Analysis of Zebrafish Larvae Skeletal Muscle Integrity with Evans Blue Dye
Summary
In this study, we describe a straightforward method to perform Evans Blue Dye (EBD) analysis on zebrafish larvae. This technique is a powerful tool for the characterization of skeletal muscle integrity and delineation of zebrafish models of muscular dystrophy, and is a valuable method for the development of novel therapeutics.
Abstract
The zebrafish model is an emerging system for the study of neuromuscular disorders. In the study of neuromuscular diseases, the integrity of the muscle membrane is a critical disease determinant. To date, numerous neuromuscular conditions display degenerating muscle fibers with abnormal membrane integrity; this is most commonly observed in muscular dystrophies. Evans Blue Dye (EBD) is a vital, cell permeable dye that is rapidly taken into degenerating, damaged, or apoptotic cells; in contrast, it is not taken up by cells with an intact membrane. EBD injection is commonly employed to ascertain muscle integrity in mouse models of neuromuscular diseases. However, such EBD experiments require muscle dissection and/or sectioning prior to analysis. In contrast, EBD uptake in zebrafish is visualized in live, intact preparations. Here, we demonstrate a simple and straightforward methodology for performing EBD injections and analysis in live zebrafish. In addition, we demonstrate a co-injection strategy to increase efficacy of EBD analysis. Overall, this video article provides an outline to perform EBD injection and characterization in zebrafish models of neuromuscular disease.
Introduction
Muscular dystrophies constitute a group of prevalent and heterogeneous human muscle diseases with specific histopathological features1,2. Symptoms typically associated with this devastating group of diseases include muscle weakness, muscle degeneration, loss of motility, and early mortality1,3. The primary pathomechanisms of muscular dystrophies are the loss of proteins that stabilize the sarcolemma, anchor transmembrane complexes, and mediate cell signaling across the membrane4-6. For example, complete loss of the protein dystrophin, a primary scaffold protein of the dystrophin-glycoprotein complex, results in destabilization of the muscle membrane in Duchenne muscular dystrophy7. Due to the fact that most muscular dystrophies result from mutations in proteins that participate in the link between the extracellular matrix and the sarcolemmal cytoskeleton, a common observation at the cellular level is the loss of sacrolemmal integrity8,9. This understanding of the primary pathomechanism(s) associated with muscular dystrophies is the product of numerous years of research employing animal model systems2,10-15. However, despite advances in the field, there are still limited therapeutic options for treatment or management of the range of dystrophy subtypes. This limitation of viable therapies is due to several key factors: 1) the difficulty of gene therapy strategies, 2) a high frequency of de-novo disease cases and the corresponding lack of translatable animal models, and 3) the lack of rigorous strategies to test the physiological consequences of putative therapeutic agents with clear and reproducible outcome measures.
To overcome some of these limitations, numerous labs including our own are employing zebrafish as a system to model and study human neuromuscular diseases2. To date, zebrafish have proven a valuable tool in disease research. The zebrafish model has been used to identify and validate novel human disease causing mutations16,17, elucidate uncharacterized disease causing mechanisms17,18, and identify novel therapeutic strategies12,19. These advances were made, in part, by the canonical strengths of the zebrafish system such as their optical clarity, ease of genetic manipulation, and ability to breed in large numbers20. Zebrafish have additionally proven amendable to large-scale drug screens21, a valuable method for the identification of novel therapeutics22-24. Regarding muscle disease research, these strengths are complemented by the ability to isolate single zebrafish skeletal muscle fibers via dissociation25 and by the ability to examine myofiber organization in vivo using the optical phenomenon called birefringence26, which collectively allows for rapid determination of macroscopic muscle integrity. Regardless of these available utilities, further tool development is continuously required to advance investigation.
We, and others, have adapted a protocol for EBD injection and analysis in the zebrafish model. EBD is a vital, cell permeable dye that is taken up by damaged, degenerating, or apoptotic cells and then visualized under fluorescence27. To date, EBD analysis has extensively been used to analyze muscle membrane integrity in mouse models of skeletal muscle and heart diseases8,9,27. However, in mammalian preparations, harvested muscle typically requires laborious sectioning or dissection prior to analysis. In zebrafish, direct analysis is possible in high numbers using live and intact animals. In this video article, we will demonstrate the methodology to perform EBD injection and analysis in live zebrafish larvae, with representative images of EBD uptake in the zebrafish dystrophy mutant line sapje15,28. Furthermore, we present a co-injection strategy that allows for increased quantification of EBD preparations.
