Handling and Tagging Techniques for Implanting Juvenile American Shad with a New Acoustic Microtransmitter
Summary
This article provides a detailed procedure for optimal handling practices and implantation of an acoustic micro transmitter into juvenile American shad. The results of our laboratory study suggest that these tagging techniques could be implemented in field studies of juvenile American shad with a high probability of survival.
Abstract
The use of telemetry techniques to better understand the behavior and survival of juvenile American shad (Alosa sapidissima), as they migrate through hydropower systems, has been challenging because shad are widely known to be particularly sensitive to handling. The goal of this study was to develop a tagging protocol using a new, acoustic micro transmitter that minimizes the detrimental effects of the tagging process and maximizes post-tagging survival of juvenile American shad. Limiting out-of-water handling and the use of brackish saltwater (7.5 parts per thousand) before and after tagging improved survival for shad tagged using a simple pectoral implantation method. This protocol provides a detailed, step-by-step procedure for tagging juvenile shad with acoustic transmitters. Fish tagged using this procedure and held in the laboratory for 60 days had an 81.5% survival rate, compared to 70% for their untagged counterparts. The successful tagging and handling practices developed in this study could be applied to field telemetry studies of juvenile shad and other sensitive species.
Introduction
American shad (Alosa sapidissima) is an anadromous fish species that is native to the East Coast of the United States. Reduced habitat availability and increased development of hydroelectric dams have resulted in population declines of shad across their native range1,2. Juvenile shad and other alosines, on their out-migration to the ocean, may be especially susceptible to injury and mortality when passing through hydroelectric structures3,4,5. Understanding the passage and survival rates of juvenile shad at hydropower dams is critical for informing the relicensing of these facilities as well as restoration efforts for the species. However, successful tagging techniques to assess the passage and survival rates of American shad as they migrate to the ocean are lacking. Shad tagged with transmitters for telemetry studies should be representative of the untagged population of inference and should not be negatively affected by the tag or the tagging process6,7.
To help improve the ability to track juvenile shad, Pacific Northwest National Laboratory (PNNL) developed a new acoustic micro transmitter for studying American shad and other fish species with similar compressiform body types. One of the common challenges of studying American shad and other alosines is their sensitivity to handling, transport, and tagging compared to other species. For example, Raquel et al.8 found that handling and trucking mortalities were consistently higher for juvenile American shad than for the other five species of juvenile fish in their study. Of the few published studies that have evaluated efforts to tag juvenile American shad, a wide range of survival has been reported, from as low as 2% after 7 days9 and up to 100% after 48 hours post-tagging10 and very little information on longer-term survival and transmitter retention for tagged juvenile shad is available.
The challenges in successfully handling and tagging sensitive species such as American shad have shed light on the knowledge gaps about their migration, behavior, and habitat use. The ability to track movement through hydropower dams would greatly advance the understanding of passage and survival rates for American shad. It would help inform management decisions for existing hydroelectric facilities and novel designs for systems that minimize effects on fish species and life stages not previously studied. As new transmitter technology is developed, understanding the effects of the transmitter and the tagging process is imperative to minimize bias and accurately assess passage and survival. The objectives of this study were to evaluate the 60-day survival of juvenile American shad tagged with a new acoustic micro transmitter and to provide a handling and tagging protocol that would reduce the negative effects of tagging on shad, thereby making them more comparable to their untagged counterparts.
Protocol
PNNL is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. American shad were handled in accordance with federal guidelines for the care and use of laboratory animals11, and the protocols for our study were conducted in compliance with and approved by PNNL's Institutional Animal Care and Use Committee.
1. Preparation of a post-tagging recovery tank
- When brackish saltwater (7.5 ppt) is not readily available, use a static system for holding shad with proper aeration and circulation for 1-2 days before the release (Figure 1).
- In a static, circular tank, install an airlift system to provide aeration. Connect a PVC pipe to the side of the tank such that a tee is located at the top, another tee is near the middle, and a screened fitting is threaded onto the bottom of the pipe.
