A Computational Modeling Approach to Investigate the Influence of Hyperthermia on the Tumor Microenvironment
Summary
The article describes a protocol to simulate the transient temperature profiles and the coupled spatiotemporal variation of the interstitial fluid pressure following the heating delivered by a dipolar radiofrequency hyperthermia system. The protocol can be used to assess the response of biophysical parameters characterizing the tumor microenvironment to interventional hyperthermia techniques.
Abstract
The biophysical properties of the tumor microenvironment differ substantially from normal tissues. A constellation of features, including decreased vascularity, lack of lymphatic drainage, and elevated interstitial pressure, diminishes the penetration of therapeutics into tumors. Local hyperthermia within the tumor can alter microenvironmental properties, such as interstitial fluid pressure, potentially leading to improvements in drug penetration. In this context, multi-physics computational models can provide insight into the interplay between the biophysical parameters within the tumor microenvironment and can guide the design and interpretation of experiments that test the bioeffects of local hyperthermia.
This paper describes a step-by-step workflow for a computational model coupling partial differential equations describing electrical current distribution, bioheat transfer, and fluid dynamics. The main objective is to study the effects of hyperthermia delivered by a bipolar radiofrequency device on the interstitial fluid pressure within the tumor. The system of mathematical expressions linking electrical current distribution, bioheat transfer, and interstitial fluid pressure is presented, emphasizing the changes in the distribution of the interstitial fluid pressure that could be induced by the thermal intervention.
Introduction
Elevated interstitial fluid pressure (IFP) is a hallmark of solid tumors1. The leakage of fluid into the interstitium from hyperpermeable blood vessels is imbalanced by the egress of fluid due to compressed intratumoral veins and absent lymphatics1,2,3. In concert with other biophysical parameters that are abnormal within the tumor microenvironment (TME), including solid stress and stiffness, elevated IFP undermines the efficacy of both systemic and local drug delivery4,5,6. Interstitial fluid pressure in solid tumors ranges from 5 mmHg (glioblastoma and melanoma) to 30 mmHg (renal cell carcinoma) compared to 1-3 mmHg in normal tissue2. High IFP is responsible for increasing the fluid flow toward the margin of the tumor and exposes stromal cells, infiltrated cells, and other extracellular components to shear stress1,4. Mechano-biological alterations sustain an immunosuppressive TME, for example, by increasing endothelial sprouting, which supports angiogenesis, cancer cell migration and invasion, transforming growth factor-β (TGF-β) expression, and stromal stiffening7,8,9.
Several studies have explored energy-based therapies with the intent of decreasing IFP, including low-intensity ultrasound, high-intensity focused ultrasound, pulsed electric fields, and thermal therapies5,10,11. Heating to temperatures in the range of 40-43 °C, referred to as mild hyperthermia, has been shown to increase tumor blood perfusion and may thus contribute to expanding compressed veins and reducing vascular pressure by facilitating intravasation and drainage of interstitial fluid11,12. Some recent studies have shown the potential of hyperthermia to reduce IFP and consequently, to facilitate the distribution of drug or contrast agents within a tumor13,14. These studies also show increased T-cell infiltration following hyperthermia compared to no treatment control groups13.
The promising results from in vivo small animal experiments motivate further studies employing computational approaches to advance the understanding of how physical parameters within the TME are affected by physical interventions4,15,16,17. Results from computational models can complement in vivo experimental studies to uncover the cause-effect relationship underlying the local heating (or other external energy sources) and the IFP. This can be particularly instructive given the challenges with measuring spatial variations in IFP with catheter- and needle-based pressure transducers, which typically provide point measurements9,16,18,19. In the context of drug delivery, an understanding of the key biophysical mechanisms is essential to define the appropriate heating protocol as well as the time window for drug injection to enhance the likelihood of effective drug distribution. Quantitative information in terms of changes in biophysical characteristics of the TME, including but not limited to the IFP, could also give insights into the interpretation of the immunological response (e.g., T-cell infiltration) to external stimuli.