Protocol
1. Preparation of Agar Injection Plates (Time: 45 min)
- Boil 2% to 3% agarose in E3 media and allow solution to cool slightly on bench. Note: The number of injection plates being prepared dictates the amount of agarose required. Each injection plate needs approximately 35 ml of the agarose solution.
- After boiling, allow the agarose to cool until desired temperature is reached (e.g., 45 °C) as per the injection mold manufacturer’s instructions.
- Pour approximately 35 ml of the cooled agarose into a 100 mm dish.
- Place one end of preferred injection mold into solution, then lay remainder of mold onto agarose solution (this will help reduce the occurrence of air bubbles).
- Allow the agarose solution to solidify either at RT or 4 °C for approximately 30 min.
- Use a spatula to separate one end of the mold from the solid agarose. Slowly remove the remainder of the mold.
2. Preparation of Evans Blue Dye (EBD) Injection Mix (Time: 30 min)
- Make a 1% stock of EBD in 1X Ringer’s solution (155 mM NaCl; 5 mM KCl; 2 mM CaCl2; 1 mM MgCl2; 2 mM Na2HPO4; 10 mM HEPES; 10 mM glucose; pH to 7.2), which can be stored at RT.
- Make a stock solution of fluorescein isothiocyanate (FITC)-dextran MW 10,000 kDa at 25 mg/mL in 1X Ringer’s solution and store at -20 °C.
- Prepare injection mix by diluting EBD to 0.1% directly in stock solution of FITC-dextran stock (i.e., for a final working volume of 100 µl: Mix 10 µl of 1% EBD in 90 µl of FITC dextran stock).
- Thoroughly vortex injection mix (it should turn green) and keep out of direct light by wrapping injection mix tube in aluminum foil.
3. EBD Injection Preparation (Time: Approximately 30 min)
Note: Protocol works best with larvae from 3-7 days post fertilization (dpf).
- Pre-warm injection plate to RT.
- Set up injection apparatus by arranging micromanipulator on metal plate and stand next to the microscope being used for injection. Turn on air driven microinjection controller. Note: The preferred injection system will vary by lab and should not change outcome of analysis.
- Back fill injection needle with approximately 2-4 µl of EBD mix.
- Calibrate injection volume to approximately 5 nl of EBD mix. Note: Injection volume calibration will depend on calibration method. A piston driven injection can be directly set to a given injection volume whereas gas pressure injectors will need the injection volume calibrated via volume bolus with the use of a micrometer.
- Wet injection plate with 1X Ringer’s solution and remove excess from wells.
- Pre-treat larvae with 0.04% ethyl 3-aminobenzoate methanesulfonate salt (tricaine) diluted in 1X Ringer’s solution to immobilize larvae prior to the start of injection. Note: Ensuring larvae are completely immotile is important as proper injection is difficult with any residual movement.
- Place anaesthetized larvae into wells of the agar injection plates using a glass pipette. Ensure that the larvae are completely within the well and lying on their side. Note: The number of larvae per well is up to the experimenter.
- After larvae are put in the wells, remove excess Ringer’s solution to minimize larvae movement within the well. Leave a residual amount of solution so that larvae do not dehydrate.
4. Pericardial Injection of Zebrafish Larvae with EBD (Time: Dependent on number of larvae injecting, estimated 1-3 hr)
- Place the injection plate containing the larvae on a dissecting scope where the injections will be performed.
- Position the injection pipette needle containing the EBD mix over a zebrafish larvae.
- Re-position the injection plate by rotating it so the injection needle is near the larvae’s heart and approximately 45° ventrally from the anterior-posterior axis.
- Insert the injection needle into the common cardinal vein (CCV) in the region of the vein at the anterior portion of the yolk where the vein is initially turning in the dorsal direction (Figure 1). Note: A magnification of up to 40x may be useful to clearly see the CCV.
- Inject 5 nl of EBD mix and keep injection needle in position for 5-8 sec to minimize immediate leakage of EBD mix. Note: A good injection will have dye coloration seen in the heart chambers (Figure 1). If EBD mix is not observed in the heart, then injecting an additional 5 nl of EBD mix may be sufficient to induce dye uptake. Alternatively, the embryo can be discarded.
