Techniques of Laparoscopic Right Posterior Sectionectomy: Glissonian Approach and a Parenchymal Transection Technique
Summary
Here, we present a protocol to perform laparoscopic right posterior sectionectomy, focusing on two key aspects: the intrahepatic Glissonian approach for inflow control and a parenchymal transection technique using an ultrasonic surgical aspirator.
Abstract
Laparoscopic liver resections (LLR) have been widely accepted as a treatment option for liver tumors. They offer several advantages over open liver resections, including less blood loss, reduced wound pain, and shorter hospital stays with a comparable oncological outcome. However, laparoscopic resection of lesions in the right posterior section of the liver is challenging due to difficulties in bleeding control and visualizing the surgical field. In the past, laparoscopic right posterior sectionectomy (LRPS) was still in the exploration phase, with undefined risks in the Second International Consensus Conference on LLR in 2014. However, recent technological advancements and increased surgical experience have shown that LRPS can be safe and feasible. It has been found to reduce hospital stay and blood loss compared to open surgery. This manuscript aims to provide a detailed description of the steps involved in LRPS. The key factors contributing to our success in this challenging procedure include proper liver retraction and exposure, the use of an intrahepatic Glissonian approach for inflow control, a technique called the ‘ultrasonic scalpel mimic Cavitron ultrasonic surgical aspirator (CUSA)’ for parenchymal transection, early identification of the right hepatic vein, and meticulous bleeding control using bipolar diathermy.
Introduction
Laparoscopic liver surgery has been shown to have several advantages over open liver resection, including reduced blood loss, decreased wound pain, and shorter hospital stay while maintaining comparable oncological outcomes1,2,3,4. Although LRPS was previously considered a relative contraindication due to its deep-seated anatomic location and challenges in bleeding control, recent advancements have proven its safety and feasibility2,5,6,7. Various strategies and surgical devices have been developed to overcome these challenges. However, no consensus exists on the best surgical technique and devices for LRPS.
In this article, we aim to provide a detailed description of the steps involved in LRPS at our centre, with a specific focus on the intrahepatic Glissonian approach for inflow control, a novel technique called the 'ultrasonic scalpel mimic Cavitron ultrasonic surgical aspirator (CUSA)' for parenchymal transection, early identification of the right hepatic vein, and meticulous bleeding control using bipolar diathermy. Machado et al. and Topal et al. reported the feasibility and safety of the laparoscopic intrahepatic Glissonian approach8,9,10. Laparoscopic techniques offer improved visualization and precision in dissecting the Glissonian pedicle. The approach maintains the blood flow to the remnant liver, reducing the risk of ischaemic injury. Additionally, this approach allows for the exact demarcation of liver segments that are being resected, making the surgery more precise and reducing the risk of bleeding. Professor Kwon introduced the 'ultrasonic scalpel mimic CUSA' technique in 201911, which has been shown to reduce intraoperative blood loss and operative time11,12.
A representative case is discussed in this study to detail the steps performed in the protocol. The patient is a 54-year-old man who is a chronic carrier of hepatitis B. During screening ultrasonography, a liver mass was identified in segment 6. A pre-operative triphasic computed tomography (CT) scan was performed, which revealed a 5.7 cm hypervascular tumor with arterial enhancement and portovenous washout in segment 7 (Figure 1). The alpha-fetoprotein (AFP) level was 2 ng/ml. The Child-Pugh score was 5 (Grade A). The indocyanine green retention at 15 min was 7.5%. The residual liver volume (RLV) was 45%. Based on the patient's hepatitis status and the radiological features of the tumor, the mass was treated as hepatocellular carcinoma. The patient was offered a laparoscopic right posterior sectionectomy, including the right hepatic vein.
Protocol
The protocol follows the guidelines of Kwong Wah Hospital's human research ethics committee.
1. Pre-operative workup
- Check triphasic CT scans to evaluate the extent of disease and vascular anatomy.
- Perform an indocyanine green (ICG) retention test to assess liver function properly. Preferably, ICG retention of less than 15% is required for patients to undergo major liver resection13.
- Perform CT volumetry to ensure adequate residual liver volume (RLV). 40% RLV is required for cirrhotic liver, while 30% RLV is necessary for the normal liver14.
