Rearing the Fruit Fly Drosophila melanogaster Under Axenic and Gnotobiotic Conditions
Summary
A method for rearing Drosophila melanogaster under axenic and gnotobiotic conditions is presented. Fly embryos are dechorionated in sodium hypochlorite, transferred aseptically to sterile diet, and reared in closed containers. Inoculating diet and embryos with bacteria leads to gnotobiotic associations, and bacterial presence is confirmed by plating whole-body Drosophila homogenates.
Abstract
The influence of microbes on myriad animal traits and behaviors has been increasingly recognized in recent years. The fruit fly Drosophila melanogaster is a model for understanding microbial interactions with animal hosts, facilitated by approaches to rear large sample sizes of Drosophila under microorganism-free (axenic) conditions, or with defined microbial communities (gnotobiotic). This work outlines a method for collection of Drosophila embryos, hypochlorite dechorionation and sterilization, and transfer to sterile diet. Sterilized embryos are transferred to sterile diet in 50 ml centrifuge tubes, and developing larvae and adults remain free of any exogenous microbes until the vials are opened. Alternatively, flies with a defined microbiota can be reared by inoculating sterile diet and embryos with microbial species of interest. We describe the introduction of 4 bacterial species to establish a representative gnotobiotic microbiota in Drosophila. Finally, we describe approaches for confirming bacterial community composition, including testing if axenic Drosophila remain bacteria-free into adulthood.
Introduction
Most animals are intimately associated with bacteria ('microbiota') from birth to death1. Comparisons of microorganism-free ('axenic') and microorganism-associated ('conventional') animals have shown microbes influence diverse aspects of animal health, including metabolic, nutritional, vascular, hepatic, respiratory, immunological, endocrine, and neurological function2. The fruit fly Drosophila melanogaster is a key model for understanding many of these processes in the presence of microbes3,4 and for studying microbiota influence on animal health5,6. No bacterial species is present in every individual ('core'), but Acetobacter and Lactobacillus species numerically dominate the microbiota of both laboratory-reared and wild-caught D. melanogaster. Other Acetobacteraceae (including Komagataeibacter and Gluconobacter), Firmicutes (such as Enterococcus and Leuconostoc), and Enterobacteriaceae are either frequently present in Drosophila individuals at low abundance, or irregularly present at high abundance7-12.
The microbiota of Drosophila and mammals is inconstant within and across generations14,19. Microbiota inconstancy can lead to phenotypic noise when measuring microbiota-dependent traits. For example, the Acetobacteraceae influence lipid (triglyceride) storage in Drosophila15-18. If Acetobacteraceae are more abundant in flies of one vial than in another19, isogenic flies can have different phenotypes20. A solution for the problem of microbiota inconstancy in mice14 has been in practice since the 1960's, by introducing a defined community of 8 dominant microbial species to mouse pups each new generation (altered Schaedler flora), ensuring that each pup is exposed to the same key members of the mouse microbiota. This practice controls for microbiota composition even when the microbiota is not the primary target of study32, and sets precedent to ensure the presence of key microbes in a variety of experimental conditions.
To define the influence of microbes on Drosophila nutrition, several protocols for deriving axenic fly lines have been developed, including hypochlorite dechorionation of embryos (either derived de novo each generation or maintained generationally by transfer to sterile diets) and antibiotic treatment13. There are benefits to different approaches, such as ease and rapidity for both of antibiotics treatment and serial transfer, versus greater control of confounding variables with de novo dechorionation (e.g., egg density, residual contaminating microbes, off-target antibiotic effects). Regardless of the method of preparation, introduction of specified microbial species to axenic embryos permits culture of Drosophila with defined ('gnotobiotic') communities. Alternatively, mimicking the use of Schaedler flora, this community could be inoculated to conventionally-laid eggs (following steps 6-7 only) to ensure the presence of trait-influencing microbes in each vial and avoid complications of microbiota inconstancy. Here we describe the protocol for raising axenic and gnotobiotic Drosophila by de novo dechorionation of embryos, and for confirming the presence of introduced or contaminating microbial taxa.
Protocol
1. Culture Bacteria (Start ~1 Week before Picking Eggs)
- Prepare modified MRS20 (mMRS) plates and broth tubes (Table 1). Pour 20 ml mMRS agar into each 100 mm Petri plate and allow to cool/dry overnight, or 5 ml mMRS broth into 18 mm test tubes.
- Streak Acetobacter pomorum, A. tropicalis, Lactobacillus brevis, and L. plantarum on mMRS agar plates. Incubate Acetobacter overnight at 30 °C. Incubate Lactobacillus anaerobically by placing the plates in an airtight container and flooding with carbon dioxide before sealing. Incubate at 30 °C overnight.
Note: L. brevis colonies may not be visible until 24-48 hr. Plates can be stored at 4 °C and colonies can be used for up to 3 weeks. - Two to three days before transferring dechorionated eggs to sterile diet (section 5), pick a single colony from the mMRS plate into a test tube containing mMRS broth. Grow Acetobacter with shaking and grow Lactobacillus statically for 24 hr or until turbid, both at 30 °C.
