Assessing the Secretory Capacity of Pancreatic Acinar Cells
Summary
Isolated pancreatic acini retain their in vivo morphology and activity and offer powerful ways for monitoring and manipulating secretion. This work demonstrates how acini can be isolated from the mouse pancreas, and how their secretory capacities can be assessed.
Abstract
Pancreatic acinar cells produce and secrete digestive enzymes. These cells are organized as a cluster which forms and shares a joint lumen. This work demonstrates how the secretory capacity of these cells can be assessed by culture of isolated acini. The setup is advantageous since isolated acini, which retain many characteristics of the intact exocrine pancreas can be manipulated and monitored more readily than in the whole animal. Proper isolation of pancreatic acini is a key requirement so that the ex vivo culture will represent the in vivo nature of the acini. The protocol demonstrates how to isolate intact acini from the mouse pancreas. Subsequently, two complementary methods for evaluating pancreatic secretion are presented. The amylase secretion assay serves as a global measure, while direct imaging of pancreatic secretion allows the characterization of secretion at a sub-cellular resolution. Collectively, the techniques presented here enable a broad spectrum of experiments to study exocrine secretion.
Introduction
The exocrine pancreas constitutes most of the mass of the mammalian pancreas and is dedicated to production and secretion of digestive enzymes1. The functional unit of the exocrine pancreas, the acinus, is a cluster of epithelial cells which forms and shares a joint lumen. Upon hormonal or neuronal stimulation, vesicles filled with enzymes are transported to the apical cell surface, fuse with it and expel their content into the lumen1-3. The secreted enzymes are then drained by a coalescing set of ducts into the small intestine, where they catalyze the breakdown of food into nutrients.
This video demonstrates how intact acini are isolated from the whole pancreas and how their secretory capacity can be assessed. Using this setup, pancreatic secretion is quantified by measuring the relative amount of amylase that was released following stimulation. Alternatively, secretion can be imaged live by the use of different sensors and dyes.
The ex vivo setup of pancreatic acini is advantageous since isolated acini, which retain many characteristics of the intact pancreas, can be manipulated and monitored more readily than in the whole animal4-6. This setup was originally developed in the late 1970’s and was since extended for the study of several exocrine tissues such as the salivary and mammary glands3-7. Notably, it allows the study of secretion with minimal interference to the complex cellular organizations of these polarized epithelia.
Protocol
NOTE: Procedures involving animal subjects have been approved by the Institutional Animal Care and Use Committee (IACUC) at the Weizmann Institute of Science.
1. Sample Preparation
- Isolation of pancreatic acini
- Prepare 300 ml of fresh Krebs-Ringer bicarbonate HEPES (KRBH) medium before starting. Mix 140 mM NaCl, 4.7 mM KCl, 1.16 mM MgCl2, 1 mM CaCl2, 11 mM Glucose, 10 mM HEPES. Add bovine serum albumin (BSA) and 0.1 mg/ml soybean trypsin inhibitor (STI) (resuspension medium). Prepare 5 – 10 ml of resuspension medium supplemented with Collagenase (~0.75 mg/ml) (digestion medium).
- Oxygenate the KRB medium for ~30 min. Add BSA after oxygenation to prevent bubbling. Bring resuspension and digestion mediums to 37 °C. NOTE: Oxygenating the medium reduces cell mortality and improves responsiveness to secretagogues. The effect of oxygenation is apparent at least for 6 hr.
- Sacrifice the mouse by cervical dislocation or CO2 asphyxiation, according to institutional animal care guidelines. Place the sacrificed animal on its back, attach it to a dissection pad and clean the abdominal surface with ethanol.
NOTE: In order to avoid contamination, autoclave the dissection tools and the water used for KRBH medium before use. Filter KRBH medium through a 0.2 µm filter. Prior to excision of the pancreas rinse the dissection tools with 70% ethanol and carry out dissection on a clean bench. Carry out viral infections in a level 3 bio-safety cabinet. Add antibiotics to the O/N culturing medium (See section 4).
- Excision of the murine pancreas
- Incision widely the abdominal skin and subcutaneous layer. Identify the liver, stomach, intestine, pancreas and spleen (Figure 1). NOTE: In the following steps, handle the pancreatic tissue gently and avoid applying excessive mechanical force to prevent auto-digestion of the tissue and cell death.
- By using two blunt forceps, pull out the small intestine. Start at the small intestine proximal end, near the exit from the stomach. Separate the pancreas from the intestine and retain it within the body cavity. Once reaching the large intestine, notice a pink/white connective tissue. Do not confuse it with the pancreas.