2. Anesthesia
- Administer pre-operative antibiotics, typically 1.2 g of amoxicillin and clavulanic acid, intravenously on induction of anesthesia.
- Perform deep venous thrombosis prophylaxis with a sequential compression device and compression stockings15.
- Place an arterial line and a central venous line.
- Control low central venous pressure to 3-8 mmHg16. An anesthetist performs this.
3. Patient positioning
- Position the patient in the Lloyd-Davis position on a split-leg table.
- Set the theatre as depicted in Figure 2, allowing the surgeon to stand between the patient's legs.
NOTE: This position allows the surgeon to operate while standing between the legs, providing ergonomic benefits during such a lengthy procedure and preventing early fatigue. - Elevate the patient to 30° left lateral position with a pillow during the operation.
4. Port site insertion and the laparoscope
- Establish access into the intraabdominal cavity with a sub-umbilical incision by open Hasson Technique17.
- Place the remaining ports as depicted in Figure 3.
- Place a 12 mm port in the right upper quadrant along the midclavicular line.
- Place 5 mm laparoscopic assistant port at epigastrium, right flank along the anterior axillary line, and left upper quadrant.
NOTE: A laparoscope with a flexible tip is preferred for a better view above the dome of the liver (Figure 4).
5. Operative steps
- Mobilization of the whole right lobe of the liver
- Expose the root of the right hepatic vein superiorly.
- Complete division of triangular ligament until bare area exposed.
- Expose the groove between the right hepatic veins and the common trunk of the middle and left hepatic veins.
- Expose the root of IVC inferiorly.
- Incise inferior peritoneal attachment and drop the adrenal gland to the retroperitoneum.
- Divide the short hepatic veins and vena cava ligament until the inferior border of the right hepatic vein is seen.
- Expose the root of the right hepatic vein superiorly.
- Right posterior pedicle control with intrahepatic Glissonian approach18.
- Dissect along the superior border of the right posterior Glissonian pedicle along the Rouviere sulcus while keeping the Laennec capsule intact.
- Transect the caudate process to expose the inferior margin of the right posterior Glissonian pedicle.
- Dissect along the inferior border of the right posterior Glissonian pedicle along the Rouviere sulcus while keeping the Laennec capsule intact.
- Control the right posterior pedicle temporarily with a bulldog clamp.
- Determination of the transection plane along the ischaemic line
- Perform Intraoperative ultrasonography (USG) to identify the course of the right hepatic vein19.
- Open the liver capsule with an ultrasonic scalpel (Table of Materials) at the dome of the liver along the left margin of the right hepatic vein to secure a correct transection plane.
- Mark the rest of the transection line along the ischaemic line with diathermy.
- Pringle maneuver
- Prepare intracorporeal tourniquet for laparoscopic Pringle maneuver by Huang's Loop using a shortened Foley catheter20.
- Parenchymal transection
- Open the liver capsule with an ultrasonic scalpel along the planned transection line.
- Perform subsequent deep parenchymal transection with the 'ultrasonic scalpel mimic CUSA' technique12.
- Keep both arms of the ultrasonic scalpel open.
- Use the active blade of the ultrasonic scalpel to dissect the tissue.
- Activate maximum and move the active blade in a horizontal swinging motion parallel to the vasculobiliary structure.
- Dissect the parenchyma while exposing the individual intrahepatic vessels and leaving them intact.
- Use a suction device with the left hand constantly to keep the surgical field dry.
- Divide small vessels with the ultrasonic scalpel.
- Divide large vessels or pedicle structures between plastic clips (Table of Materials) or metal clips.
- Right posterior pedicle transection
- Encircle the right posterior pedicle with vascular tape.
- Transect the right posterior pedicle with a vascular stapler.
- Right hepatic vein transection
- Transect the right hepatic vein with a vascular stapler.
- Bleeding control
- The right posterior pedicle is temporarily controlled as a form of partial Pringle's maneuver during parenchymal transection. Meticulous dissection and identification of intrahepatic vessels are critical. Divide portal and hepatic venules with clips or ultrasonic scalpel.
- Use bipolar diathermy for bleeding with simultaneous suctioning to clear the surgical field.
