Modeling Ovarian Cancer Multicellular Spheroid Behavior in a Dynamic 3D Peritoneal Microdevice
Summary
To study ovarian tumor progression in a physiologically relevant model, multicellular spheroids were cultured in a microdevice under simulated fluid flow. This dynamic 3D model emulates the intraperitoneal environment with the cellular and mechanical components where ovarian cancer metastasis occurs.
Abstract
Ovarian cancer is characterized by extensive peritoneal metastasis, with tumor spheres commonly found in the malignant ascites. This is associated with poor clinical outcomes and currently lacks effective treatment. Both the three-dimensional (3D) environment and the dynamic mechanical forces are very important factors in this metastatic cascade. However, traditional cell cultures fail to recapitulate this natural tumor microenvironment. Thus, in vivo-like models that can emulate the intraperitoneal environment are of obvious importance. In this study, a new microfluidic platform of the peritoneum was set up to mimic the situation of ovarian cancer spheroids in the peritoneal cavity during metastasis. Ovarian cancer spheroids generated under a non-adherent condition were cultured in microfluidic channels coated with peritoneal mesothelial cells subjected to physiologically relevant shear stress. In summary, this dynamic 3D ovarian cancer-mesothelium microfluidic platform can provide new knowledge on basic cancer biology and serve as a platform for potential drug screening and development.
Introduction
Ovarian cancer is the most lethal gynecological cancer and is characterized by widespread peritoneal dissemination and the formation of malignant ascites1. This extensive peritoneal metastasis represents a major clinical challenge and is associated with poor clinical outcomes. Unlike most solid carcinomas that metastasize via the blood, ovarian cancer primarily disseminates within the peritoneal cavity. Tumor cells exist as multicellular aggregates/spheroids during the process of metastasis2. The fact that suspension culture can enrich ovarian cancer stem/tumor-initiating cells further suggests that these spheroids may be associated with both tumor aggressiveness and enhanced chemoresistance3,4. There are differences in drug response between 2D and 3D cultures, which presumably have different molecular mechanisms5.
The essential interaction with the mesothelium constructs the primary microenvironment for ovarian tumor progression. These mesothelial cells lie on an extracellular matrix (ECM), where fibronectin is a ubiquitous constituent. A link between the increased expression of mesothelial cell-derived fibronectin and tumor progression has been shown. Fibronectin is abundantly present in malignant ascites6,7. Ovarian cancer cells are also able to induce the secretion of fibronectin from mesothelial cells in order to promote early ovarian cancer metastasis8.
Emerging evidence shows that mechanical stimuli, including shear stress, can modulate cell morphology, gene expression, and, thus, the phenotypes of tumor cells9,10,11. As malignant ascites develop and accumulate during tumor progression, ovarian tumor cells are exposed to fluid flow and the resulting shear stress. A number of groups, ours included, have shown the impact of shear stress on ovarian cancer progression, including cytoskeleton modifications, epithelial-to-mesenchymal transitions, and cancer stemness12,13,14,15. Thus, a physiologically relevant microenvironment is important for the investigation of tumor peritoneal metastasis. However, current in vitro hydrodynamic culture systems have limitations on mimicking and controlling a constant, low, physiologically relevant shear stress16,17,18,19. Conventional in vitro approaches focusing on either the cellular or mechanical environment are still limited in mimicking the complexity of the intraperitoneal microenvironment with proper physiological relevance.
Here, in order to engineer a new model of the peritoneum to overcome the limitations of conventional strategies and to advance the study of the intraperitoneal compartment in cancer metastasis, a 3D microfluidic-based platform with controlled fluid flow was designed. In this model, ovarian cancer spheroids were co-cultured with primary human peritoneal mesothelial cells in microfluidic chips under continuous fluidic flow (Figure1A). The mesothelial cells were plated on fibronectin. Non-adherent ovarian cancer spheroids were seeded into microfluidic channels with continuous flow medium perfused by a syringe pump. Both the 3D environment and the dynamic mechanical forces are very important factors of the metastatic cascade. This platform can be used for investigating the intraperitoneal microenvironment in terms of complex cellular and co-culture interactions, as well as with regard to dynamic mechanical cues.
Protocol
1. Microfluidic Device Design and Fabrication
- Microfluidic master design
- Design and draw the microfluidic channel pattern with any computer-aided design (CAD) software.
