Antigen-Capture Enzyme-Linked Immunosorbent Assay for Specific Detection of Mycoplasma pneumoniae
Summary
In Mycoplasma pneumoniae infection, serology tests can generate good results, yet with low specificity because of immunological cross-reaction. The in-house antigen-capture ELISA, described in this paper, guarantees high species specificity and has been shown to be a reliable screening test for accurate diagnosis of M. pneumoniae.
Abstract
Mycoplasma pneumoniae is a cell wall-deficient prokaryote, mainly known to colonize the human respiratory tract and to be endemic, with epidemic peaks every 6 years, in older children and young adults. Diagnosis of M. pneumoniae is challenging because of the fastidious nature of the pathogen and the possibility of asymptomatic carriage. Laboratory diagnosis of M. pneumoniae infection based on antibody titration in the serum samples of patients remains the most practiced method. Because of the potential problem of immunological cross-reactivity with the use of polyclonal serum for M. pneumoniae, an antigen-capture enzyme-linked immunosorbent assay (ELISA) has been developed to improve the specificity of serological diagnosis. ELISA plates are coated with M. pneumoniae polyclonal antibodies, raised in rabbits and rendered specific after adsorption against a panel of heterologous bacteria that share antigens with M. pneumoniae species and/or are known to colonize the respiratory tract. The reacted M. pneumoniae homologous antigens are then specifically recognized by their corresponding antibodies in the serum samples. Further optimization of the physicochemical parameters to which the antigen-capture ELISA is subjected led to a highly specific, sensitive, and reproducible ELISA.
Introduction
Mycoplasmas are among the smallest and simplest known prokaryotes. They are mainly distinguished from other bacteria by the lack of a cell wall structure. Thus, Mycoplasmas were classified in a separate class named Mollicutes1. The cell wall deficiency confers intrinsic resistance to these microorganisms against some antimicrobial agents and is largely responsible for their polymorphism. Mycoplasmas have small genome and reduced size, which limits their metabolic and biosynthetic capabilities and explains their parasitic and saprophytic nature1.
Mycoplasma pneumoniae is one of the Mycoplasmas that infect man and is thought to be the most virulent2. M. pneumoniae colonizes the upper respiratory tract, leading to atypical pneumonia in children and young adults. The clinical signs engendered by M. pneumoniae infection are flu-like, with headache, fever, and cough3. The cytadherence of M. pneumoniae to host cells is mediated by an attachment organelle including P1 major adhesion and several accessory proteins4,5. More clinical manifestations may occur because of local inflammation and stimulation of the host immune system resulting from the intimate adherence of M. pneumoniae to the airway mucosa6. Although pneumonia is a hallmark of M. pneumoniae infection, it has been revealed that infection with this bacterium can also be responsible for a wide spectrum of non-pulmonary manifestations in different anatomical sites such as the central nervous system, heart, skin, and joints7.
As for all Mycoplasma species, the diagnosis of M. pneumoniae is challenging. The clinical signs evoking mycoplasmosis are mostly inapparent and non-characteristic8. Since it is very hard to diagnose M. pneumoniae infection by only relying on clinical manifestations and symptoms, laboratory screening is of particular interest9. Detecting M. pneumoniae colonies by culture is the gold standard method for a proper diagnosis. However, the fastidious growth requirements and the long time needed for the delivery of definitive results (1-2 weeks) complicate the culture, and thus means it is rarely used for routine diagnosis10. Nucleic acid amplification technologies were validated in terms of speed and efficiency, although because of their relatively high cost and unavailability in some health care facilities, these molecular techniques are not considered first-line diagnostic tests. It is true that commercial PCR tests are widely used to diagnose M. pneumoniae infections, but they still cannot replace serology. Also, the frequent occurrence of both false negative and false positive results has limited the use of PCR9. Routinely, serology remains the most practiced in laboratories for the diagnosis of M. pneumoniae infection. Several serology approaches have been reported for decades, such as cold hemagglutinins, complement fixation test11, indirect hemagglutination test12, immunofluorescence13, and the technology of ELISA, which was first applied to mycoplasma serology in the early 1980's14,15,16. One of the major issues encountered when performing ELISA serodiagnosis of M. pneumoniae infection is cross-reactions, which considerably lowers the specificity of the technique. Nonspecific adsorption of human sera with M. pneumoniae antigens was previously reported; in fact, many of the antibodies detected by ELISA in human sera may not always be bound to mycoplasmal antigens17, due to the commonality of M. pneumoniae with some bacteria18,19 and some animal and human tissues20.
