Biaxial Basal Tone and Passive Testing of the Murine Reproductive System Using a Pressure Myograph
Summary
This protocol utilized a commercially available pressure myograph system to perform pressure myograph testing on the murine vagina and cervix. Utilizing media with and without calcium, the contributions of the smooth muscle cells (SMC) basal tone and passive extracellular matrix (ECM) were isolated for the organs under estimated physiological conditions.
Abstract
The female reproductive organs, specifically the vagina and cervix, are composed of various cellular components and a unique extracellular matrix (ECM). Smooth muscle cells exhibit a contractile function within the vaginal and cervical walls. Depending on the biochemical environment and the mechanical distension of the organ walls, the smooth muscle cells alter the contractile conditions. The contribution of the smooth muscle cells under baseline physiological conditions is classified as a basal tone. More specifically, a basal tone is the baseline partial constriction of smooth muscle cells in the absence of hormonal and neural stimulation. Furthermore, the ECM provides structural support for the organ walls and functions as a reservoir for biochemical cues. These biochemical cues are vital to various organ functions, such as inciting growth and maintaining homeostasis. The ECM of each organ is composed primarily of collagen fibers (mostly collagen types I, III, and V), elastic fibers, and glycosaminoglycans/proteoglycans. The composition and organization of the ECM dictate the mechanical properties of each organ. A change in ECM composition may lead to the development of reproductive pathologies, such as pelvic organ prolapse or premature cervical remodeling. Furthermore, changes in ECM microstructure and stiffness may alter smooth muscle cell activity and phenotype, thus resulting in the loss of the contractile force.
In this work, the reported protocols are used to assess the basal tone and passive mechanical properties of the nonpregnant murine vagina and cervix at 4-6 months of age in estrus. The organs were mounted in a commercially available pressure myograph and both pressure-diameter and force-length tests were performed. Sample data and data analysis techniques for the mechanical characterization of the reproductive organs are included. Such information may be useful for constructing mathematical models and rationally designing therapeutic interventions for women’s health pathologies.
Introduction
The vaginal wall is composed of four layers, the epithelium, lamina propria, muscularis, and adventitia. The epithelium is primarily composed of epithelial cells. The lamina propria has a large amount of elastic and fibrillar collagen fibers. The muscularis is also composed of elastin and collagen fibers but has an increased amount of smooth muscle cells. The adventitia is comprised of elastin, collagen, and fibroblasts, albeit in reduced concentrations compared to the previous layers. The smooth muscle cells are of interest to biomechanically motivated research groups as they play a role in the contractile nature of the organs. As such, quantifying the smooth muscle cell area fraction and organization is key to understanding the mechanical function. Previous investigations suggest that the smooth muscle content within the vaginal wall is primarily organized in the circumferential and longitudinal axis. Histological analysis suggests that the smooth muscle area fraction is approximately 35% for both the proximal and distal sections of the wall1.
The cervix is a highly collagenous structure, that until recently, was thought to have minimal smooth muscle cell content2,3. Recent studies, however, have suggested that smooth muscle cells may have a greater abundance and role in the cervix4,5. The cervix exhibits a gradient of smooth muscle cells. The internal os contains 50-60% smooth muscle cells where the external os only contains 10%. Mouse studies, however, report the cervix to be composed of 10-15% smooth muscle cells and 85-90% fibrous connective tissue with no mention of regional differences6,7,8. Given that the mouse model differs from the frequently reported human model, further investigations concerning the mouse cervix are needed.
The purpose of this protocol was to elucidate the mechanical properties of the murine vagina and cervix. This was accomplished by using a pressure myograph device that enables assessment of mechanical properties in the circumferential and axial directions simultaneously while maintaining native cell-matrix interactions and organ geometry. The organs were mounted on two custom cannulas and secured with silk 6-0 sutures. Pressure-diameter tests were performed around the estimated physiological axial stretch to determine the compliance and tangent moduli9. Force-length tests were conducted to confirm the estimated axial stretch and to ensure that mechanical properties were quantified in the physiological range. The experimental protocol was performed on the nonpregnant murine vagina and cervix at 4-6 months of age in estrus.
The protocol is divided into two main mechanical testing sections: basal tone and passive testing. A basal tone is defined as the baseline partial constriction of smooth muscle cells, even in the absences of external local, hormonal, and neural stimulation10. This baseline contractile nature of the vagina and cervix yields characteristic mechanical behaviors which are then measured by the pressure myograph system. The passive properties are assessed by removing the intercellular calcium that maintains the baseline state of contraction, resulting in relaxation of the smooth muscle cells. In the passive state, collagen and elastin fibers provide the dominant contributions for the mechanical characteristics of the organs.
The murine model is used extensively to study pathologies in women’s reproductive health. The mouse offers several advantages for quantifying the evolving relationships between ECM and mechanical properties within the reproductive system11,12,13,14. These advantages include short and well-characterized estrous cycles, relatively low cost, ease of handling, and a relatively short gestational time15. Additionally, the genome of laboratory mice is well-mapped and genetically-modified mice are valuable tools to test mechanistic hypotheses16,17,18.
Commercially available pressure myograph systems are used extensively to quantify the mechanical responses of various tissues and organs. Some notable structures analyzed on the pressure myograph system include elastic arteries19,20,21,22, veins and tissue engineered vascular grafts23,24, the esophagus25, and the large intestines26. The pressure myograph technology permits simultaneous assessment of properties in the axial and circumferential directions while maintaining the native cell-ECM interactions and in vivo geometry. Despite the extensive use of myograph systems in soft tissue and organ mechanics, a protocol utilizing the pressure myograph technology had not previously been developed for the vagina and cervix. Prior investigations into the mechanical properties of the vagina and cervix were assessed uniaxially27,28. These organs, however, experience multiaxial loading within the body29,30, thus quantifying their biaxial mechanical response is important.
Moreover, recent work suggests smooth muscle cells may play a potential role in soft tissue pathologies5,28,31,32. This provides another attraction of utilizing the pressure myograph technology, as it preserves the native cell-matrix interactions, thus permitting delineation of the contribution that smooth muscle cells play in physiological and pathophysiological conditions. Herein, we propose a protocol to quantify the multiaxial mechanical properties of the vagina and cervix under both basal tone and passive conditions.
Protocol
Nulliparous 4-6 months female C57BL6J mice (29.4 ± 6.8 grams) at estrus were used for this study. All procedures were approved by the Institute Animal Care and Use Committee at Tulane University. After delivery, the mice acclimated for one week before euthanasia and were housed under standard conditions (12-hour light/dark cycles).
1. Mouse sacrifice at estrus
- Determine the estrous cycle: The estrous cycle was monitored by visual assessment in accordance to previous studies15,33,34. The estrous cycle consists of four stages: proestrus, estrus, metestrus, and diestrus. During the proestrus phase the genitals are swollen, pink, moist, and wrinkled. The estrus phase is wrinkly but less swollen, pink, and moist. Metestrus and diestrus are both reported as exhibiting no swelling and wrinkling, lacking in a pink hue, and dry34,35.
- Perform experiment at estrus: All mechanical tests were performed while the mice were at estrus, as this is the easiest to visualize and provides a consistent and repeatable timepoint.
- For mice undergoing basal tone testing, euthanize via guillotine. For mice tested only under the passive conditions, euthanize using carbon dioxide (CO2) inhalation. The guillotine serves to preserve the function of smooth muscle cells of the reproductive tract, as the CO2 gas alters the contractile properties of the smooth muscle cells36,37,38,39,40,41,42. It is imperative to perform the dissection within 30 minutes to minimize the chance of cell apoptosis.
2. Reproductive system dissection
- Set up: Place an absorbent pad on the workstation and fill a Petri dish and syringe with 4 °C Hank’s Balanced Salt Solution (HBSS) solution. Use a wipe for adipose tissue disposal. Place the mouse ventral side up and tape the paws and tail. Turn the microscope lights on and set out micro-scissors, scissors, two pairs of straight tweezers, and two pairs of curved tweezers.