NOTE: Normally, the CAD drawing can be sent to a photomask company to produce the photomask. The microfluidic design consists of three identical parallel channels, each with the following dimensions: 4 mm × 25 mm × 250 µm (width × length × height) and set 2 mm apart. Both channel ends were designed with 127° angles to facilitate liquid entrance and exit (Figure 1B). The channel used in this protocol was reported in a previous publication13. - Wash the silicon wafer with ultrasonication (500 W/42 kHz) in acetone, isopropanol, and deionized water for 15 min each. Dry the wafer with pressurized nitrogen gas. Heat the silicon wafer on a hot plate at 250 °C for 15 min to completely dry it.
- Spin-coat a layer of SU-8 2075 photoresist (125 µm-thick) onto the silicon wafer with a spinning speed of 1,800 rpm. Bake the photoresist-coated wafer directly on a hot plate at 65 °C and then at 95 °C, to remove excess solvent, for 5 and 25 min, respectively (according to the SU-8 product manual).
- Repeat step 3 and spin-coat another layer of SU-8 2075 photoresist to obtain a final thickness of 250 µm. Bake the photoresist-coated wafer directly on a hot plate at 65 °C for 7 min and then at 95 °C for 30 min. Slowly cool the wafer to room temperature. Do not cool with pressurized air.
- Align and put the photomask directly on top of the wafer. Expose it to UV light for 20 s to crosslink the pattern.
- Bake the wafer with photoresist at 65 °C and 95 °C on a hotplate for 5 and 12 min, respectively, to remove the solvent for polymerization (according to the SU-8 product manual). Afterwards, cool the wafer to room temperature.
- Remove the non-crosslinked photoresist by immersing the wafer in SU-8 developer for 25 min. Rinse the wafer with isopropanol and dry it with pressured air.
NOTE: If a milky solution was observed when rinsing with isopropanol, indicating incomplete development, immerse the wafer back into SU-8 developer for a longer development time. - Place the wafer on a hot plate at 180 °C for 10 min to heal the pattern from potential cracks. Turn off the hot plate and slowly cool down the wafer to room temperature by leaving it on the hot plate.
- Add a few drops of trichloro (1H, 1H, 2H, 2H-perfluorooctyl) silane in a vial cap. Put the vial cap in the vacuum desiccator. Place the wafer next to the vial cap to silanize the wafer for 10 min.
- Design and draw the microfluidic channel pattern with any computer-aided design (CAD) software.
- PDMS chip fabrication
- Wrap the wafer with aluminum foil to create a holder.
- Mix the polydimethylsiloxane (PDMS) base and curing agent at a 10:1 mass ratio with a centrifugal mixer under the mixing and degas function. Pour the PDMS mixture slowly onto the wafer to a height of around 5 mm. Degas the PDMS under vacuum for 40 min. Cure the PDMS in an oven at 65 °C for 2 h.
- Carefully peel the PDMS slab off the master and trim the PDMS along the channels until it is the size of a glass slide. Punch the PDMS with a 1-mm biopsy punch to create the inlets and outlets of the device.
- Clean the PDMS surface with pressurized air and adhesive tape to remove dust and PDMS debris.
- Place the PDMS slab, with the channel side facing upwards, into a plasma cleaner. Place a clean glass slide into the plasma cleaner alongside the PDMS slab.
- Treat the PDMS and glass slide under air plasma at high RF level for 1 min. Bind the PDMS to the glass slide to create an irreversible covalent bond.
- Place the PDMS chip on a hot plate at 150 °C for 1 h to enhance the bonding strength and to return the hydrophobicity of the PDMS before use.
2. Seeding the Microfluidic Chip with Primary Peritoneal Mesothelial Cells (HPMCs)
- Fibronectin coating of microfluidic channels
- Sterilize the microfluidic channel with 30 µL of 75% ethanol. Remove the ethanol and rinse twice with 30 µL of sterilized phosphate-buffered saline (PBS) using a pipette tip. Thoroughly aspirate PBS in the channels.
NOTE: To minimize contamination, the microfluidic cell culture should be carefully conducted under fully aseptic conditions through the use of a laminar flow hood. - Prepare a 10 µg/mL fibronectin solution in serum-free M199:MCDB105 (1:1) medium. Pipette the solution up and down, with special care to avoid bubble formation. Do not vortex.