- Increase carbon dioxide pneumoperitoneum (15-20 mmHg) to slow down the bleeding from hepatic veins and use intracorporal suture for significant bleeding, for example, bleeding from the right hepatic vein.
- Use a suture to secure bleeding from large veins or arteries.
6. Specimen retrieval
- The specimen is put into a plastic bag and retrieved through the Pfannenstiel incision.
Representative Results
In the representative case, the total operative time was 738 min, with an estimated blood loss of 400 mL. The patient was nursed in the intensive care unit for 2 days. The recovery was uneventful, and the patient was discharged on postoperative day 5. Histopathological examination of the specimen revealed moderately differentiated cholangiocarcinoma measured 8.0 cm x 5.5 cm x 4.5 cm. There was no perineural or lymphovascular permeation. The resection margin was 14 mm. The American Joint Committee on Cancer (AJCC) staging (8th edition) was pT1bN0M0. He was regularly followed up after the operation. The follow-up protocol is a 3-monthly follow-up in the first 2 years and then 6-monthly subsequently. The follow-up included clinical examination, liver function test and serum alpha-fetoprotein, and 6-monthly chest X-ray and contrast-enhanced computed tomography. All these details are listed in Table 1. The patient's first postoperative imaging is depicted in Figure 1C. It has been 2 years and 7 months post operation with no signs of recurrence and a normal serum alpha-fetoprotein level.
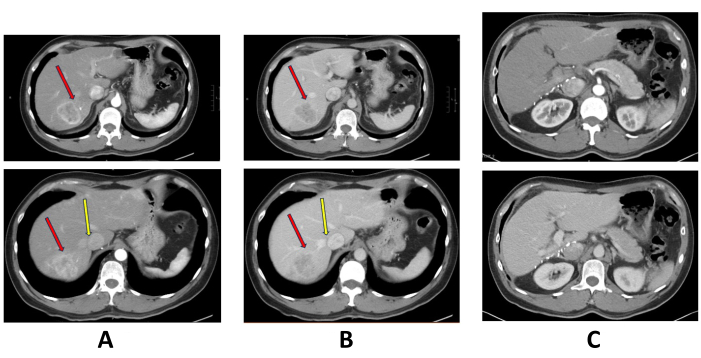
Figure 1: Pre-operative and postoperative triphasic computed tomography scan. (A) Arterial enhancing mass at segment 7 of the liver. The red arrow points at the tumor, and the yellow arrow points at the right hepatic vein. (B) The segment 7 tumor showed portovenous washout. The red arrow points at the tumor, and the yellow arrow points at the right hepatic vein. (C) Postoperative computed tomography scan. Please click here to view a larger version of this figure.
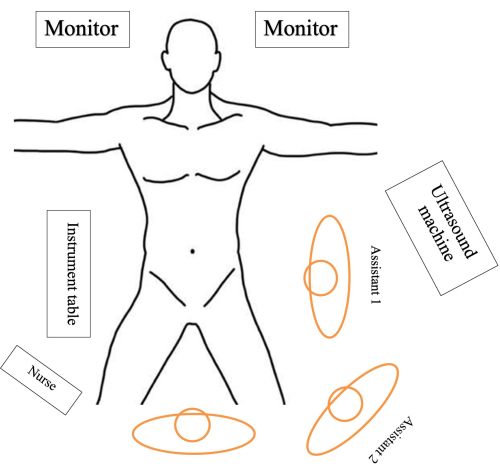
Figure 2. Patient positioning and theatre setup. The patient is placed in the Lloyd-Davis position on a split-leg table with the surgeon standing between the patient's leg. The first assistant stands on the patient's left side while the second assistant, responsible for holding the camera, stands between the surgeon and the first assistant. The instrumental table is placed on the right side while the scrub nurse standing next to the surgeon passes instruments to the surgeon. Please click here to view a larger version of this figure.
