A Rabbit Aortic Valve Stenosis Model Induced by Direct Balloon Injury
Summary
An appropriate animal model is needed to understand the pathologic mechanisms underlying aortic valve stenosis (AVS) and to evaluate the efficacy of therapeutic interventions. The present protocol describes a new procedure for developing the AVS rabbit model via a direct balloon injury in vivo.
Abstract
Animal models are emerging as an important tool to understand the pathologic mechanisms underlying aortic valve stenosis (AVS) because of the lack of access to reliable sources of diseased human aortic valves. Among the various animal models, AVS rabbit models are one of the most commonly used in large animal studies. However, traditional AVS rabbit models require a long-term period of dietary supplementation and genetic manipulation to induce significant stenosis in the aortic valve, limiting their use in experimental studies. To address these limitations, a new AVS rabbit model is proposed, in which stenosis is induced by a direct balloon injury to the aortic valve. The present protocol describes a successful technique for inducing AVS in New Zealand white (NZW) rabbits, with step-by-step procedures for the preparation, the surgical procedure, and the post-operative care. This simple and reproducible model offers a promising approach for studying the initiation and progression of AVS and provides a valuable tool for investigating the underlying pathological mechanisms of the disease.
Introduction
It is increasingly recognized that the use of appropriate animal models can contribute to a better understanding of the pathological mechanisms underlying aortic valve stenosis (AVS) due to the lack of access to reliable sources of diseased human aortic valves associated with the progression of aortic stenosis (AS). Among the various animal models for studying AVS, rabbits are one of the most commonly used large-animal AVS models, and the AVS rabbit model is induced either through cholesterol/vitamin D2 supplementation or genetic manipulation1,2,3,4.
Although rabbit AVS models have provided significant insight into the development and progression of AVS, it still remains challenging to induce AVS consistently and reproducibly, as seen in our preliminary experiments.
In addition to diet-induced and genetically susceptible animal models, a new model of AVS has been established through direct mechanical injury in mice5,6. The mechanical injury model successfully induces aortic stenosis and represents a simple and reproducible AVS model in wild-type mice. To the best of our knowledge, there have been no prior studies examining the effects of a mechanical injury on the aortic valve in rabbit models. Thus, this study provides a new procedure for inducing AVS in male New Zealand white rabbits through a direct balloon injury to the aortic valve, which can accurately mimic the condition of valvular aortic stenosis. This protocol includes step-by-step descriptions of the preparation, the surgical procedure, and the post-operative care, which are useful for inducing reproducible AVS rabbit models.
Protocol
All animal research procedures were approved and performed in accordance with the Laboratory Animals Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Guidelines and Policies for Animal Experiments provided by the Institutional Animal Care and Use Committee (IACUC) at the College of Medicine of The Catholic University of Korea (approval number: CUMC-2021-0176-05). The present study utilized 3 month old male New Zealand white (NZW) rabbits weighing 3.5-4.0 kg, which were maintained under standard conditions in individual cages. The rabbits were fed either a normal diet or a 0.5% cholesterol-enriched diet supplemented with 50,000 U of vitamin D2 (see Table of Materials). The experimental design and analysis methods for the induction of the AVS rabbit model are depicted in Figure 1.
1. Preparation for the operation
- Ensure that all the medical and surgical instruments (see Table of Materials) are sterilized at the beginning of the operation.
- Prepare the dilation balloon catheter set following the steps below.
- Connect the in-deflation device filled with a mixture of saline and commercially available contrast medium (1:1) to the luer lock part of the balloon catheter (see Table of Materials).
- Fill the balloon with inflation solution, and remove any air from the balloon catheter.
NOTE: For the present study, the inflation solution consisted of 30% iodixanol with 0.9% saline (see Table of Materials). - Verify proper balloon inflation by purging the balloon lumen with the inflation solution.
2. Surgical procedure for the aortic valve injury
- Administer an intramuscular injection of tiletamine & zolazepam (15 mg/kg) and xylazine (5 mg/kg) (see Table of Materials) to anesthetize the animal.
NOTE: Before administering the anesthesia, the rabbits were pre-treated with a subcutaneous glycopyrrolate injection (0.05 mg/kg) as a pre-anesthetic anticholinergic agent. The adequate anesthesia level was determined by a combination of criteria, including a lack of response to a toe pinch and a steady respiratory rate. - Insert a 24 G intravenous (IV) catheter into the marginal auricular vein, and connect an infusion set with heparinized saline (100 U/kg heparin).