Note: In some situations, the heart may stop beating. If this occurs, continue to monitor the larvae for 20-40 sec. Typically, the heart resumes beating as the dye moves through the circulatory system. - Move onto the next larvae and repeat.
- Identify successfully injected embryos by observing the presence of FITC-dextran in the vasculature immediately after injection (Figure 2).
5. Incubation and EBD Uptake (Time: 4-6 hr)
- After the desired number of larvae are injected, return injected larvae to 1X Ringer’s solution without tricaine in 100 mm dishes.
- Keep dishes wrapped in aluminum foil. Note: Keeping injected larvae in the dark significantly increases survival rates and ensures the greatest consistency in signal strength. Wrapping in aluminum foil is especially important for the period of time that the larvae are outside of the incubator.
- Allow larvae to incubate at 28.5 °C for 4-6 hr to ensure sufficient EBD uptake.
6. Visualization of EBD in the Muscle (Time: Dependent on number of larvae injecting and type of microscopy, estimated 0.5-3 hr)
- Prior to imaging, anesthetize the larvae with 0.04% tricaine to prevent movement.
- View larvae under red fluorescence to determine if EBD uptake is occurring in the skeletal muscle (Figure 3).
Representative Results
The EBD injection mix was injected into the CCV of sapje homozygous mutants and wild-type siblings at 3 dpf. Injections that filled the heart chambers (Figure 1B) were then analyzed for successful injection by visualizing FITC-dextran in the vasculature under green fluorescence (Figure 2).
After a 4 hr incubation period, EBD uptake was examined at the somite level using fluorescence microscopy. Wild type siblings exhibited no EBD fluorescence within any visible muscle fibers (Figure 3A), whereas the sapje homozygous mutants showed EBD uptake, indicating damage to the muscle membrane15 (Figure 3B).
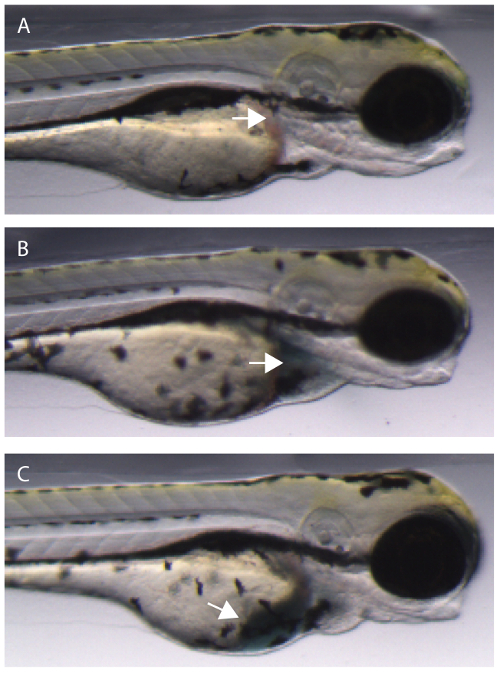
Figure 1. Injecting EBD injection mix into the common cardinal vein (CCV) of a zebrafish embryo. (A) Uninjected embryo. Arrow denotes ideal location for CCV injection. (B) Successful injection into the CCV. The dye enters the heart chambers (arrow) and begins to be pumped through the vasculature. (C) An unsuccessful CCV injection will result in some or all of the dye entering the yolk sac of the embryo (arrow). Please click here to view a larger version of this figure.
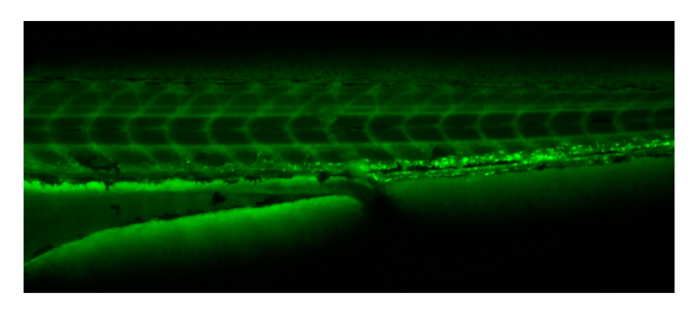
Figure 2. Embryos can be sorted for successful injection by observing FITC-dextran distribution under green fluorescence throughout the vasculature immediately following injection and prior to EBD uptake. Please click here to view a larger version of this figure.