Because of the high background readings observed in the conventional ELISA test that was practiced in the laboratory, the interpretation of results was often complicated, and thus the delivery of a proper M. pneumoniae diagnosis was a tough assignment. While facing this issue, we opted to improve the M. pneumoniae ELISA by removing nonspecific reactions of M. pneumoniae antigens with antibodies to be tested. For this purpose, we worked on selective depletion of the nonspecific M. pneumoniae antigens using the adsorption technique. In fact, the main goal of the antigen-capture ELISA is to specifically detect M. pneumoniae immunoglobulin (Ig) G in human serum samples. The concept of this ELISA consists mainly of the selective capture of M. pneumoniae-specific antigens, before adding the human serum samples. This selectivity is insured by incubating M. pneumoniae crude antigen with a M. pneumoniae polyclonal antiserum, produced in rabbits in the laboratory and rendering it species-specific by adsorption against a panel of heterologous bacteria, belonging or not belonging to the Mollicutes class, sharing antigens with M. pneumoniae species and/or known to colonize the respiratory tract. The adsorption procedure was repeated thrice, and its efficiency to eliminate cross-reactivity was tested by immunoblotting. The developed ELISA assay is a combination of sandwich and indirect ELISA. Briefly, the wells of the ELISA plate are first coated with a polyclonal antiserum specific to M. pneumoniae. Then, M. pneumoniae antigen is added and trapped between the antiserum and the antibodies present in the serum sample to be tested. The formed immunological complex is detected by a secondary enzyme-conjugated antibody (peroxidase-conjugated IgG). The reactions are visualized by the addition of chromogenic substrate, and the absorbance is measured spectrophotometrically. This in-house ELISA is schematically presented in Figure 1. The homemade ELISA proved to be efficient in specifically detecting M. pneumoniae infection and is currently one of the most practiced tests in routine diagnostic activity.
Protocol
The present study has been conducted in conformity with ethical aspects established by the ethical committee of Pasteur Institute of Tunis.
1. Pre-ELISA steps: Prerequisites and preprocessing
- Bacterial strains and growth media
NOTE: Mollicutes species and the walled bacteria used in the present study and their growth media are listed in Table 1.- Growth of Mollicutes species
- Inoculate 200 µL from the glycerol stock of each species in 1,800 µL of the media.
- Grow Mollicutes species statically at 37 ˚C with 5% CO2 for 2-4 days until a pH change is observed (the pH indicator is phenol red).
- Scale up the cultures to 10 mL by adding a 1/10th dilution of the culture to the media and allow them to grow again in the same conditions.
NOTE: According to the metabolism, some human Mollicutes species (M. pneumoniae, M. genitalium, and M. fermentans) acidify the medium due to glucose fermentation, and some others (M. hominis and Ureaplasma) alkalinize the medium by arginine hydrolysis or urea hydrolysis, respectively. Regarding avian mycoplasmas, the acidification of Frey's medium demonstrates the presence of M. gallisepticum, while its alkalinization proves the growth of M. imitans. - Spread 50 µL of the cultures on agar plates to confirm the growth of the bacteria. Maintain the agar plates at 37 ˚C with 5% CO2 and observe them regularly under the microscope for the appearance of typical fried egg colonies (Mycoplasmas) and dark urchin-like colonies (Ureaplasmas).
- Growth of non-Mollicutes species
- Culture the non-Mollicutes bacteria in 3 mL of the media at 37 ˚C overnight (shaking at 200 rpm).
- Compare the turbidity of the cultures with the control media to confirm growth.
- Scale up the cultures to 10 mL by adding a 1/100th dilution of the culture to the media and allow them to grow again in the same conditions.
- Growth of Mollicutes species
- Antigen preparation
- Antigen preparation of Mollicutes species
- Harvest the whole proteins (present in confirmed cultures of M. pneumoniae and the other Mollicutes species) by centrifugation at 40,000 x g at 4 ˚C for 30 min.
- Discard the supernatant and wash the pellets three times with 1 mL of 1x phosphate-buffered saline (PBS, pH 7.4) by a series of three centrifugations with the same conditions.
- Resuspend each antigen in 500 µL of PBS.
- Antigen preparation of non-Mollicutes species
- Centrifuge the overnight grown cultures of the non–Mollicutes bacteria at 1,500 x g for 15 min.
- Resuspend each bacterial pellet in 500 µL of PBS.
NOTE: Protein concentration is determined using the classic Bradford quantitation method with bovine serum albumin as standard21. The protein concentration of Mollicutes species usually ranges between 0.7-4.8 mg/mL. For the other bacterial species, protein concentration can reach 20 mg/mL. All antigens are stored at -20 ˚C until their subsequent usage.
- Antigen preparation of Mollicutes species
- Cross-reactivity screening by immunoblotting
- Denature the bacterial proteins by mixing 8 µL of each antigen with an equal volume of sample buffer (0.5 M Tris-HCl; pH 6.8, 10% glycerol [v/v], 10% SDS [w/v], and 0.2% bromophenol blue), incubate the mixture at 100 ˚C for 5 min.
NOTE: Denaturation is performed to cover the proteins with negative charge and to break their secondary structures, which allows them to run across the polyacrylamide gel and to be separated according to their molecular weights. - Subject the denatured proteins (100 µg protein/well) to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) by Laemmli's method22 with 12% separating gel and 5% stacking gel. After that, transfer the proteins electrophoretically to the nitrocellulose membrane by Towbin et al.'s method23.