2. Prepare Sterile Diet
- Prepare sterile diet (Table 2) in a 2 L Erlenmeyer flask. Microwave the diet until it has boiled 3 sequential times; mix in between each boil.
- Place the flask on a stir plate to maintain stirring while transferring diet to conical centrifuge tubes. Transfer 7.5 ml of diet to 50 ml centrifuge tubes. Loosely cap the tubes, and place in a covered, autoclavable polypropylene rack.
- Sterilize fly diet using an autoclave at 121 °C and 15 psi for 25 min. Remove racks from autoclave, and immediately shake each rack horizontally to ensure diet does not separate during cooling. Be careful to not shake the racks vertically to prevent transfer of diet to the lid or rims of the tubes. Allow the diet to cool on a shaker for exactly 45 min and again shake the diet horizontally by hand.
Note: Diet can be stored at 4-15 °C for up to a week. - As an alternative to using covered autoclavable racks (steps 2.1-2.3), or to raise flies on diets containing acid preservative, perform the following steps:
- Prepare 1 L liquid diet in a 2 L flask with a stir bar in the flask. Autoclave the diet.
- After autoclaving, add 10 ml preservative and stir continuously on a heated stir plate. When the agar has cooled to 50-60 °C, move the flask to a heated stir plate in a biosafety cabinet and maintain flask temperature by heating at 50-60 °C.
- In the biosafety cabinet, pipet ~7.5 ml individually into conical tubes.
3. Prepare Egg-laying Cages
- Make grape-juice agar plates by microwaving 100 ml water, 10 g brewer's yeast, 10 g glucose, and 1 g of agar. Bring to a boil 3 times in step 1.1 and add 10 g of frozen grape juice concentrate to increase visibility of eggs on the agar plate.
- When agar has cooled to 55 °C, pour 20 ml into 100 mm Petri dishes and allow to solidify.
- Cover the surface of the agar plates with a yeast paste by mixing 1 g brewer's yeast with 15 g water. Pour yeast paste onto the agar plate making sure the surface is covered, then pour off the excess, leaving a thin yeast residue behind. If multiple plates are used, the paste can be sequentially poured to multiple plates.
- Transfer agar plates into the bottom of a cage.
Note: A 32 oz deli container is a good substitute for acrylic fly cages.- Make lids for 32 oz deli containers by cutting a hole in the top and gluing a breathable mesh over the hole with non-toxic glue. If the glue is water soluble prevent water exposure to the lid.
- Transfer 200-300 flies into the container and cover with lid. Cover the mesh-protected hole with an empty Petri dish lid to prevent evaporation from the agar surface and add a moist tissue paper inside the lid if desired. Incubate flies at 25 °C overnight for 16-20 hr.
4. Collect Eggs
- Prepare a sieve for egg collection by placing nylon mesh into a plastic bushing.
- To retrieve the plate with eggs on it, remove flies from the cage by transferring to an empty container. If same flies will be used the next day, transfer immediately to a new cage containing a freshly yeasted grape-juice agar plate. Flies can be used for 3-5 sequential days
- Remove dead flies from the agar with a clean paintbrush, being careful to not break up the agar.
- Collect eggs by rinsing the agar plate with distilled water, gently brushing eggs from the agar surface, and pouring the slurry over the mesh. Repeat 3-4 times until all or most of the eggs have been removed from the agar plate.
5. Dechorionate Eggs and Transfer to Sterile Diet
- Prepare the biosafety cabinet by spraying the inside (including sides) with 70% ethanol. Wipe the bottom with a lab tissue, and sterilize the hood with UV light for ~15 min. Sterilize all non-biological supplies (specimen cups, paintbrush, forceps, waste container, 400 ml sterilized water, and 100 ml of 0.6% sodium hypochlorite) by spraying with ethanol and immediately placing in the biosafety cabinet. Sterilize with UV light for 15 min.
- Start the first of 2 sodium hypochlorite washes by placing the bushing with the eggs into a 120 ml specimen cup or other sterile container. Slowly pour ~90 ml of 0.6% sodium hypochlorite solution into the bushing until just below the rim.
- Rinse eggs for 2.5 min. Periodically re-suspend the eggs by using forceps to move the bushing up and down in the hypochlorite solution.
- Transfer the bushing directly into a second specimen cup, pre-filled with 90 ml bleach, inside the biosafety cabinet.
- Repeat step 5.3 inside the biosafety cabinet. At the end of the second bleach treatment, the eggs should begin to adhere to the sides of the bushing.
- Carry out steps 5.7-5.8 in the biosafety cabinet.
- Discard the bleach and wash the bushing with sterile water 3 times. Re-suspend the eggs several times during each washing by moving the bushing with forceps. By the end of the third washing most eggs should be attached to the side of the bushing.