We present a protocol for computational modeling of thermally mediated changes to tumor IFP profiles. Specifically, the protocol details how to model a custom small-animal apparatus for delivering controlled thermal therapy with radiofrequency current, simulate transient temperature profiles following heating, and couple fluid dynamic simulations to compute the spatio-temporal variation of tumor IFP in response to thermal therapy. This model mirrors the essential features of the experimental setup we used in a subcutaneous tumor model (McArdle RH7777, ATCC) in a prior experimental study20.
Figure 1 shows the computational model we implemented to calculate thermally induced changes in IFP in a tumor surrounded by normal tissue. A pair of hypodermic needles inserted in the tumor is modeled to deliver heating with radiofrequency current at 500 kHz. A porous material is assumed in the tumor domain, composed of two phases: the solid phase represents the solid extracellular matrix, and the fluid phase represents the interstitial fluid. In the case of a pressure change or a matrix deformation resulting from an external stimulus, for example increase of temperature, the solid and fluid components rearrange. This causes the movement of the interstitial fluid through the extracellular solid matrix16,17,21.
From the poroelasticity theory, the stress tensor S (Pa) (equation [1]) is the combination of the elastic term describing the change in volume of the solid component relative to the initial conditions, and a porous term describing the stress induced by the hydrostatic pressure of the fluid component.
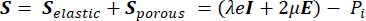 (1)
(1)
Where, λ, μ (Pa) are the Lamé parameters, E is the strain tensor, e is the volumetric strain tensor, Pi (Pa) is the interstitial fluid pressure (I is the identity matrix). Steady-state conditions are assumed for the solid component under poroelastic stress, which means that the stress tensor components are orthogonal, 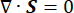 .
.
Figure 2 shows the system of mathematical equations implemented in the described poroelastic model and the interplay between the components of the presented multi-physics model. The workflow of the computational simulations includes:
Electrical problem equations. The solution of the electrical problem equations provides the time-averaged RF heat source Q (Joule heating). To this end, a quasi-static approximation to Maxwell's equations is used to calculate the distribution of the time-averaged electric field E (V/m) (Figure 2, block 1).
Thermal problem equations. The solution of the Pennes bioheat equation (Figure 2, block 2) provides the spatial and temporal variation of the temperature T (°C) as a result of the heat source (Q) linked to the absorbed electromagnetic energy, the passive heating linked to the thermal conduction of the tissues (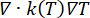 ), and the heat sink effect of the tissue blood perfusion (cWb(T) (T – Tb)). The heat sink term approximates the heat exchange between the blood flowing in the microvasculature and adjacent tissue where electromagnetic power is absorbed. The heat transfer equation also includes the advection term (
), and the heat sink effect of the tissue blood perfusion (cWb(T) (T – Tb)). The heat sink term approximates the heat exchange between the blood flowing in the microvasculature and adjacent tissue where electromagnetic power is absorbed. The heat transfer equation also includes the advection term (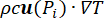 ), which describes the change in the temperature caused by the movement of interstitial fluid through the extracellular matrix of the poroelastic model. However, this term has a negligible impact on the temperature profile compared to the other mechanisms responsible for the temperature change.
), which describes the change in the temperature caused by the movement of interstitial fluid through the extracellular matrix of the poroelastic model. However, this term has a negligible impact on the temperature profile compared to the other mechanisms responsible for the temperature change.
Fluid-dynamic problem equations. The conservation of mass equation (Figure 2, block 3) combined with Darcy's law (Figure 2, block 4) gives as an output the spatial and temporal variation of the interstitial fluid pressure Pi resulting from the balance between the source (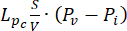 ) and the sink (
) and the sink (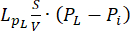 ) of the fluid. The transient pressure term on the left side of the mass conservation equation,
) of the fluid. The transient pressure term on the left side of the mass conservation equation, 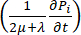 , describes the rearrangement of the fluid and solid components in the poroelastic material. This is caused by the variation of the interstitial fluid pressure, Pi, driven by the variation of the vascular pressure Pv as a function of the temperature.
