissue morphology and growth in vitro, but an excellent system for investigating the roles of multiple bioactive ligands on cell fate. The ability to pattern multiple biochemical cues in distinct and highly ordered patterns establishes the groundwork for building culture substrates capable of instructing the formation of tissue structures with multiple cell types organized at the micron scale. This, coupled with the ability to modify micropatterned substrates in situ, could allow for unparalleled control of tissue morphogenesis and cell fate in culture. The patterning techniques described here provide a versatile system for manufacturing culture substrates that could one day facilitate rational and reproducible production of organotypic tissue structures in vitro.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
Funding for this work, GTK, TK, and JDM were provided by the Wisconsin Institute for Discovery and the Wisconsin Alumni Research Foundation.
Materials
| Name | Company | Catalog Number | Comments |
| SCARA | Epson | LS3-401ST | Higher end models with increased precision are available if desired. |
| (TRIDECAFLUORO-1,1,2,2-TETRAHYDROOCTYL)TRICHLOROSILANE | Gelest | SIT8174.0 | CAUTION, Should only be handled in a chemical fume hood. When silanizing wafers no one should enter the hood until all silane has been evaporated. |
| Sylgard 184 Silicone Elastomer Kit | Ellsworth Adhesive Co | NC9020938 | Thouroughly degass solutions via vacuum exposure before use. Alternative kits such as Kit 182 are acceptable. |
| 24mm X 50 mm #1 Cover Glass Slides | Fisher Scientific | 48393106 | These can be purchased from a number of suppliers with varying dimensions to suit need. |
| CHA-600 Telemark Electron Beam Evaporator | Telemark | SEC-600-RAP | Requries specialized training. |
| EPSON LS3 SCARA | EPSON | LS3-401ST | |
| ω-mertcaptoundecyl bromoisobutyrate | Prochimia | FT 015-m11-0.2 | Store at -20°C. Other ATRP initiators may be used as this R-μCP platform is applicable to all micropatterning modalities. |
| Schlenk Tube Flask 50 mL | Synthware | 60003-078 | Requires rubber stoppers with diaphram. |
| Poly(ethylene glycol) methyl ether methacrylate | Sigma Aldrich | 447943 | Shipped containing MEHQ and BHT free readical inhibitors. |
| Methanol (Certified ACS) | Fisher Scientific | A412-4 | CAUTION, only handle in chemical fume hood. |
| Copper(II) Bromide | Sigma Aldrich | 437867 | CAUTION, limit exposure with surgical mask. |
| 2',2-Bipyridine | Sigma Aldrich | D216305 | CAUTION, limit exposure with surgical mask. |
| Sodium L-Ascorbate | Sigma Aldrich | A4034 | |
| 20mL Borosilicate Glass Scintillation Vials | Fisher Scientific | 03-340-4E | |
| Sodium Azide | Sigma Aldrich | S2002 | CAUTION, limit exposure with surgical mask. |
| N,N-dimethyformamide | Sigma Aldrich | 227056 | CAUTION, only handle in chemical fume hood. |
| Ethanolamine | Sigma Aldrich | 398136 | CAUTION, only handle in chemical fume hood. |
| Triethylamine | Sigma Aldrich | T0886 | CAUTION, only handle in chemical fume hood. |
| Dimethylsulfoxide | Sigma Aldrich | 276855 | CAUTION, only handle in chemical fume hood. |
| Propargylamine | Sigma Aldrich | P50900 | CAUTION, only handle in chemical fume hood. |
| 200 Proof Ethanol | University of Wisconsin Material Distribution Services | 2292 | CAUTION, only handle in chemical fume hood. |
| Azide-PEG3-Biotin | ClickChemistryTools | AZ104-100 | Solubilized in DMF |
| Copper(II) Sulfate | Sigma Aldrich | C1297 | CAUTION, limit exposure with surgical mask. |
| Tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) | Sigma Aldrich | 678937 | |
| L-Ascorbic Acid | Sigma Aldrich | A7506 | |
| Phosphate Buffer Saline | Invitrogen | 14190144 | |
| Donkey Serum | Sigma Aldrich | D9663 | Donkey serum contaminated items are considered bio-hazardous material and should be disposed of accordingly. Various other compounds (e.g. BSA) are available and serve this purpose. |
| 12-Well Polystyrene Plate | Thermo Scientifit – NUNC | 07-200-81 | Plates can be purchased form a number of suppliers with varying dimensions. |
| DBCO-PEG4-Biotin | Clickchemistytools | A105P4-10 | Solubilized in DMF |
| Streptavidin, Alexa Fluor 488 Conjugate | Life Technologies | S-11223 | Solubilized in PBS |
| Streptavidin, Alexa Fluor 546 conjugate | Life Technologies | S-11225 | Solubilized in PBS |
| Nikon A1-R Confocal Microscope | Nikon | Nikon Eclipse Ti, A1R | An epifluorescent microscope is sufficient to image functionalized micropatterned substrates. |
Riferimenti
- Senaratne, W., Andruzzi, L., Ober, C. K. Self-Assembled Monolayers and Polymer Brushes in Biotechnology: Current Applications and Future Perspectives. Biomacromolecules. 6 (5), 2427-2448 (2005).
- Hucknall, A., Kim, D. -. H., Rangarajan, S., Hill, R. T., Reichert, W. M., Chilkoti, A. Simple Fabrication of Antibody Microarrays on Nonfouling Polymer Brushes with Femtomolar Sensitivity for Protein Analytes in Serum and Blood. Advanced Materials. 21 (19), 1968-1971 (2009).