- Next, connect an air stone to a compressed air line and place the air stone at the bottom of the pipe near the screen.
NOTE: The screen prevents fish from swimming into the airlift system. - Fill the tank with fresh water until the exit port (middle PVC tee) of the airlift is approximately halfway submerged. Then, turn off the water.
- Next, turn on and increase the air supply until the aerated water exiting the port creates a directional flow for the fish to orient.
- Add commercial sea salt to make a 7.5 ppt brackish saltwater solution and stir until dissolved.
2. Preparation of saltwater fish source bucket and saltwater anesthetic solution
- Measure 7.5 g of sea salt for every 0.5 L of water and dissolve in a bucket.
NOTE: In step 3.5, an equal volume of freshwater containing the shad is added to create a final concentration of 7.5 ppt. - In another bucket, measure and dissolve 7.5 g of sea salt for every liter of water.
- Add 120 mg of tricaine methanesulfonate (MS-222) buffered with 120 mg of sodium bicarbonate per liter of saltwater.
- Add supplemental air to the anesthetic buckets.
3. Collection of shad with a water-to-water transfer into brackish saltwater
- Partially fill another bucket with fresh water and place it sideways into the pre-tagging source tank.
- Use a net or hand to gently guide shad to swim into the bucket.
- Once an appropriate number of fish are in the bucket, turn the bucket upright, and secure it with a perforated lid.
- Pour out any excess water through the lid, keeping the fish contained in the target amount of fresh water (i.e., half the volume of the final 7.5 ppt saltwater source bucket).
- Gently pour the shad and the freshwater into the saltwater source bucket prepared in step 2.1.
NOTE: The final salinity will be 7.5 ppt. - Provide supplemental air to the pre-tag fish source bucket using an aquarium air pump and air stone to maintain dissolved oxygen at an acceptable level (>90% saturation is ideal).
4. Implantation of an acoustic transmitter in a shad
- Disinfect transmitters in 70% ethanol for 20 min and rinse with sterile water before use.
- Using a dip net with smooth, extra-fine mesh (~0.4 mm), net a fish from the source bucket and into the anesthetic bucket. Shad should lose equilibrium and spinal reflexes in ~2-3 min, depending on water temperature and other water quality parameters.
- Once fully anesthetized to stage four12, use a gloved hand to gently place the fish on a wet measuring board to obtain its length.
- Move the fish into a water-filled weigh boat on a tared scale to obtain its weight.
- Record length and weight, acoustic tag code, and any comments on the fish's condition prior to tagging, such as scale loss or hemorrhaging.
- Place the fish in a transfer container filled with anesthetic saltwater and deliver it, along with the acoustic transmitter, to the fish surgeon.
- Place the fish, left side facing down, on a wet, water-resistant foam pad prepared with a v-groove (Figure 2A).
NOTE: The v-groove keeps the fish from sliding too much during the procedure and allows water to pool around the fish's mouth so it can actively draw water over its gills. - Supply fresh water to the fish's mouth via tubing attached to a gravity-fed water reservoir.
- Using a disinfected or brand new #11 stainless steel surgical blade, make a 3 mm-long incision vertically between the myomeres near the distal end of the pectoral fin.
- If needed, remove a scale at the blade tip if it is obstructing the fish's skin.
- Carefully insert the transmitter into the incision and push it posteriorly until the entire tag rests inside the body cavity (Figure 2B).
- If needed, use the blunt end of the scalpel (or fine-tipped forceps) to carefully insert the tag fully.
- Place the tagged fish in a small container of 7.5 ppt saltwater with aeration to allow the fish to recover from anesthesia.
- Once the fish has regained equilibrium, make a water-to-water transfer from the recovery container to the post-tagging holding tank containing 7.5 ppt saltwater.
- Allow the tagged fish to recover in saltwater for 1-2 days prior to the release.