- Using angled tweezers and scissors, lift the skin around the abdomen and make an incision at the base of the abdomen, above the pubic bone. The incision should be shallow enough to not puncture the abdominal muscle wall. Continue using the scissors to cut superiorly towards the rib cage and deep through the abdominal muscles.
- Remove superficial fat by pulling lightly on the fat with the curved tweezers and micro-scissors. Adipose tissue will reflect light heterogeneously with a glitter-like appearance. Place all the removed fat and tissue on the wipe. Identify both uterine horns and the pubic bone.
- Place closed scissors between the vaginal wall and the pubic bone. Carefully cut the middle of the pubic bone (pubic symphysis). Place curved tweezers on both ends of the cut pubic bone. Pull both cut ends laterally to allow for better access to the reproductive organs.
- Remove the bladder and the urethra from the vaginal wall. This can be done by using straight tweezers and micro-scissors. Hold the bladder with straight tweezers to create tension and use blunt dissection techniques to separate the surrounding tissue from the vagina. Once the bladder and urethra are dissected away, cut the base and remove from the body cavity.
- Identify the reproductive system: The uterine horns bifurcate from the cervix. The cervix can be identified from the vagina due to differences in geometry and stiffness. The outer diameter of the cervix is smaller than the vagina. The cervix is stiffer than the vagina and feels similar to that of a bead (Figure 1).
- Use ink and calipers to mark 3 mm dots along the organs. Start below the ovaries on the uterine tubes and mark dots inferiorly to reach the cervix. Use the center cervix dot to start a dot path down to the vagina introitus.
- Allow the ink to dry and separate the reproductive organs from surrounding adipose tissue, connective tissue, and the colon. Clean the vagina as close to the vaginal introitus as possible. Using scissors, cut around the vaginal introitus.
NOTE: It is possible for organs to dry out during this process. If this is a concern, a syringe filled with 4 °C HBSS may be used to add moisture to the organs. - Cut the uterine horns immediately inferior to the ovaries. Note that the organs will retract from the post explant length as the connective tissue is removed and the organ recoils. Place the dissected reproductive organs in a Petri dish filled with 4 °C HBSS. This change in length can be used in for calculating the estimated in vivo length (section 5).
NOTE: We have identified that using HBSS at this temperature during the dissection and cannulation does not affect the smooth muscle cell viability. Maintaining a pH of 7.4, however, is imperative for maintaining the viability of the smooth muscle cells. At this temperature, the HBSS has a pH level of 7.4. - After a 15-minute equilibration period in 4 °C HBSS, measure the space between dots using calipers. Record the measurements for each distance into a spreadsheet. These values will be used to calculate the in vivo stretch ratio (original length/explanted length).
- Set the wipe that contains the discarded tissue on the abdominal region with the excess tissue facing the inside of the mouse and soak the wipe in 4 °C HBSS. Wrap the mouse and excess tissue in foil and place in a freezer safe bag to be stored at -20 °C. Passive mechanical behavior on the vagina was not found to be significantly different after one freeze-thaw cycle43. All organs tested were used immediately after euthanasia or after one freeze-thaw cycle.
3. Cannulating
- Determine the proper cannula size for the organ type. In a typical C57BL6J mouse, the vagina uses cannulas that are both 3.75 mm in diameter and riveted. The cervix uses one cannula that is 3.75 mm for the vaginal end and a cannula 0.75 mm in diameter for the uterine end (Figure 2) The 0.75 mm cannula is smooth.
NOTE: The diameter sizes denoted above are used for typical nulliparous 4-6 months C57BL6 mice, C57BL6 x 129SvEv, and nonparous mice aged 7-9 months. However, certain circumstances, such as prolapse or pregnancy, may require a larger size cannula. - With each organ, mount the cervical side on the force transducer portion of the cannulation device. Mount the opposite end of the organ (vaginal or uterine) on the micrometer portion of the device. Tighten both ends with sutures.
- Due to the difference in thickness and degree of contractility among the vagina and cervix, varying techniques may be utilized to perform the most effective cannulation. For the vagina, place 2 sutures in between the 2nd and 3rd rivets of the cannula in a “X” fashion. When cannulating the cervix, the cannula is not riveted so the organ is best placed at the back of the cannula with 3 horizontal sutures on the uterine end and 4 sutures on the external os. For both organs, maximum length should be no more than 7 mm between the sutures (Figure 3).
4. Pressure myograph set up
- In order to set up the pressure myograph system, power on the testing system and fill the reservoir bottle with 200 mL of HBSS (Figure 4). Turn the heat to “on” and allow the HBSS in the reservoir bottle to heat up. Next, turn on the microscope and open the computer program. Ensure that the image of the cannulated organ, pressure interface, flow meter readings, and the sequencer function tool are all visible (Figure 5).
5. Basal tone mechanical testing
NOTE: The cervix exhibited a phasic nature during the beginning stages of testing. However, this diminished after preconditioning. Basal tone testing is done utilizing Krebs Ringer Buffer (KRB) in the basin of the DMT device. The buffer is aerated with 95% O2 and 5% CO2. After the basal tone portion is complete, calcium free KRB is utilized.
- Finding the unloaded geometry: Stretch the organ so that the wall is not in tension. For the vagina, observe the grooves on the vaginal wall. For the cervix, cut immediately below the ink dots that located above and below the central cervix mark. This devises a repeatable method for a cervical in situ length of 6 mm44. Measure the length from suture to suture with calipers
- Finding the unloaded pressure (UP): Increase the pressure from 0 to 10 mmHg in increments of 1 mmHg. Determine the pressure in which the organ is no longer collapsed. This can be determined as the largest jump in the outer diameter at a given pressure, as exhibited on the program monitor. After recording the pressure and outer diameter, note this as the first point wherein the organ is not collapsed and zero the force.
- Estimated in vivo stretch: Calculate the estimated in vivo stretch by dividing the length measured in vivo by the length measured post explant:
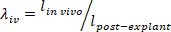
- Pressure-diameter pre-conditioning: Set the pressure to 0 mmHg, the length to the estimated in vivo length
 and the gradient to 1.5 mmHg/s. Run a sequence that takes the pressure from 0 mmHg to the in vivo pressure + unloaded (Table 1), hold for 30 seconds, and take the pressure to 0 mmHg with a 30 second hold period. After repeating for a total of 5 cycles, press Stop in the computer program and save the file.
and the gradient to 1.5 mmHg/s. Run a sequence that takes the pressure from 0 mmHg to the in vivo pressure + unloaded (Table 1), hold for 30 seconds, and take the pressure to 0 mmHg with a 30 second hold period. After repeating for a total of 5 cycles, press Stop in the computer program and save the file. - Finding the experimental in vivo stretch: Adjust the organ to be at the estimated in vivo length while at the unloaded pressure and press Start. Assess pressure vs force values for pressure values ranging from the unloaded pressure to the maximum pressure (Table 1). Press the Stop button in the computer program and save the file.
NOTE: The measured stretch value is calculated in situ. This is accompanied by the limitation that it can only be measured after disarticulating the pubic symphysis. As a result, the natural tethering is lost, which may modify the length. The theoretical stretch, however, is based on the previously introduced theory that the organ will experience minimal changes in force when exposed to physiological pressures to conserve energy45. In the protocol, the measured in vivo stretch will be the stretch value calculated using the experimentally identified length wherein there is minimal change in force when exposed to a physiological range of pressures. - Pressure-diameter pre-conditioning: Set the pressure to 0 mmHg, the length to the experimental in vivo length, and the gradient of 1.5 mmHg/s. Run a sequence that takes the pressure from 0 mmHg to the maximum pressure + UP , hold for 30 seconds, and back to 0 mmHg with an additional 30 second hold period. After repeating this for a total of 5 cycles, press the Stop button in the program interface and save the file.