- Immerse the nitrocellulose membrane in 5% skim milk in PBS for 30 min to block the unoccupied surfaces. Incubate the protein blots at room temperature for 2 h with rabbit polyclonal M. pneumoniae antiserum (produced at Pasteur Institute of Tunis) diluted to 1/200 in PBS-Tween 20, then with horseradish peroxidase (HRP)-conjugated anti-rabbit IgG diluted to 1/2,000 in PBS-Tween 20 for 1 h at room temperature.
- Wash off the unbound antibodies with PBS and obtain the results by exposing the nitrocellulose sheet to the substrate solution (4-chloro-1-naphthol, H2O2). Stop the reaction by adding water after 5-15 min.
- Denature the bacterial proteins by mixing 8 µL of each antigen with an equal volume of sample buffer (0.5 M Tris-HCl; pH 6.8, 10% glycerol [v/v], 10% SDS [w/v], and 0.2% bromophenol blue), incubate the mixture at 100 ˚C for 5 min.
- Adsorption procedure
NOTE: To eliminate cross-reactions between polyclonal M. pneumoniae antiserum and heterologous bacteria antigens, the adsorption procedure previously described by Ben Abdelmoumen and Roy24 is employed.- Prepare an antigen pool of the 12 heterologous bacteria (suspected to share antigens with M. pneumoniae), mix it in a microcentrifuge tube, and adjust the protein concentration corresponding to half of the antiserum to be adsorbed in the experiment.
NOTE: The volume and the concentration vary between the different assays. This step mainly depends on the concentration of the antibody to be adsorbed. For example, if the antibody concentration = 3.56 mg/mL, the concentration of the antigen pool (composed of the 12 heterologous bacteria) should be 1.78 mg/mL (therefore, approximately 0.148 mg of each antigen). - Centrifuge the antigen mixture at 14,000 x g for 10 min at 4 °C, collect the pooled pellet, and incubate it with the purified polyclonal anti-M. pneumoniae IgG for 2 h at 37 °C with slow agitation.
- After incubation, centrifuge the suspension at 14,000 x g for 10 min at 4 °C and recover the supernatant (which corresponds to the specific polyclonal M. pneumoniae antiserum).
NOTE: To ensure specificity of the polyclonal M. pneumoniae antiserum, repeat the adsorption procedure three times. Before it can be used in the ELISA assay, the adsorbed polyclonal M. pneumoniae antiserum should be tested by immunoblotting for the absence of cross-reactions against the included set of bacteria.
- Prepare an antigen pool of the 12 heterologous bacteria (suspected to share antigens with M. pneumoniae), mix it in a microcentrifuge tube, and adjust the protein concentration corresponding to half of the antiserum to be adsorbed in the experiment.
2. ELISA steps: The assay itself
- Microplate coating with the capture antibody
- Dilute the capture antibody (M. pneumoniae pre-adsorbed polyclonal antiserum) to a concentration of 10 µg/mL in 0.1 M carbonate-bicarbonate buffer (pH 9.6).
- Coat the 96-well ELISA plate by adding 100 µL of the diluted capture antibody to each well.
- After overnight incubation at 4 ˚C, remove the coating solution and wash the plate five times with the washing buffer (composition [per L]: 146.29 g of NaCl, 39.4 g of Tris-HCl, 0.2 g of Thimerosal, and 0.5 mL of Tween 20; pH 7.3).
- Blocking
- Block the remaining binding surfaces of the coated wells by adding 100 µL of the blocking solution (0.5% casein in PBS) to each well.
- Incubate for 1 h at room temperature.
- Remove the blocking solution with a pipette and wash the plate five times with the washing buffer.
- Application of the antigen
- Add an equal amount of M. pneumoniae whole proteins to all coated wells (10 ng/well).
- Allow the reaction to take place for 2 h at room temperature.
- Remove the excess of the antigen solution and wash the plate five times with the washing buffer.
- Application of the test samples and controls
- Dilute the test samples (human sera) and controls (reference positive and negative sera) to 1/200, 1/400, and 1/800 in PBS-Tween 20.
- Add 100 µL of the diluted samples and controls into the appropriate wells. Carry out each reaction in duplicate. Mark the wells containing neither antigen nor sera as blank.
- After incubating the plate for 90 min at room temperature, remove the serum solutions and wash the plate five times with the washing buffer.
- Application of the enzyme-conjugated detection antibody
- Dilute the enzyme-conjugated detection antibodies, HRP-conjugated anti-rabbit, and anti-human IgG to 1/10,000 in PBS-Tween 20.
- Pipette 100 µL of the appropriately diluted detection antibody to each well.
- After incubating the plate for 1 h at room temperature in the dark, remove the unbound detection antibodies and wash the plate five times with the washing buffer.
- Detection and data analysis
- Add 100 µL of the 3,3', 5,5' Tetramethyl-benzidine (TMB) chromogen solution to visualize the antigen-antibody binding.
- Allow the plate to develop for 30 min at room temperature then add 100 µL of the stop solution (7.5% H2SO4) to stop the enzymatic reaction.
- Read the absorbance at 450 nm wavelength using the microplate reader and sort out the results based on the calculation of the index of positivity (IP).