- Clear as much pancreatic tissue as possible without collecting fat or connective tissue. Remove the pancreas by detaching it from the spleen and stomach and cutting the abdominal blood vessels beneath it (Figure 1A-C).
NOTE: Since the yield of pancreatic acini from a single animal is high, one can refrain from collecting all the pancreatic tissue in order to prevent collecting other tissues.
- Collagenase digestion
- Rinse the pancreas a few times in resuspension medium heated to 37 °C and remove remaining parts of non-pancreatic tissues. Immerse the pancreas in ~5 ml of pre-heated digestion medium in a 20 ml scintillation vial. Mince the pancreas to pieces of 1-3mm. Place it in a heated water bath (optimally under agitation of ~100 rpm). To accelerate digestion, pipette the digested tissue up and down once every few minutes.
NOTE: The duration of collagenase digestion is dependent mainly on the collagenase concentration and on the amount of mechanical force applied on the tissue. Use of 0.75 mg/ml collagenase for approximately 15 min and pipetting the tissue with a 5 ml polystyrene pipette with a 2 mm orifice every 3 – 5 min, resulting in acini of 10 – 30 cells. Following digestion, the suspension appears turbid containing visible pieces of tissue. Since collagenases efficiencies varies between different commercial types and lots, determine the timing of digestion empirically. - Washing and filtering
- Transfer the digested tissue into a 50 ml tube and terminate digestion by adding 45 ml of resuspension medium. Filter the suspension through a coarse (~1 mm) metal mesh into a fresh tube. Centrifuge the flow-through at 100 × g for 3 min.
- Discard the supernatant and resuspended the digested pancreas in 50 ml of resuspension medium. Filter the suspension through a 70 or 100 µm nylon mesh. Centrifuge as before.
- In order to remove floating, damaged acini, discard the supernatant and resuspend the pancreatic tissue in 3 – 5 ml of resuspension medium. Apply 1 ml portions of the pancreatic acini into 15 ml conical tubes filled with 7 ml of resuspension medium. Allow the acini to settle for 2 – 3 min. Collect the acini that have settled at the bottom of the tube with a 1 ml pipette, and repeat this step 2 – 3 times.
- View the pancreatic acini under a light or dissecting microscope to test their condition. In Figure 2, observe the purity of isolation and damages inflicted upon the cells during isolation. Use these acini immediately or culture for up to 20 hr.
- Rinse the pancreas a few times in resuspension medium heated to 37 °C and remove remaining parts of non-pancreatic tissues. Immerse the pancreas in ~5 ml of pre-heated digestion medium in a 20 ml scintillation vial. Mince the pancreas to pieces of 1-3mm. Place it in a heated water bath (optimally under agitation of ~100 rpm). To accelerate digestion, pipette the digested tissue up and down once every few minutes.
2. Amylase Secretion Assay
NOTE: The amylase secretion assay serves as a global measure for the secretory capacity of pancreatic acini. This is achieved by collecting the medium in which the pancreatic acini were incubated, and determining the amylase activity of the medium, usually by using a commercial kit.
- Determine the number of experimental conditions to be examined (e.g. non-stimulated vs. stimulated). Use at least three replicates for each condition. In addition, add a set of replicates to determine the total cellular amylase content (‘total’ sample) and a set for the amylase activity of the medium (‘zero’ sample). Label a multi-well plate in which the assay will be conducted, and two sets of microtubes for each of the replicates to be used.
NOTE: 12- or 24-well plates can be used to monitor 1 and 0.5 ml of acinar suspensions, respectively. - Wash the acini twice in 30 ml of KRBH resuspension medium and incubate them in resuspension medium for 30 min at 37 °C. To the multi-well plate add a 10µl drop of secretagogue to reach the appropriate final concentration.
- Pancreatic acini respond to a gradient of secretagogue in a bi-phasic manner. The concentrations with optimal secretion values are 50 – 100 pM of Cholecystokinin (CCK) and 1µM of Carbachol (Figure 3A)5.
- Wash and resuspend the acini in resuspension medium. Ensure that the acini are distributed homogenously in the suspension and aliquot equal amounts of suspension into the multi-well plate and into the ‘total’-labeled microtubes. Incubate the plate for 30 min at 37 °C. (Preferably in a water bath under mild agitation (~50 rpm)).
NOTE: The exact number of acini may vary between experiments but should be similar in replicates within each experiment. Thus, precise aliquoting of the acini suspension is critical for comparing between different replicates and conditions. - During the incubation, produce the ‘total’ and ‘zero’ samples. Centrifuge the ‘total’ microtubes for few seconds at 5,000 × g and remove 50 µl from the supernatant into ‘zero’-labeled microtubes and place them at 4°C. Lyse the acini in the ‘total’ samples by adding Triton X-100 to the microtubes to reach a final concentration of 1%. Vortex the microtubes vigorously.