NOTE: 5.4 is imperative for achieving a more consistent axial force reading with increasing pressure. This step aids in finding the correct in vivo stretch, which is often underestimated based on visual cues. 5.6 serves as a precautionary step to minimize hysteresis and to achieve a consistent, repeatable, mathematically interpretable response of the organ. - Force-length pre-conditioning: Enter 1/3 max pressure + UP for both the inlet and outlet pressure. Adjust the organ to -2% of the in vivo length and press Start. Adjust the length to +2% in vivo length then back down to -2% at 10 µm/s. Repeat axial extension for a total of 5 cycles. Press Stop in the computer program and save the file.
- Equilibration: With the organ at the determined in vivo length, set both the inlet and outlet pressure at 1/3 of the maximum pressure + UP. Equilibrate the organ for 10 minutes. Slowly bring both pressures back down to 0 mmHg with the gradient set as 1.5 mmHg/s.
- Re-evaluate the unloaded geometry: Set the organ to the in vivo length and the pressure to the unloaded pressure. Decrease the axial length towards the estimated unloaded length at a rate of 10 µm/s until there is minimal change in the force. This corresponding length is known as the unloaded length, or where the organ is not in tension nor compression. Before zeroing the force, record the unloaded length, outer diameter, and the force value.
NOTE: The prior unloaded geometry was determined by visual cues, which is purely qualitative. A re-evaluation is necessary for a quantitative method and to account for possible changes in length that may occur during the preconditioning. This geometry will be used in section 8. - Ultrasound Setup: Use the general imaging abdominal package to visualize the organs in the testing device. (Figure 6). Before testing, minimize artifacts from the bottom of the pressure myograph metal basin. Adjust the cannula to a height that is the maximum distance from the bottom with the tissue still being fully submerged in the testing solution. A custom holder is 3D printed to stabilize the transducer in a vertical position during imaging.
- Ultrasound Imaging: Identify the cannula near the force transducer and adjust the stage of the microscope to image along the length of the tissue. Throughout the testing process, the middle region along the length is tracked (Figure 6A,C). Following imaging, review the image “Cine store” loop that consists of a series of B-mode frames and identify the frame with the largest outer diameter. The thickness calculations made will be used in section 8.
- Pressure diameter testing (-2% in vivo length): Press Start and adjust the organ so that it is -2% of the in vivo length, set the pressure to 0 mmHg and gradient to 1.5 mmHg/s. Increase the pressure from 0 mmHg to the maximum pressure. Bring the pressure back down to 0 mmHg with a 20 second hold period. Repeat this for 5 cycles.
- Pressure diameter testing (in vivo length): Press Start and adjust the organ so that it is at the vivo length, set the pressure to 0 mmHg, and gradient to 1.5 mmHg/s. Increase the pressure from 0 mmHg to the maximum pressure. Bring the pressure back down to 0 mmHg with a 20 second hold period. Repeat this for 5 cycles.
- Pressure diameter testing (+2% in vivo length): Adjust the organ so that it is +2% in vivo length, set the pressure to 0 mmHg, and gradient to 1.5 mmHg/s. Increase the pressure from 0 mmHg to the maximum pressure and then back down to 0 mmHg with a 20 second hold period. Repeat this for 5 cycles. The pressure data from all three lengths will be used in section 8.
- Force-length testing (Nominal pressure): Set the pressure to the unloaded pressure and the organ to -2% of the in vivo length. Stretch the organ to +2% of the in vivo length and return to -2% the in vivo length at rate of 10 µm/s. Repeat for a total of 3 cycles.
- Force-length testing (1/3 maximum pressure + UP): Set the pressure to 1/3 of the maximum pressure + UP and adjust the organ to -2% the in vivo length. After pressing Start, stretch the organ to +2% the in vivo length and back to -2% the in vivo length at a rate of 10 µm/s. After repeating for a total of 3 cycles, press Stop and save the data.
- Force-length testing (2/3 maximum pressure + UP): Set the pressure to 2/3 of the maximum pressure + UP and adjust the organ to -2% the in vivo length. Press Start and stretch the organ to +2% the in vivo length and back to -2% the in vivo length at a rate of 10 µm/s. After repeating for a total of 3 cycles, press Stop and save the data.
- Force-length testing (maximum pressure + UP): Set the pressure to the maximum pressure + UP and adjust the organ to -2% the in vivo length. At a rate of 10 µm/s, stretch the organ to +2% of the in vivo length and back to -2% the in vivo length. After repeating for a total of 3 cycles, save the data. All force data will be used in section 8.
- Remove KRB testing media and wash with calcium-free KRB. Replace the media with calcium free KRB solution supplemented with 2 mM EGTA. Incubate the tissue for 30 minutes. Remove the solution and replace the media with fresh calcium-free KRB.
6. Passive mechanical testing
NOTE: If starting with passive testing start at step 1. If basal tone testing was performed prior to passive start at step 6. If starting with frozen tissue, allow a 30-minute equilibration period at room temperature before cannulating the organ.
- Finding the unloaded geometry: Stretch the organ so the wall of the organ is not in tension. Measure the cannulated organ from suture to suture and record this as the unloaded length.
- Finding the unloaded pressure: After pressing Start, increase the pressure from 0 to 10 mmHg in increments of 1 mmHg. While going through this process, determine the pressure in which the organ is not in tension. Using the computer program monitor, this can be determined from the largest jump in the outer diameter. After zeroing the force, record this pressure as well as the outer diameter and note this as the first point in which the organ is not collapsed.
- Estimated in vivo stretch: Calculate the estimated in vivo stretch by dividing the length measured in vivo by the length measured post-explant.
- Pressure diameter pre-conditioning: After pressing Start, set the pressure set to 0 mmHg, the length as the estimated in vivo length, and gradient to 1.5 mmHg/s. Begin running a sequence that takes the pressure from 0 mmHg to the maximum pressure and back to 0 mmHg. Repeat this process through 5 cycles with a 30 second hold time.
- Force-length preconditioning: Adjust the organ to the in vivo length and manually enter the unloaded pressure in the computer program for both pressures. After pressing Start, set the gradient to 2 mmHg and the pressure to 1/3 of the maximum. Stretch the organ up to +2% and back down to -2% stretch at 10 µm/s. Repeat this cycle for a total of 5 times and press Stop.
- Finding the experimental in vivo length: Find and plot force values at -2% of the in vivo length, the in vivo length, and +2% of the in vivo length. Take forces at evenly spaced pressures ranging from 0 mmHg to the maximum pressure. The experimental in vivo stretch will be the stretch value that exhibits a relatively flat line over a range of pressures.
- Repeat the pressure diameter and axial pre-conditioning steps at the new in vivo length.
- Equilibration: With the organ at the determined in vivo length, set the inlet and outlet pressure to the unloaded pressure. Let the organ re-equilibrate for 15 minutes. After 15 minutes, slowly bring the inlet and outlet pressure back down to 0 mmHg.
- Re-evaluate unloaded configuration: Bring the organ to the unloaded length and re-estimate the unloaded length. Record the unloaded length and the outer diameter while the pressure is 0 mmHg, the unloaded pressure, and 1/3 the maximum pressure. Zero the force at the unloaded pressure. The diameter at the unloaded pressure is the in vivo diameter.
NOTE: Re-estimating the unloaded length is necessary as small plastic deformations were observed previously in soft biological tissues following preconditioning. This unloaded configuration will be the one utilized in section 8. - Ultrasound: Perform ultrasound B-mode imaging at the unloaded length and pressure.
- Pressure-diameter testing: With the organ at -2% of the experimentally determined in vivo length and the pressure at 0 mmHg, press Start. Increase the pressure from 0 mmHg to the maximum pressure and back to 0 mmHg. Hold the 2-0 mmHg step for 20 seconds. After repeating for a total of 5 times, press the Stop button in the interface and save the file.
NOTE: Repeat at the experimental in vivo length, +2% of the experimental in vivo length. - Force-length testing: Set the pressure to nominal pressure and adjust the organ to -2% of the in vivo length. Stretch the organ up to +2% of the in vivo length and back to -2% of the in vivo length at a rate of 10 µm/s. After repeating for a total of 3 times, save the data. Repeat this for 1/3 max pressure, 2/3 max pressure, and at the max pressure.