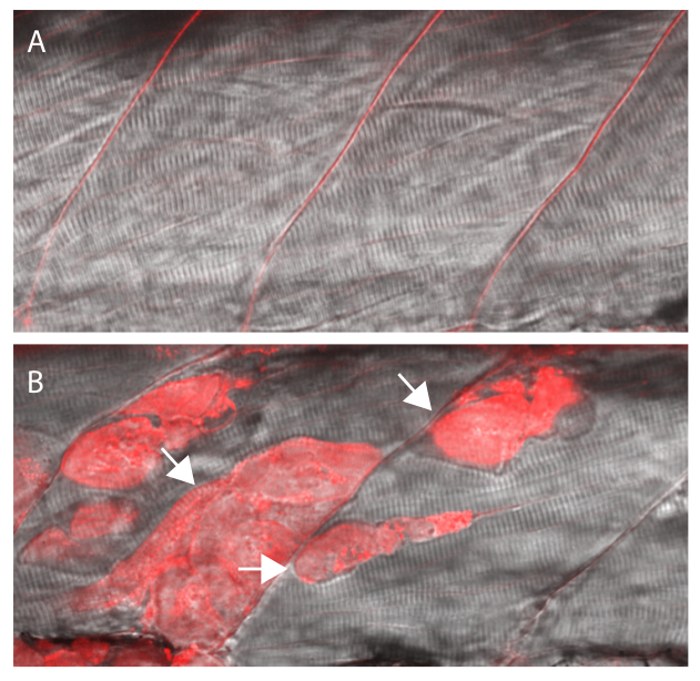
Figure 3: EBD will be taken up by fibers with damaged membranes. (A) Wildtype siblings showing no EBD fluorescence in muscle fibers. (B) Sapje homozygous mutant with EBD fluorescence within multiple muscle fibers (arrows). All larvae were injected with the EBD injection mix and analyzed after a 4 hr incubation period at 3 dpf. Siblings and mutants were sorted by muscle fiber detachment prior to CCV injection. Please click here to view a larger version of this figure.
Discussion
Zebrafish are emerging as a powerful tool for the study of neuromuscular disease2,29. To date, the zebrafish system has been used to validate new muscle disease-causing mutations16,17,30, elucidate novel pathomechanisms18, and identify potentially new therapeutic drugs12,24. These collective efforts have established the utility of the zebrafish to model human neuromuscular diseases. However, despite the advances made with zebrafish and mammalian models, there are limited treatment options for patients within the wide spectrum of neuromuscular conditions. Therefore, a high demand exists for therapy development for this group of devastating diseases. Paralleling this demand for therapeutics is the corresponding need for ongoing experimental innovation, as well as rigorous analysis to verify new animal models and putative therapeutic strategies.
EBD analysis is commonly used in mouse models to study tissue and cellular damage in brain, heart, and skeletal muscle27,31. Most notably, EBD is used extensively in mouse models of various muscular dystrophy subtypes to show the severity of muscle membrane instability and damage8. The use of EBD to reveal muscle membrane damage is a supportive parameter establishing similarities of the animal model to the human disease state9. The power of EBD in mouse has led several labs, including our own, to develop and apply EBD to zebrafish models of neuromuscular disease. Due to the applicability of EBD analysis, this technique is actively being implemented to corroborate zebrafish models to the human disease state11,15,22,24,32. Larvae with damaged muscle membranes will have EBD uptake and therefore red fluorescence within muscle fibers. Fluorescence observed in the inter-fiber space, but not within individual muscle fibers may also be informative of fibers detaching from the basement membrane in the absence of membrane damage, providing useful diagnostic detail. EBD analysis has potential application beyond animal model validation. Efforts from our lab have recently demonstrated that EBD analysis is beneficial in validating potentially novel therapeutic drugs24. Determining if potential therapeutic treatments reduce or abolish EBD uptake in neuromuscular disease models can signify relevant therapeutic action8. This type of analysis can help establish the mechanism(s) of therapeutics and expands the application of EBD analysis.