Representative Results
Two series of tagging evaluations were conducted to address the efficacy of tagging juvenile shad – preliminary trials in 2020 and a long-term holding study in 2021. Preliminary laboratory evaluations were conducted at PNNL in November 2020 to determine a preferred method for implanting American shad with a novel acoustic microtransmitter. Prototype transmitter designs (n = 4, P1-P4) were paired with different tagging locations (gastric, pectoral, pelvic, and dorsal) for a combined total of 4 unique acoustic transmitter-tagging location treatments (n = 40 fish per treatment, Table 1). All test fish were randomly assigned to a treatment and holding tank. Test fish were held in 2 holding tanks with equal numbers of fish from each treatment (i.e., 20 fish per treatment) per tank for 14 days. For the first 2 days of the evaluation, shad were held in brackish saltwater (7.5 ppt) and allowed to recover from tagging and handling. Then the tanks were switched to flow-through fresh water for the remainder of the evaluation period.
For the preliminary evaluation, tagged fish and fin-clipped controls ranged in size from 50-80 mm in fork length. Juvenile shad survival and tag retention were highest for fish implanted via a pectoral incision compared to the other tagging techniques (Figure 3). Additional pilot evaluations also demonstrated that handling techniques such as water-to-water transfers and holding fish in brackish saltwater before and after stressful events, such as tagging, were critical to increasing survival rates.
Using the successful tagging and handling protocols from the preliminary evaluation, a laboratory study was conducted at PNNL in 2021 to evaluate the long-term 60 d survival and tag retention of juvenile American shad implanted with an acoustic transmitter using the pectoral incision tagging method. The long-term evaluation used the dummy transmitter P5 (Figure 4), an improved prototype design similar in form and size to the P1 design used in the preliminary evaluation. The average dimensions and weight of the dummy P5 tag were 7.6 mm long × 2.3 mm in diameter and a weight in air of 0.058 g (standard deviation 0.002 g), which resulted in a tag burden of <1%. The prototype acoustic transmitter with functional components (Figure 4) has dimensions of 7.6 mm long x 2.0 mm in diameter and a weight in air of 0.050 g.
Juvenile American shad used in the long-term evaluation had been held in captivity for 4 months at the time of testing. While the experiment was designed to have equal numbers of treatment and control fish held in two tanks for a 60 day period, the remaining shad numbers at the time of tagging were low. Therefore, more shad were randomly assigned to the tagged treatment group than the control group to get a better understanding of the long-term efficacy of the tagging technique on shad. Each of the two tanks held 27 treatment fish and 9 or 10 control fish. However, since survival from Tank A (13.8%) was significantly worse than Tank B (78.4%; Fisher's exact test, p < 0.001) and there was no difference in survival between the tagged and control groups within each tank, only the results for tank B are included here.
Shad (fork length 69-105 mm; weight 3.9-11.7 g) were either tagged with the P5 transmitter using the pectoral incision (n = 27) or assigned to the control group (n = 10). Control fish were handled using the same procedures, including being placed on the surgical pad for ~20 s, but they did not receive a fin clip or an incision nor were they implanted with a transmitter. After tagging, both treatment groups were held in brackish saltwater (7.5 ppt) for 1 day and then switched to flow-through river water for the remainder of the study. Survival at 60 days was 81.5% for the tagged group and 70% for the untagged controls (Figure 5). Survival for tagged fish in this evaluation was defined as both survival and tag retention because tag expulsion cannot be differentiated from a mortality event in a telemetry study. There was no significant difference in survival between the two groups (Fisher's exact test, P = 0.884); however, the power to detect a difference was 38.4% due to the small sample sizes. Although the power to detect a difference between the treatments was low, the results of the long-term evaluation show that this handling and tagging protocol can be used with moderate success to implant American shad with acoustic transmitters.

Figure 1: Post-tagging recovery tank filled with brackish saltwater. An airlift system supplies oxygen to the static tank. Please click here to view a larger version of this figure.
