Intraluminal Middle Cerebral Artery Occlusion (MCAO) Model for Ischemic Stroke with Laser Doppler Flowmetry Guidance in Mice
Summary
The intraluminal middle cerebral artery occlusion (MCAO) model is the most frequent used model among experimental ischemic stroke models. Here we will demonstrate the entire model in detail with the guide of Laser Doppler flowmetry, and its representative results.
Abstract
Stroke is the third leading cause of death and the leading cause of disability in the world, with an estimated cost of near $70 billion in the United States in 20091,2. The intraluminal middle cerebral artery occlusion (MCAO) model was developed by Koizumi4 in 1986 to simulate this impactful human pathology in the rat. A modification of the MCAO method was later presented by Longa3. Both techniques have been widely used to identify molecular mechanisms of brain injury resulting from ischemic stroke and potential therapeutic modalities5. This relatively noninvasive method in rats has been extended to use in mice to take advantage of transgenic and knockout strains6,7. To model focal cerebral ischemia, an intraluminal suture is advanced via the internal carotid artery to occlude the base of the MCA. Retracting the suture after a specified period of time mimics spontaneous reperfusion, but the suture can also be permanently retained. This video will be demonstrating the two major approaches for performing intraluminal MCAO procedure in mice in a stepwise fashion, as well as providing insights for potential drawbacks and pitfalls. The ischemic brain tissue will subsequently be stained by 2,3,5-triphenyltetrazolium chloride (TTC) to evaluate the extent of cerebral infarction8.
Protocol
This protocol was approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Florida and is in compliance with the “Principle of Laboratory Animal Care” (NIH publication No. 86-23, revised 1985).
1. Materials
- Animals: Male C57BL/6 mice (Harlan Laboratories, Indianapolis, IN, USA) weighing 20–26 grams at the time of surgery.
- Anesthesia:
- Anesthesia machine: VetEquip inhalation anesthesia system (VetEquip Inc., Pleasanton, CA, USA).
- Inhalation anesthetic: Isoflurane (Baxter Pharmaceutics, Deerfield, IL).
- Surgical tools: SuperCut Iris scissors, straight (World Precision Instruments, Inc., Sarasota, FL, USA); Vannas micro-scissors, straight (World Precision Instruments, Inc., Sarasota, FL, USA); Dumont forceps (fine tip, 45° bent tips, World Precision Instruments, Inc., Sarasota, FL, USA); Bipolar coagulation unit (High Frequency Desiccator 900, World Precision Instruments, Inc., Sarasota, FL, USA); micro vessel clip (World Precision Instruments, Inc., Sarasota, FL, USA)
- Surgical supplies: Sterile cotton tips; Wipes; Gauze sponges; Suture (7-0 Silk suture, Ethicon, Inc., Somerville, NJ, USA); Tissue adhesive (Vetbond, 3M Company, St Paul, MN, USA); Surgical scrub: Bethadine, 70% ethanol, and sterile 0.9% saline; Eye ointment (Puralube Vet Ointment, Pharmaderm, Melville, NY, USA).
- Occlusion suture preparation for MCAO:
- Filament for MCA occlusion: 7-0 surgical Nylon monofilament suture (Ethicon, Inc., Somerville, NJ, USA).
- Heat source for blunting suture tip (Thermal Cautery Unit, World Precision Instruments, Inc., Sarasota, FL).
- Liquid silicone rubber (Silicone RTV adhesive, World Precision Instruments, Inc., Sarasota, FL, USA).
- Surgical/Dissecting microscope (Leica Stereo Microscope MZ6).
- Fiber optic illuminator (Fiber Optic illuminator FO1–150, TechniQuip Corp., Livermore, CA, USA).
- Laser doppler flowmetry (PeriFlux System 5000, Perimed, Stockholm, Sweden).
- Flexible microtip (MT B500-0 Straight Microtip, Perimed, Stockholm, Sweden)
- Temperature parameters:
- Rectal temperatures monitor (BAT-12, World Precision Instruments, Inc., Sarasota, FL).
- Thermal blanket (T/PUMP, TP600, Gaymar, NY, USA).
- Staining and quantitative measurement of volume infarct:
- 2% solution 2,3,5-triphenyltetrazolium chloride (2% TTC, Sigma-Aldrich Co., St Louis, MO, USA).