- Slowly fill up each channel with 30 µL of fibronectin solution. Seal the channels with tape and incubate the chip overnight at 4 °C.
- Sterilize the microfluidic channel with 30 µL of 75% ethanol. Remove the ethanol and rinse twice with 30 µL of sterilized phosphate-buffered saline (PBS) using a pipette tip. Thoroughly aspirate PBS in the channels.
- Seeding HPMCs into the microfluidic channels
- Culture HPMCs in 10% fetal bovine serum (FBS) culture medium (M199:MCDB105 supplemented with 10% FBS and 100 U/mL penicillin/streptomycin) under 5% CO2 at 37 °C until 80% confluence.
- Rinse the HPMCs with 3 mL of PBS and trypsinize with 2 mL of 0.05% trypsin/0.01% EDTA (TE) solution for 30 s. Neutralize with 4 mL of 10% FBS culture medium and spin down the cells at 1,000 x g for 5 min.
- Resuspend the HPMCs in 2 mL of 10% FBS culture medium. Count the cell number with a hemocytometer and adjust the cell concentration to 3.5×106/mL.
- Warm the fibronectin-coated microfluidic chip in a 37 °C incubator for 5 min before use. Completely aspirate the fibronectin solution.
- Slowly pipette 30 µL of HPMC suspension into each channel. Seal the channels with tape and place the devices in the CO2 incubator overnight.
3. Ovarian Cancer Spheroid Formation and Co-culture
- Ovarian cancer spheroid formation
- Maintain the human epithelial ovarian cancer cell line SKOV-3 in 5% FBS culture medium (M199:MCDB105 with 5% FBS and 100 U/mL penicillin/streptomycin) under 5% CO2 at 37 °C before the experiments.
- Dissolve 0.5% agarose in sterilized water by boiling. Dispense 50 µL of agarose solution into each well of 96-well plates. Leave the plates in the hood for 20 min to allow the agarose to solidify; solidified agarose coating the wells can prevent cell adhesion and indicates that the plates are ready for use.
- Rinse the SKOV-3 cells with 3 mL of PBS. Trypsinize with 2 mL of TE solution for 3 min. Neutralize with 4 mL of 5% FBS culture medium and spin down the cells at 1,000 x g for 5 min.
- Count the SKOV-3 cell number with a hemocytometer and resuspend the cells in 5% FBS culture medium at a density of 5,000 cells/mL. Transfer 200 µL of cell suspension into each agarose-coated well and culture under 5% CO2 at 37 °C for 48 h.
- Cancer spheroid loading
- Transfer cancer spheroids to a 50-mL centrifuge tube pre-wetted with 1 mL of serum-free medium and spin down at 750 x g for 5 min.
NOTE: Pre-wetted pipette tips and centrifuge tubes can prevent undesired spheroid attachment onto the centrifuge tubes. - Prepare 2.5 µg/mL of 5-chloromethylfluorescein diacetate (CMFDA) solution in serum-free M199:MCDB105 medium. Resuspend cancer spheroids in 1 mL of CMFDA solution and incubate at 37 °C for 30 min. Centrifuge at 750 x g for 5 min.
- Rinse the cancer spheroids with 2 mL of serum-free medium and centrifuge at 750 x g for 5 min. Repeat this step twice to remove the remaining fluorescent dye from the medium.
NOTE: The fluorescent signal can be retained in spheroids for more than 96 h. - Resuspend the cancer spheroids in serum-free M199:MCDB105 medium and count the spheroid number with a hemocytometer. Adjust the concentration to 2,000 spheroids/mL with serum-free medium.
- Prior to spheroid loading, slowly inject 100 µL of serum-free M199:MCDB105 medium to wash away the non-adhered mesothelial cells inside the channels.
NOTE: A fast injection, aspiration, or bubble formation will lead to the detachment of the mesothelial monolayer. - Load 30 µL of fluorescent-labeled spheroid suspension into each channel. Place the microfluidic chip into the CO2 incubator.
- Transfer cancer spheroids to a 50-mL centrifuge tube pre-wetted with 1 mL of serum-free medium and spin down at 750 x g for 5 min.