IP = Absorbance of the tested serum / Absorbance of the cut-off
IP < 0.7: Negative result
IP = 0.7: Test should be repeated within 2 weeks
IP > 0.7: Positive result
NOTE: The IP formula is conceived in the laboratory and the 0.7 value is set up after optimization. For duplicate reactions, the mean value of absorbance is calculated. Optimum serum dilution and antigen concentration are established by the ELISA checkerboard titration method (data not shown). Cut-off value = OD (optical density) of a reference positive serum (diluted at the same dilution as the sample). This OD should be at least three times higher than the OD value of the negative control.
3. Post-ELISA steps: Results assessment and test validation
- The ability of the in-house ELISA to specifically diagnose M. pneumoniae infection in the tested patients is evaluated through supplementary immunoblots using antigens of M. pneumoniae and the heterologous bacteria to confirm the positivity of the tested human sera in M. pneumoniae antibodies and their negativity in the other suspected bacteria (data not shown).
Representative Results
The immunoblotting activity of the non-adsorbed polyclonal Mycoplasma pneumoniae antiserum to heterologous bacteria
Cross-reactivity does indeed exist as showcased in the immunoblotting results (Figure 2) and compared to the positive control (Lane 1), some of the M. pneumoniae antigens are shared with the screened bacteria. The intensity of these cross-reactions was variable. For instance, M. gallisepticum and M. imitans antigens displayed the strongest reactivity with M. pneumoniae antiserum (Lanes 9 and 10), besides being avian Mycoplasmas. Opposingly, neither of the two human clinical isolates of Ureaplasma urealyticum showed any reactivity (Lanes 7 and 8). However, antigens of the remaining human genital Mycoplasma species M. hominis, M. fermentans, and M. genitalium yielded considerable cross-reactions with M. pneumoniae antiserum (Lanes 11, 12, and 13, respectively). Escherichia coli (Lane 2), Pseudomonas aeruginosa (Lane 4), and Klebsiella pneumoniae (Lane 6) also reacted with polyclonal M. pneumoniae antibodies, and their antigenic profile pattern was very similar. This profile was slightly different from those of the two cocci bacteria species Streptococcus pneumoniae and Staphylococcusaureus; the reactivity was less with S. pneumoniae (Lane 3) and even lesser with S.aureus (Lane 5).
Specificity of Mycoplasma pneumoniae antibodies guaranteed by adsorption procedure
After the adsorption of M. pneumoniae polyclonal antiserum against the heterologous bacterial antigens, the specificity was tested by immunoblotting (Figure 3). Apart from some proteins of M. pneumoniae antigen revealed in Lane 1 (positive control), the other antigens were almost completely non-detected (Lanes 2-13). The revealed proteins in Lane 1 are actually the specific proteins detected by the specific M. pneumoniae antibodies remaining after adsorption. Based on this result, the efficiency of the adsorption procedure was proved, since the encountered nonspecific reactions of M. pneumoniae polyclonal antiserum with the heterologous antigens, described above, were eliminated. M. pneumoniae antiserum rendered specific was then used in all the subsequent serological and immunoblotting tests.
Capture-antigen ELISA: Valid assay for routine laboratory serodiagnostic activity
Since all sera were tested in duplicate wells for the different dilutions, OD mean values were calculated and a bar graph correlating these absorbance values to their corresponding serum dilutions was plotted (Figure 4). Based on IP calculation, the set of human sera tested and presented in this paper proved positive for the M. pneumoniae IgG. Several other serum sets were also tested using this homemade ELISA, and at each time the assay proved efficient in specifically detecting the M. pneumoniae IgG and thus distinguishing the M. pneumoniae-infected patients from those uninfected (data not shown).
ELISA results were always confirmed by several immunoblot analyses simultaneously showing the positivity in M. pneumoniae and the negativity in all the other bacteria of any tested serum found to be positive by the in-house capture-antigen M. pneumoniae ELISA (data not shown).
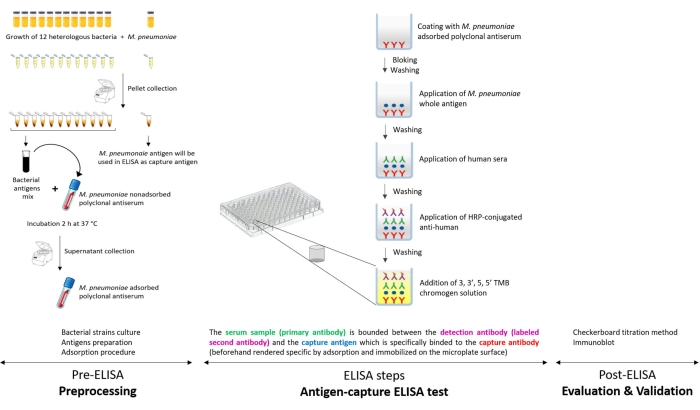
Figure 1: Schematic presentation of the antigen-capture ELISA developed in the laboratory for specific detection of Mycoplasma pneumoniae IgG. Please click here to view a larger version of this figure.