- Connect the rabbit with a multiparameter veterinary monitor (see Table of Materials) to continuously monitor the vital signs, such as the oxygen saturation signal (SpO2), temperature, and blood pressure.
NOTE: For the SpO2 monitoring, attach the SpO2 sensor to the rabbit's tongue. For the temperature monitoring, insert the probe into the rabbit's rectum. For the blood pressure monitoring, place the cuff on the forelimb. - Place the rabbit in a supine position on an operating table equipped with a C-arm fluoroscopy (see Table of Materials), and remove the hair from the ventral neck area using animal hair clippers (Figure 2A).
- Sterilize the incision area with iodine, and cover the rabbit with surgical towels.
- Position the rabbit's heart at the center of the C-arm image.
NOTE: All researchers must wear protective gear with attached thermoluminescent dosimeters (TLDs) to reduce radiation exposure while performing the C-arm-guided surgery.- Turn on the C-arm, and select the fluoroscopic mode for cardiac imaging.
- Adjust the position of the rabbit to ensure that the heart is at the center of the imaging field.
- Make a longitudinal incision of approximately 3 cm in the skin of the neck, and use surgical scissors to cut the fascia and fat tissue.
- Expose the left common carotid artery (LCCA) by carefully separating the muscles until approximately 3-3.5 cm of the LCCA is exposed (Figure 2B).
- Ligate the LCCA with a 3-0 silk suture (see Table of Materials) at the top and end of the exposed LCCA to stop the blood flow.
- Insert a 22 G IV catheter into the LCCA, and introduce a guide wire (0.016 in, see Table of Materials) into the left ventricle (LV) through the IV catheter, ensuring that the tip of the catheter is properly positioned in the imaging field of the C-arm.
NOTE: When inserting the IV catheter, carefully loosen the ligature suture on the way down to the aortic valve to allow for the advancement of the catheter. - Withdraw the IV catheter, leaving the guide wire, and place a 4-F sheath (see Table of Materials) over the guide wire into the LCCA to introduce the balloon catheter (Figure 2C).
NOTE: After replacing the IV catheter with the sheath, any trapped air should be removed from the sheath device. - Carefully insert the 8 mm balloon catheter over the guidewire into the aortic valve under C-arm fluoroscopic guidance (Figure 2D).
- Place the balloon catheter tip approximately 1-2 cm distal to the aortic valve, and inflate the balloon by purging the inflation solution with a pressure inflator at 6 atm.
- Advance the balloon into the LV apex, and pull it back into the LV outlet. Repeat this procedure five times, and then deflate the balloon (Figure 2E, F).
- Repeat steps 2.8-2.9 three times to ensure adequate valve injury.
- Withdraw the balloon catheter and guidewire. Slowly remove the sheath from the LCCA, and immediately tie the LCCA with the suture on the way down to the aortic valve.
- Clean the incision area with saline to remove the blood clots, and inspect the punctured site for arterial bleeding.
- Close the muscle and skin with a 3-0 non-absorbable suture, and sterilize all the sides of the wound with iodine.
3. Post-operative care
- Remove the monitoring patches and clips, and keep the rabbit in an intensive care incubator.
NOTE: After surgery, the rabbits were closely observed for 1 day in an intensive care incubator and then moved to a home cage. - Manage post-operative pain with 5 mg/kg tramadol and 3 mg/kg ketoprofen and administer antibiotics (4 mg/kg gentamicin) twice daily for 3 days via subcutaneous injection (see Table of Materials).
NOTE: Post-operative pain management should adhere to veterinary and IACUC guidelines (e.g., opioid, NSAID, local anesthetic or combination). - Feed a 0.5% cholesterol-enriched diet with 50,000 U of vitamin D2 (HC + VitD2) for 8 weeks.
4. Echocardiography
- After 8 weeks of balloon injury, anesthetize the rabbit using the same procedure described in step 2.1.
- Visualize the aortic valves using two-dimensional transthoracic views, and record M-mode images in short-axis and long-axis views.
- Place the rabbit in a supine position on an echo table.
- Shave the chest area using clippers and hair removal cream.
- Apply ultrasound transducer gel (see Table of Materials) to the chest.
- Adjust the transducer to obtain the parasternal long-axis view and the parasternal short-axis view of the aortic valve.
- Use M-mode imaging to record images of the aortic valve in both long-axis and short-axis views, and save the images for later analysis.