- Mice brain matrix slicer (Zivic-Miller Lab., Inc., Allison Park, PA, USA).
- Digital camera (V1253, Kodak Co., Rochester, NY, USA).
- Image J software (ImageJ 1.42q software, U.S. National Institutes of Health, Bethesda, MA, USA).
2. Pre-surgical steps
- Prior to surgery, the mice are housed under a 12:12 light/dark cycle with free access to water and rodent chow.
- Sutures for MCAO are prepared by blunting 1-2mm of the suture tip with heat and coating 3-4mm of suture shaft from its tip with liquid silicone rubber, allowing it to dry (7-0; tip diameter, 0.20-0.25mm; coating length, 3-4mm; suture length 11-13mm). Sterility should be preserved in all of the aforementioned steps.
- After weighing the mouse, anesthesia is induced with 4% isoflurane in an induction chamber and maintained during the procedure with 1.5-2% isoflurane in 30% O2 and 70% N2O gas mixture via a facemask.
- The mouse is placed in a prone position on a gauze pad lying on a temperature-controlled operating surface (thermal blanket); the head fur is shaved.
- Lubricant ophthalmic ointment is applied to both eyes, and eyelids are closed to prevent eye desiccation during the surgical process.
- A midline incision (1 cm) is made on skin overlying the calvarium from superior nuchal line to the nasion and the skin is pulled laterally to affix a flexible microtip (MT B500-0 Straight Microtip; 0.5mm diameter). The microtip is placed perpendicular to the surface of the right parietal skull (1 mm posterior and 5 mm lateral to the bregma) to monitor blood flow in the MCA territory by laser doppler flowmetry.
- Make sure that the flexible microtip is attached to skull firmly before the animal is turned to a supine position. The animal’s neck is placed on a soft pad and its nose is held in nose cone of the anesthetic apparatus. This will result in having enough space for the flexible microtip.
- A rectal temperature probe is inserted to maintain a constant animal core temperature of 37±0.5°C.
- Both forepaws and hindpaws are taped to the operating platform surface, and the tail is taped to the rectal tube.
3. MCA occlusion surgery:
- Under the operating microscope, a midline neck incision is made with scissors between the manubrium and the jaw by gently lifting the loose skin.
- The paired underlying submandibular glands are bluntly divided, with the left gland left in situ. The right submandibular gland is retracted cranially and secured along with the sternocleidomastoid muscle.
- The exposed division of the omohyoid muscle covering the carotid sheath is carefully divided with pickups.
- The right common carotid artery (CCA) is carefully separated and isolated from the vagus nerve and its sheath (located lateral to CCA).
- Hereafter, the 2 surgical methods that have been used frequently to perform MCA occlusion diverge:
Koizumi’s4 method
- Three loose collar sutures (7-0 silk) are prepared around the right CCA.
- The external carotid artery (ECA) is identified, isolated, and cauterized (2-3mm distal to bifurcation). It can subsequently be divided with micro-scissors. To avoid inadvertent peripheral damages at the cauterization site, the power should be set at 1 using the power knob. This is consistent for all cauterizations.
- Sufficient tension is applied to the most proximal collar suture on the CCA to occlude the blood flow (permanent ligation). The flow occlusion is verified by a ~50% drop of baseline cortical perfusion values.
- Then a vessel clip is placed just before the CCA bifurcation to avoid retrograde flow at the time of arteriotomy.
- An arteriotomy is performed between the proximal ligated suture and the vessel clip on the CCA with micro-scissors (closer to ligation).
- The heat-blunted silicone-coated 7-0 nylon suture is introduced via the arteriotomy and advanced till it reaches the vessel clip. The 2 remaining loose collar sutures are gently tightened around the CCA between the arteriotomy and the vessel clip to avoid back flow leakage through CCA, with caution not to traumatize the arterial wall; then the vessel clip is withdrawn.
Longa’s3 method
- The ECA is identified, dissected as much as possible (near its bifurcation into lingual and maxillary arteries), cauterized (with power set to 1), and divided with micro-scissors.
- Two loose collar sutures (7-0 silk) are prepared around the proximal ICA just above the CCA bifurcation.
- The CCA is temporarily closed with a vessel clip. The flow occlusion is verified by a ~50% drop of baseline cortical perfusion values.