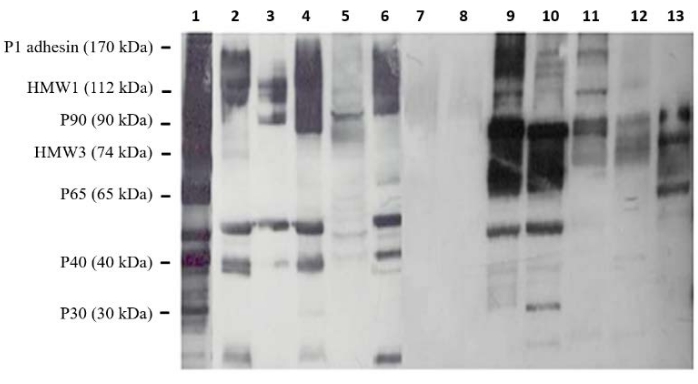
Figure 2: Immunoblot analysis of cross-reactions between Mycoplasma pneumoniae and a set of heterologous bacteria. The reactivity of M. pneumoniae non-adsorbed polyclonal antiserum was tested against antigens of Escherichia coli (Lane 2), Streptococcus pneumoniae (Lane 3), Pseudomonas aeruginosa (Lane 4), Staphylococcus aureus (Lane 5), Klebsiella pneumoniae (Lane 6), two Ureaplasma urealyticum isolates (Lanes 7 and 8), M. imitans (Lane 9), M. gallisepticum (Lane 10), M. hominis (Lane 11), M. fermentans (Lane 12), and M. genitalium (Lane 13). Lane 1: M. pneumoniae (positive control). Molecular weights of some major M. pneumoniae proteins were given on the left. Please click here to view a larger version of this figure.

Figure 3: Immunoblot confirmation of the efficiency of the adsorption procedure to eliminate cross-reactions. Reactivity of all the selected heterologous bacteria; Escherichia coli, Streptococcus pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus, Klebsiella pneumoniae, two Ureaplasma urealyticum isolates, M. imitans, M. gallisepticum, M. hominis, M. fermentans, and M. genitalium (Lanes 2-13, respectively) was tested against the adsorbed M. pneumoniae polyclonal antiserum. M. pneumoniae antigen was loaded in Lane 1 as positive control (only specific proteins were revealed). Lane MW: pre-stained protein ladder. Please click here to view a larger version of this figure.
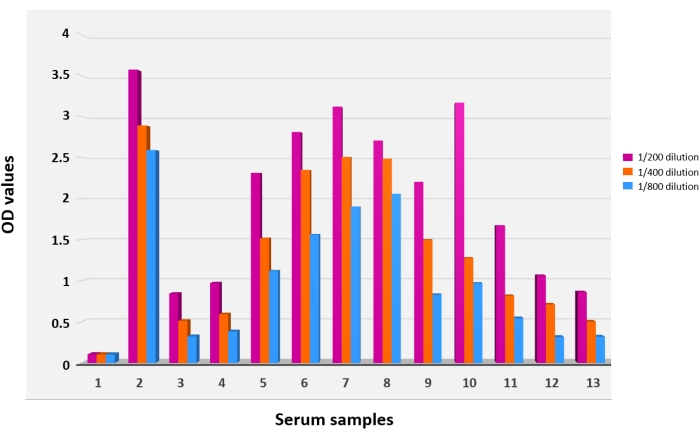
Figure 4: Results of the antigen-capture ELISA. The ELISA was performed using the adsorbed Mycoplasma pneumoniae polyclonal antiserum as a coating agent. Bar graphs 1 and 2 depict negative and positive controls, respectively. Bar graphs 3-13 depict the sera of a group of tested patients. Each serum was tested at three different dilutions: 1/200 (purple bars), 1/400 (orange bars), and 1/800 (blue bars). OD values served to calculate the index of positivity. Each reaction was performed in duplicate. Please click here to view a larger version of this figure.
| Microorganism | Strain* | Medium | Reference | |
| Mycoplasma and ureaplasma species | Mycoplasma pneumoniae | ATCC 160 20030 | SP4 | [35] |
| Mycoplasma genitalium | CIP 103767T | SP4 | ||
| Mycoplasma fermentans | ATCC 160 20026 | SP4 | ||
| Mycoplasma hominis | CIP 103715T | SP4 supplemented with arginine | ||
| Ureaplasma urealyticum | 2 clinical isolates** | SP4 supplemented with urea | ||
| Mycoplasma gallisepticum | CIP S6 15302 | Frey | [36] | |
| Mycoplasma imitans | ATCC 160 20037 | Frey | ||
| Other bacteria | Escherichia coli | ATCC 25922 | LB broth | [37] |
| Klebsiella pneumoniae | Clinical isolate*** | LB broth | ||
| Pseudomonas aeruginosa | ATCC 27853 | LB broth | ||
| Staphylococcus aureus | ATCC 25923 | LB broth | ||
| Streptococcus pneumoniae | Clinical isolate*** | Blood broth | ||
| * ATCC: American Type Culture Collection, CIP: Collection Institut Pasteur | ||||
| ** Isolates were collected from clinical samples voluntarily shipped to the Laboratory of Mycoplasmas (Pasteur Institute of Tunis) for routine diagnostic | ||||
| *** Isolates were collected from clinical samples voluntarily shipped to the Laboratory of Bacteriology (Pasteur Institute of Tunis) for routine diagnostic | ||||
Table 1: List of bacteria strains used in this study.