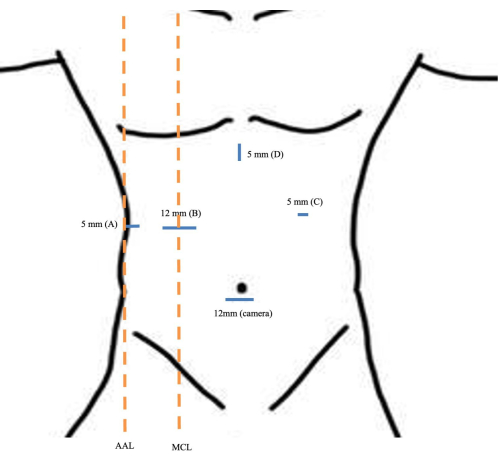
Figure 3. Port placement. 12 mm sub-umbilical port (camera port). (A) 5 mm port at the right flank along the anterior axillary line. (B) 12 mm port at the right upper quadrant along the mid-clavicular line. (C) 5 mm port at the left upper quadrant (liver retractor/assistant). (D) 5 mm port at epigastrium. Abbreviations: MCL = mid clavicular line; AAL = anterior axillary line. Please click here to view a larger version of this figure.
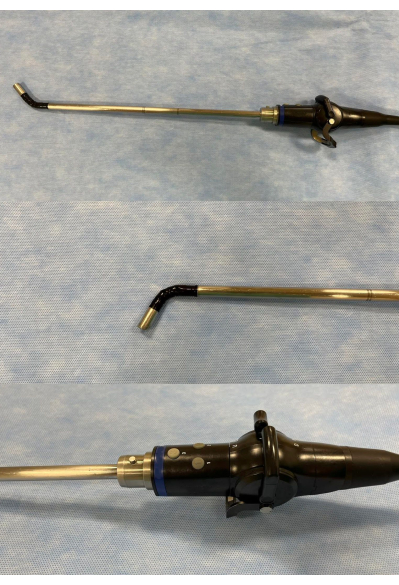
Figure 4. Flexible tip laparoscope. Its flexible tip (up-down and left-right) facilitates visualization of the anatomically obscured area, i.e., the dome. However, it requires a skillful and trained camera holder to operate this laparoscope21. Please click here to view a larger version of this figure.

Figure 5. Difference between the two models of the ultrasonic scalpel. This figure has been modified with permission from Welling et al.22. Please click here to view a larger version of this figure.
| Total Operative Time | 738 min | ||
| Estimated blood loss | 400 mL | ||
| Day of discharge | Post-operative day 5 | ||
| Histopathological examination | Moderately differentiated cholangiocarcinoma | ||
| Size of tumour: 8.0 cm x 5.5 cm x 4.5 cm | |||
| Resection margin: 14 mm | |||
| No perineural or lymphovascular permeation | |||
| (American Joint Committee on Cancer (AJCC) staging (8th edition): pT1bN0M0) | |||
| First post-operative CT | See Figure 1C | ||
| Follow-up | Total follow-up period: 2 years 7 months | ||
| Status: No recurrence, normal serum alpha-fetoprotein level | |||
| Follow up protocol | |||
| · 3-monthly follow-up in first two years, then 6-monthly subsequently | |||
| · Clinical examination, liver function test and serum alpha-fetoprotein level, chest X-ray and contrast-enhanced computed tomography 6-monthly | |||
Table 1: Surgical outcomes and post operative details of the patient.
Discussion
The critical components of the surgery include the intrahepatic Glissonian approach for inflow control, a 'ultrasonic scalpel mimic CUSA' parenchymal transection technique, early identification of right hepatic vein and meticulous bleeding control by bipolar diathermy.
The first critical step in this protocol is identifying and controlling the right posterior pedicle. The Glissonian approach was first introduced as extrahepatic Glissonian pedicle control in open liver surgery23 and has been associated with reduced blood loss and faster than intra-fascial approach24,25,26. With the advancement of laparoscopic liver surgery, the laparoscopic Glissonian approach has become a safe and effective alternative to traditional inflow control. Cho et al. demonstrated that the laparoscopic extrahepatic Glissonian approach is feasible and safe in hepatectomy5. Machado et al. and Topal et al. also reported the feasibility and safety of the laparoscopic intrahepatic Glissonian approach8,9,10. This intrahepatic Glissonian approach, also known as the extra-fascial approach with minor hepatotomy27, involves making a hepatotomy on the right edge of the gallbladder bed and a vertical incision on the segment seven perpendicular to the hepatic hilum. Rouviere's sulcus is a reliable anatomical landmark housing the right posterior pedicle28. After making the hepatotomies, blunt dissection around the Rouviere's sulcus readily exposes the right posterior pedicle, which can be temporarily controlled with a vascular clamp, serving as a partial Pringle's maneuver. Ultrasonographic confirmation of normal vascular flow in other segments and the appearance of a demarcation line indicates the correct placement of the vascular clamp.