, describes the rearrangement of the fluid and solid components in the poroelastic material. This is caused by the variation of the interstitial fluid pressure, Pi, driven by the variation of the vascular pressure Pv as a function of the temperature.
The difference between the vascular pressure (Pv) and the interstitial fluid pressure (Pi) is the source of the fluid that flows through the extracellular matrix. The sink term is linked to the pressure difference between the lymphatic vessels (PL) and the interstitial space (Pi). In normal tissue, the pressure in the lymphatic vasculature (~ -6-0 mmHg) is up to two-fold lower than the interstitial fluid pressure13. This pressure difference ensures the efficacy of the lymphatic vessels to drain the excess of the fluid extravasating from the wall of blood vessels into the interstitium. For the tumor model presented here, we neglected the contribution of the lymphatic system4,16,22.
Mathematical expressions from Equations (2) to (5) are used to describe the temperature dependence of the electrical and thermal conductivity of tissue and tissue blood perfusion23,24. Two different mathematical models are used to describe the temperature dependence of blood perfusion in the normal and tumor tissue domains, respectively24,25. The models show that the blood perfusion increases with the temperature up to nine times compared to the baseline in the normal tissue and only approximately two times of the baseline value in the tumor domain. For both models, the increase in the blood perfusion is limited to the temperatures within the mild hyperthermia range (below 45 °C). It is worth mentioning that the mathematical expressions, Equations (4) and (5), do not fully describe the mechanisms underlying the temperature-dependent changes in the blood perfusion in the two different types of tissue. However, they help represent the limited perfusion that typically characterizes the tumor microenvironment compared to normal tissues.
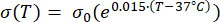 (2)
(2)
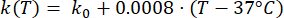 (3)
(3)
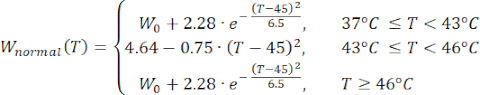 (4)
(4)
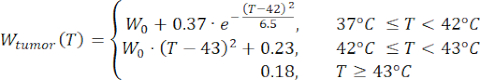 (5)
(5)
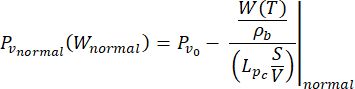 (6)
(6)
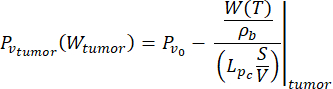 (7)
(7)
In this study, we used Equations (6) and (7) to model the vascular pressure as a function of the blood perfusion both for normal and tumor tissue models26. From Equations (4) and (5), the blood flow rate can be expressed as the ratio between the blood perfusion and the blood density. The relationship between blood flow and vascular pressure is well established in the literature3: the blood flow rate and the geometrical resistance (or conductivity, Lp) of the vasculature determine the pressure difference within the blood vessel. The vascular pressure can be expressed as a function of the temperature (Equations (6) and (7)), leveraging on this relationship and the temperature-dependent model of the blood perfusion (Equations (4) and (5)).
The implementation of the computational workflow (Figure 2) and the temperature-dependent properties of the tissue models are described in detail in the following section. All material properties and their descriptions and baseline values (i.e., at body temperature) are listed in Table 1. See the Table of Materials for details about COMSOL Multiphysics installed on a computer used to implement this computational protocol. The electrical problem was modeled using the AC/DC module; bioheat transfer was modeled using heat transfer physics; and the fluid-dynamics problem was modeled using the Mathematics interface.
Protocol
Representative Results
Discussion
We present a computational modeling protocol to couple transient electric-thermal simulations with fluid-dynamic simulations to study the impact of RF-hyperthermia on thermal and interstitial fluid pressure profiles in tumors. The key aspect is in the building of a numerical workflow capable of capturing the relationship existing between temperature and vascular pressure, which in turn drives the changes in interstitial fluid pressure.