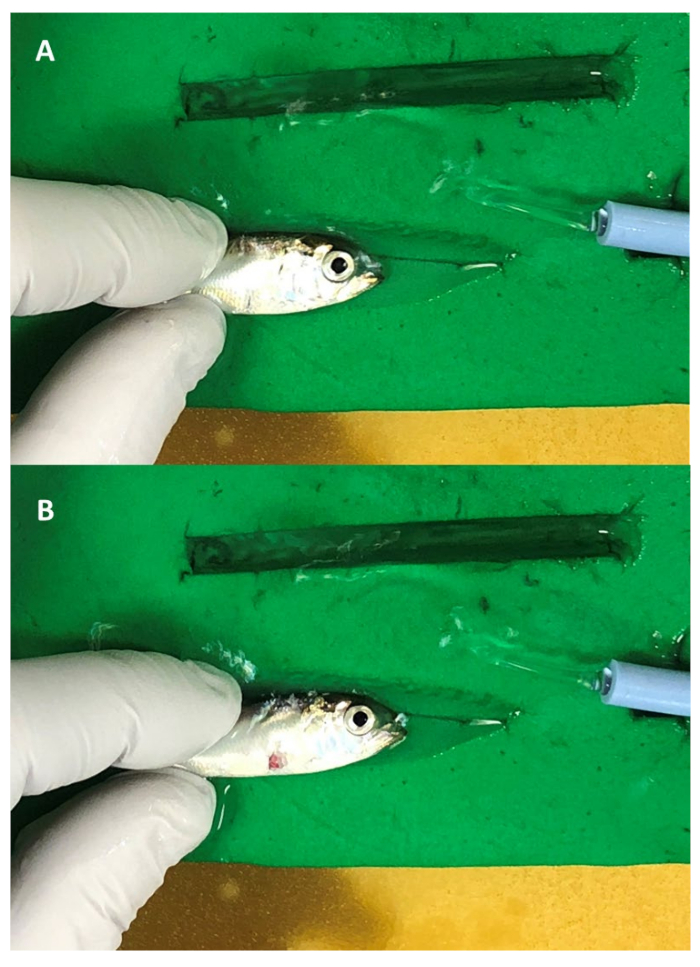
Figure 2: Acoustic transmitter implantation of a Juvenile American shad. Juvenile American shad (A) with a pectoral incision and (B) with the dummy P5 transmitter inserted into the incision. Note, the mouth of the shad is partially submerged in water flowing from the blue tubing. Please click here to view a larger version of this figure.
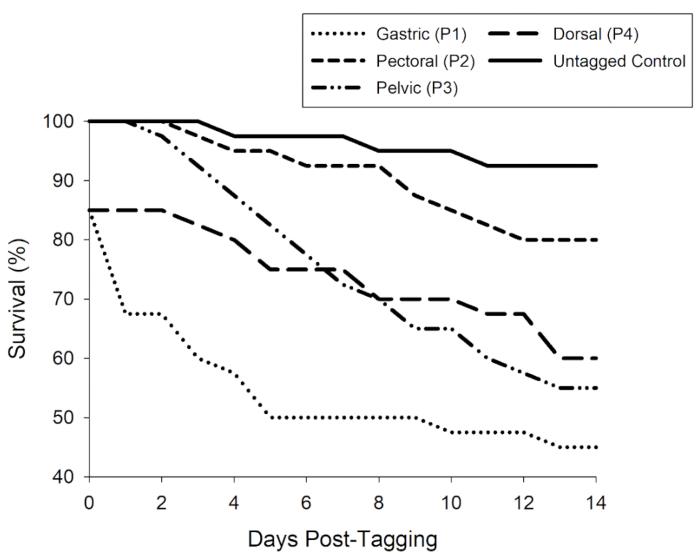
Figure 3: Survival percentage over a preliminary 14 d evaluation with one group of untagged controls and four tagged groups of juvenile American shad. The tagged treatments consisted of four tagging locations (gastric, pectoral, pelvic, and dorsal) each paired with a unique transmitter prototype (P1-P4). Survival of the tagged fish was defined as both survival and tag retention. Please click here to view a larger version of this figure.