This approach can allow anatomical segmental resection, which is shown to be associated with a lower recurrence rate than non-anatomical resection29. It also provides rapid and direct access to the right posterior pedicle, avoiding the time-consuming and tedious hilar dissection. Compared with the extrahepatic approach, which, as proposed by the Korean group, is not always feasible, especially for right posterior sectionectomy, as the extrahepatic course for the right pedicle is short5. However, aberrant vascular and biliary anatomy may pose potential difficulties and danger in the intrahepatic Glissonian approach. Pre-operative studying of any imaging and liberal use of intra-operative cholangiogram and ultrasonography are essential. Laparoscopic encircling of the Glissonian pedicle may be difficult due to the constraint of movement of the laparoscopic instrument. The approach used in this case temporarily controls the pedicle with a Bulldog clamp, leaving the encircling part until the parenchymal transection is completed. This modification to Machado et al.'s proposed method is a safe and easier way of performing major hepatectomy30. One limitation of this technique is that when the tumor is closed to the liver hilum, the intrahepatic Glissonian approach may cause the tumor to rupture, compromising the resection margins.
The current consensus on the best practice for laparoscopic liver parenchymal transection is still lacking29. Several surgical devices are most commonly used, including the ultrasonic scalpel, vessel sealer, CUSA, diathermy, monopolar sealer (saline drip), argon beam coagulator, radiofrequency pre-coagulator, microwave pre-coagulator and water-jet31,32. Mechanical methods include the crush-clamp approach and stapler31,32. The choice of liver parenchymal transection technique ultimately depends on the surgeon's preference. Professor Kwon introduced a novel technique called the 'ultrasonic scalpel mimic CUSA' technique in 201911. This technique was initially used only for superficial parenchymal transection, while CUSA was used for deeper tissue. However, another study used this technique for the entire parenchymal transection12. The retrospective series by Yang et al. showed promising results. Their study showed that the 'ultrasonic scalpel mimic CUSA' group resulted in less intraoperative blood loss and shorter operative time when compared to the CUSA group. This technique is best for the cirrhotic liver because CUSA would inadvertently damage vessels and bile ducts easily in such a situation.
The model of the ultrasonic scalpel affects the accuracy of tissue dissection. We prefer the specific model stated in Table of Materials (Figure 5) because of a blunter and shorter active blade that will cause less inadvertent tissue, bile duct, or vessel injury.
Identification and constant visualization of a clear transection plane are crucial for safe parenchymal transection when operating in this anatomically challenging location where visibility may be limited. The success of our surgery relies on two essential tips. Firstly, early identification of the right hepatic vein is vital. Anatomical resection should always follow the plane along the hepatic veins. However, bleeding from veins may occur during the parenchymal transection. Low central venous pressure should be maintained during the dissection along the hepatic veins. Minor bleeding can usually be controlled by gauze packing and bipolar diathermy, with the surgeon's assisting hand maintaining suction to keep the field clear. However, in case of significant bleeding from the right hepatic vein and even IVC, the surgeon should be familiar with intracorporal suturing techniques to control the bleeding effectively. Secondly, we used a flexible tip laparoscope. A skillful camera operator should be able to manipulate the laparoscope smoothly and accurately to provide a clear view of the field around the corners and in tight spaces, improving the surgeons' ability to identify and maintain a correct transection plane.
The intrahepatic Glissonian approach and the 'ultrasonic scalpel mimic CUSA' parenchymal transection method are promising techniques in helping surgeons overcome the difficulties in this challenging operation. Early identification of hepatic veins and meticulous hemostasis by bipolar diathermy minimize blood loss. Laparoscopic right posterior sectionectomy is safe and feasible in preserving the liver parenchymal while achieving anatomical resection for hepatic tumors.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The study is self-sponsored.