- Calculate the unloaded thickness from ultrasound images B-mode image. Using imaging software, draw a line to denote the penetration depth. Set the scale to the length of the line (i.e., 2000 µm as shown in Figure 6B and 6D).
- Wall thickness calculations: Using a computer software, trace and measure the inner and outer diameter of the organ. Then, draw and measure a line between the diameters. Draw a total of 25 transmural lines. Average all data points and repeat for a total of 3 times.
7. Clean up
- Ensure that the pressure is 0 mmHg and turned off. Close the main inlet and outlet off for both three-way valves. Aspirate the remaining fluid from the basin of the cannulation device.
- Remove the organ from the stage and fill the reservoir bottle with deionized water. Using a syringe, rinse the cannula with water. Connect the tubing to bypass the cannula.
- Turn the pressure and flow on, set the inlet pressure to 200 mmHg, the outlet pressure to 0 mmHg, gradient to 10 mmHg/s, and let the flow run for 5 minutes. Allow the system to run while the reservoir bottle is empty and let the air run for 5 minutes or until the lines are dry.
8. Data analysis
- For pressure diameter testing, collect data from where the pressure begins to increase from the minimum value until the maximum. For force-length testing, collect data from just below the maximum peak in force until the force stopped decreasing.
- Open the data file for each pressure-diameter test and select the mean pressure tab. Navigate to the loading region of the last curve, 0 mmHg to the maximum pressure, and drop the data into a spreadsheet. Select the same region on the outer diameter, inlet pressure, outlet pressure, force, temperature, pH, and flow tab placing each item in the same document.
- Open the data for each Force-length test. Navigate to the loading region of the curve, -2% to +2%, and drag and drop the data into a spreadsheet. Select the same region for the other measured variables and place each item in the same spreadsheet.
- For the pressure diameter and force length test subtract the UP from all pressure values.
- Average the pressure-diameter data every 1 mmHg (i.e., 0+/- 0.5, 1+/-0.5, 2+/- 0.5).
- Find the unloaded volume of the organ (V). Equation 1 can be utilized to find V, given that R02 is the unloaded outer radius measured by the microscope, L is the unloaded length, and H is the unloaded thickness as detected by the ultrasound. The assumption of incompressibility is leveraged, meaning that the organ conserves volume while subjected to deformations.
NOTE: The unloaded length is measured with calipers from suture to suture. The unloaded diameter is measured via the microscope, camera, and software followed by calculation of the radius (Figure 5) The unloaded thickness is calculated from the ultrasound images (Figure 6).
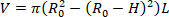 Equation 1
Equation 1 - Using the assumption of incompressibility, use the unloaded volume, deformed outer radius (
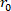 ), and length (
), and length (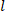 ) to determine the deformed inner radius
) to determine the deformed inner radius 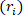 .
.
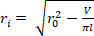 Equation 2
Equation 2 - Use Equations 3, 4, and 5 to calculate each stress, respectively. In equations 3-5, P is defined as the intraluminal pressure and Ft is the force measured by the transducer.
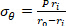 Equation 3
Equation 3
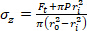 Equation 4
Equation 4
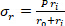 Equation 5
Equation 5 - Plot the pressure-diameter relationship, force-pressure relationship, circumferential stress-circumferential stretch relationship, and the axial stress and circumferential stretch values (Figure 7, Figure 8). The stretch values can be calculated using the midwall radius. Calculations of the circumferential and axial stresses can be found in Equations 6 and 7, respectively.
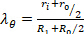 Equation 6
Equation 6
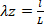 Equation 7
Equation 7 - Calculate compliance near the physiological pressure range and at the in vivo stretch. The lower pressure bound (LPB) is 1 standard deviation below the mean measured pressure. The upper pressure bound (UPB) is 1 standard deviation above the mean measured pressure9.
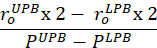
- Calculate the tangent moduli to quantify the material stiffness. Identify the calculated circumferential stress that corresponds to the lower pressure bound and upper bound pressure. Fit a linear line to the circumferential stress- circumferential stretch curve within the identified stress range at the in vivo length. Calculate the slope of the line9.
Representative Results
Successful analysis of the mechanical properties of the female reproductive organs is contingent on appropriate organ dissection, cannulation, and testing. It is imperative to explant the uterine horns to the vagina without any defects (Figure 1). Depending on the organ type, the cannula size will vary (Figure 2). Cannulation must be done so that the organ cannot move during the experiment but also not damage the wall of the organ during the procedure (Figure 3). Failure of either step will result in inability of the vessel to hold pressure. Testing procedure standardization is vital to the success of the protocol in order to yield consistent and repeatable results.
Once the organ is dissected and cannulated properly, power on the pressure myograph system. The setup of the pressure myograph systems involves a controller unit, flow meter, and stage (Figure 4). The pressure myograph system is used to monitor various aspects of the organ as it undergoes mechanical testing (Figure 5). An ultrasound system, or equivalent, is used to measure the thickness of the organs in the unloaded state with and without basal tone (Figure 6). After mechanical testing, the tangent moduli may be calculated for the circumferential and axial directions (Table 2).
Both basal tone testing and passive testing yield key mechanical properties of the reproductive tract, with and without the contractile contribution of smooth muscle cells (Figure 7, Figure 8). Scaling between the organs requires a few adjustments to the protocols (Table 1), as the cervix and vagina experience different loads in vivo46-48. Such variations may be monitored through techniques such as pressure catherization. Pressure catherization is a method used previously to monitor the in vivo conditions within the vagina and uterus49-53. Models in the previous studies range from mice, rabbits, and humans. The same principles would apply similarly to the cervical and vaginal pressure specific for the murine model. Though, regardless which organ is being tested, the same materials are needed for the protocols (Table 3).
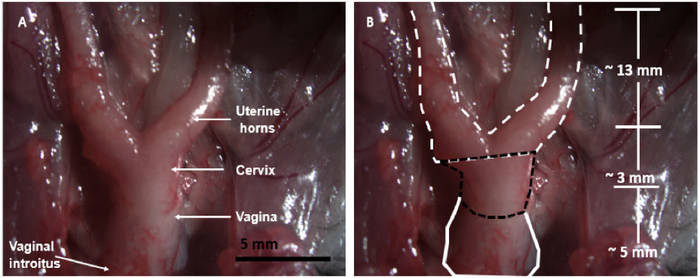
Figure 1: Murine dissection diagram. The mouse dissection for the reproductive organs: both uterine horns, cervix, and the vagina. In the figure, the bladder and urethra are removed from the anterior of the vagina. The intestines and abdominal muscles were reflected superiorly. Please click here to view a larger version of this figure.

Figure 2: Size comparison of the two cannula. Size comparison of the two cannulas used for cannulation of the reproductive organs. The larger cannula (D = 3.75 mm) is used for the vaginal tissue (A). The smaller cannula (D = 0.75 mm) is used for cannulating cervical tissue (B). The cervical cannula is smooth while the vaginal cannula has two grooves. Please click here to view a larger version of this figure.