We used the relationship between vascular pressure and blo…
Divulgazioni
The authors have nothing to disclose.
Acknowledgements
The study was supported by grants from the National Science Foundation (no. 2039014) and the National Cancer Institute (R37CA269622).
Materials
| COMSOL Multiphysics (v. 6.0) | COMSOL AB, Stockholm, Sweden | Software used to implement the computational workflow described in the protocol | |
| Dell 1.8.0, 11th Gen Intel(R) Core(TM) i7-11850H @ 2.50GHz, 2496 Mhz, 8 Core(s), 16 Logical Processor(s), 32 GB RAM | Dell Inc. | Laptop used to run computational simulations |
Riferimenti
- Nia, H. T., Munn, L. L., Jain, R. K. Physical traits of cancer. Science. 370 (6516), 546-556 (2020).
- Heldin, C. -. H., Rubin, K., Pietras, K., Östman, A. High interstitial fluid pressure – an obstacle in cancer therapy. Nature Reviews Cancer. 4 (10), 806-813 (2004).
- Jain, R. K. Determinants of tumor blood flow: a review. Ricerca sul cancro. 48, 2641-2658 (1988).
- Stylianopoulos, T., Munn, L. L., Jain, R. K. Reengineering the physical microenvironment of tumors to improve drug delivery and efficacy: from mathematical modeling to bench to bedside. Trends in Cancer. 4 (4), 292-319 (2018).
- Sheth, R. A., Hesketh, R., Kong, D. S., Wicky, S., Oklu, R. Barriers to drug delivery in interventional oncology. Journal of Vascular and Interventional Radiology. 24 (8), 1201-1207 (2013).
- Chauhan, V. P., Stylianopoulos, T., Boucher, Y., Jain, R. K. Delivery of molecular and nanoscale medicine to tumors: transport barriers and strategies. Annual Review of Chemical and Biomolecular Engineering. 2 (1), 281-298 (2011).
- Li, R., et al. Interstitial flow promotes macrophage polarization toward an M2 phenotype. Molecular Biology of the Cell. 29 (16), 1927-1940 (2018).
- Stine, C. A., Munson, J. M. Autologous gradient formation under differential interstitial fluid flow environments. Biophysica. 2 (1), 16-33 (2022).
- Provenzano, P. P., et al. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell. 21 (3), 418-429 (2012).
- Pal, K., Sheth, R. A. Engineering the tumor immune microenvironment through minimally invasive interventions. Cancers. 15 (1), 196 (2022).
- Dunne, M., Regenold, M., Allen, C. Hyperthermia can alter tumor physiology and improve chemo- and radio-therapy efficacy. Advanced Drug Delivery Reviews. 163-164, 98-124 (2020).
- Vaupel, P., et al. From localized mild hyperthermia to improved tumor oxygenation: physiological mechanisms critically involved in oncologic thermo-radio-immunotherapy. Cancers. 15 (5), 1394 (2023).
- Stapleton, S., et al. Radiation and heat improve the delivery and efficacy of nanotherapeutics by modulating intratumoral fluid dynamics. ACS Nano. 12 (8), 7583-7600 (2018).
- Li, Q., Zhou, Y., Zhang, F., McGregor, H., Yang, X. Radiofrequency hyperthermia enhances locally delivered oncolytic immuno-virotherapy for pancreatic adenocarcinoma. CardioVascular and Interventional Radiology. 45 (12), 1812-1821 (2022).
- Mpekris, F., et al. Combining microenvironment normalization strategies to improve cancer immunotherapy. Proceedings of the National Academy of Sciences. 117 (7), 3728-3737 (2020).