Materials
| 3D ENDOEYE Flex | Olympus | LTF-S190-10-3D | Flexible tip laparoscopic camera |
| 5 mm ROBI Bipolar Grasping Forceps | KARL STORZ | 38851 ON | atraumatic, fenestrated forceps |
| AESCULAP Challenger Ti-P | Barun | PL520L | Pneumatic driven multi-fire clip applicator. 5 mm metal clips |
| Endo GIA Reloads with Tri-Staple Technology, 30 mm, Tan colour | Medtronic | SIG30AVM | Tristaple system that has stepped cartridge face that delivers graduated compression and three rows of varied height staples. Staple height 2 mm, 2.5 mm, 3 mm. |
| Endo GIA Ultra Universal Stapler | Medtronic | EGIAUSTND | Manual stapler that compresses tissue while it simultaneously lays down a staple line and transects the tissue |
| HARMONIC ACE+7 Shears | Ethicon | HARH36 | Curved tip, energy sealing and dissecting, diameter 5 mm, length 36 cm |
| Hem-o-lok Clips L | Weck Surgical Instruments, Teleflex Medical, Durham, NC | 544240 | Vascular clip 5–13 mm Size Range |
| Hem-o-lok Clips ML | Weck Surgical Instruments, Teleflex Medical, Durham, NC | 544230 | Vascular clip 3–10 mm Size Range |
| Hem-o-Lok Polymer Ligation System | Weck Surgical Instruments, Teleflex Medical, Durham, NC | 544965 | |
| Profocus 2202 Ultraview 800 | BK Medical | N/A | Intraoperative Ultrasonography |
References
- van der Heijde, N., et al. Laparoscopic versus open right posterior sectionectomy: an international, multicenter, propensity score-matched evaluation. Surgical Endoscopy. 35 (11), 6139-6149 (2021).
- Cheng, K. C., Yeung, Y. P., Ho, K. M., Chan, F. K. Laparoscopic right posterior sectionectomy for malignant lesions: An anatomic approach. Journal of Laparoendoscopic & Advanced Surgical Techniques. Part A. 25 (8), 646-650 (2015).
- Fretland, A. A., et al. Laparoscopic versus open resection for colorectal liver metastases: The OSLO-COMET randomized controlled trial. Annals of Surgery. 267 (2), 199-207 (2018).
- Landi, F., et al. Short-term outcomes of laparoscopic vs. open liver resection for hepatocellular adenoma: a multicenter propensity score adjustment analysis by the AFC-HCA-2013 study group. Surgical Endoscopy. 31 (10), 4136-4144 (2017).
- Cho, A., et al. Safe and feasible extrahepatic Glissonean access in laparoscopic anatomical liver resection. Surgical Endoscopy. 25 (4), 1333-1336 (2011).
- Oztas, M., Lapsekili, E., Fatih Can, M. Laparoscopic liver right posterior sectionectomies; surgical technique and clinical results of a single surgeon experience. Turkish Journal of Surgery. 38 (1), 18-24 (2022).
- Siddiqi, N. N., et al. Laparoscopic right posterior sectionectomy (LRPS): surgical techniques and clinical outcomes. Surgical Endoscopy. 32 (5), 2525-2532 (2018).
- Machado, M. A., Makdissi, F. F., Galvao, F. H., Machado, M. C. Intrahepatic Glissonian approach for laparoscopic right segmental liver resections. The American Journal of Surgery. 196 (4), e38-e42 (2008).
- Machado, M. A., Kalil, A. N. Glissonian approach for laparoscopic mesohepatectomy. Surgical Endoscopy. 25 (6), 2020-2022 (2011).
- Topal, B., Aerts, R., Penninckx, F. Laparoscopic intrahepatic Glissonian approach for right hepatectomy is safe, simple, and reproducible. Surgical Endoscopy. 21 (11), 2111 (2007).
- Park, J., et al. Safety and Risk factors of pure laparoscopic living donor right hepatectomy: Comparison to open technique in propensity score-matched analysis. Transplantation. 103 (10), e308-e316 (2019).
- Yang, Y., et al. Laparoscopic liver resection with "ultrasonic scalpel mimic CUSA" technique. Surgical Endoscopy. 36 (12), 8927-8934 (2022).