- Using a paintbrush sterilized in ethanol, transfer eggs from the side of the bushing to the sterile diet. Transfer eggs individually or in small batches. Aim for 30-50 eggs per vial. Leave the caps loose to allow oxygen to enter the tube. If vials are to remain axenic, transfer to an insect incubator; otherwise, add bacteria as below.
6. Make Gnotobiotic Flies Using 4 Bacterial Species
- Prepare Bacteria
- Prepare a sterilized biosafety cabinet with necessary supplies (pipettes, pipette tip boxes, sterilized centrifuge tubes, MRS broth, and test tube racks) as in step 4.1. Wipe the outside of test tubes with an ethanol-soaked laboratory wipe before placing in biosafety cabinet.
- Pellet the bacteria by first transferring 500 µl of overnight growth to a sterile microfuge tube. If bacterial density is low, add up to 1.5 ml to each tube or sequentially remove supernatant and add extra culture to the same tube. Remove samples from the biosafety cabinet and centrifuge for 10 min at 10,000 x g. Use filter tips to avoid contamination between samples.
- Determine the density of each culture by measuring OD600. If using a multi-well plate reader, transfer 200 µl of each culture to a 96-well plate in 1-, 2-, and 4- fold dilutions.
- Determine the amount of mMRS in which to dilute each cell pellet (5.2.2) using a plate reading spectrophotometer and the following equations. Plan to add enough broth to inoculate 50 μl to each fly vial.
- Collect OD600 readings for a 1:1, 1:2, and 1:4 dilution of each bacterial culture on a plate-reading spectrophotometer. Select the dilution for each bacterial strain that produces an OD600 value between 0.1 and 0.2 and use this value and its corresponding dilution factor as 'O' and 'D' in the formulas given in 6.1.4.2 or 6.1.4.3.
- If using the 4 species described here, normalize cells to equivalent colony forming unit (CFU)/ml densities (OD600 to CFU conversion determined previously20) using this equation:
E = ((O-B) x V x D)/C
where E = volume to resuspend pellet in (μl), O = OD600 bacteria, B = OD600 blank media, D = fold-dilution, V = µl bacterial culture prior to centrifugation, C = OD600 of predetermined constant. See Supplemental Code File for examples of calculations using these equations. For spectrophotometers that automatically blank, use "O" in place of "O-B".
Note: The predetermined constants (units OD600, normalized to 107 CFU ml-1, constants derived in20) are as follows: A. tropicalis (0.053), A. pomorum (0.038), L. brevis (0.077), L. plantarum (0.056). - If using other bacterial species (no CFU/OD600 constant is available), normalize density to OD600 = 0.1 using this equation:
E = ((O-B) x V x D)/0.1 OD600
Note: Units are the same as in step 6.1.4.2. See Supplemental Code File for examples of calculations using these equations.
- In the biosafety cabinet, remove supernatant with a pipet tip and resuspend the pellet in fresh mMRS or PBS as calculated in step 6.1.4.2.
- Inoculate Bacteria
- Transfer 50 µl of the bacteria to the conical tubes with sterile diet and dechorionated eggs in biosafety cabinet. Add bacteria after egg transfer to prevent contamination between vials.
- Place inoculated tubes in an incubator at 25° C.
7. Measure CFU Load/Test for Sterility
- To measure the CFU load in whole body fly homogenates, transfer 5 flies (5-7 days post eclosion) to a 1.7 ml microfuge tube containing 125 µl of ceramic beads and 125 µl of mMRS broth. Homogenize flies using a tissue homogenizer for 30 sec at 4.0 M/sec.
- Alternatively, omit beads and hand homogenize in microcentrifuge tubes with plastic pestles for 1 min.
- If quantifying the gut microbiota, surface sterilize the flies to remove exogenous microbes22. Transfer flies to a microcentrifuge tube containing 100 µl 70% ethanol for 1 min, aspirate ethanol, and transfer to a new microcentrifuge tube for homogenizations. If the DNA content of the gut will be measured, rinse for 1 min with 0.6% sodium hypochlorite before the ethanol wash.
- Dilute the homogenate with 875 μl mMRS, vortex for 5 sec, and pipet 120 µl of homogenate into the first well of a microtiter plate.
- Perform two sequential 1:8 dilutions using 10 µl homogenate and 70 µl MRS in the next two wells.
- Remove 10 µl from the first well and add it to the second well containing 70 µl MRS. Mix the contents of the second well thoroughly, transfer 10 µl from the second well to the third well containing 70 µl MRS, and mix thoroughly. This leads to 3 total concentrations of the original 1,000 μl homogenate: undiluted, 1:8, and 1:64.
- Transfer 10 µl of each dilution to a mMRS plate (using a multi-channel pipet if desired). Slightly incline the dish to spread the dilution several millimeters down the agar surface and allow liquid to dry before moving the plate. The liquid dries on the plate quickly if the plates are 2 days old, reducing mixing of two neighboring droplets.