Figure 4: Acoustic and dummy transmitters for tagging juvenile American shad. (A) Functional acoustic microtransmitter and (B) the dummy P5 prototype transmitter, that was used in the 60 d laboratory survival study. Note the numbers 4-7 on the ruler represent centimeters. Please click here to view a larger version of this figure.
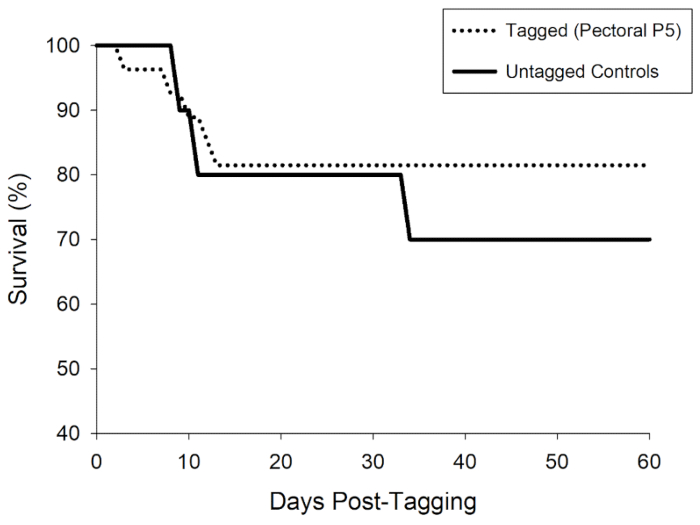
Figure 5: Survival percentage of American shad over a long-term 60 d holding study. Juvenile shad were either untagged (Untagged Controls; solid line) or tagged (Tagged [Pectoral P5]; dashed line) with a dummy transmitter. Survival of the tagged group was defined as both survival and tag retention. Please click here to view a larger version of this figure.
| Tag Type | Tag Location | N | Fork Length (mm) | Mean Tag Weight (SD; g) | Tag Burden (%) | Survival (%) | Mean Time to Tag/Clip (s) | |
| Range | Mean (SD) | |||||||
| P1 | Gastric | 40 | 50 – 76 | 60 (6.0) | 0.058 (0.003) | 1.5 – 5.2 | 45 | 12 |
| P2 | Pectoral | 40 | 50 – 78 | 60 (7.3) | 0.039 (0.001) | 1.0 – 3.2 | 80 | 23 |
| P3 | Pelvic | 40 | 50 – 70 | 58 (5.3) | 0.039 (0.001) | 1.0 – 4.1 | 55 | 26 |
| P4 | Dorsal | 40 | 50 – 80 | 61 (6.8) | 0.088 (0.004) | 0 | 60 | 57 |
| Control | NA (Clip) | 40 | 50 – 80 | 59 (5.7) | NA | 0 | 92.5 | 14 |
Table 1:Tagging and survival information for American shad implanted with prototype transmitters (P1–P4) or marked with upper caudal and lower caudal fin clips (Control) as part of the preliminary evaluation. Tag location for the control group is Not Applicable (NA) as these fish only received fin clips (Clip). Note, that the tag type P4 was a neutrally buoyant design. The standard deviation (SD) of the mean is listed in parentheses.
Discussion
The need to study juvenile American shad movements around hydropower facilities has prompted the development of a handling and tagging protocol to improve the survival of tagged shad. At PNNL, initial attempts to implant juvenile shad with a novel acoustic transmitter, without the use of saltwater resulted in 100% mortality within 24 hours. The subsequent, carefully developed handling and tagging protocol demonstrated that American shad can be implanted with an acoustic microtransmitter and held long-term in a laboratory setting with a high survival rate (81.5%). Minimizing out-of-water handling and the use of brackish saltwater before and after tagging was essential to the success of tagging juvenile American shad.