As with many techniques, EBD analysis does have several caveats to be observed during experimental design and practice. For example, it can be challenging to identify the CCV due to the thickening of the tissue with age. Additionally, it is easy to damage larvae in preparation before and during the pericardial injection, reducing experimental counts and increasing the need to prep large numbers of larvae. Furthermore, physical damage done to the larvae during handling and injection could result in false positives as damaged muscle can take up EBD. In order to overcome some of these obstacles, we have described a co-injection strategy in this video article that allows easy and reliable identification of larvae with successful dye infusion immediately following injection and prior to subsequent analysis. The FITC-dextran co-injection controls for successful injection by allowing confirmation of EBD in the vasculature prior to its uptake by the muscle fibers. This can be particularly useful as EBD fluorescence becomes highly diffuse in larvae after several hours if not collected in the muscle fibers; as such, it can be difficult to detect. In addition, missing the CCV and injecting EBD into the yolk or body cavity can, after incubation, result in diffuse red fluorescence similar to control embryos, yet with the reduced likelihood of uptake by damaged muscle fibers. Collectively, these caveats suggest EBD injection requires patience and practice in order to achieve consistent and reliable results.
In all, we describe a practical and straightforward method to perform EBD analysis on zebrafish larvae. To date, the use of zebrafish as a model system, especially as a human disease model, has been rapidly expanding. This expansion is partially due to the continued development and modification of experimental techniques that improve upon the current advantages of the zebrafish system. The EBD injection technique provides an additional and powerful tool to a researcher’s arsenal for the validation and study of zebrafish muscle disease models. The ongoing implementation and modification of this technique has the potential to help uncover novel therapeutic strategies as well as disease causing mechanisms.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
We wish to thank Trent Waugh for his technical assistance. We also acknowledge the Department of Pediatrics at the Hospital for Sick Children and Cure Congenital Muscular Dystrophy (CMD) for their generous funding for this project.
Materials
| Fluorescein isothiocyanate-dextran MW 10,000 | Sigma | FD10S | |
| Evan's Blue Dye | Sigma | E2129 | |
| Ethyl 3-aminobenzoate methanesulfonate salt | Sigma | A5040 | |
| 100 mm Petri dish | Fischerbrand | FB0875712 | Injection mold base |
| Thin wall glass capillaries | World Precision Instruments | TW100F-4 | For Injection needle |
| Agarose | Bioshop | AGA001 | Injection mold |
| Microinjection mold | Adaptive Science Tools | TU-1 | Injection mold |
| Sodium chloride | Bioshop | SOD001 | Ringer's solution |
| Potassium chloride | Bioshop | POC888 | Ringer's solution |
| Magnessium chloride hexahydrate | Sigma | M2670 | Ringer's solution |
| Sodium phosphate monobasic monohydrate | Sigma | S9638 | Ringer's solution |
| HEPES | Sigma | H4034 | Ringer's solution |
| Glucose | BioBasic | GB0219 | Ringer's solution |
| Calcium chloride | Sigma | C1061 | Ringer's solution |
Riferimenti
- Verma, S., Anziska, Y., Cracco, J. Review of Duchenne muscular dystrophy (DMD) for the pediatricians in the community. Clin Pediatr (Phila. 49, 1011-1017 (2010).
- Gibbs, E. M., Horstick, E. J., Dowling, J. J. Swimming into prominence: the zebrafish as a valuable tool for studying human myopathies and muscular dystrophies). FEBS J. 280, 4187-4197 (2013).
- Lancet, . , 381-845 (2013).
- Lapidos, K. A., Kakkar, R., McNally, E. M. The dystrophin glycoprotein complex: signaling strength and integrity for the sarcolemma. Circ Res. 94, 1023-1031 (2004).
- Campbell, K. P., Kahl, S. D. Association of dystrophin and an integral membrane glycoprotein. Nature. 338, 259-262 (1989).
- Cohn, R. D., Campbell, K. P. Molecular basis of muscular dystrophies. Muscle Nerve. 23, 1456-1471 (2000).
- Yoshida, M., Ozawa, E. Glycoprotein complex anchoring dystrophin to sarcolemma. J Biochem. 108, 748-752 (1990).
- Straub, V., Rafael, J. A., Chamberlain, J. S., Campbell, K. P. Animal models for muscular dystrophy show different patterns of sarcolemmal disruption. J Cell Biol. 139, 375-385 (1997).
- Matsuda, R., Nishikawa, A., Tanaka, H. Visualization of dystrophic muscle fibers in mdx mouse by vital staining with Evans blue: evidence of apoptosis in dystrophin-deficient muscle. J Biochem. 118, 959-964 (1995).