- Incubate at 30 °C for 1-2 days. Remove plates from the incubator once distinct, individual colonies are visible, and count from a dilution with 10-100 isolated colonies.
- Calculate CFU per fly using the equation E = C x D/P x V/F, where E =CFU per fly, C = number of colonies counted, D = dilution, P = µl plated, V = volume of fly homogenate, and F = number of flies homogenized.
Representative Results
Successful rearing of axenic flies is confirmed by isolation of no CFUs from whole-body homogenizations of D. melanogaster adults (Figure 1). Alternatively, if the plated homogenate yields colonies, the vials are contaminated and should be discarded. For gnotobiotic flies, each of the four bacterial isolates were isolated from pools of 5 adult males, demonstrating differences in total viable CFUs associated with adult flies (Figure 1). Each bacterial species has a distinct morphology and can be distinguished visually (Figure 2). If one or more colony types are not detected there may have been errors in preparing the bacterial inoculum (e.g., washing, normalizing, mixing) or the corresponding species may not be compatible with culture conditions (e.g., Drosophila density23 or genotype24). To rule out technical errors we recommend plating a portion of the bacterial mixture immediately after inoculating fly vials (step 6.2).
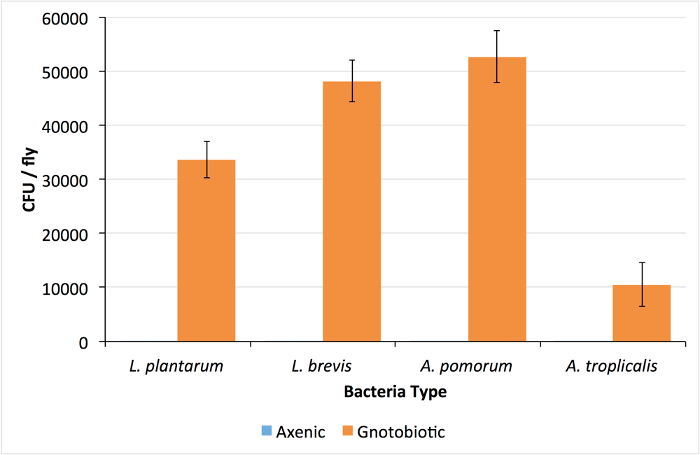
Figure 1: Colony Forming Units Found in Axenic and Gnotobiotic Drosophila. The number of colony forming units (CFU) per fly in whole-body homogenates of 4-species gnotobiotic and axenic D. melanogaster. Lack of colonies in the axenic homogenates confirms D. melanogaster sterility. Values are presented as mean ±SEM of 24 replicate values. Please click here to view a larger version of this figure.
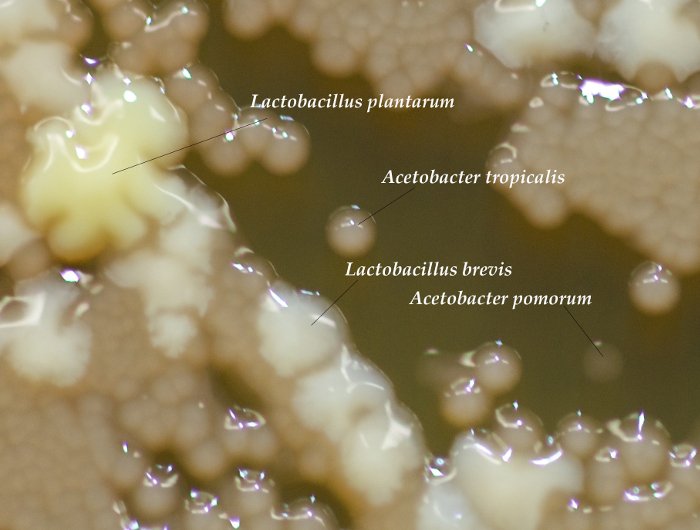
Figure 2: Differentiating Bacterial Colonies. CFUs from a sample homogenization showing different morphologies for each of the 4-species in the gnotobiotic D. melanogaster. Acetobacter colonies are a clear, light brown color and come in varying sizes depending on the species. ~24-36 hr after plating, A. tropicalis colonies are opaque, whereas A. pomorum is transparent, though over time the difference becomes less pronounced. L. brevis colonies are small and white and L. plantarum colonies are large and yellow. If necessary, growth from the homogenate can be compared to the original bacteria plates to help distinguish each type of colony20. Please click here to view a larger version of this figure.
| mMRS Recipe | ||
| Amount in g | Notes | |
| Distilled Water | 1,000 | |
| Universal Peptone | 12.5 | |
| Yeast Extract | 7.5 | |
| Glucose | 20 | |
| Dipotassium Phosphate | 2 | |
| Ammonium Citrate | 2 | |
| Sodium Acetate | 5 | |
| Magnesium Sulfate | 0.1 | |
| Manganous Sulfate | 0.05 | |
| Agar | 12 | Do not add to broth |
Table 1: mMRS recipe.
| Yeast-glucose diet recipe | |
| Amount in g | |
| Distilled water | 500 |
| Brewer's Yeast | 50 |
| Glucose | 50 |
| Agar | 6 |
Table 2: Yeast-glucose diet recipe.