- Then the ICA is temporarily closed with a vessel clip above the two loose collar sutures to avoid retrograde flow at the time of arteriotomy.
- An arteriotomy is performed using micro-scissors in the reflected ECA close to the stump.
- The heat-blunted silicone-coated 7-0 nylon suture is introduced via the arteriotomy in ECA and advanced till it reaches the applied vessel clip in ICA. The 2 loose collar sutures in proximal ICA are gently tightened around the proximal ICA to avoid reflux blood flow through ICA, with enough attention not to traumatize the arterial wall; then the vessel clip is withdrawn.
The rest of the procedure is largely convergent:
- The occluding suture is slowly advanced through internal carotid artery (ICA) toward the cranial base until a mild resistance is felt. A concurrent drop in cerebral blood flow value as a percentage (= 80%) relative to baseline is confirmed by laser doppler flowmetry. The 2 collar sutures are tightened around the inserted filament.
- For transient cerebral ischemia experiments, the occluding suture is withdrawn gently after 45 minutes. This time point can be altered for various experimental aims.
- After withdrawing the suture filament, the arteriotomy site is closed to avoid post-surgical bleeding. In Koizumi’s method, this involves securely tightening the collar sutures around the CCA. In Longa’s technique, the arteriotomy site at the base of ECA is cauterized, then the collar sutures at the proximal ICA are loosened and removed to restore the carotid flow.
- To close the incision, the submandibular gland and SCM are laid over the operative field. The wound edges are then approximated with tissue adhesive in cervical and scalp areas. Alternatively, an absorbable suture or metal staple could be used to close the wound especially when long-term survival of the animal is intended.
- After incision closure, the animal receives 1 ml warm saline subcutaneously as volume replenishment. A dose of appropriate analgesics (i.e. Buprenorphine at 0.05-0.1 mg/kg) should be used to minimize animal’s pain and discomfort during recovery period. The animal is removed from the surgical suite and placed in a warm recovery area/cage to prevent hypothermia, with free, easy access to soft food and water.
4. Post-Op neurological evaluation
After the animal regains total consciousness, neurological deficits can be evaluated by a simple scale (five-point scale) as follows3:
- no observable deficits
- failure to extend contralateral forepaw (mild focal neurologic deficit)
- circling in a direction contralateral to infarct (moderate focal neurologic deficit)
- falling in a direction contralateral to infarct (severe focal neurologic deficit)
- depressed level of consciousness without spontaneous movement
5. Staining and quantitative measurement of brain infarct volume
- After 24 hours of MCA reperfusion, the surviving mouse is weighed, deeply anesthetized with 4% isoflorane, and sacrificed according to approved protocol.
- The brain is carefully removed and placed in a mouse brain matrix slicer, and the entire cerebrum is coronally sectioned at 2 mm intervals.
- The sections are immersed in 2% TTC in phosphate buffered saline (PBS) at 37 °C for 20 minutes. After TTC staining, infracted area is shown as a white (unstained) tissue adjacent to red brick (viable) tissue (Figure 1).
- Serial sections are photographed using a digital camera and the area of infarct is measured with the image J program.
- The area of infarct (white, unstained), the area of ipsilateral hemisphere (white and red brick, stained), and the area of the contralateral hemisphere (red brick, stained) are measured for each section. The volume is calculated by summing the representative areas in all sections and multiplying by the slice thickness. After correcting for edema, the volume of infarction is calculated as follows9:
Corrected Infarct Volume (CIV), % = [contralateral hemisphere volume – (ipsilateral hemisphere volume – infarct volume)]/contralateral hemisphere volume x 100
6. Representative Results
After occlusion and reperfusion of the MCA via left CCA to establish transient cerebral ischemia, animals are sacrificed and their coronally sectioned brains are stained with TTC to evaluate the volume of infarct (Figure 1).
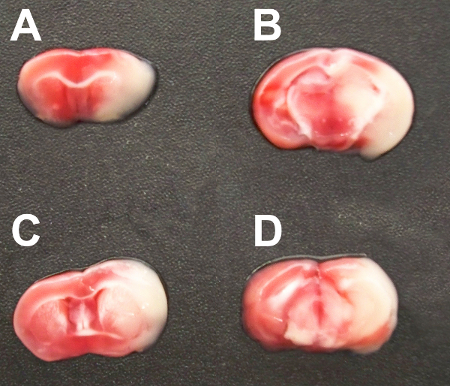
Figure 1. TTC-stained serial coronal brain sections (2 mm) from mice subjected to MCA occlusion.