- Hucknall, A., Rangarajan, S., Chilkoti, A. In Pursuit of Zero: Polymer Brushes that Resist the Adsorption of Proteins. Advanced Materials. 21 (23), 2441-2446 (2009).
- Rozkiewicz, D. I., Jańczewski, D., Verboom, W., Ravoo, B. J., Reinhoudt, D. N. Click” Chemistry by Microcontact Printing. Angewandte Chemie International Edition. 45 (32), 5292-5296 (2006).
- Jewett, J. C., Bertozzi, C. R. Cu-free click cycloaddition reactions in chemical biology. Chemical Society Reviews. 39 (4), 1272-1279 (2010).
- Sha, J., Lippmann, E. S., McNulty, J., Ma, Y., Ashton, R. S. Sequential Nucleophilic Substitutions Permit Orthogonal Click Functionalization of Multicomponent PEG Brushes. Biomacromolecules. 14 (9), 3294-3303 (2013).
- Tugulu, S., Silacci, P., Stergiopulos, N., Klok, H. -. A. RGD—Functionalized polymer brushes as substrates for the integrin specific adhesion of human umbilical vein endothelial cells. Biomaterials. 28 (16), 2536-2546 (2007).
- Ashton, R. S., et al. High-Throughput Screening of Gene Function in Stem Cells Using Clonal Microarrays. Stem Cells. 25 (11), 2928-2935 (2007).
- Koepsel, J. T., Murphy, W. L. Patterned Self-Assembled Monolayers: Efficient, Chemically Defined Tools for Cell Biology. ChemBioChem. 13 (12), 1717-1724 (2012).
- Mrksich, M., Dike, L. E., Tien, J., Ingber, D. E., Whitesides, G. M. Using microcontact printing to pattern the attachment of mammalian cells to self-assembled monolayers of alkanethiolates on transparent films of gold and silver. Experimental cell research. 235 (2), 305-313 (1997).
- Ma, H., Hyun, J., Stiller, P., Chilkoti, A. Non-Fouling” Oligo(ethylene glycol)- Functionalized Polymer Brushes Synthesized by Surface-Initiated Atom Transfer Radical Polymerization. Advanced Materials. 16 (4), 338-341 (2004).
- Bou Chakra, E., Hannes, B., Dilosquer, G., Mansfield, D. C., Cabrera, M. A new instrument for automated microcontact printing with stamp load adjustment. Review of Scientific Instruments. 79 (6), (2008).
- Trinkle, C. A., Lee, L. P. High-precision microcontact printing of interchangeable stamps using an integrated kinematic coupling. Lab on a Chip. 11 (3), 455 (2011).
- McNulty, J., et al. High-precision robotic microcontact printing (R-μCP) utilizing a vision guided selectively compliant articulated robotic arm. Lab on a Chip. , (2014).
- Qin, D., Xia, Y., Whitesides, G. M. Soft lithography for micro- and nanoscalepatterning. Nature Protocols. 5 (3), 491-502 (2010).
- Nam, Y., Chang, J. C., Wheeler, B. C., Brewer, G. J. Gold-Coated Microelectrode Array With Thiol Linked Self-Assembled Monolayers for Engineering Neuronal Cultures. IEEE Transactions on Biomedical Engineering. 51 (1), 158-165 (2004).
- Ma, H., Wells, M., Beebe, T. P., Chilkoti, A. Surface-Initiated Atom Transfer Radical Polymerization of Oligo(ethylene glycol) Methyl Methacrylate from a Mixed Self-Assembled Monolayer on Gold. Advanced Functional Materials. 16 (5), 640-648 (2006).
- Scadden, D. T. The stem-cell niche as an entity of action. Nature. 441 (7097), (2006).
- Codelli, J. A., Baskin, J. M., Agard, N. J., Bertozzi, C. R. Second-Generation Difluorinated Cyclooctynes for Copper-Free Click Chemistry. Journal of the American Chemical Society. 130 (34), 11486-11493 (2008).
- Debets, M. F., van Berkel, S. S., Schoffelen, S., Rutjes, F. P. J. T., van Hest, J. C. M., van Delft, F. L. Aza-dibenzocyclooctynes for fast and efficient enzyme PEGylation via copper-free (3+2) cycloaddition. Chemical Communications. 46 (1), 97 (2010).
- DeForest, C. A., Polizzotti, B. D., Anseth, K. S. Sequential click reactions for synthesizing and patterning three-dimensional cell microenvironments. Nature Materials. 8 (8), 659-664 (2009).
- Roth, E. A., Xu, T., Das, M., Gregory, C., Hickman, J. J., Boland, T. Inkjet printing for high-throughput cell patterning. Biomaterials. 25 (17), 3707-3715 (2004).
- Xu, T., Zhao, W., Zhu, J. M., Albanna, M. Z., Yoo, J. J., Atala, A. Biomaterials. Biomaterials. 34 (1), 130-139 (2013).
- Brouzes, E., et al. Droplet microfluidic technology for single-cell high-throughput screening. Proceedings of the National Academy of Sciences. 106 (34), 14195-14200 (2009).
- Meitl, M. A., et al. Transfer printing by kinetic control of adhesion to an elastomeric stamp. Nature Materials. 5 (1), 33-38 (2005).