NOTE: Aliquote the ‘total’ replicates similar to the replicates of the experiment. Lysing the acini, leads to the release of the total amylase content. Alternatively, collect the acini at each condition and lyse at the end of the experiment, to determine the cellular content of each replicate. - Collect most of the acinar suspension into the first set of the pre-labeled microtubes. Centrifuge the microtubes for a few seconds at 5,000 × g. Transfer the supernatant into the second set of the pre-labeled microtubes.
- Assay the supernatant for amylase activity with a commercial kit. Calculate the percentage of amylase secreted.
NOTE: It is crucial to read the amylase activity at its linear range. Present the percentage of amylase secreted as the percent of amylase released from initial total content. Subtract the background amylase level of the medium from each triplicate and divide it by the similarly normalized initial total content.
3. Live Imaging of Pancreatic Secretion
Secretion from pancreatic acini can be monitored directly by live imaging. Using various fluorescent dyes and sensors, live imaging allows the characterization of secretion at a sub-cellular resolution.
- Prepare freshly oxygenated resuspension medium as in step 1.1 and bring it to 37 ºC. Set the microscope, and if equipped with a temperature controller, adjust it to 37 ºC.
- Wash the acinar cells in 30 ml of resuspension medium and place them in a glass or plastic slide. Note: If lipophilic dyes are to be used to label For plasma membrane labeled with lipophilic dyes, stain the acini for not more than 5 min to prevent labeling of internal membranes by endocytosis. Throughout the work, handle the acini gently and avoid excessive pipetting.
- Once at the microscope, image acini whose morphology is clear, and avoid imaging of acini which display basolateral blebs or intracellular vacuoles. Add secretagogue drop wise or by using a perfusion chamber, either during or immediately prior to the initiation of imaging.
NOTE: A high magnification lenses with high numeric apertures (e.g. ×63/1.4 or ×100/1.4) is recommended to observe subcellular structures.
4. O/N Culture of Pancreatic Acini (Alternate Cell Culture Technique)
NOTE: O/N culturing is often used for Adeno virus infection. Infection is carried out at 106-107 pfu/ml for 9 – 16 hr before examination.
- Culture pancreatic acini for up to 20 hr. Resuspended acini obtained from a single pancreas in 25 ml culture medium (DMEM supplemented with fetal calf serum (FCS) (5%), sodium-pyruvate (1%), antibiotics (1%), L-glutamine (0.5%), BSA (10 mg/ml) and STI (0.2 mg/ml), and divided to five 10cm plates. Infect the acini with virus in a level 3 bio-safety cabinet.
- Incubated pancreatic acini at 37ºC in a 5% CO2 humidified air. Allow acini to recover in oxygenated resuspension medium for 30 min before subsequent examination3,8.
Representative Results
Pancreatic acini that were isolated properly display a stereotypic morphology when viewed by transmitted light. Their basolateral domains should appear round and devoid of blebs. The apical domain is surrounded by hundreds of secretory vesicles and appears darker in color (Figure 2A, B). The nuclei are located basal to the vesicular area. Cell debris and components of the pancreatic ductal system and of the endocrine pancreas, which can be detected at early stages of acini isolation (Figure 2C, D), should be absent from the final acinar suspension (Figure 2A, B). Improper isolation may result in generation of basolateral blebs and breakage of cells which discharge their digestive enzymes into the medium (Figure 2E, F).
Amylase release from pancreatic acini depicts a bi-phasic curve when plotted against the concentration of secretagogues (Figure 3A). Without stimulation about 5% of the initial content is released during 30 min of incubation. Following stimulation, this number is elevated by up to 5 fold. Acini which were cultured O/N, display a lower ratio of stimulated vs. basal amylase release when compared to freshly isolated acini. Importantly, cultured acini that were infected by Adeno viruses display a high basal amylase secretion and a sensitized stimulated secretion (Figure 3B, C). It is thus critical to infect control acini with a similar dose of control virus.
Live imaging of acini should allow clear identification of the basolateral and apical domains of acinar cells. Lipophilic dyes can be instrumental for distinguishing between the narrow apical domain and the lateral and basolateral aspects of the cells (Figure 4). Under physiological stimulation, secretion is directed exclusively to the apical surface1,9. Use of the F-actin probe Lifeact-GFP allows live visualization of secretory vesicles, since pancreatic zymogen vesicles undergo actin coating shortly before exocytosis (Figure 4).