- Perfusion platform assembly and co-culture
- Attach a syringe loaded with fresh, serum-free M199:MCDB105 medium to a 1 m-long tube.
- Place the syringe on the syringe pump and secure the plunger and the body of the syringe. Install the syringe pump in a horizontal position at the same height as the microfluidic chip inside the incubator.
NOTE: The number and size of syringes depend on the number of channels used and the duration of the experiment. - Quickly infuse the whole tube with medium by pressing and holding the fast forward button until the medium overflows the tubing.
- Run the injection at the desired flow rate. Estimate the wall shear stress using the following equation:
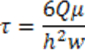
where τ represents the wall shear stress inside the chamber, Ǫ = flow rate, μ = viscosity (0.01 g cms-1); h = flow chamber height, and w = flow chamber width. - Connect the tubing to the inlet of the channel. Place the excess inlet tubing in the incubator to ensure that the medium is warmed before injected into the channels. To facilitate the connection between the tubing and the channel, appropriate connectors can be chosen. NOTE: Beware not to introduce any bubble to the syringe, tubing, and connections.
- Connect the outlet of the channel to a 50-mL conical tube to collect the outflow medium with a short output tube.
NOTE: The complete dynamic co-culture system is shown in Figure 1D.
4. Observation of HPMCs and Cancer Spheroids after Perfusion
- Observe the HPMCs and cancer spheroids with a bright-field or fluorescence microscope and take pictures after 1 h of perfusion.
Representative Results
Using this protocol, a microfluidic platform was set up to model ovarian cancer spheroids with mesothelial cells under hydrodynamic conditions. Primary human peritoneal mesothelial cells were cultured in the microdevice for 16 h and observed under a bright-field microscope. As shown in Figure 2A, the channel bottom was successfully covered with a monolayer of HPMCs. It is important to note that bubble formation during fibronectin or HPMC patterning will lead to channel coating failure. Through non-adherent suspension culture, multicellular spheroids with diameters of approximately 100 µm, which are similar in size to those found in patients' ascites, were generated and labeled with green fluorescent dye, CMFDA. The spheroids were then transferred to HPMC-coated microfluidic channels and remained in suspension. Cell morphologies were observed under the microscope and images were captured (Figure 2B). The fluorescent-labeled cancer spheroids can be easily differentiated from the mesothelial cells under a fluorescence microscope (Figure 2C).
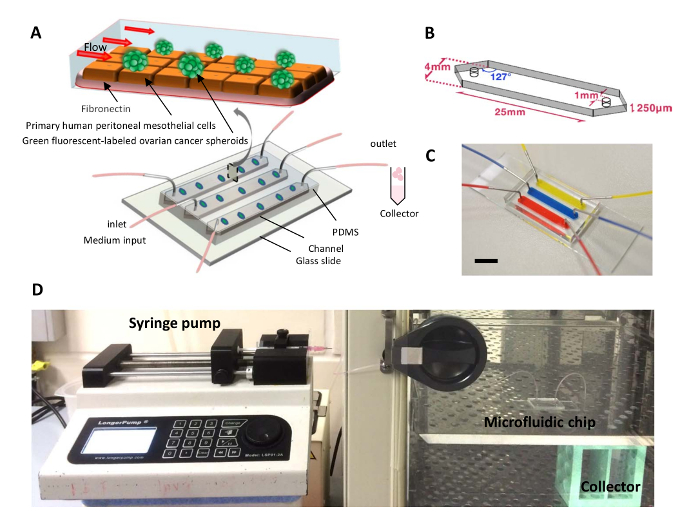
Figure 1. A Dynamic 3D Peritoneal Microdevice for Modeling Ovarian Cancer Multicellular Spheroid Behaviors. (A) Schematic diagram of a microfluidic chip used to co-culture ovarian cancer spheroids with HPMCs under flow condition. (B) Schematic diagram showing the design of the microfluidic channels. (C) Photograph of a microfluidic chip used in the protocol, with its channels infused with dye solution for clear visualization. Bar = 1 cm. (D) Photograph showing the experimental setup. Please click here to view a larger version of this figure.