5. Histological analysis
- After the echocardiography, euthanize the rabbit by administering an intravenous injection of potassium chloride (KCl, 3 g/20 mL, 1 mL).
- Open the thoracic cavity, harvest the heart with the ascending aorta7, and place it on ice in phosphate-buffered saline (PBS).
- Immediately immerse the heart in a 4% paraformaldehyde (PFA) solution, and embed it in a paraffin block (see Table of Materials).
- Cut the paraffin-embedded heart block into 4 µm thick sections using a microtome, and stain the sections with Masson's trichrome (MT), Alizarin Red, and von Kossa (see Table of Materials) to assess the collagen deposition and valve calcification, respectively8,9.
Representative Results
Rabbit AVS model induced by aortic valve injury
To induce the rabbit AVS model, male NZW rabbits weighing 3.5-4.0 kg were used for this study. According to the surgical procedures described in step 2 (Figure 2), the AVS model was established by aortic valve injury, which resulted in mechanical aortic valve degeneration and calcification. The control group included rabbits fed with a 0.5% cholesterol-enriched diet (high-cholesterol, HC) and 50,000 U of vitamin D2 (VitD2), which is known as the diet-induced AVS model.
Assessment of the aortic valve
To assess structural changes in the aortic valve, the leaflet mobility and thickness were evaluated using echocardiographic short-axis and long-axis views. At 8 weeks after the aortic valve injury, the echocardiography revealed that the cusps were thickened and the motion was restricted in the injured rabbits fed with the HC + VitD2 diet compared to the control rabbits, including the wild-type (WT) rabbits and the rabbits fed the HC + VitD2 diet without valve injury (Figure 3).
Histological analysis
To evaluate the histological changes in the aortic valve, the rabbits were sacrificed at 8 weeks after the aortic valve injury, and a histological analysis was performed with the excised hearts (Figure 4). As shown in Figure 4A, the aortic valve stained with Masson's trichrome (MT) showed increased thickness of the aortic valve cusps in the injured group compared to the WT and HC + VitD2 diet-induced groups. Additionally, to compare the degree of valvular calcium deposits, Alizarin Red staining and von Kossa staining were performed, as shown in Figure 4B,C. While the HC + VitD2 diet-induced group exhibited negligible calcium deposits in the valvular leaflets, significant calcific deposits were observed in the balloon-injured group.
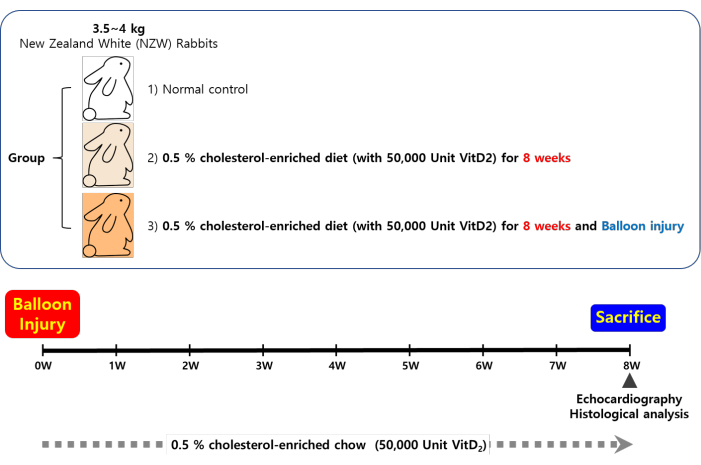
Figure 1: Scheme of the experimental timeline. A rabbit model of aortic valve stenosis was established by direct balloon injury on the aortic valve in male New Zealand white (NZW) rabbits (3.5-4.0 kg), followed by a high-cholesterol/vitamin D2 diet (0.5% cholesterol-enriched diet + 50,000 U of vitamin D2; HC + VitD2) for 8 weeks. Please click here to view a larger version of this figure.