Discussion
This paper presents a general description of an in-house ELISA mainly developed to ensure specific screening of M. pneumoniae infection. The details about the protocol of the ELISA assay itself as well as some pre- and post-processing steps are provided. The specificity of this assay is ensured by the adsorption technique. This procedure was previously described in ELISA tests developed for the diagnosis of human and avian Mycoplasmas17,24. It was also employed in ELISA assays for the diagnosis of other bacterial infections such as Lyme disease25. In these three papers, adsorption was proved to help in increasing ELISA specificity without scarifying or decreasing the sensitivity17,24,25. In the case of the present ELISA assay, the appropriate bacteria species selected for adsorption can be partitioned into two groups: the first group contains some pathogenic human and avian Mollicutes species (M. genitalium, M. fermentans, M. hominis, U. urealyticum, M. gallisepticum, and M. imitans), while the second contains the most frequent bacteria known to infect humans (E. coli, P. aeruginosa, and S. aureus) and those notoriously known for their tropism to the respiratory tract (K. pneumoniae and S. pneumoniae). The wide range selection of heterologous bacteria was based on the fact that the dogma of species and organ specificity is not respected anymore. Many reports on the detection of microorganisms in unfamiliar habitats have been previously published. For instance, M. pneumoniae has been isolated from many extra-pulmonary sites and found to be associated with a varied range of extra-pulmonary complications with mild to severe symptoms, although it was assumed to be specific to the respiratory tract6,7. The opposite case (isolating genital Mycoplasma species from the respiratory tract) was signalled for M. hominis and M. fermentans26,27. The inclusion of avian Mycoplasma spp. in the serodiagnosis test of human infection was based on previous studies reporting the possibility of infection of humans (notably immunocompromised persons and veterinarians) with Mycoplasma spp. originated from domestic animals28,29,30. It is true that, until now, no paper was found evoking the isolation of M. gallisepticum and M. imitans (the two species included in the adsorption procedure in this paper) from humans. Despite this, nothing is impossible, especially since these species are among the most regularly handled species by veterinarians and are phylogenetically too close to M. pneumoniae. In fact, these two avian Mycoplasma spp. have shown the strongest reactivity with M. pneumoniae antiserum (as illustrated in Figure 2). Briefly, while designing the adsorption procedure, a relatively large spectrum of suspicious bacteria that could share antigens with M. pneumoniae was included. The choices made were based on phylogenetic relatedness and tropism to the respiratory tract. Nevertheless, due to human flora diversity and antigenic variability, it is impossible to predict and include all the possible heterologous bacterial species and strains that could cross-react with M. pneumoniae antibodies. Thus, the possibility of cross-reaction between M. pneumoniae antiserum, even after adsorption, and other bacteria remains probable. Moreover, since M. pneumoniae is commonly known to infect children and young adults, the adsorption procedure can be further be improved in the future by the addition of other pediatric respiratory pathogens such as Haemophilus influenzae31 and Streptococcus pyogenes32 into the adsorption bacterial shortlist.
The purpose of this ELISA is to detect the presence of the target antibody (specific M. pneumoniae-IgG) in the human sera. So, to be able to both use the serum as a sample (since it is the easiest to collect and the less painful for patients) and to ensure specificity, the in-house ELISA is based on the principle of two ELISA types: indirect and sandwich ELISA. The serum sample (primary antibody) is bound between the detection antibody (labeled second antibody) and the capture antigen, which was specifically linked to the capture antibody (beforehand rendered specific by adsorption and immobilized on the microplate surface). Given these unique characteristics, it is believed that this ELISA is effective and special. Nevertheless, this test is not perfect, and troubles can occur. False positive and false negative results can still be encountered, sometimes because of the serum quality or the infection phase, and sometimes because of errors during the protocol application in the laboratory. For example, ELISA serodiagnosis results can be affected even if the wells are not sufficiently washed. Also, the incubation (temperature and time) could influence the test outcomes. Microplate and blocking agent selection was also reported to be a critical step during ELISA development33,34. For all these reasons, it is important to continuously work to improve this ELISA test in order to determine the optimal parameters and conditions, such as the dilution of the different antibodies (serum samples, capture antibody, detecting antibody), the incubation and washing steps, and the inclusion of the appropriate negative and positive controls.
The culture of bacteria and the production of their antigens to perform the adsorption procedure could be considered a heavy step, especially for Mollicutes species. Nevertheless, it is believed that this is important as this step greatly helps in enhancing the ELISA specificity and in avoiding further confirmation tests such as immunoblotting or PCR. Furthermore, in the context of M. pneumoniae infection, an effective and reliable diagnosis is definitely required in order to establish an adequate and appropriate antibiotic therapy, as the use of beta-lactam drugs in the treatment of community-acquired pneumonia is ineffective against M. pneumoniae8. After years of being used in routine diagnostic activity in the laboratory, it is believed that the antigen-capture ELISA, described in this paper, represents a valid tool for the specific screening of M. pneumoniae infection.