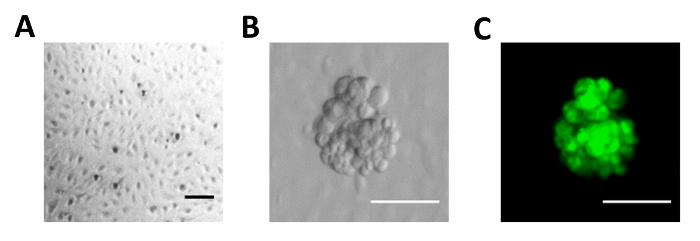
Figure 2. Images of the Ovarian Cancer Spheroid-mesothelial Cell Dynamic Co-culture Model. (A) Image of the HPMC monolayer grown in the microfluidic channel. (B) Images of ovarian cancer spheroids in the microfluidic channel coated with peritoneal mesothelial cells under phase contrast microscopy (left panel) or (C) fluorescence microscopy (Green, CMFDA; right panel). Bar = 100 µm. Please click here to view a larger version of this figure.
Discussion
This assay offers a flexible and physiologically relevant model that can be incorporated with various biochemical and cell-based assays, including, but not limited to, adhesion assays, mesothelial clearance assays, and drug screening. It can be applied to the evaluation of the effect of the intraperitoneal microenvironment on cancer progression. However, several experimental conditions might need to be optimized, depending on the aims of the project (e.g., the number of HPMCs and cancer spheroids seeded per channel, the co-culture time, etc.). Other cell types in the intraperitoneal microenvironment, such as fibroblasts, endothelial cells, or immune cells, can also be included in the system in order to study the essential interactions between ovarian cancer spheroids and nearby cells. Other ECM components, such as laminin, vitronectin, or collagen, can also be used. One weakness of this protocol that may need to be considered is that, while the coupling of nutrient replenishing and shear stress is reminiscent of that seen in vivo, there is no separate control of nutrient replenishing and shear stress in our current system. Moreover, techniques such as on-chip trypsinization combined with fluorescence-activated cell sorting will be needed to separately collect ovarian cancer cells and HPMCs for further molecular studies about different types of cells.
This technique exhibits several advantages when compared with traditional approaches. First, 3D co-culture better mimics the in vivo intercellular communication between ovarian cancer cells and mesothelial cells. Second, cells are cultured under fluidic flow but are not in static condition, generating a cellular response that is potentially representative of the in vivo situation12,13. Furthermore, the fluid flow can be precisely controlled, mimicking the hydrodynamic conditions at different stages of tumor progression.
In summary, the dynamic 3D ovarian cancer-mesothelium co-culture model presented here provides a flexible framework for modeling tumor behavior in the peritoneal cavity. With this model, the interaction between cancer cells and mesothelial cells under a dynamic hydrodynamic condition can be comprehensively analyzed. It can greatly advance our understanding of the underlying mechanisms of metastatic progression and might also facilitate therapeutic development.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by Hong Kong Research Grant Council (grants 17122014, C1013-15G, 719813E, and 17304514). A. S. T. Wong is a recipient of the Croucher Senior Research Fellowship.
Materials
| Silicon wafer | University wafer | #1196 | 100mm |
| SU-8 2075 photoresist | Microchem | ||
| SU-8 developer | Microchem | 108-65-6 | |
| Trichloro (1H, 1H, 2H, 2H-perfluorooctyl) silane | Sigma | 448931 | |
| Sylgard 184 | Dow Corning | 1673921 | Polydimethylsiloxane (PDMS) + curing agent kit |
| Biopsy punch | Miltex | 33-31AA | 1 mm diameter |
| Plasma cleaner | Harrick Plasma | PDC-002 | |
| Polyethylene tubing | SCI | BB31695-PE/5 | 0.86mm (inner diameter) |
| Syringe | Terumo | ||
| Syringe pump | Longer precision pump | LSP01-2A | |
| Medium 199 | Invitrogen | 31100-035 | Add 2.2g/L sodium bicarbonate |
| MCDB 105 Medium | Sigma | M6395 | |
| Fetal bovine serum (FBS) | Hyclone | SH30068.02 | |
| Penicillin/streptomycin | Invitrogen | 15070-063 | |
| Trypsin EDTA solution | Gibco | 25300-054 | 0.05% Trypsin -0.01% EDTA, phenol red |
| Fibronectin human | BD | 354008 | |
| Agarose | Invitrogen | 15510-027 | |
| 5-chloromethylfluorescein diacetate | Life technologies | C7025 | Green CMFDA |
| CO2 incubator | SANYO | MCO-18AIC | |
| Centrifuge | Hitachi | CT15RE | |
| Fluorescent microscope | Nikon | Model: 80i or ECLIPSE Ti; software: SPOT | |
| SKOV-3 | Gift from Dr. N Auersperg (University of British Columbia) |
References
- Jemal, A., et al. Global cancer statistics. CA: Cancer J. Clin. 61, 69-90 (2011).