In the preliminary evaluation, shad as small as 50 mm were tagged with a dummy transmitter using four implantation methods. Gastric tagging, one of the most common methods for tagging adult shad13,14,15 had promising results during pilot testing but had a high incidence of tag loss during the preliminary evaluation. Implantation through a pelvic incision has been used successfully to study adult Twaite shad movements16 and dorsal attachments have been used for short-term monitoring of juvenile American shad10. More recently, tag implantation through a pectoral incision was used to study longer-term movements of adult shad in both riverine and marine environments17. In the preliminary evaluation at PNNL, the pectoral incision location performed better than the three other locations evaluated, and the 7-d post-tagging survival was over 90%.
Overall, the results from these evaluations showed that the survival of tagged shad was comparable to the survival of untagged shad beyond the duration of the acoustic transmitter's battery life, which is expected to be ~30 days with an acoustic signal transmitted every 5 seconds.
This transmitter design and tagging protocol show great promise for studying small, sensitive, and threatened fish species like American shad in field applications, allowing researchers to gain valuable information on fish movements near hydroelectric facilities. For example, this tagging technique will be used in an upcoming field application to study the behavior of acoustic-tagged juvenile shad as they approach the spillway and powerhouse of a hydroelectric dam. The results gained from in-river studies can better inform management decisions at these facilities and can help conserve the species throughout their juvenile life stage. Future studies should evaluate the effectiveness of this procedure for tagging and tracking run-of-river fish in field conditions. In addition, these techniques are easily transferable to implanting shad or other sensitive species with Passive Integrated Transponder (PIT) tags, which can provide long-term monitoring throughout their life history.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This study was funded by the U.S. Department of Energy (DOE) Water Power Technologies Office. The laboratory studies were conducted at PNNL, which is operated by Battelle for the DOE under Contract DE-AC05-76RL01830. The authors would like to thank Dana McCoskey and Tim Welch of DOE, Eric Francavilla, Ryan Harnish, Huidong Li, Stephanie Liss, Brian Mason, Megan Nims, Brett Pflugrath, and Ashlynn Tate of PNNL for their assistance with the study and the manuscript, and U.S. Army Corps of Engineers and Pacific States Marine Fisheries Commission for their help in collecting the juvenile shad.
Materials
| #11 stainless steel surgical blade | Exel | 29502 | purchased from Med-Vet International; no real preference on blade vendor |
| #11 stainless steel surgical blade | Miltex | MIL4-311 | purchased from Med-Vet International; no real preference on blade vendor |
| 2 gallon bucket | Leaktite | #2GL White Pail | |
| acoustic transmitter for American shad | Pacific Northest National Laboratory | Patent-Pending | BattelleIPID: 32500 |
| air stone | Pentair | AS3 | |
| aquarium air pump | Tetra | Whisper | |
| dissolved oxygen meter | YSI | ProODO or ProSolo | |
| ethanol | Decon Laboratories | 2805HC | |
| fine mesh net | Blue Ribbon | ABLEC8 | |
| fish holding tank | Reiff Manufacturing | NA | round aquaculture tank |
| foam garden kneeler | Tommyco | 12003 | |
| plastic storage container | Ziploc | discontinued; 8oz container with lid | |
| PVC cement | Oatey | 30821 | |
| PVC pipe | Charlotte Pipe | NA | PVC Schedule 40 2" diameter |
| PVC primer | Oatey | 30757 | |
| PVC tee | Charlotte Pipe | NA | 2" PVC Schedule 40 S x S x S Tee |
| sea salt | InstantOcean | SS15-10 | |
| silicone tubing 3/16" | Pentair | tp30s | tubing to supply water during tagging |
| sodium bicarbonate | Fisher Chemical | S233-500 | |
| sterile water | NA | NA | water is sterilized using an autoclave |
| tricaine methanesulfonate | Syndel USA | 15650 | |
| tubing for airline | Hydromaxx | 1403038050 | |
References
- Dadswell, M. J., Rulifson, R. A. Macrotidal estuaries: A region of collision between migratory marine animals and tidal power development. Biological Journal of the Linnean Society. 51, 93-113 (1994).