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
This study was funded by the Tunisian Ministry of Health and the Tunisian Ministry of Higher Education and Scientific Research.
Materials
| 4-chloro-1-naphtol | Sigma-Aldrich | C6788-50 TAB | |
| Bacto peptone | BD | 211677 | |
| Bacto tryptone | BD | 211705 | |
| Bovine serum albumin | Sigma-Aldrich | A9647-50 G | |
| Carbonate bicarbonate buffer | Sigma-Aldrich | C3041-50 Cap | |
| Casein | Sigma-Aldrich | C7078-1 KG | |
| CMRL1066 | VWR | P0058-N1L | |
| D-Glucose | Sigma-Aldrich | G7528-1KG | |
| Difco PPLO Broth | BD | 255420 | Frey media |
| ELISA plate-Reader | MULTISKAN GO, Thermo Scientific | Ref: 51119200 | |
| Fetal bovine serum | Capricorn Scientific | FBS-12A | |
| Goat peroxidase-conjugated anti-human IgG | Abcam | ab6759 | |
| Goat peroxidase-conjugated anti-rabbit IgG | Life Technologies | 656120 | |
| Hydrochloric Acid 37% | Prolab | 2025.290 | |
| Hydrogen peroxide (H2O2), solution 30% | Scharlau | HI01361000 | |
| L-arginin | Sigma-Aldrich | A5006-1KG | |
| LB broth | Prepared in Pasteur Institute of Tunis | Provided by the laboratory of bacteriology of the Pasteur Institute of Tunis | |
| Mycoplasma pneumoniae polyclonal antiserum produced in rabbit | Produced in Pasteur Institute of Tunis | Serum was produced by rabbit immunization at the Pasteur Institute of Tunis | |
| Nicotinamide adenine dinucleotide | Sigma-Aldrich | N7004-10G | |
| Nunc Maxisorp flat-bottom 96-well microtiter plate | Invitrogen | 44-2404-21 | |
| Penicillin G sodium (1 MIU) | PANPHARMA | ||
| Phenol red | fluka chemika | 77660 | |
| Skim milk | MP Biomedicals | 902887 | |
| Sodium bicarbonate | Sigma-Aldrich | S6297-1KG | |
| Sodium carbonate | Sigma-Aldrich | S7795-1KG | |
| Sodium chloride (NaCl) | Novachim | PS02805 | |
| Sulfuric acid (H2SO4) 95-97% | Merck | 1007311011 | |
| Thimerosal | USB | 22215 | |
| TMB substrate (3,3’, 5,5’ TetraMethyl-Benzidine solution) | Abcam | ab142042 | |
| Trizma base | Sigma-Aldrich | T6791-1 KG | |
| Tween 20 | Sigma-Aldrich | 1379-500 ML | |
| Yeast extract, powder, Ultrapure | Thermo Scientific | J23547 |
Riferimenti
- Razin, S., Yogev, D., Naot, Y. Molecular biology and pathogenicity of mycoplasmas. Microbiology and Molecular Biology Reviews. 62 (4), 1094-1156 (1998).
- Chanock, R. M. Mycoplasma infections of man. The New England Journal of Medicine. 273 (22), 1199-1206 (1965).
- Braun, G. S., Wagner, K. S., Huttner, B. D., Schmid, H. Mycoplasma pneumoniae: Usual suspect and unsecured diagnosis in the acute setting. The Journal of Emergency Medicine. 30 (4), 371-375 (2006).
- Baseman, J. B., Cole, R. M., Krause, D. C., Leith, D. K. Molecular basis for cytadhesion of Mycoplasma pneumoniae. Journal of Bacteriology. 151, 1514-1522 (1982).
- Waldo, R. H., Krause, D. C. Synthesis, stability, and function of cytadhesin P1 and accessory protein B/C complex of Mycoplasma pneumoniae. Journal of Bacteriology. 188 (2), 569-575 (2006).
- Waites, K. B., Balish, M. F., Atkinson, T. P. New insights into the pathogenesis and detection of Mycoplasma pneumoniae infections. Future Microbiology. 3 (6), 635-648 (2008).
- Narita, M. Pathogenesis of extrapulmonary manifestations of Mycoplasma pneumoniae infection with special reference to pneumonia. Journal of Infection and Chemotherapy. 16 (3), 162-169 (2010).
- Kashyap, S., Sarkar, M. Mycoplasma pneumonia: Clinical features and management. Lung India. 27 (2), 75-85 (2010).
- Zhang, L., Zong, Z. Y., Liu, Y. B., Ye, H., Lv, X. J. PCR versus serology for diagnosing Mycoplasma pneumoniae infection: A systematic review & meta-analysis. Indian Journal of Medical Research. 134 (3), 270-280 (2011).
- Saraya, T. Mycoplasma pneumoniae infection: Basics. Journal of General and Family Medicine. 18 (3), 118-125 (2017).
- Jacobs, E. Serological diagnosis of Mycoplasma pneumoniae infections: a critical review of current procedures. Clinical Infectious Diseases. 17, 79-82 (1993).