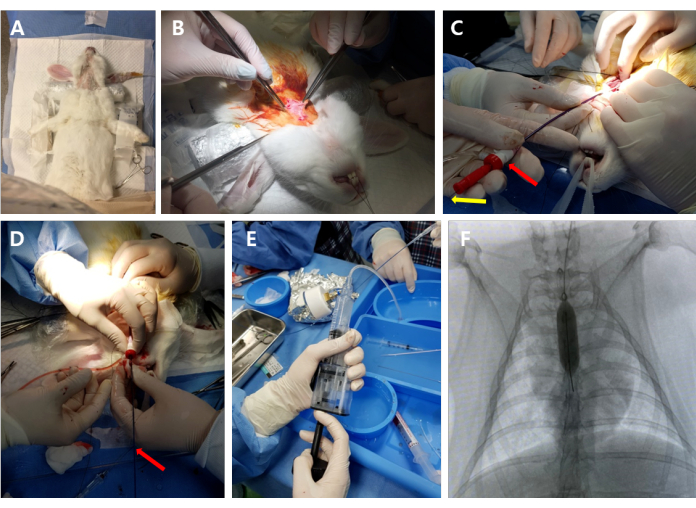
Figure 2: Outline of the operative procedure. (A) Under anesthesia, the rabbit was placed in a supine position on the operating table. (B) The left common carotid artery (LCCA) was exposed by carefully separating the skin and muscles. (C) The 4-F sheath and guide wire were inserted into the LCCA. Red arrow: sheath; yellow arrow: guide wire. (D) The balloon catheter was introduced over the guide wire into the aortic valve. Red arrow: balloon catheter. (E,F) The balloon catheter was inflated and advanced/pulled back between the left ventricular apex and outlet under C-arm fluoroscopic guidance. Please click here to view a larger version of this figure.
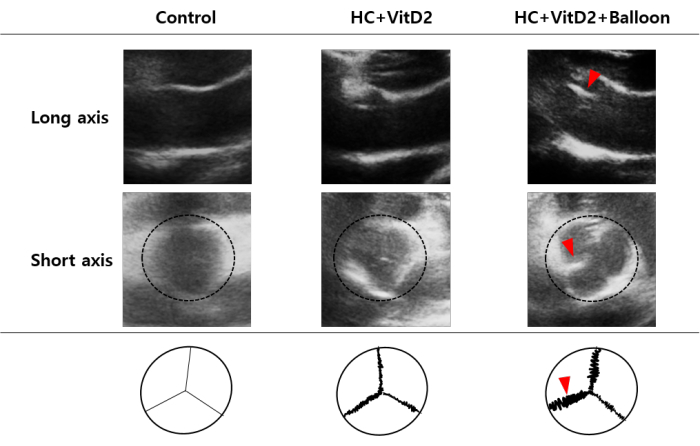
Figure 3: Echocardiographic analysis of the aortic valve stenosis. Representative images of the long-axis (upper panels) and short-axis (middle panels) views in the echocardiogram and a schematic diagram of the degree of valvular stenosis (lower panels) in the WT (n = 3), HC + VitD2-diet (n = 3), and HC + VitD2 diet with valve injury (n = 3) groups. Dotted circle: aortic valve; red arrowhead: thickened leaflets. Please click here to view a larger version of this figure.
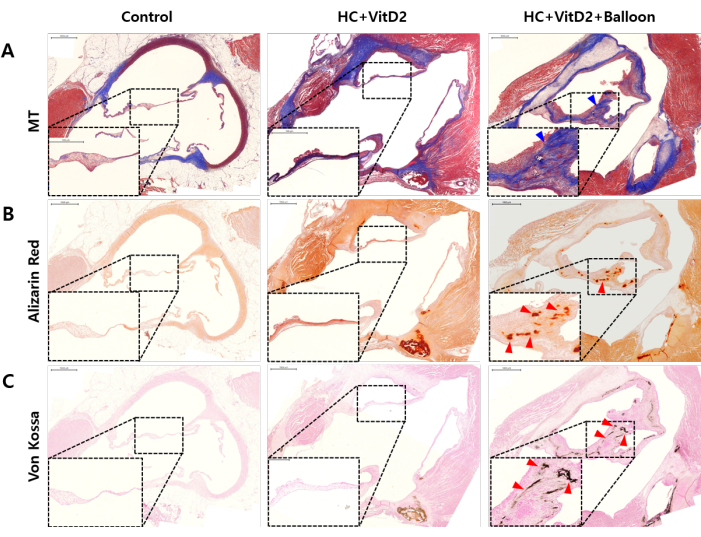
Figure 4: Histological analysis of the aortic valves. Representative images of (A) Masson's trichrome, (B) Alizarin Red, and (C) von Kossa staining in the WT, HC + VitD2-diet, and HC + VitD2-diet with valve injury groups. Blue arrowhead: thickened leaflets; red arrowhead: calcified leaflets. Scale bars = 1 mm. Please click here to view a larger version of this figure.