Supplemental Code File: Sample Calculations. Please click here to download this file.
Discussion
The method described here is one of several approaches for embryo dechorionation8,11,18,25,26,27, together with alternative methods of rearing axenic flies, including serial transfer of axenic adults18,27 or antibiotic treatment13,18. Other dechorionation methods include ethanol washes and reduce11,25,26 or extend8 hypochlorite treatment. Different wash steps may aid rearing different fly genotypes: in a previous study most of ~100 Drosophila genotypes were axenic when reared as outlined here (without ethanol washes), but some lines were contaminated and discarded24. Perhaps some contaminated lines would have been axenic if reared with ethanol washes or longer hypochlorite treatment. If the method outlined here does not lead to isolation of axenic flies for particular host genotypes we recommend ethanol rinses or longer hypochlorite treatment to remedy the problem.
When serial transfers of Drosophila are used, a parental axenic generation is made axenic by egg dechorionation as described here, and subsequent generations are maintained by aseptic transfer in a biosafety cabinet, with or without antibiotics18,27. Serial transfer is faster than deriving axenic flies anew each generation, and transferred flies can remain axenic for multiple generations. One complication of serial transfers is maintaining matched densities of conventional/gnotobiotic and axenic flies since axenic flies tend to lay fewer eggs over a comparable time interval (data not shown). Since Drosophila density influences multiple traits, including bacterial composition in fly diet23,28,29, transferring eggs anew each generation may be a superior approach for fly density-sensitive traits. De novo dechorionation also avoids the possibility of bacterial contamination during transfers. Thus, while serial transfers can save time, dechorionation allows more control of complicating variables.
Antibiotics can also be used to create axenic flies, although in contrast to dechorionation, antibiotic treatment is usually insufficient to completely remove colonizing microbes13. Additionally, antibiotics may influence the host directly. For example, raising flies on a diet with antibiotics decreases their fecundity and protein content, but these effects were not observed in flies raised from dechorionated embryos13. We note that antibiotics are necessary to eliminate endosymbiotic bacteria that are transmitted vertically and are not affected by surface sterilization, such as Wolbachia33.
Several steps are critical to the success of preparing dechorionated axenic or gnotobiotic flies. First, it is crucial to shake the fly diet as in steps 2.3. If the racks are not shaken by hand for about 15 sec each before and after an exact 45 min interval on the shaker, the yeast and agar will settle, reducing fly access to yeast and making the agar surface too soft for fly culture. Second, the thickness of yeast paste and the agar concentration of grape juice plates influences egg removal (step 3.3). A thin layer produced by a 1:15 yeast:water dilution will prevent eggs from embedding in the agar when rinsing and removing from the food plates (step 4.4). Third, if grape juice plates are too soft and break up during egg collection, firmness of plates can be increased by adding less grape juice concentrate to the next batch of plates. Fourth, egg yields are higher if flies have 24 hr to acclimatize to the cage environment: this can be addressed by placing flies in the cage ~40 hours in advance of collection, including transfer to a new collection cage with a fresh agar plate <20 hr before the desired collection date. Fifth, if the eggs do not adhere to the side of the bushing during dechorionation (step 5.7), the rinsing period should be extended by 15-30 sec (Caution: extending the rinse time too long may kill the eggs). Also, egg loss into the liquid rinses can be prevented by checking the mesh-bushing seal for complete closure and verify that eggs do not spill outside of the sieve during re-suspension. If the bushing is well sealed, spilled eggs can be recovered by pouring the wash through the sieve as the wash is discarded. Sixth, after flies have hatched, a presumptive test to determine if the flies are axenic is to examine the color of the diet. If the flies are axenic, the top layer of diet will be a dark-brown coffee color and no air-bubbles will be present throughout the diet. Air bubbles or tan color of the top diet layer indicate bacterial presence. Bacterial presence should still be confirmed by homogenization. Alternatively, PCR amplification of the 16S rRNA gene can also be performed to detect unculturable microbes (e.g., strict anaerobes)30. Finally, after homogenization, ceramic beads can reused by rocking in a solution of 2% hypochlorite + 0.05 M potassium hydroxide for 30 min, rinsing generously (10 times or more) in H2O, washing once with 100% ethanol (to facilitate drying), and drying at 65 °C. If all steps are followed carefully, axenic flies should be isolated every time the protocol is performed.