- Limburg, K. E., Waldman, J. R. Dramatic declines in North Atlantic diadromous fishes. BioScience. 59, 955-965 (2009).
- Castro-Santos, T., Mulligan, K. B., Kieffer, M., Haro, A. J. Effects of plunge pool configuration on downstream passage survival of juvenile blueback herring. Aquaculture and Fisheries. 6 (2), 135-143 (2021).
- Dubois, R. B., Gloss, S. P. Mortality of juvenile American shad and striped bass passed through Ossberger crossflow turbines at a small-scale hydroelectric site. North American Journal of Fisheries Management. 13 (1), 178-185 (1993).
- Pflugrath, B. D., et al. The susceptibility of Juvenile American shad to rapid decompression and fluid shear exposure associated with simulated hydroturbine passage. Water. 12 (2), 586 (2020).
- Brown, R. S., et al. An evaluation of the maximum tag burden for implantation of acoustic transmitters in juvenile Chinook salmon. North American Journal of Fisheries Management. 30 (2), 499-505 (2010).
- Skalski, J. R., et al. Status after 5 years of survival compliance testing in the Federal Columbia River Power System (FCRPS). North American Journal of Fisheries Management. 36 (4), 720-730 (2016).
- Raquel, P. F. Effects of handling and trucking on chinook salmon, striped bass, American shad, steelhead trout, threadfin shad, and white catfish salvaged at the John E. Skinner delta fish protective facility (Vol. 19). Interagency Ecological Study Program for the Sacramento-San Joaquin Estuary. , (1989).
- Kleinschmidt, G., Sullivan, E. Relicensing Study 3.3.3. Evaluate downstream passage of juvenile American Shad. Interim Study Report: Northfield Mountain Pumped Storage Project (No. 2485) and Turners Falls Hydroelectric Project (No. 2485) and Turners Falls Hydroelectric Project (No. 1889). FirstLight. , (2016).
- Heisey, P. G., Mathur, D., Rineer, T. A reliable tag-recapture technique for estimating turbine passage survival: application to young-of-the-year American shad (Alosa sapidissima). Canadian Journal of Fisheries and Aquatic Sciences. 49, 1826-1834 (1992).
- National Research Council. . Guide for the Care and Use of Laboratory Animals.: Eighth Edition. , (2011).
- Summerfelt, R. C., Smith, L. C., Schreck, C. B., Moyle, P. B. Anesthesia, surgery, and related techniques. Methods for Fish Biology. , 213-263 (1990).
- Bailey, M. M., Isely, J. J., Bridges, W. C. Movement and population size of American shad near a low-head lock and dam. Transactions of the American Fisheries Society. 133 (2), 300-308 (2004).
- Grote, A. B., Bailey, M. M., Zydlewski, J. D. Movements and demography of spawning American Shad in the Penobscot River, Maine, prior to dam removal. Transactions of the American Fisheries Society. 143 (2), 552-563 (2014).
- Harris, J. E., Hightower, J. E. Movement patterns of American shad transported upstream of dams on the Roanoke River, North Carolina and Virginia. North American Journal of Fisheries Management. 31 (2), 240-256 (2011).
- Bolland, J. D., et al. Refinement of acoustic-tagging protocol for twaite shad Alosa fallax (Lacépède), a species sensitive to handling and sedation. Fisheries Research. 212, 183-187 (2019).
- Gahagan, B. I., Bailey, M. M. Surgical implantation of acoustic tags in American Shad to resolve riverine and marine restoration challenges. Marine and Coastal Fisheries. 12 (5), 272-289 (2020).