Discussion
Animal AVS models are commonly used to study the pathological aspects of AVS, including the initiation and progression of AVS. This protocol introduces a new rabbit AVS model induced by a direct balloon injury to the aortic valve. In this study, the aortic valve injury model showed significant leaflet thickening and calcification. Compared to the mild AVS model induced by dietary supplementation, the aortic valve in the direct balloon injury model was selectively injured, leading to thickened cusps and restricted motion, as well as thickened and calcified leaflets. These results are consistent with the general characteristics of AVS10,11.
The commonly used AVS rabbit models induced by dietary supplementation and genetic manipulation have several limitations in experimental studies12,13,14. The development of significant stenosis in rabbit models often requires a longer feeding period than in mice, which can cause significant inflammation and hepatic toxicity. Additionally, dietary supplementation, such as with hypercholesterolemic diets and VitD2, in these models does not always induce consistent and significant valvular stenosis. In comparison, the direct balloon injury described in this protocol can cause mechanical damage to the operated aortic valve leaflets, thereby specifically inducing a reproducible remodeling response. Moreover, this protocol allows for the manipulation of the severity of AVS by adjusting the injury intensity. To the best of our knowledge, this is the first time the effect of mechanical injury on the aortic valve in rabbit models has been validated in vivo.
Despite these advantages, this protocol has limitations in inducing consistent and reproducible AVS models. Firstly, the surgical procedure requires a lot of surgical experience with animal models. Secondly, it is necessary to establish detailed conditions for optimizing the AVS severity, such as in terms of the injury intensity and the duration of dietary supplementation. Thirdly, this protocol is limited in its ability to provide information on the effects of the balloon injury alone on aortic valve stenosis, as this study only investigated the effects of balloon injury in combination with a cholesterol-enriched diet. Including a group receiving the balloon injury without a cholesterol-enriched diet would be informative, and we will consider it for future studies. Nevertheless, this work demonstrates a new protocol for direct balloon injury on the aortic valve in the rabbit model, which is useful for studying the pathological mechanisms underlying AVS and can potentially be used for developing therapeutic options.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2020R1A4A3079570), the Ministry of Education (No. 2021R1I1A1A01051425), and the Industrial Strategic Technology Development Program (No. 20014873) funded by the Ministry of Trade, Industry & Energy, Republic of Korea.
Materials
| 3-0 Silk suture | AILEE | SK312 | |
| 4% paraformaldehyde(PFA) | Intron | IBS-BP031-2 | |
| Alizarin red Solution | Millpore | TMS-008-C | |
| ASAHI SION BLUE | ASAHI | Guide wire | |
| Back Table Cover | Yuhan kimberly | 80101-30 | |
| Balloon In-deflation Device | Demax Medical | DID30s | |
| Bionet Veterinary monitor | BIONET | BM3 VET | |
| C-Arm | SIEMENS Healthcare GmbH | Cios alpha | |
| Certified Rabbit Diet | Purina | 5322 | 4.7% Hydrogenated Coconut Oil, 0.5% Cholesterol, & 1% Molasse |
| Curadle Smart Incubator | Autoelex | CS-CV206 | Intensive Care Unit (ICU) |
| Ergocalciferol | Sigma-aldrich | E5750 | Vitamin D2 |
| Fechtner conjunctiva forceps titanium | WORLD PRECISSION Instrument | WP1820 | |
| Forceps | HEBU | HB203 | |
| Gentamicin | Shin Poong | ||
| Glycopyrrolate | SamChunDang | ||
| Greenflex NS | DAI HAN PHARM | Normal saline 500 mL | |
| Hematoxylin solution | Sigma-aldrich | HT1079-1 SET | |
| Heparin | JW pharmaceutical | 25,000 U | |
| Infusion set for single use | SWOON MEDICAL | ||