This work outlines a method for re-associating sterile Drosophila embryos with a 4-species microbiota that is representative of the bacterial communities of laboratory flies raised on a yeast-glucose diet. Previous work has used a 5-species community including the 4-species here and Lactobacillus fructivorans, which has been numerically abundant in several fly surveys9,19. We recommend omitting L. fructivorans for several reasons, including that phenotypes in D. melanogaster monoassociated with L. fructivorans are largely congruent with axenic phenotypes 31 and L. fructivorans is more fastidious than other fly isolates. If L. fructivorans is included, it should be cultured as described for L. plantarum and L. brevis: static liquid culture; and in an airtight container flooded with CO2 for solid culture (the latter step reduces ambient oxygen levels to support Lactobacillus growth).
The approaches outlined here can be readily varied to raise Drosophila under diverse gnotobiotic conditions. For example, monoassociated flies can be reared by inoculating sterile Drosophila embryos with one microbial species at a time15,25,31. Multi-species associations are formed by inoculating multiple species in equivalent ratios20,24. Although we provided CFU/OD600 constants for each of the 4-species to facilitate normalization, it may not be necessary to derive a constant for most species mixtures. It was previously shown that the abundance of microbes in 5-7 d.p.e. adults was not significantly different in 2-species associations when the starting density of bacteria was varied over three orders of magnitude 20. Also, the Drosophila gut is highly permissive, and many species that are readily cultured on fly diet can associate with Drosophila at high densities in monoassociation (e.g., E. coli, B. subtilis25) enabling genetic dissection of microbial influence by microbes with extensive genetic and genomic resources. Finally, studies of Drosophila phenotypes that are influenced by the microbiota could adapt the approach of Altered Schaedler Flora in mice by inoculating naturally laid conventional eggs with a defined microbial community to ensure each vial has access to a specific set of microbes.
Disclosures
The authors have nothing to disclose.
Acknowledgements
Some details of this protocol were optimized with the assistance of Dr. Adam Dobson, who also provided helpful comments on the manuscript. This work was supported by the Foundation for the National Institutes of Health (FNIH) grant number R01GM095372 (JMC, A(CN)W, AJD, and AED). FNIH grant number 1F32GM099374-01 (PDN), and Brigham Young University startup funds (JMC, MLK, MV). Publication costs were supported by the Brigham Young University College of Life Sciences and Department of Plant and Wildlife Sciences.
Materials
| Brewer's Yeast | MP Biomedicals, LLC. | 903312 | http://www.mpbio.com/product.php?pid=02903312 |
| Glucose | Sigma Aldrich | 158968-3KG | http://www.sigmaaldrich.com/catalog/product/aldrich/158968?lang=en®ion=US |
| Agar | Fisher–Lab Scientific | fly802010 | https://www.fishersci.com/shop/products/drosophila-agar-8-100mesh-10kg/nc9349177 |
| Welch's 100% Grape Juice Concentrate | Walmart or other grocery store | 9116196 | http://www.walmart.com/ip/Welch-s-Frozen-100-Grape-Juice-Concentrate-11.5-oz/10804406 |
| Cage: 32 oz. Translucent Round Deli Container | Webstaurant Store | 999L5032Y | http://www.webstaurantstore.com/newspring-delitainer-sd5032y-32-oz-translucent-round-deli-container-24-pack/999L5032Y.html |
| Translucent Round Deli Container Lid | Webstaurant Store | 999YNL500 | http://www.webstaurantstore.com/newspring-delitainer-ynl500-translucent-round-deli-container-lid-60-pack/999YNL500.html |
| Stock Bottles | Genesee Scientific | 32-130 | https://geneseesci.com/shop-online/product-details/?product=32-130 |
| Droso-Plugs | Genesee Scientific | 49-101 | https://geneseesci.com/shop-online/product-details/?product=49-101 |
| Nylon Mesh | Genesee Scientific | 57-102 | https://geneseesci.com/shop-online/product-details/715/?product=57-102 |
| Plastic Bushing | Home Depot | 100343125 | http://www.homedepot.com/p/Halex-2-1-2-in-Rigid-Insulated-Plastic-Bushing-75225/100343125 |
| Specimen Cup | MedSupply Partners | K01-207067 | http://www.medsupplypartners.com/covidien-specimen-containers.html |
| Repeater M4 | Eppendorf | 4982000322 | https://online-shop.eppendorf.us/US-en/Manual-Liquid-Handling-44563/Dispensers–Burettes-44566/Repeater-M4-PF-44619.html |