Discussion
The MCAO model is an established model that replicates cerebral ischemia in murine species. This model was first used in rats and thereafter
adapted to mice. Avoiding craniectomy to eliminate the influence of surgical manipulation on blood brain barrier permeability and intracranial pressure is an advantage
of this technique. However, many variables, such as strain-related differences10, size of suture tip11, duration of occlusion6,
body temperature12, anesthesia13, and other factors can lead to inconsistency in infarct volumes in this model.
There are specific technical details that warrant mention:
- Coating the end of suture will create a smooth and manageable suture for occluding the MCA with decreased perforation risk.
- Delicately separate and secure the vagus nerve from the adjacent arteries so it is not inadvertently restricted in a collar suture. Make sure to use two collar sutures to avoid retrograde bleeding through the arteriotomy while ascending the occluding suture and tighten them gently to avoid traumatizing the arterial wall.
- The pterygopalatine artery diverges from the ICA after CCA bifurcation. To avoid deviating the tip of ascending suture into the pterygopalatine artery instead of the ICA, set up your surgical platform to keep the mouse head and neck in a straight direction. This deviation is common to both Koizumi’s and Longa’s technique, so care must be taken in either procedure.
- Laser doppler flowmetry is a method of real-time monitoring of blood flow in various anatomical regions, and is applied in our experiment to measure blood flow in the area of the brain supplied via MCA. It significantly increases the accuracy of suture positioning, and thus ensures obtaining consistent results. This is a noted advantage over less precise techniques of measuring the length of the suture to be inserted, which is susceptible to the interference of anatomical variations.
- A temporary increase in laser doppler flowmetry while inserting your suture through CCA and ICA ensures that you are moving in the right direction and close to the MCA ostium.
- The occipital artery most often arises from the proximal ECA (although sometimes from the ICA) in a posterolateral direction. It should be identified (in some cases ligated) to avoid later confusion while inserting the suture.
- To decrease variability of infarction, keep the mouse body temperature steady during the surgery and avoid hypothermia after surgery.
- Use TTC staining up to 72 hours after MCAO surgery for analyzing the infarct volume appropriately. For longer survival time points use other staining methods such as cresyl violet staining to determine the infarct size.
| Koizumi’s Method | Longa’s Method | |
| Site of suture insertion | Ipsilateral Common Carotid artery | Ipsilateral External Carotid artery |
| Permanent arterial sacrifice | Common Carotid and External Carotid arteries | External Carotid artery |
| Risk of lethal intraoperative arterial rupture/hemorrhage | Less concern | Greater concern |
| Possibility of suture diversion into Pterygopalatine artery | Yes | Yes |
| Procedural time | Generally quicker | Generally longer |
| Laser Doppler Flowmetry: Percentage drop from baseline | >= 80% | >= 80% |
| Laser Doppler Flowmetry: occlusion/reperfusion trend |
|
|
Table 1. Comparison of two methods of intraluminal middle cerebral artery occlusion (MCAO) in mice
Disclosures
The authors have nothing to disclose.