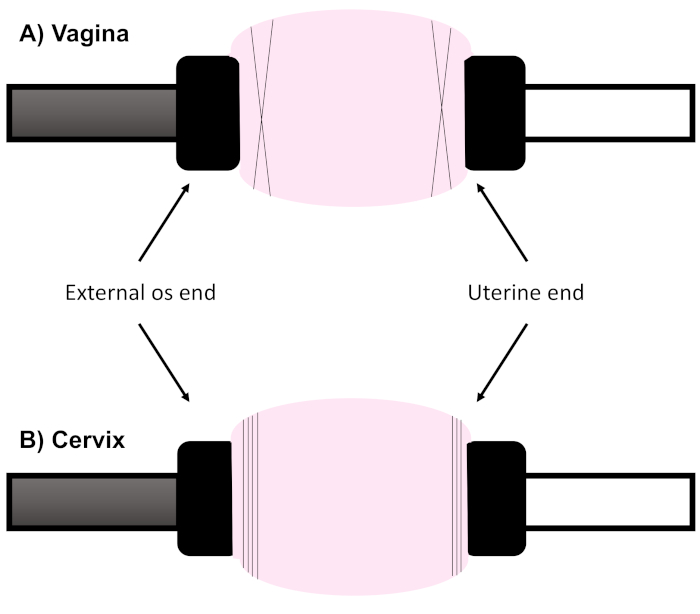
Figure 3: Cannulation method for vagina and cervix. Due to the varying geometry and thickness of the reproductive organs, they are most effectively cannulated in distinct manners. For the vagina, place two sutures in an “X” fashion. When cannulating the cervix, place 3 horizontal sutures on the uterine end and 4 sutures on the external os. Please click here to view a larger version of this figure.
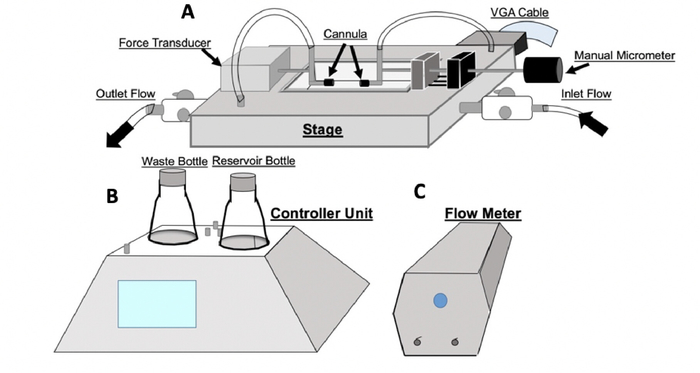
Figure 4: Setup for pressure myograph device. The setup of the DMT device utilized for both basal and passive testing. The DMT is composed of three main hubs: the stage (A), controller unit (B), and flow meter (C). Within the controller unit, there is a reservoir bottle and a waste bottle. The reservoir bottle is initially filled with fluid that empties as the experiment is carried out. The waste bottle, which is initially empty, collects the fluid that runs through the experiment. The controller unit interfaces with the DMT software on the computer and controls the pressure, temperature, and flow. The controller unit reads the outputs from the force and pressure transducers within the stage through a VGA interface cable. The stage component of the system contains an inlet and outlet flow of the system. The inlet and outlet flow have corresponding inlet and outlet pressures measured by the system. Please click here to view a larger version of this figure.
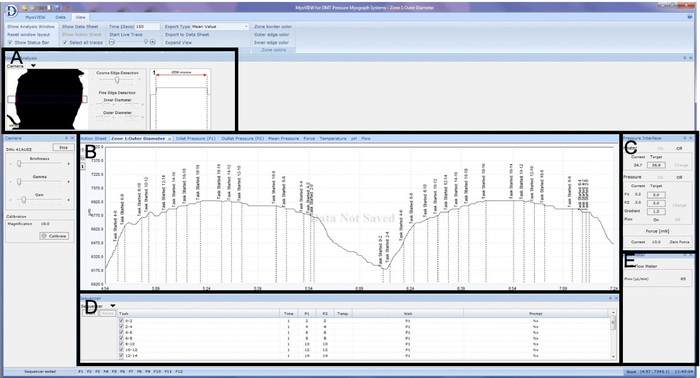
Figure 5: File setup on the pressure myograph program. Display of computer software set-up. A box is drawn around the region of interest and outer diameter of the tissue is optically tracked in real-time (A). Data obtained during mechanical testing is recorded and displayed real-time in the outer diameter, inlet pressure, outlet pressure, mean pressure, force, temperature, pH, and flow tab (B). Within the pressure interface pressure (mmHg), gradient (mmHg/s), and flow is controlled. Further, the axial force (mN) measured by the in-line force transducer is displayed. Flow rate (µL/min) is reported in the flow meter tab (C). Pressure sequencing is shown and controlled in the sequencer tab (D). Data recorded during mechanical testing is recorded and displayed real-time in the outer diameter, inlet pressure, outlet pressure, mean pressure, force, temperature, pH, and flow tab (E). A representative Pressure Diameter test of the vagina is displayed showing outer diameter as a function of time on the outer diameter tab. Please click here to view a larger version of this figure.
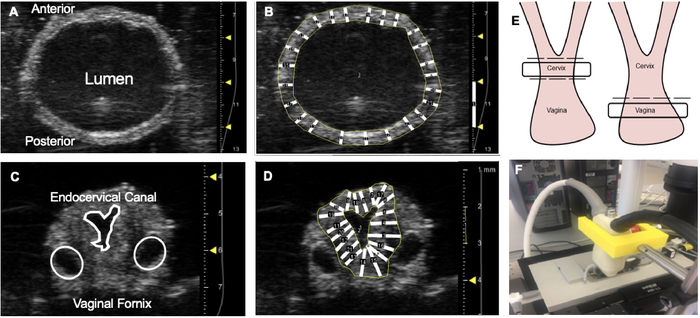
Figure 6: Ultrasound Imaging. Ultrasound imaging of the murine reproductive organs. All images were taken using the ultrasound system on the short-axis-B mode. A representative image of the vagina at the unloaded length and pressure (A). Vaginal wall thickness was calculated in ImageJ. A vertical line was drawn along the depth scale (mm) to calibrate the number of pixels per µm. The polygon tool was used to trace the inner and outer diameter. Then transmural lines were drawn to calculate the thickness and averaged (B). This was performed 3 times. A representative image of the cervix at the unloaded length and pressure (C). Wall thickness was then calculated using Image J and the polygon tool in a similar manner to that of the vagina (D). Within the reproductive complex, the outer diameter is tracked at two different locations (E). Throughout the imaging process, the transducer is stabilized by a 3-D printed holder (F). Please click here to view a larger version of this figure.
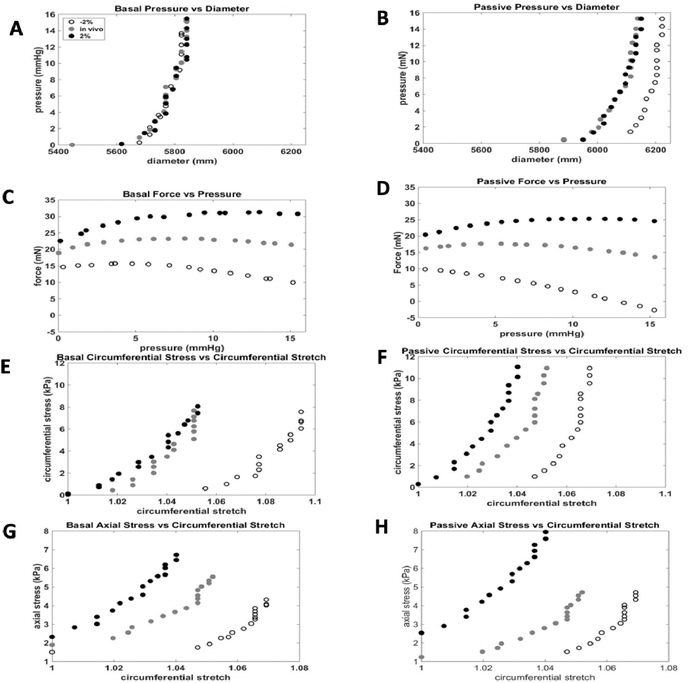
Figure 7: Representative results for vaginal testing. The representative mechanical testing results of the vaginal basal and passive protocols. With the data obtained by the DMT system, several mechanical relationships can be derived. A) Basal Pressure-Diameter, B) Passive Pressure-Diameter, C) Basal Force-Pressure, D) Passive Force-Pressure, E) Basal circumferential stress-circumferential stretch, F) Passive circumferential stress-circumferential stretch, G) Basal axial stress-circumferential stretch, H) Passive axial stress-circumferential stretch. Please click here to view a larger version of this figure.