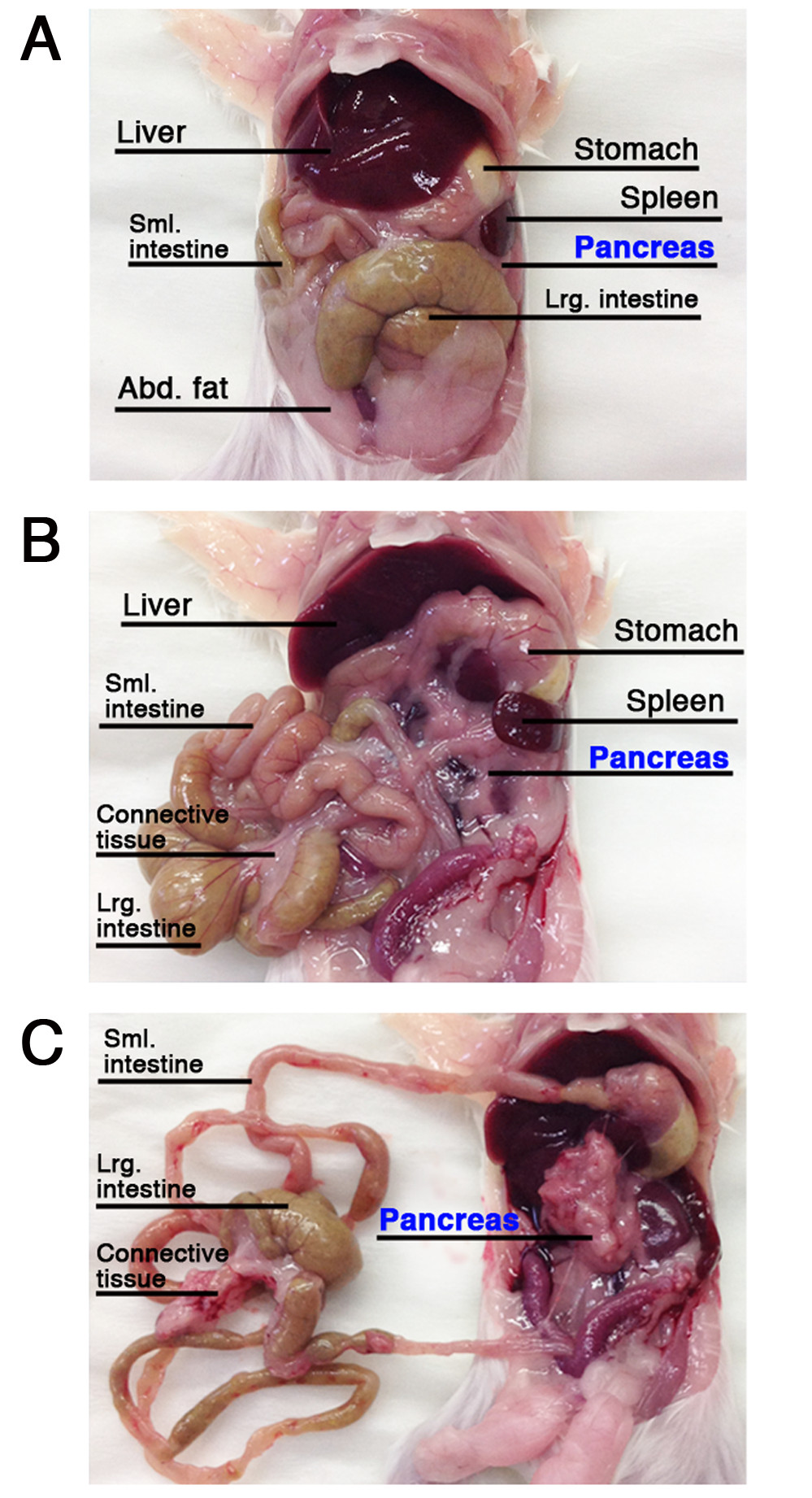
Figure 1. Localization of the pancreas and adjacent organs. (A-C) The pancreas and adjacent organs after the incision of the abdominal skin and subcutaneous layer (A), before and after pancreatic separation from neighboring organs, (B) and (C), respectively. Please click here to view a larger version of this figure.