| 50 ml Centrifuge Tubes | Laboratory Product Sales | TR2003 | https://www.lpsinc.com/Catalog4.asp?catalog_nu=TR2003 |
| Food Boxes | USA Scientific | 2316-5001 | http://www.usascientific.com/search.aspx?find=2316-5001 |
| Lysing Matrix D Bulk | MP Biomedicals, LLC. | 116540434 | http://www.mpbio.com/search.php?q=6540-434&s=Search |
| Filter Pipette Tips, 300μl | USA Scientific | 1120-9810 | http://www.usascientific.com/search.aspx?find=1120-9810 |
| Petri Dishes | Laboratory Product Sales | M089303 | https://www.lpsinc.com/Catalog4.asp?catalog_nu=M089303 |
| Ethanol | Decon Laboratories, INC. | 2701 | http://www.deconlabs.com/products.php?ID=88 |
| Paintbrush | Walmart | 5133 | http://www.walmart.com/ip/Chenille-Kraft-5133-Acrylic-Handled-Brush-Set-Assorted-Sizes-colors-8-Brushes-set/41446005 |
| Forceps | Fisher | 08-882 | https://www.fishersci.com/shop/products/fisherbrand-medium-pointed-forceps-3/p-128693 |
| Household Bleach (6-8% Hypochlorite) | Walmart | 550646751 | http://www.walmart.com/ip/Clorox-Concentrated-Regular-Bleach-121-fl-oz/21618295 |
| Universal Peptone | Genesee Scientific | 20-260 | https://geneseesci.com/shop-online/product-details/?product=20-260 |
| Yeast Extract | Fisher Scientific | BP1422-500 | https://www.fishersci.com/shop/products/fisher-bioreagents-microbiology-media-additives-yeast-extract-3/bp1422500?matchedCatNo=BP1422500 |
| Dipotassium Phosphate | Sigma Aldrich | P3786-1KG | http://www.sigmaaldrich.com/catalog/search?term=P3786-1KG&interface=All&N=0&mode=match%20partialmax&lang=en®ion=US&focus=product |
| Ammonium Citrate | Sigma Aldrich | 25102-500g | http://www.sigmaaldrich.com/catalog/search?term=25102-500g&interface=All&N=0&mode=match%20partialmax&lang=en®ion=US&focus=product |
| Sodium Acetate | VWR | 97061-994 | https://us.vwr.com/store/catalog/product.jsp?catalog_number=97061-994 |
| Magnesium Sulfate | Fisher Scientific | M63-500 | https://www.fishersci.com/shop/products/magnesium-sulfate-heptahydrate-crystalline-certified-acs-fisher-chemical-3/m63500?matchedCatNo=M63500 |
| Manganese Sulfate | Sigma Aldrich | 10034-96-5 | http://www.sigmaaldrich.com/catalog/search?term=10034-96-5&interface=CAS%20No.&N=0&mode=match%20partialmax&lang=en®ion=US&focus=product |
| MRS Powder | Sigma Aldrich | 69966-500G | http://www.sigmaaldrich.com/catalog/product/sial/69966?lang=en®ion=US |
| 96 Well Plate Reader | BioTek (Epoch) | NA | http://www.biotek.com/products/microplate_detection/epoch_microplate_spectrophotometer.html |
| 1.7 ml Centrifuge Tubes | USA Scientific | 1615-5500 | http://www.usascientific.com/search.aspx?find=1615-5500 |
| Filter Pipette Tips, 1000μl | USA Scientific | 1122-1830 | http://www.usascientific.com/search.aspx?find=1122-1830 |
| 96 Well Plates | Greiner Bio-One | 655101 | https://shop.gbo.com/en/usa/articles/catalogue/article/0110_0040_0120_0010/13243/ |
| Ceramic Beads | MP Biomedicals, LLC. | 6540-434 | http://www.mpbio.com/product.php?pid=116540434 |
| Tissue Homogenizer | MP Biomedicals, LLC. | 116004500 | http://www.mpbio.com/product.php?pid=116004500 |
| Class 1 BioSafety Cabinet | Thermo Scientific | Model 1395 | http://www.thermoscientific.com/en/product/1300-series-class-ii-type-a2-biological-safety-cabinet-packages.html |
References
- McFall-Ngai, M. J. Giving microbes their due–animal life in a microbially dominant world. J Exp Biol. 218, 1968-1973 (2015).
- Smith, K., McCoy, K. D., Macpherson, A. J. Use of axenic animals in studying the adaptation of mammals to their commensal intestinal microbiota. Semin Immunol. 19 (2), 59-69 (2007).
- Rieder, L. E., Larschan, E. N. Wisdom from the fly. Trends Genet. 30 (11), 479-481 (2014).
- Arias, A. M. Drosophila melanogaster and the development of biology in the 20th century. Methods Mol Biol. 420, 1-25 (2008).
- Lee, W. J., Brey, P. T. How microbiomes influence metazoan development: insights from history and Drosophila modeling of gut-microbe interactions. Annu Rev Cell Dev Biol. 29, 571-592 (2013).
- Erkosar, B., Leulier, F. Transient adult microbiota, gut homeostasis and longevity: novel insights from the Drosophila model. FEBS Lett. 588 (22), 4250-4257 (2014).
- Chandler, J. A., Lang, J. M., Bhatnagar, S., Eisen, J. A., Kopp, A. Bacterial communities of diverse Drosophila species: ecological context of a host-microbe model system. PLoS Genet. 7 (9), e1002272 (2011).
- Broderick, N. A., Buchon, N., Lemaitre, B. Microbiota-induced changes in drosophila melanogaster host gene expression and gut morphology. MBio. 5 (3), 01117 (2014).