Materials
| Name of the reagent | Type | Company | Catalogue number | Comments (optional) |
|---|---|---|---|---|
| Male C57BL/6 mice | Animal | Harlan Laboratories, Indianapolis, IN, USA | 20-26 grs | |
| Dissection microscope | Microscope | Leica stereo Microscope MZ6 | 6.3:1 zoom | |
| Fiber optic illuminator | Illuminator | TechniQuip Corp., Livermore, CA, USA | FO1–150 | |
| Isoflurane | Anesthesia | Baxter Pharmaceutics, Deerfield, IL | 1001936060 | |
| SuperCut Iris scissor, straight | Surgical tool | World Precision Instruments, Inc., Sarasota, FL, USA | 14218 | |
| Vannas micro-scissor, straight | Surgical tool | World Precision Instruments, Inc., Sarasota, FL, USA | 14003 | |
| Dumont fine tip forceps | Surgical tool | World Precision Instruments, Inc., Sarasota, FL, USA | 503290 | |
| Vessel clip | Surgical tool | World Precision Instruments, Inc., Sarasota, FL, USA | 14120 | |
| High Frequency Desiccator 900 | Surgical tool | World Precision Instruments, Inc., Sarasota, FL, USA | 500397 | |
| 7-0 surgical Nylon monofilament suture | Suture | Ethicon, Inc., Somerville, NJ, USA | 1647G | Suture for occlusion |
| 7-0 surgical Silk suture | Suture | Ethicon, Inc., Somerville, NJ, USA | 7733G | Suture for ligation |
| Silicone RTV adhesive | Silicone rubber | World Precision Instruments, Inc., Sarasota, FL, USA | 1571 | |
| Vetbond | Tissue adhesive | 3M Company, St Paul, MN, USA | 70200746587 | |
| Puralube Vet Ointment | Eye ointment | Pharmaderm, Melville, NY, USA | ||
| Buprenorphine hydrochlorode | Analgesics | Hospira Inc., Lake Forest, IL, USA | NDC 0409-2012-32 | |
| Thermal Cautery Unit | World Precision Instruments, Inc., Sarasota, FL, USA | 501292 | ||
| PeriFlux System 5000 | TCD flowmetry | Perimed, Stockholm, Sweeden | ||
| BAT-12 | Rectal thermometer | World Precision Instruments, Inc., Sarasota, FL, USA | BAT-12R | |
| T/PUMP, TP600 | Thermal blanket | Gaymar, NY, USA | ||
| 2,3,5-triphenyltetrazolium chloride | Reagent | Sigma-Aldrich Co., St Louis, MO, USA | T8877 | |
| Mice brain matrix slicer | Brain slicer | Zivic-Miller Lab., Inc., Allison Park, PA, USA | BSMAS001-1 |
References
- . Recommendations on stroke prevention, diagnosis, and therapy. Report of the WHO Task Force on Stroke and other Cerebrovascular Disorders. Stroke; a journal of cerebral circulation. 20, 1407-1431 (1989).
- Lloyd-Jones, D. Heart disease and stroke statistics–2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 119, e21-e181 (2009).
- Longa, E. Z., Weinstein, P. R., Carlson, S., Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke; a journal of cerebral circulation. 20, 84-91 (1989).
- Koizumi, J., Yoshida, Y., Nakazawa, T., Ooneda, G. Experimental studies of ischemic brain edema. I. A new experimental model of cerebral embolism in rats in which recirculation can be introduced in the ischemic area. Jpn J Stroke. 8, 1-8 (1986).
- Clark, W. M., Lessov, N. S., Dixon, M. P., Eckenstein, F. Monofilament intraluminal middle cerebral artery occlusion in the mouse. Neurol Res. 19, 641-648 (1997).
- Fujii, M. Strain-related differences in susceptibility to transient forebrain ischemia in SV-129 and C57black/6 mice. . Stroke; a journal of cerebral circulation. 28, 1805-1810 (1997).
- Bederson, J. B. Evaluation of 2,3,5-triphenyltetrazolium chloride as a stain for detection and quantification of experimental cerebral infarction in rats. Stroke; a journal of cerebral circulation. 17, 1304-1308 (1986).
- Qi, X., Hosoi, T., Okuma, Y., Kaneko, M., Nomura, Y. Sodium 4-phenylbutyrate protects against cerebral ischemic injury. Mol Pharmacol. 66, 899-908 (2004).
- Barone, F. C., Knudsen, D. J., Nelson, A. H., Feuerstein, G. Z., Willette, R. N. Mouse strain differences in susceptibility to cerebral ischemia are related to cerebral vascular anatomy. J Cereb Blood Flow Metab. 13, 683-692 (1993).
- Tureyen, K., Vemuganti, R., Sailor, K. A., Dempsey, R. J. Ideal suture diameter is critical for consistent middle cerebral artery occlusion in mice. Neurosurgery. 56, 196-200 (2005).
- Barber, P. A., Hoyte, L., Colbourne, F., Buchan, A. M. Temperature-regulated model of focal ischemia in the mouse: a study with histopathological and behavioral outcomes. Stroke; a journal of cerebral circulation. 35, 1720-1725 (2004).
- Kapinya, K. J., Prass, K., Dirnagl, U. Isoflurane induced prolonged protection against cerebral ischemia in mice: a redox sensitive mechanism?. Neuroreport. 13, 1431-1435 (2002).