| Iodine | Green pharmaceutical | ||
| Iodixanol | GE Healthcare | Visipaque | Inflation solution (contrast agent) |
| IV catheter 22 G | BD | 382423 | |
| IV catheter 24 G | BD | 382412 | |
| Ketoprofen | SamChunDang | ||
| Luer-Lok syringe 10 mL | Becton Dickinson Medical | ||
| Luer-Lok syringe 3 mL | Becton Dickinson Medical | ||
| Microscope | OLYMPUS | SZ61 | |
| Microtome | ThermoFisher Scientific | HM 325 | |
| MT stain kit | Sigma-aldrich | HT15-1kt | |
| Needel holder | Solco | 009-1304 | |
| Needle Holder with Lock and Suture | JEUNGDO BIO & PLANT | H-1222-18 | |
| Paraffin | LK LABKOREA | H06-660-107 | |
| PBS | Gibco | 10010-023 | |
| Potassium chloride 40 | Daihan Pharm | KCl | |
| Prelude Ideal Hydrophilic Sheath | MERIT MEDICAL | PID4F11018SS | Sheath 4F |
| PTA Balloon Dilatation catheter | Boston Scientific | H749-3903280208-0 | Balloon catheter 8.0 mm |
| Rompun | Elanco | Xylaxine | |
| sterile Gauze | DAE HAN Medical | 10 cm x 20 cm | |
| Surgical Gloves | Ansell | Ansell | |
| Surgical Gown | Yuhan kimberly | 90002-02 | |
| Surgical Scissors | Nopa, Germany | AC020/16 | |
| Surgical Tape | 3M micopore | 1530-1 | |
| Syringe 1 mL | Shin Chang Medical | ||
| Syringe 10 mL | Shin Chang Medical | ||
| Tissue cassette | Scilav korea | Cas3003 | |
| Transducer gel | SUNGHEUNG | SH102 | |
| Tridol | Yuhan Corp. | Tramadol HCl | |
| Ultrasound system | Philps | Affiniti 50 | |
| Von Kossa stain kit | Abcam | ab105689 | |
| Zoletil 50 | Virbac korea | Tiletamine & zolazepam |
References
- Aliev, G., Burnstock, G. Watanabe rabbits with heritable hypercholesterolaemia: A model of atherosclerosis. Histology and Histopathology. 13 (3), 797-817 (1998).
- Cimini, M., Boughner, D. R., Ronald, J. A., Aldington, L., Rogers, K. A. Development of aortic valve sclerosis in a rabbit model of atherosclerosis: An immunohistochemical and histological study. Journal of Heart Valve Disease. 14 (3), 365-375 (2005).
- Drolet, M. C., Couet, J., Arsenault, M. Development of aortic valve sclerosis or stenosis in rabbits: role of cholesterol and calcium. Journal of Heart Valve Disease. 17 (4), 381-387 (2008).
- Sider, K. L., Blaser, M. C., Simmons, C. A. Animal models of calcific aortic valve disease. International Journal of Inflammation. 2011, 364310 (2011).
- Honda, S., et al. A novel mouse model of aortic valve stenosis induced by direct wire injury. Arteriosclerosis, Thrombosis, and Vascular Biology. 34 (2), 270-278 (2014).
- Niepmann, S. T., et al. Graded murine wire-induced aortic valve stenosis model mimics human functional and morphological disease phenotype. Clinical Research in Cardiology. 108 (8), 847-856 (2019).
- Robbins, N., Thompson, A., Mann, A., Blomkalns, A. L. Isolation and excision of murine aorta; A versatile technique in the study of cardiovascular disease. Journal of Visualized Experiments. (93), e52172 (2014).
- Wirrig, E. E., Gomez, M. V., Hinton, R. B., Yutzey, K. E. COX2 inhibition reduces aortic valve calcification in vivo. Arteriosclerosis, Thrombosis, and Vascular Biology. 35 (4), 938-947 (2015).
- Jung, S. H., et al. Spatiotemporal dynamics of macrophage heterogeneity and a potential function of Trem2(hi) macrophages in infarcted hearts. Nature Communications. 13 (1), 4580 (2022).
- Freeman, R. V., Otto, C. M. Spectrum of calcific aortic valve disease: Pathogenesis, disease progression, and treatment strategies. Circulation. 111 (24), 3316-3326 (2005).
- Lindman, B. R., et al. Calcific aortic stenosis. Nature Reviews Disease Primers. 2, 16006 (2016).
- Cuniberti, L. A., et al. Development of mild aortic valve stenosis in a rabbit model of hypertension. Journal of the American College of Cardiology. 47 (11), 2303-2309 (2006).
- Marechaux, S., et al. Identification of tissue factor in experimental aortic valve sclerosis. Cardiovascular Pathology. 18 (2), 67-76 (2009).
- Hara, T., et al. Progression of calcific aortic valve sclerosis in WHHLMI rabbits. Atherosclerosis. 273, 8-14 (2018).