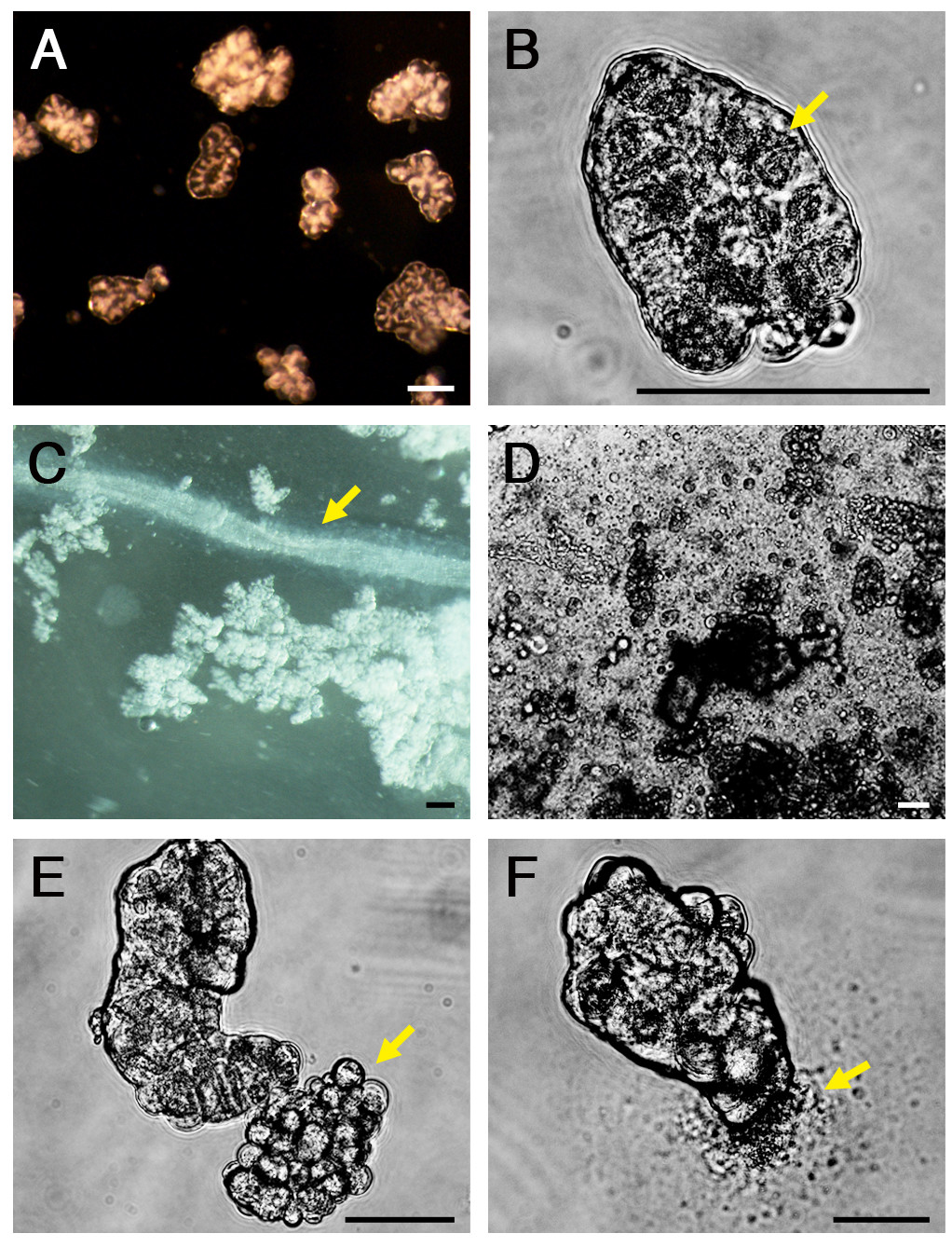
Figure 2. Isolation of pancreatic acini. (A) and (B) Pancreatic acini isolated properly display a stereotypic morphology; their basolateral domains appear round and devoid of blebs, while their apical domain are surrounded by hundreds of secretory vesicles and appears darker in color (arrow). (C) and (D) Cell debris and components s of the pancreatic ductal system can be detected at early stages of acini isolation (arrow points to an exocrine duct). (E) and (F) Improper isolation results in generation of basolateral blebs (E, arrow), and breakage of cells, which discharge their digestive enzymes into the medium (F, arrow). Scale bars represent 100 µm. Please click here to view a larger version of this figure.