- Wong, C. N., Ng, P., Douglas, A. E. Low-diversity bacterial community in the gut of the fruitfly Drosophila melanogaster. Environ Microbiol. 13 (7), 1889-1900 (2011).
- Staubach, F., Baines, J. F., Kunzel, S., Bik, E. M., Petrov, D. A. Host species and environmental effects on bacterial communities associated with Drosophila in the laboratory and in the natural environment. PLoS One. 8 (8), e70749 (2013).
- Brummel, T., Ching, A., Seroude, L., Simon, A. F., Benzer, S. Drosophila lifespan enhancement by exogenous bacteria. Proc Natl Acad Sci U S A. 101 (35), 12974-12979 (2004).
- Cox, C. R., Gilmore, M. S. Native microbial colonization of Drosophila melanogaster and its use as a model of Enterococcus faecalis pathogenesis. Infect Immun. 75 (4), 1565-1576 (2007).
- Ridley, E. V., Wong, A. C., Douglas, A. E. Microbe-dependent and nonspecific effects of procedures to eliminate the resident microbiota from Drosophila melanogaster. Appl Environ Microbiol. 79 (10), 3209-3214 (2013).
- Rogers, G. B., et al. Functional divergence in gastrointestinal microbiota in physically-separated genetically identical mice. Sci Rep. 4, 5437 (2014).
- Chaston, J. M., Newell, P. D., Douglas, A. E. Metagenome-wide association of microbial determinants of host phenotype in Drosophila melanogaster. MBio. 5 (5), 01631-01714 (2014).
- Huang, J. H., Douglas, A. E. Consumption of dietary sugar by gut bacteria determines Drosophila lipid content. Biology Letters. , (2015).
- Shin, S. C., et al. Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling. Science. 334 (6056), 670-674 (2011).
- Storelli, G., et al. Lactobacillus plantarum promotes Drosophila systemic growth by modulating hormonal signals through TOR-dependent nutrient sensing. Cell Metab. 14 (3), 403-414 (2011).
- Wong, A. C., Chaston, J. M., Douglas, A. E. The inconstant gut microbiota of Drosophila species revealed by 16S rRNA gene analysis. ISME J. 7 (10), 1922-1932 (2013).
- Newell, P. D., Douglas, A. E. Interspecies interactions determine the impact of the gut microbiota on nutrient allocation in Drosophila melanogaster. Appl Environ Microbiol. 80 (2), 788-796 (2014).
- Broderick, N. A., Lemaitre, B. Gut-associated microbes of Drosophila melanogaster. Gut Microbes. 3 (4), 307-321 (2012).
- Ren, C., Webster, P., Finkel, S. E., Tower, J. Increased internal and external bacterial load during Drosophila aging without life-span trade-off. Cell Metab. 6 (2), 144-152 (2007).
- Wong, A. C., et al. The Host as the Driver of the Microbiota in the Gut and External Environment of Drosophila melanogaster. Appl Environ Microbiol. 81 (18), 6232-6240 (2015).
- Dobson, A. J., et al. Host genetic determinants of microbiota-dependent nutrition revealed by genome-wide analysis of Drosophila melanogaster. Nat Commun. 6, 6312 (2015).
- Bakula, M. The persistence of a microbial flora during postembryogenesis of Drosophila melanogaster. J Invertebr Pathol. 14 (3), 365-374 (1969).
- Ryu, J. H., et al. Innate immune homeostasis by the homeobox gene caudal and commensal-gut mutualism in Drosophila. Science. 319 (5864), 777-782 (2008).
- Blum, J. E., Fischer, C. N., Miles, J., Handelsman, J. Frequent replenishment sustains the beneficial microbiome of Drosophila melanogaster. MBio. 4 (6), 00860 (2013).
- Bitner-Mathe, B. C., Klaczko, L. B. Plasticity of Drosophila melanogaster wing morphology: effects of sex, temperature and density. Genetica. 105 (2), 203-210 (1999).
- Edward, D. A., Chapman, T. Sex-specific effects of developmental environment on reproductive trait expression in Drosophila melanogaster. Ecol Evol. 2 (7), 1362-1370 (2012).
- Ridley, E. V., Wong, A. C., Westmiller, S., Douglas, A. E. Impact of the resident microbiota on the nutritional phenotype of Drosophila melanogaster. PLoS One. 7 (5), e36765 (2012).
- Newell, P. D., et al. In vivo function and comparative genomic analyses of the Drosophila gut microbiota identify candidate symbiosis factors. Front Microbiol. 5, 576 (2014).
- Dewhirst, F. E., et al. Phylogeny of the defined murine microbiota: altered Schaedler flora. Appl Environ Microbiol. 65 (8), 3287-3292 (1999).
- Min, K. T., Benzer, S. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc Natl Acad Sci U S A. 94 (20), 10792-10796 (1997).