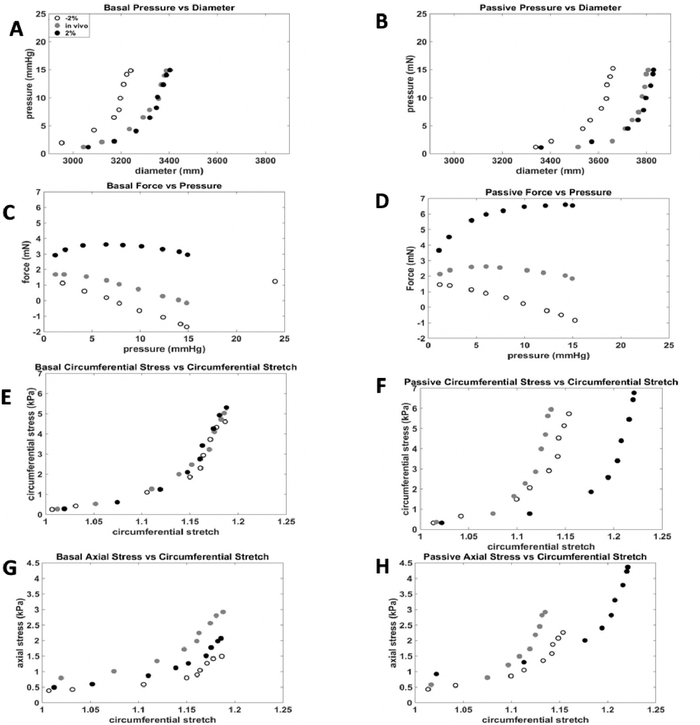
Figure 8: Representative results for cervical testing. The representative mechanical testing results of the cervical basal and passive protocols. With the data obtained by the DMT system, several mechanical relationships can be derived. A) Basal Pressure-Diameter, B) Passive Pressure-Diameter, C) Basal Force-Pressure, D) Passive Force-Pressure, E) Basal circumferential stress-circumferential stretch, F) Passive circumferential stress-circumferential stretch, G) Basal axial stress-circumferential stretch, H) Passive axial stress-circumferential stretch. Please click here to view a larger version of this figure.
| In Vivo Pressure | Maximum Pressure | 1/3 Max Pressure | 2/3 Max Pressure | Axial Stretch | Cannula Size | Recommended number of sutures |
|
| Vagina | 7 mmHg | 15 mmHg | 5 mmHg | 10 mmHg | -2%, in vivo, +2% | 3.75 mm | 2– in an "X" fashion |
| Cervix | 10 mmHg | 200 mmHg | 66 mmHg | 133 mmHg | -2%, in vivo, +2% | 0.75 mm for uterine end 3.75 mm for vaginal end |
3 horizontal sutures on the uterine end 4 sutures on the vaginal external os |
Table 1: Summary of information for scaling the mechanical testing methods for each organ. The unloaded pressure values were measured using catherization techniques under anesthesia (4% isoflurane in 100% oxygen). A balloon catheter was utilized for the vaginal measurements and a 2F catheter for the cervix.
| Vagina | Cervix | |
| Basal Circumferential (kPa) |
127.94 | 188 |
| Basal Axial (kPa) |
56.8 | 75.44 |
| Passive Circumferential (kPa) |
246.03 | 61.26 |
| Passive Axial (kPa) |
112.74 | 19.26 |
Table 2: The representative results for the tangent moduli of the vagina and cervix. The tangent moduli were calculated for both basal and passive conditions as well as for both circumferential and axial directions. All measurements provided are in units of kPa.
Discussion
The protocol provided in this article presents a method for determining the mechanical properties of the murine vagina and cervix. The mechanical properties analyzed in this protocol include both the passive and basal tone conditions of the organs. Passive and basal tone conditions are induced by altering the biochemical environment in which the organ is submerged. For this protocol, the media involved in basal testing contains calcium. Testing the basal tone condition permits isolation of the smooth muscle cell mechanical contribution within the female reproductive organs54,55. When performing passive mechanical testing, the media does not contain calcium. The lack of calcium inhibits the smooth muscle cells from contracting. This permits elucidation of other ECM components, such as collagen and elastic fibers, which largely dictate the passive mechanical properties. When combined with biochemical and histological analysis, these results permit elucidation of relationships between ECM microstructural composition and mechanical function. This then allows for delineation of the structural and mechanical mechanisms of pathologies relevant to women’s reproductive health.
Previously, the vagina and cervix were tested uniaxially27,28. The vagina and cervix, however, demonstrate anisotropic properties and experience multiaxial loading in vivo29,30 . Hence, pressure myograph systems used herein provide quantitative information on multiaxial loading that may aid in understanding the etiologies of reproductive pathologies, as well as the subsequent design of potential treatments. Further, pressure myography permits assessment of multiaxial properties while preserving the in vivo organ geometry and the native cell-matrix interaction56 . In vivo, the cells actively remodel the surrounding ECM in response to changes in biomechanical and biochemical cues57,58,59. The protocol used herein is advantageous as it permits monitoring of subsequent changes in bulk organ properties under physiologically relevant conditions. This aids in providing a platform to generate systematic datasets of multiaxial active and passive mechanical properties. Further, the data collected in these experiments may be leveraged to formulate and validate microstructurally-motivated nonlinear constitutive models to describe and predict the mechanical response of the female reproductive organs in healthy and pathological states16,60.
An additional system component that was advantageous to the protocol was the use of ultrasound imaging to measure the thickness of the organ walls. The thickness is crucial information for calculating stress experienced while undergoing testing.
With any experimental set up, there are some limitations to this procedure. This protocol currently only considers the elastic response of the vagina and cervix and not the viscoelastic response. A potential method to mitigate this limitation in the future is to modify the existing protocol to include creep and stress relaxation assays61. A second limitation is assuming the organs are incompressible. Within this study, thickness was solely measured at the unloaded configuration, as motivated by prior studies that demonstrate nonpregnant murine tissue exhibits minimal changes in volume during osmotic loading62. Furthermore, additional studies have operated under the same assumption of incompressibility44,60,63. Ideally, an ultrasound would be performed for the entirety of the experiment in order to remove the need for the incompressibility assumption and to better inform finite element models. A final limitation is the lack of quantified in vivo cervical pressure to inform the loading protocols. Literature suggests that cervical pressure in human women is 37 mmHg53. Mice, however, may exhibit different cervical pressure from that of humans. A difference in vaginal pressure was demonstrated between rodent models and human samples64,65. Further studies are needed to quantify pressure in the non-pregnant murine cervix. Towards this end, intra-uterine pressure was recently reported throughout pregnancy49.
The commercially available pressure myograph system utilized in this procedure measures the force properties of elastic, hollow organs. This protocol is easily adaptable to other various organs and tissues by modifying the chemical additives in the bath, cannula size, and suture thickness.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The work was funded by NSF CAREER award grant #1751050.