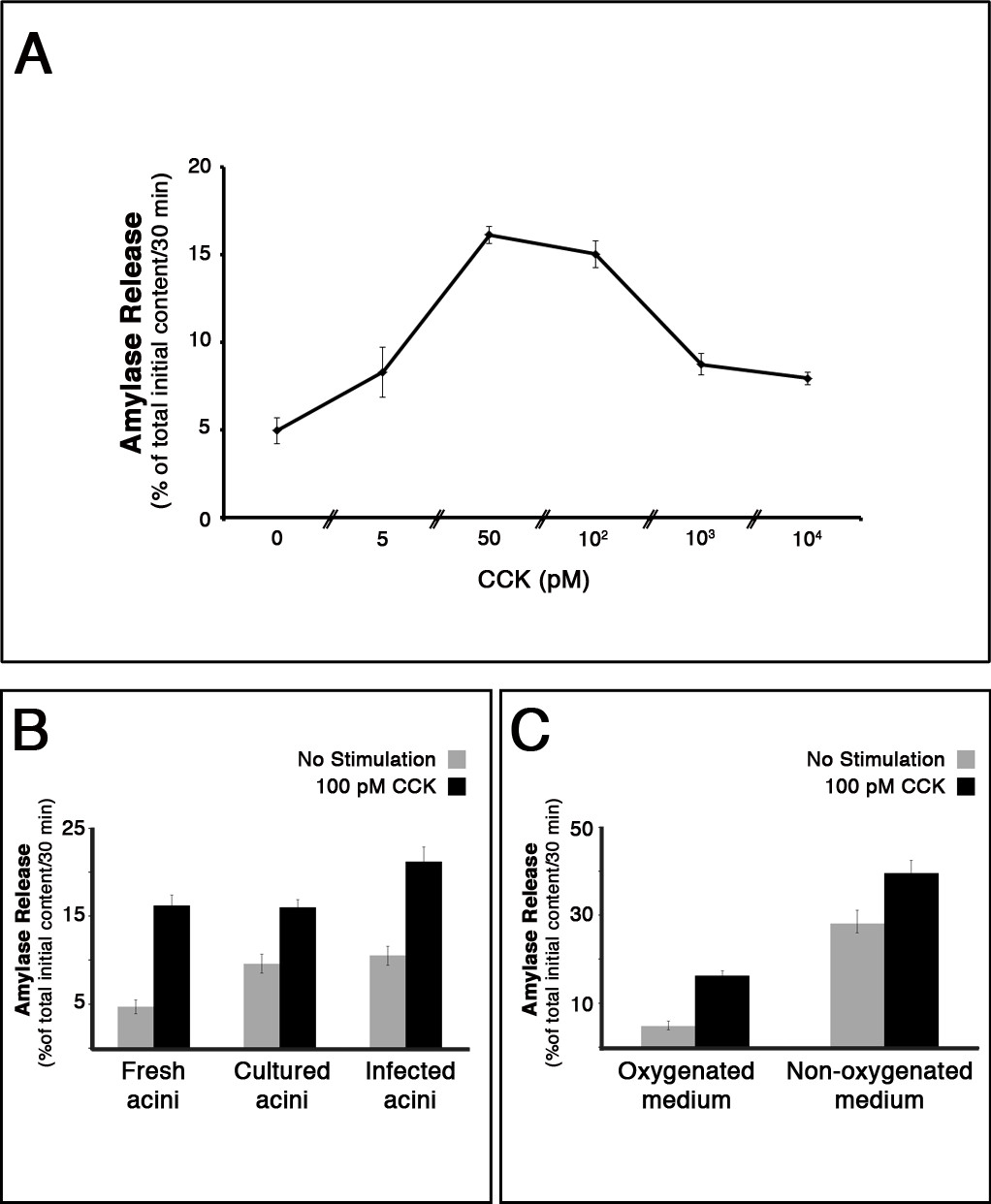
Figure 3. Measurement ofamylase release from isolated acini. (A) Amylase release from pancreatic acini depicts a bi-phasic curve when plotted against the concentration of a secretagogue. Acini were isolated and left to recover for 30 min before stimulation with the indicated concentrations of Cholecystokinin (CCK). Results represent the average and s.e.m. of three independent experiments. (B) Acini which were cultured O/N, display a lower ratio of stimulated vs. basal amylase release when compared to freshly isolated acini. Cultured acini that were infected by Adeno viruses display a high basal amylase secretion and a sensitized stimulated secretion. (C) Non-oxygenating culture conditions lead to excessive discharge of amylase, which masks the stimulatory effect of the secretagogue.