- Kitano, S., Kim, Y. I. ICG clearance in assessing cirrhotic patients with hepatocellular carcinoma for major hepatic resection. HPB Surgery. 10 (3), 182-183 (1997).
- Lim, M. C., Tan, C. H., Cai, J., Zheng, J., Kow, A. W. C. CT volumetry of the liver: Where does it stand in clinical practice. Clinical Radiology. 69 (9), 887-895 (2014).
- Sachdeva, A., Dalton, M., Lees, T. Graduated compression stockings for prevention of deep vein thrombosis. Cochrane Database of Systematic Reviews. 11 (11), CD001484 (2018).
- Liu, T. S., et al. Application of controlled low central venous pressure during hepatectomy: A systematic review and meta-analysis. Journal of Clinical Anesthesia. 75, 110467 (2021).
- Ellison, E. C., Zollinger, R. M. . Zollinger’s Atlas of Surgical Operations. , (2016).
- Kato, Y., et al. Minimally invasive anatomic liver resection for hepatocellular carcinoma using the extrahepatic Glissonian approach: Surgical techniques and comparison of outcomes with the open approach and between the laparoscopic and robotic approaches. Cancers (Basel). 15 (8), 2219 (2023).
- Kamiyama, T., Kakisaka, T., Orimo, T. Current role of intraoperative ultrasonography in hepatectomy. Surgery Today. 51 (12), 1887-1896 (2021).
- Huang, J. W., Su, W. L., Wang, S. N. Alternative laparoscopic intracorporeal Pringle maneuver by Huang’s loop. World Journal of Surgery. 42 (10), 3312-3315 (2018).
- . Laparoscopes articulating HD 3D videoscope ENDOEYE FLEX 3D (LTF-190-10-3D) Available from: https://medical.olympusamerica.com/products/laparoscopes/endoeye-flex-3d (2023)
- Welling, A., Scoggins, P., Cummings, J., Clymer, J., Amaral, J. Superior dissecting capability of a new ultrasonic device improves efficiency and reduces adhesion formation. Global Surgery. 3 (1), (2017).
- Lortat-Jacob, J. L., Robert, H. G., Henry, C. Case of right segmental hepatectomy. Memoires. Academie de Chirurgie (Paris). 78 (8-9), 244-251 (1952).
- Takasaki, K. Glissonean pedicle transection method for hepatic resection: a new concept of liver segmentation. Journal of Hepato-Biliary-Pancreatic Surgery. 5 (3), 286-291 (1998).
- Ramacciato, G., et al. Effective vascular endostapler techniques in hepatic resection. International Surgery. 83 (4), 317-323 (1998).
- Figueras, J., et al. Hilar dissection versus the "glissonean" approach and stapling of the pedicle for major hepatectomies: a prospective, randomized trial. Annals of Surgery. 238 (1), 111-119 (2003).
- Jegadeesan, M., Jegadeesan, R. Anatomical basis of approaches to liver resection. Acta Scientific Gastrointestinal Disorders. 3 (12), 17-23 (2020).
- Eikermann, M., et al. Prevention and treatment of bile duct injuries during laparoscopic cholecystectomy: the clinical practice guidelines of the European Association for Endoscopic Surgery (EAES). Surgical Endoscopy. 26 (11), 3003-3039 (2012).
- Imamura, H., et al. Prognostic significance of anatomical resection and des-gamma-carboxy prothrombin in patients with hepatocellular carcinoma. British Journal of Surgery. 86 (8), 1032-1038 (1999).
- Lee, N., et al. Application of temporary inflow control of the Glissonean pedicle method provides a safe and easy technique for totally laparoscopic hemihepatectomy by Glissonean approach. Annals of Surgical Treatment and Research. 92 (5), 383-386 (2017).
- Otsuka, Y., et al. What is the best technique in parenchymal transection in laparoscopic liver resection? Comprehensive review for the clinical question on the 2nd International Consensus Conference on Laparoscopic Liver Resection. Journal of Hepato-Biliary-Pancreatic Sciences. 22 (5), 363-370 (2015).
- Jia, C., et al. Laparoscopic liver resection: a review of current indications and surgical techniques. Hepatobiliary Surgery and Nutrition. 7 (4), 277-288 (2018).