- Bassett, D., Currie, P. D. Identification of a zebrafish model of muscular dystrophy. Clin Exp Pharmacol Physiol. 31, 537-540 (2004).
- Gupta, V., et al. The zebrafish dag1 mutant: a novel genetic model for dystroglycanopathies. Hum Mol Genet. 20, 1712-1725 (2011).
- Kawahara, G., et al. Drug screening in a zebrafish model of Duchenne muscular dystrophy. Proc Natl Acad Sci U S A. 108, 5331-5336 (2011).
- Guyon, J. R., et al. Modeling human muscle disease in zebrafish. Biochim Biophys Acta. 1772, 205-215 (2007).
- Cavanna, J. S., et al. Molecular and genetic mapping of the mouse mdx locus. Genomics. 3, 337-341 (1988).
- Bassett, D. I., et al. Dystrophin is required for the formation of stable muscle attachments in the zebrafish embryo. Development. 130, 5851-5860 (2003).
- Horstick, E. J., et al. Stac3 is a component of the excitation-contraction coupling machinery and mutated in Native American myopathy. Nat Commun. 4, (1952).
- Davidson, A. E., et al. Novel deletion of lysine 7 expands the clinical, histopathological and genetic spectrum of TPM2-related myopathies. Brain. 136, 508-521 (2013).
- Telfer, W. R., Nelson, D. D., Waugh, T., Brooks, S. V., Dowling, J. J. Neb: a zebrafish model of nemaline myopathy due to nebulin mutation. Dis Model Mech. 5, 389-396 (2012).
- Dowling, J. J., et al. Oxidative stress and successful antioxidant treatment in models of RYR1-related myopathy. Brain. 135, 1115-1127 (2012).
- Detrich, H. W., Westerfield 3rd, ., M, L. I., Zon, . Overview of the Zebrafish system. Methods Cell Biol. 59, 3-10 (1999).
- Whitfield, T. T., et al. Mutations affecting development of the zebrafish inner ear and lateral. , 123-241 (1996).
- Kawahara, G., Guyon, J. R., Nakamura, Y., Kunkel, L. M. Zebrafish models for human FKRP muscular dystrophies. Hum Mol Genet. 19, 623-633 (2010).
- Baraban, S. C., Dinday, M. T., Hortopan, G. A. Drug screening in Scn1a zebrafish mutant identifies clemizole as a potential Dravet syndrome treatment. Nat Commun. 4, (2013).
- Waugh, T. A., et al. Fluoxetine prevents dystrophic changes in a zebrafish model of Duchenne muscular dystrophy. Hum Mol Genet. 23 (17), 4651-4662 .
- Horstick, E. J., Gibbs, E. M., Li, X., Davidson, A. E., Dowling, J. J. Analysis of embryonic and larval zebrafish skeletal myofibers from dissociated preparations. J Vis Exp. 50259, (2013).
- Smith, L. L., Beggs, A. H., Gupta, V. A. Analysis of skeletal muscle defects in larval zebrafish by birefringence and touch-evoke escape response assays. J Vis Exp. 50925, (2013).
- Hamer, P. W., McGeachie, J. M., Davies, M. J., Grounds, M. D. Evans Blue Dye as an in vivo. marker of myofibre damage: optimising parameters for detecting initial myofibre membrane permeability. J Anat. 200, 69-79 (2002).
- Guyon, J. R., et al. Genetic isolation and characterization of a splicing mutant of zebrafish dystrophin. Hum Mol Genet. 18, 202-211 (2009).
- Santoriello, C., Zon, L. I. Hooked! Modeling human disease in zebrafish. J Clin Invest. 122, 2337-2343 (2012).
- Majczenko, K., et al. Dominant Mutation of CCDC78 in a Unique Congenital Myopathy with Prominent Internal Nuclei and Atypical Cores. Am J Hum. , (2012).
- Wooddell, C. I., et al. Use of Evans blue dye to compare limb muscles in exercised young and old mdx mice. Muscle Nerve. 41, 487-499 (2010).
- Hall, T. E., et al. The zebrafish candyfloss mutant implicates extracellular matrix adhesion failure in laminin alpha2-deficient congenital muscular dystrophy. Proc Natl Acad Sci U S A. 104, 7092-7097 (2007).