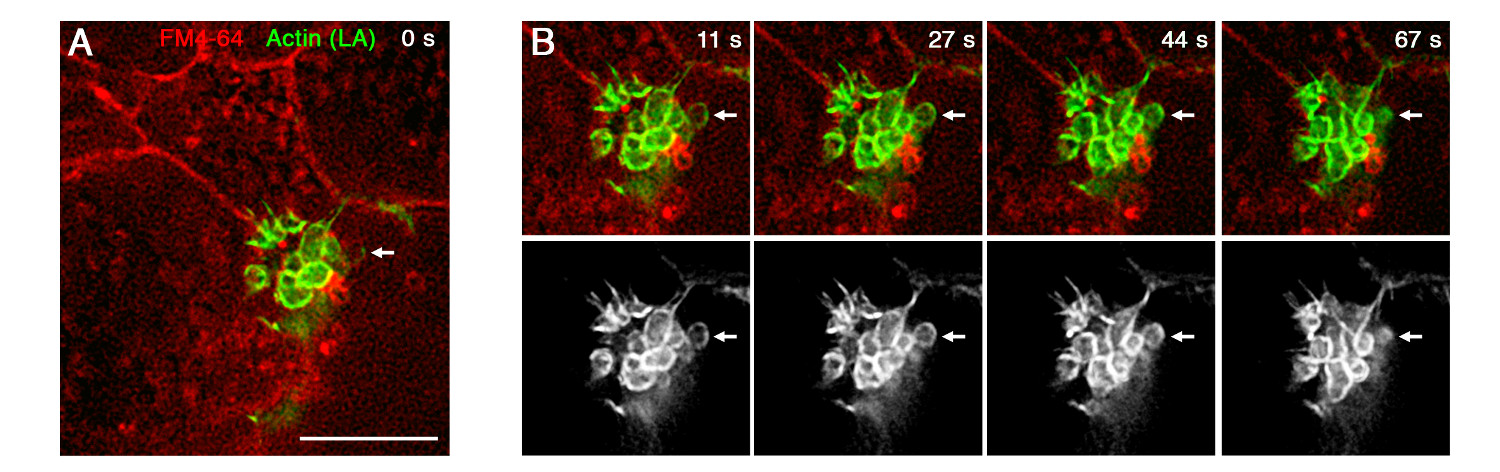
Figure 4. Live imaging of pancreatic secretion. Live stimulated acinar cells, expressing Lifeact-GFP (LA, green, gray) and stained with the lipophilic dye FM4-64 (red). Use of the Lifeact probe allows live visualization of single fusion events of actin-coated vesicles (arrows). Cells were infected O/N with adenovirus-Lifeact-GFP (Ad-Lifeact-GFP), stained and stimulated briefly with 100 pM CCK before the initiation of imaging. Scale bar represents 5 µm. Please click here to view a larger version of this figure.
Discussion
Besides the ex vivo culture, alternative setups for the study of exocrine secretion include intravital microscopy and measurement of fluids from the pancreatic duct. Intravital microscopy was shown to operate well in the mouse salivary glands10. This method can record secretion in real time in the intact animal, but is limited in its resolution and by the means for manipulating the exocrine tissue. Assessing secretion by collecting fluids from the pancreatic duct was achieved in anesthetized rats11. However, since cannulation of the pancreatic duct is extremely difficult, this method is seldom used.
Compared with these setups, the ex vivo culture of pancreatic acini is simple, rapid and allows accessible means for monitoring and manipulating the cells, without interfering with the organization of the intact tissue4,5. Nevertheless, this setup has its shortcomings. Pancreatic acinar cells are delicate in nature and very sensitive to variations in culture conditions. Since the cells are loaded with proteolytic enzymes, they are prone to lysis and cell death. As a consequence, this culture is short lived and usually does not last over 20 hr.
It is always best to identify and troubleshoot problems as they arise. The key methodological step is to isolate intact pancreatic acini without significant signs of cell damage and mortality. Such signs are readily apparent, and include excessive cell debris and acini with basolateral blebs or intracellular vacuoles. In the amylase secretion assay, cell damage will lead to a low stimulated/non-stimulated secretion ratio. In order to prevent such damage, it is recommended to handle the tissue gently, add BSA and STI to the medium, and to enrich it with oxygen. For example, without oxygenation, excessive cell discharge of amylase masks the stimulatory effect of the secretagogue (Figure 3C). Yet, due to the fragility of the exocrine tissue, some tissue damage is almost inevitable – such preparations should be discarded.
For the amylase secretion assay, the supernatant, which contains the secreted amylase but is free from cells, can be kept at 4 °C for 1 – 2 days. This supernatant should then be assayed by a commercial kit. Many available kits rely on a cleavage of an artificial amylase substrate and the subsequent release of a chromophore. Thus, higher amylase concentrations lead to a faster change in the solution’s color. Since the secreted amylase will eventually release all the chromophore from the substrate, it is essential to follow the reaction over time, and use only the data collected during the linear phase.