- Netti, P. A., Baxter, L. T., Boucher, Y., Skalak, R., Jam, R. K. Time-dependent behavior of interstitial fluid pressure in solid tumors: implications for drug delivery. Ricerca sul cancro. 15 (55), 5451-5458 (1995).
- Andreozzi, A., Iasiello, M., Netti, P. A. Effects of pulsating heat source on interstitial fluid transport in tumour tissues. Journal of The Royal Society Interface. 17 (170), 612-626 (2020).
- Leunig, M., Goetz, A. E., Messmer, K. Interstitial fluid pressure in solid tumors following hyperthermia: possible correlation with therapeutic response. Ricerca sul cancro. 52, 487-490 (1992).
- Muñoz, N. M., et al. Immune modulation by molecularly targeted photothermal ablation in a mouse model of advanced hepatocellular carcinoma and cirrhosis. Scientific Reports. 12 (1), 14449 (2022).
- Bottiglieri, A., et al. RF-hyperthermia to modulate tumor interstitial fluid pressure: an in vivo pilot study. 38th Annual Society for Thermal Medicine Meeting. , (2023).
- Baxter, L. T., Jain, R. K. Transport of fluid and macromolecules in tumors. I. Role of interstitial pressure and convection. Microvascular Research. 37 (1), 77-104 (1989).
- Stapleton, S., et al. A mathematical model of the enhanced permeability and retention effect for liposome transport in solid tumors. PLoS ONE. 8 (12), 1-10 (2013).
- Rossmann, C., Haemmerich, D. Review of temperature dependence of thermal properties, dielectric properties, and perfusion of biological tissues at hyperthermic and ablation temperatures. Critical Reviews in Biomedical Engineering. 42 (6), 467-492 (2014).
- Song, C. W., Lokshina, A., Rhee, J. G., Patten, M., Levitt, S. H. Implication of blood flow in hyperthermic treatment of tumors. IEEE Transactions on Biomedical Engineering. 31 (1), 9-16 (1984).
- Tompkins, D. T., et al. Temperature-dependent versus constant-rate blood perfusion modelling in ferromagnetic thermoseed hyperthermia: results with a model of the human prostate. International Journal of Hyperthermia. 10 (4), 517-536 (1994).
- Andreozzi, A., Iasiello, M., Netti, P. A. A thermoporoelastic model for fluid transport in tumour tissues. Journal of The Royal Society Interface. 16 (154), 0030-0046 (2019).
- Hasgall, P. A., et al. . IT’IS Database for thermal and electromagnetic parameters of biological tissues. , (2022).
- Cavagnaro, M., et al. Influence of the target tissue size on the shape of ex vivo microwave ablation zones. International Journal of Hyperthermia. 31 (1), 48-57 (2015).
- Munson, J., Shieh, A. Interstitial fluid flow in cancer: implications for disease progression and treatment. Cancer Management and Research. 19 (6), 317-328 (2014).
- Muñoz, N. M., et al. Influence of injection technique, drug formulation and tumor microenvironment on intratumoral immunotherapy delivery and efficacy. Journal for ImmunoTherapy of Cancer. 9 (2), 0018-0027 (2021).
- Swartz, M. A., Lund, A. W. Lymphatic and interstitial flow in the tumour microenvironment: linking mechanobiology with immunity. Nature Reviews Cancer. 12 (3), 210-219 (2012).
- Mehta, A., Oklu, R., Sheth, R. A. Thermal ablative therapies and immune checkpoint modulation: can locoregional approaches effect a systemic response. Gastroenterology Research and Practice. 2016, 1-11 (2016).
- Song, C. W., Park, H., Griffin, R. J. Improvement of tumor oxygenation by mild hyperthermia. Radiation Research. 155 (4), 515-528 (2001).
- Dewhirst, M. W., Oleson, J. R., Kirkpatrick, J., Secomb, T. W. Accurate three-dimensional thermal dosimetry and assessment of physiologic response are essential for optimizing thermoradiotherapy. Cancers. 14 (7), 1701 (2022).

.