Materials
| 2F catheter | Millar | SPR-320 | catheter to measure cervical pressure |
| 6-0 Suture | Fine Science Tools | 18020-60 | larger suture ties |
| CaCl2 (anhydrous) | VWR | 97062-590 | HBSS concentration: 140 mg/ mL |
| CaCl2-2H20 | Fischer chemical | BDH9224-1KG | KRB concentration: 3.68 g/L |
| Dextrose (D-glucose) | VWR | 101172-434 | HBSS concentration: 1000 mg/mL KRB concentration: 19.8 g/L |
| Dumont #5/45 Forceps | Fine Science Tools | 11251-35 | curved forceps |
| Dumont SS Forceps | Fine Science Tools | 11203-25 | straight forceps |
| Eclipse | Nikon | E200 | microscope used for imaging |
| Flow meter | Danish MyoTechnologies | 161FM | flow meter within the testing apparatus |
| Force Transducer – 110P | Danish MyoTechnologies | 100079 | force transducer |
| ImageJ | SciJava | ImageJ1 | used to measure volume |
| Instrument Cases | Fine Science Tools | 20830-00 | casing to hold dissection tools |
| KCl | Fisher Chemical | 97061-566 | HBSS concentration: 400 mg/ mL KRB concentration: 3.5 g/L |
| KH2PO4 | G-Biosciences | 71003-454 | HBSS concentration: 60 mg/ mL |
| MgCl2 | VWR | 97064-150 | KRB concentration: 1.14 g/L |
| MgCl2-6H2O | VWR | BDH9244-500G | HBSS concentration: 100 mg/ mL |
| MgSO4-7H20 | VWR | 97062-134 | HBSS concentration: 48 mg/ mL |
| Mircosoft excel | Microsoft | 6278402 | program used for spreadsheet |
| Na2HPO4 (dibasic anhydrous) | VWR | 97061-588 | HBSS concentration: 48 mg/mL KRB concentration: 1.44 g/L |
| NaCl | VWR | 97061-274 | HBSS concentration: 8000 mg/mL KRB concentration: 70.1 g/L |
| NaHCO3 | VWR | 97062-460 | HBSS concentration: 350 mg/ mL KRB concentration: 21.0 g/L |
| Pressure myograph systems | Danish MyoTechnologies | 110P and 120CP | Pressure myograph system: prorgram, cannulation device, and controller unit |
| Pressure Transducer | Danish MyoTechnologies | 100106 | pressure transducer |
| Student Dumont #5 Forceps | Fine Science Tools | 91150-20 | straight forceps |
| Student Vannas Spring Scissors | Fine Science Tools | 91500-09 | micro-scissors |
| Tissue dye | Bradley Products | 1101-3 | ink to measure in vivo stretch |
| Ultrasound transducer | FujiFilm Visual Sonics | LZ-550 | ultrasound transducer used; 256 elements, 40 MHz center frequency |
| VEVO2100 | FujiFilm Visual Sonics | VS-20035 | ultrasound used for imaging |
| Wagner Scissors | Fine Science Tools | 14069-12 | larger scissors |
References
- Capone, D., et al. Evaluating Residual Strain Throughout the Murine Female Reproductive System. Journal of Biomechanics. 82, 299-306 (2019).
- Danforth, D. The fibrous nature of the human cervix, and its relation to the isthmic segment in gravid and nongravid uteri. American Journal of Obstetrics and Gynecology. 53 (4), 541-560 (1947).
- Hughesdon, P. The fibromuscular structure of the cervix and its changes during pregnancy and labour. Journal of Obstetrics and Gynecology of the British Commonwealth. 59, 763-776 (1952).
- Bryman, I., Norstrom, A., Lindblo, B. Influence of neurohypophyseal hormones on human cervical smooth muscle cell contractility in vitro. Obstetrics and Gynecology. 75 (2), 240-243 (1990).
- Joy, V., et al. A New Paradigm for the Role of Smooth Muscle Cells in the Human Cervix. Obstetrics. 215 (4), e471-e478 (2016).
- Xu, X., Akgul, Y., Mahendroo, M., Jerschow, A. Ex vivo assessment of mouse cervical remodeling through pregnancy via Na (23) MRS. NMR Biomedical. 23 (23), 907-912 (2014).
- Leppert, P. Anatomy and Physiology of cervical ripening. Clinical Obstetrics and Gynecology. 43 (43), 433-439 (2000).
- Schlembach, D., et al. Cervical ripening and insufficiency: from biochemical and molecular studies to in vivo clinical examination. European Journal of Obstetrics, Gynecology, and Reproductive Biology. 144, S70-S79 (2000).
- Stoka, K., et al. Effects of Increased Arterial Stiffiness on Atherosclerotic Plaque Amounts. Journal of Biomechanical Engineering. 140 (5), (2018).
- Mohram, D., Heller, L. Ch. 7. Cardiovascular Physiology. , (2006).
- Yoshida, K., et al. Quantitative Evaluation of Collagen Crosslinks and Corresponding Tensile Mechanical Properties in Mouse Cervical Tissue during Normal Pregnancy. PLoS One. 9, e112391 (2014).
- Mahendroo, M. Cervical remodeling in term and preterm birth: insight from an animal model. Society for Reproduction and Fertility. 143 (4), 429-438 (2012).
- Elovitz, M., Miranlini, C. Can medroxyprogesterone acetate alter Toll-like receptor expression in a mouse model of intrauterine inflammation?. American Journal of Obstetrics and Gynecology. 193 (3), 1149-1155 (2005).
- Ripperda, C., et al. Vaginal estrogen: a dual-edged sword in postoperative healing of the vaginal wall. North American Menopause Society. 24 (7), 838-849 (2017).
- Nelson, J., Felicio, P., Randall, K., Sims, C., Finch, E. A Longitudinal Study of Estrous Cyclicity in Aging C57/6J Mice: Cycle, Frequency, Length, and Vaginal Cytology. Biology of Reproduction. 27 (2), 327-339 (1982).
- Ferruzzi, J., Collins, M., Yeh, A., Humphrey, J. Mechanical assessment of elastin integrity in fibrillin-1-deficient carotid arteries: implications for Marfan Syndrome. Cardiovascular Research. 92 (2), 287-295 (2011).
- Mariko, B., et al. Fribrillin-1 genetic deficiency leads to pathological ageing of arteries in mice. The Journal of Pathology. 224 (1), 33-44 (2011).
- Rahn, D., Ruff, M., Brown, S., Tibbals, H., Word, R. Biomechanical Properties of The Vaginal Wall: Effect of Pregnancy, Elastic Fiber Deficiency, and Pelvic Organ Prolapse. American Urogynecological Society. 198 (5), (2009).
- Caulk, A., Nepiyushchikh, Z., Shaw, R., Dixon, B., Gleason, R. Quantification of the passive and active biaxial mechanical behavior and microstructural organization of rat thoracic ducts. Royal Society Interface. 12 (108), 20150280 (2015).
- Amin, M., Le, V., Wagenseil, J. Mechanical Testing of Mouse Carotid Arteries: from Newborn to Adult. Journal of Visualized Experiments. (60), e3733 (2012).
- Sokolis, D., Sassani, S., Kritharis, E., Tsangaris, S. Differential histomechanical response of carotid artery in relation to species and region: mathematical description accounting for elastin and collagen anisiotropy. Medical and Biological Engineering and Computing. 49 (8), 867-879 (2011).
- Kim, J., Baek, S. Circumferential variations of the mechanical behavior of the porcine thoracic aorta during the inflation test. Journal of Biomechanics. 44 (10), 1941-1947 (2011).
- Faury, G., et al. Developmental adaptation of the mouse cardiovascular system to elastin haploinsufficency. Journal of Clinical Investigation. 11 (9), 1419-1428 (2003).
- Naito, Y., et al. Beyond Burst Pressure: Initial Evaluation of the Natural History of the Biaxial Mechanical Properties of Tissue-Engineered Vascular Grafts in the Venous Circulation Using a Murine Model. Tissue Engineering. 20, 346-355 (2014).
- Sommer, G., et al. Multaxial mechanical response and constitutive modeling of esophageal tissues: Impact on esophageal tissue engineering. Acta Biomaterialia. 9 (12), 9379-9391 (2013).
- Sokolis, D., Orfanidis, I., Peroulis, M. Biomechanical testing and material characterization for the rat large intestine: regional dependence of material parameters. Physiological Measurement. 32 (12), 1969-1982 (2011).
- Martins, P., et al. Prediction of Nonlinear Elastic Behavior of Vaginal Tissue: Experimental Results and Model Formation. Computational Methods of Biomechanics and Biomedical Engineering. 13 (3), 317-337 (2010).
- Feola, A., et al. Deterioration in Biomechanical Properties of the Vagina Following Implantation of a High-stiffness Prolapse Mesh. BJOG: An International Journal of Obstetrics and Gynaecology. 120 (2), 224-232 (2012).