Live imaging of pancreatic acini can be a frustrating experience for the researcher. Some cell preparations, especially those of infected acini, may not be suitable for live imaging due to compromised morphology of the acini or to insufficient levels of the fluorescent sensors. Even in suitable preparations, it may take time to locate appropriate specimens. Thus imaging may require considerable time. Lifeact-GFP, an F-actin probe, is noteworthy12. This tool allowed us to follow the dynamics of F-actin during pancreatic secretion for up to 16 hr after infection, at a level of resolution that could not be attained in fixed acinar samples. In addition, use of the Lifeact probe enabled tracking of single exocytosis events, as pancreatic zymogen vesicles undergo actin coating shortly before exocytosis3,13.
Collectively, when mastered, the techniques described here offer the researcher means to evaluate pancreatic secretion, and by harness it to identify novel participants in this multi-step process. In addition, in view of the structural and functional similarities between the pancreas and other exocrine organs, these techniques can be applied to other exocrine tissues as well3.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by a grant from the US-Israel BSF to B.S. and E.D.S. B.S. is an incumbent of the Hilda and Cecil Lewis professorial chair in Molecular Genetics.
Materials
| ICR mice | Harlan | Any strain will do | |
| HBSS | Sigma Aldrich | H8264 | Can substitude for KRB |
| BSA | Sigma Aldrich | A7906 | Fraction V |
| Trypsin inhibitor | Sigma Aldrich | T9003 | |
| Collagenase XI | Sigma Aldrich | C0130 | |
| Nylon mesh | BD Falcon | 352360 | 70-100µm |
| Amylase infinity reagent | Thermo | TR25421 | |
| CCK | Sigma Aldrich | C2175 | |
| FM4-64 | Lifetechnologies | F34653 | Diluted to 16µM |
| 20mL scintillation vial | Sigma Aldrich | Z190527 | |
| DeltaVision system | Applied Precision | Consisting of an inverted microscope IX71 equipped with 60x/1.4 or 100x/1.3 objectives (Olympus) | |
| Zeiss 510 or 710 confocal microscope | Zeiss | 60x/1.4 or 100x/1.4 objectives | |
| Ultra Microplate reader | BioTek | ELx808 |
References
- Williams, J. A. Regulation of pancreatic acinar cell function. Current Opinion in Gastroenterology. 22, 498-504 (2006).
- Schnekenburger, J., et al. The role of kinesin, dynein and microtubules in pancreatic secretion. Cellular and Molecular Life Sciences : CMLS. 66, 2525-2537 (2009).
- Geron, E., Schejter, E. D., Shilo, B. Z. Directing exocrine secretory vesicles to the apical membrane by actin cables generated by the formin mDia1. Proceedings of the National Academy of Sciences of the United States of America. 110, 10652-10657 (2013).
- Amsterdam, A., Jamieson, J. D. Structural and functional characterization of isolated pancreatic exocrine cells. Proceedings of the National Academy of Sciences of the United States of America. 69, 3028-3032 (1972).
- Williams, J. A., Korc, M., Dormer, R. L. Action of secretagogues on a new preparation of functionally intact, isolated pancreatic acini. The American journal of physiology. 235, 517-524 (1978).
- Jerdeva, G. V., et al. Actin and non-muscle myosin II facilitate apical exocytosis of tear proteins in rabbit lacrimal acinar epithelial cells. Journal of Cell Science. 118, 4797-4812 (2005).
- Menozzi, D., Jensen, R. T., Gardner, J. D. Dispersed pancreatic acinar cells and pancreatic acini. Methods in Enzymology. , 192-271 (1990).
- Bi, Y., Williams, J. A. A role for Rho and Rac in secretagogue-induced amylase release by pancreatic acini. Am J Physiol Cell Physiol. 289, C22-C32 (2005).
- Palade, G. Intracellular aspects of the process of protein synthesis. Science. 189, 867 (1975).
- Masedunskas, A., et al. Role for the actomyosin complex in regulated exocytosis revealed by intravital microscopy. Proceedings of the National Academy of Sciences of the United States of America. 108, 13552-13557 (2011).
- Petersen, H., Grossman, M. I. Pancreatic exocrine secretion in anesthetized and conscious rats. The American Journal of Physiology. 233, E530-E536 (1977).
- Riedl, J., et al. Lifeact: a versatile marker to visualize F-actin. Nat Methods. 5, 605-607 (2008).
- Porat-Shliom, N., Milberg, O., Masedunskas, A., Weigert, R. Multiple roles for the actin cytoskeleton during regulated exocytosis. Cellular and Molecular Life Sciences : CMLS. 70, 2099-2121 (2013).