- Burleson, K. M., et al. Ovarian carcinoma ascites spheroids adhere to extracellular matrix components and mesothelial cell monolayers. Gynecol. Oncol. 93, 170-181 (2004).
- Chau, W. K., Ip, C. K., Mak, A. S., Lai, H. C., Wong, A. S. c-Kit mediates chemoresistance and tumor-initiating capacity of ovarian cancer cells through activation of Wnt/beta-catenin-ATP-binding cassette G2 signaling. Oncogene. 32, 2767-2781 (2013).
- Zhang, S., et al. Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res. 68, 4311-4320 (2008).
- Tang, M. K. S., Zhou, H. Y., Yam, J. W. P., Wong, A. S. T. c-Met overexpression contributes to the acquired apoptotic resistance of nonadherent ovarian cancer cells through a cross talk mediated by phosphatidylinositol 3-kinase and extracellular signal-regulated kinase 1/2. Neoplasia. 12, 128-144 (2010).
- Ksiazek, K., et al. Senescent peritoneal mesothelial cells promote ovarian cancer cell adhesion: the role of oxidative stress-induced fibronectin. Am. J. Pathol. 174, 1230-1240 (2009).
- Hafter, R., Klaubert, W., Gollwitzer, R., Vonhugo, R., Graeff, H. Crosslinked Fibrin Derivatives and Fibronectin in Ascitic Fluid from Patients with Ovarian-Cancer Compared to Ascitic Fluid in Liver-Cirrhosis. Thromb Res. 35, 53-64 (1984).
- Kenny, H. A., et al. Mesothelial cells promote early ovarian cancer metastasis through fibronectin secretion. J. Clin. Invest. 124, 4614-4628 (2014).
- Jain, R. K. Normalization of tumor vasculature: An emerging concept in antiangiogenic therapy. Science. 307, 58-62 (2005).
- Chang, S. F., et al. Tumor cell cycle arrest induced by shear stress: Roles of integrins and Smad. Proc. Natl. Acad. Sci. USA. 105, 3927-3932 (2008).
- Rutkowski, J. M., Swartz, M. A. A driving force for change: interstitial flow as a morphoregulator. Trends Cell Biol. 17, 44-50 (2007).
- Rizvi, I., et al. Flow induces epithelial-mesenchymal transition, cellular heterogeneity and biomarker modulation in 3D ovarian cancer nodules. Proc. Natl. Acad. Sci. USA. 110, E1974-E1983 (2013).
- Ip, C. K., et al. Stemness and chemoresistance in epithelial ovarian carcinoma cells under shear stress. Sci. Rep. 6, 26788 (2016).
- Avraham-Chakim, L., et al. Fluid-flow induced wall shear stress and epithelial ovarian cancer peritoneal spreading. PloS one. 8, e60965 (2013).
- Burkhalter, R. J., et al. Peritoneal mechanobiology and metastatic success in epithelial ovarian cancer. Faseb Journal. 26, (2012).
- Lane, W. O., et al. Parallel-plate flow chamber and continuous flow circuit to evaluate endothelial progenitor cells under laminar flow shear stress. Journal of visualized experiments : JoVE. , (2012).
- Botta, G. P., Manley, P., Miller, S., Lelkes, P. I. Real-time assessment of three-dimensional cell aggregation in rotating wall vessel bioreactors in vitro. Nat. Protoc. 1, 2116-2127 (2006).
- Ismadi, M. Z., et al. Flow characterization of a spinner flask for induced pluripotent stem cell culture application. PloS one. 9, e106493 (2014).
- Yu, W., et al. A microfluidic-based multi-shear device for investigating the effects of low fluid-induced stresses on osteoblasts. PloS one. 9. 9, e89966 (2014).