- Huntington, A., Rizzuto, E., Abramowitch, S., Prete, Z., De Vita, R. Anisotropy of the Passive and Active Rat Vagina Under Biaxial Loading. Annals of Biomedical Engineering. 47, 272-281 (2018).
- Tokar, S., Feola, A., Moalli, P., Abramowitch, S. Characterizing the Biaxial Mechanical Properties of Vaginal Maternal Adaptations During Pregnancy. ASME 2010 Summer Bioengineering Conference, Parts A and B. , 689-690 (2010).
- Feloa, A., et al. Impact of Pregnancy and Vaginal Delivery on the Passive and Active Mechanics of the Rat Vagina. Annals of Biomedical Engineering. 39 (1), 549-558 (2010).
- Baah-Dwomoh, A., Alperin, M., Cook, M., De Vita, R. Mechanical Analysis of the Uterosacral Ligament: Swine vs Human. Annual Biomedical Engineering. 46 (12), 2036-2047 (2018).
- Champlin, A. Determining the Stage of the Estrous Cycle in the Mouse by the Appearance. Biology of Reproduction. 8 (4), 491-494 (1973).
- Byers, S., Wiles, M., Dunn, S., Taft, R. Mouse Estrous Cycle Identification Tool and Images. PLoS One. 7 (4), e35538 (2012).
- McLean, A. Performing Vaginal Lavage, Crystal Violet Staining and Vaginal Cytological Evaluation for Mouse Estrous Cycle Staging Identification. Journal of Visualized Experiments. 67, e4389 (2012).
- Bugg, G., Riley, M., Johnston, T., Baker, P., Taggart, M. Hypoxic inhibition of human myometrial contractions in vitro: implications for the regulation of parturition. European Journal of Clinical Investigation. 36 (2), 133-140 (2006).
- Taggart, M., Wray, S. Hypoxia and smooth muscle function: key regulatory events during metabolic stress. Journal of Physiology. 509, 315-325 (1998).
- Yoo, K., et al. The effects of volatile anesthetics on spontaneous contractility of isolated human pregnant uterine muscle: a comparison among sevoflurane, desflurane, isoflurane, and halothane. Anesthesia and Analgesia. 103 (2), 443-447 (2006).
- de Souza, L., et al. Effects of redox disturbances on intentional contractile reactivity in rats fed with a hypercaloric diet. Oxidative Medicine and Cellular Longevity. , 6364821 (2018).
- Jaue, D., Ma, Z., Lee, S. Cardiac muscarinic receptor function in rats with cirrhotic cardiomyopathy. Hepatology. 25, 1361-1365 (1997).
- Xu, Q., Shaffer, E. The potential site of impaired gallbladder contractility in an animal mode of cholesterol gallstone disease. Gastroenterology. 110 (1), 251-257 (1996).
- Rodriguez, U., et al. Effects of blast induced Neurotrauma on pressurized rodent middle cerebral arteries. Journal of Visualized Experimentals. (146), e58792 (2019).
- Rubod, C., Boukerrou, M., Brieu, M., Dubois, P., Cosson, M. Biomechanical Properties of Vaginal Tissue Part 1: New Experimental Protocol. Journal of Urology. 178, 320-325 (2007).
- Robison, K., Conway, C., Desrosiers, L., Knoepp, L., Miller, K. Biaxial Mechanical Assessment of the Murine Vaginal Wall Using Extension-Inflation Testing. Journal of Biomechanical Engineering. 139 (10), 104504 (2017).
- Van loon, P. Length-Force and Volume-Pressure Relationships of Arteries. Biorheology. 14 (4), 181-201 (1977).
- Fernandez, M., et al. Investigating the Mechanical Function of the Cervix During Pregnancy using Finite Element Models Derived from High Resolution 3D MRI. Computational Methods Biomechanical and Biomedical Engineering. 19 (4), 404-417 (2015).
- House, M., Socrate, S. The Cervix as a Biomechanical Structure. Ultrasound Obstetric Gynecology. 28 (6), 745-749 (2006).
- Martins, P., et al. Biomechanical Properties of Vaginal Tissue in Women with Pelvic Organ Prolapse. Gynecologic and Obstetrics Investigation. 75, 85-92 (2013).
- Rada, C., Pierce, S., Grotegut, C., England, S. Intrauterine Telemetry to Measure Mouse Contractile Pressure In vivo. Journal of Visualized Experiments. (98), e52541 (2015).
- Lumsden, M. A., Baird, D. T. Intra-uterine pressure in dysmenorrhea. Acta Obstectricia at Gynecologica Scandinavica. 64 (2), 183-186 (1985).
- Milsom, I., Andersch, B., Sundell, G. The Effect of Flurbiprofen and Naproxen Sodium On Intra-Uterine Pressure and Menstrual Pain in Patients With Primary Dysmennorrhea. Acta Obstetricia et Gynecologica Scandinavica. 67 (8), 711-716 (1988).
- Park, K., et al. Vasculogenic female sexual dysfunction: the hemodynamic basis for vaginal engorgement insufficiency and clitoral erectile insufficiency. International Journal of Impotence Journal. 9 (1), 27-37 (1997).
- Bulletti, C., et al. Uterine Contractility During Menstrual Cycle. Human Reproduction. 15, 81-89 (2000).
- Kim, N. N., et al. Effects of Ovariectomy and Steroid Hormones on Vaginal Smooth Muscle Contractility. International Journal of Impotence Research. 16, 43-50 (2004).
- Giraldi, A., et al. Morphological and Functional Characterization of a Rat Vaginal Smooth Muscle Sphincter. International Journal of Impotence Research. 14, 271-282 (2002).
- Gleason, R., Gray, S. P., Wilson, E., Humphrey, J. A Multiaxial Computer-Controlled Organ Culture and Biomechanical Device for Mouse Carotid Arteries. Journal of Biomechanical Engineering. 126 (6), 787-795 (2005).
- Swartz, M., Tscumperlin, D., Kamm, R., Drazen, J. Mechanical Stress is Communicated Between Different Cell Types to Elicit Matrix Remodeling. Proceedings of the National Academy of Sciences of the United States of America. 98 (11), 6180-6185 (2001).
- Rachev, A. Remodeling of Arteries in Response to Changes in their Mechanical Environment. Biomechanics of Soft Tissue in Cardiovascular Systems. 441, 221-271 (2003).
- Lee, E. J., Holmes, J., Costa, K. Remodeling of Engineered Tissue Anisotropy in Response to Altered Loading Conditions. Annals of Biomedical Engineering. 36 (8), 1322-1334 (2008).
- Akintunde, A., et al. Effects of Elastase Digestion on the Murine Vaginal Wall Biaxial Mechanical Response. Journal of Biomechanical Engineering. 141 (2), 021011 (2018).
- Griffin, M., Premakumar, Y., Seifalian, A., Butler, P., Szarko, M. Biomechanical Characterization of Human Soft Tissues Using Indentation and Tensile Testing. Journal of Visualized Experiments. (118), e54872 (2016).
- Myers, K., Socrate, S., Paskaleva, A., House, M. A Study of the Anisotripy and Tension/Compression Behavior of Human Cervical Tissue. Journal of Biomechanical Engineering. 132 (2), 021003 (2010).
- Murtada, S., Ferruzzi, J., Yanagisawa, H., Humphrey, J. Reduced Biaxial Contractility in the Descending Thoracic Aorta of Fibulin-5 Deficent Mice. Journal of Biomechanical Engineering. 138 (5), 051008 (2016).
- Berkley, K., McAllister, S., Accius, B., Winnard, K. Endometriosis-induced vaginal hyperalgesia in the rat: effect of estropause, ovariectomy, and estradiol replacement. Pain. 132, s150-s159 (2007).
- van der Walt, I., Bø, K., Hanekom, S., Rienhardt, G. Ethnic Differences in pelvic floor muscle strength and endurance in South African women. International Urogynecology Journal. 25 (6), 799-805 (2014).