- Kok, T. W., Marmion, B. P., Varkanis, G., Worswick, D. A., Martin, J. Laboratory diagnosis of Mycoplasma pneumoniae infection: 3. Detection of IgM antibodies to M. pneumoniae by a modified indirect haemagglutination test. Epidemiology and Infection. 103 (3), 613-623 (1989).
- Smith, T. F. Mycoplasma pneumoniae infections: diagnosis based on immunofluorescence titer of IgG and IgM antibodies. Mayo Clinic Proceedings. 61 (10), 830-831 (1986).
- Busolo, F., Tonin, E., Conventi, L. Enzyme-linked immunosorbent assay for detection of Mycoplasma pneumoniae antibodies. Journal of Clinical Microbiology. 12 (1), 69-73 (1980).
- Van Griethuysen, A. J., et al. Use of the enzyme-linked immunosorbent assay for the early diagnosis of Mycoplasma pneumoniae infection. European Journal of Clinical Microbiology. 3 (2), 116-121 (1984).
- Raisanen, S. M., Suni, J. I., Leinikki, P. Serological diagnosis of Mycoplasma pneumoniae infection by enzyme immunoassay. Journal of Clinical Pathology. 33 (9), 836-840 (1980).
- Sasaki, T., Bonissol, C., Stoilikovic, B., Ito, K. Demonstration of cross-reactive antibodies to mycoplasmas in human sera by ELISA and immunoblotting. Microbiology and Immunology. 31 (7), 639-648 (1987).
- Brunner, H., et al. Unexpectedly high frequency of antibody to Mycoplasma pneumoniae in human sera as measured by sensitive techniques. Journal of Infectious Diseases. 135 (4), 524-530 (1977).
- Plackett, P., Marmion, B. P., Shaw, E. J., Lemcke, R. M. Immunochemical analysis of Mycoplasma pneumoniae: 3. Separation and chemical identification of serological active lipids. Australian Journal of Experimental Biology and Medical Science. 47 (2), 171-195 (1969).
- Ponka, A. The occurrence and clinical picture of serologically verified Mycoplasma pneumoniae infections with emphasis on central nervous system, cardiac and joint manifestations. Annals of Clinical Research. 11, 1-60 (1979).
- Bradford, M. M. A Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry. 72 (1-2), 248-254 (1976).
- Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227 (5259), 680-685 (1970).
- Towbin, H., Staehlin, T., Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets. Procedure and some applications. Proceedings of National Academy of Sciences. 76 (9), 4350-4354 (1979).
- Ben Abdelmoumen, B., Roy, S. An enzyme-linked immunosorbent assay for detection of avian mycoplasmas in culture. Avian Diseases. 39, 85-93 (1995).
- Fawcett, P. T., O’Brien, A. E., Doughty, R. A. An adsorption procedure to increase the specificity of enzyme-linked immunosorbent assays for lyme disease without decreasing sensitivity. Arthritis and Rheumatism. 32 (8), 1041-1044 (1989).
- Nicolson, G. L., Nasralla, M. Y., Nicolson, N. L. The pathogenesis and treatment of mycoplasmal infections. Antimicrobics and Infectious Disease Newsletter. 17 (11), 81-88 (1999).
- Meyer, R. D., Clough, W. Extragenital Mycoplasma hominis infections in adults: emphasis on immunosuppression. Clinical Infectious Diseases. 17, 243-249 (1993).
- Pitcher, D. G., Nicholas, R. A. J. Mycoplasma host specificity: Fact or fiction. Veterinary Journal. 170 (3), 300-306 (2005).
- Lierz, M., Jansen, A., Hafez, H. M. Mycoplasma lipofaciens transmission to veterinarian. Emerging Infectious Diseases. 14 (7), 1161-1163 (2008).
- Baker, A. S., Ruoff, K. L., Madoff, S. Isolation of Mycoplasma species from a patient with seal finger. Clinical Infectious Diseases. 27 (5), 1168-1170 (1998).
- Slack, M., P, E. A review of the role of Haemophilus influenzae in community-acquired pneumonia. Pneumonia. 6 (1), 26-43 (2015).
- Janira Avire, N., Whiley, H., Ross, K. A review of Streptococcus pyogenes: public health risk factors, prevention and control. Pathogens. 10 (2), 248 (2021).
- Steinitz, M. Quantitation of the blocking effect of Tween 20 and bovine serum albumin in ELISA microwells. Analytical Biochemistry. 282 (2), 232-238 (2000).
- Tully, J. G., Whitcomb, R. F., Clark, H. F., Williamson, D. L. Pathogenic mycoplasmas: cultivation and vertebrate pathogenicity of a new spiroplasma. Science. 195 (4281), 892-894 (1977).
- Frey, M. L., Hanson, R. P., Andrson, D. P. A medium for the isolation of avian mycoplasmas. American Journal of Veterinary Research. 29 (11), 2163-2171 (1968).
- Bertani, G. Studies on lysogenesis. I. The mode of phage liberation by lysogenic Escherichia coli. Journal of Bacteriology. 62 (3), 293-300 (1951).

