An In Vitro Model to Study the Effect of 5-Aminolevulinic Acid-mediated Photodynamic Therapy on Staphylococcus aureus Biofilm
Summary
This manuscript describes a protocol to study the antimicrobial effect of 5-aminolevulinic acid-mediated photodynamic therapy (ALA-PDT) on a Staphylococcus aureus biofilm. This protocol can be used to develop an in vitro model to study the treatment of bacterial biofilms with PDT in the future.
Abstract
Staphylococcus aureus (S. aureus) is a common human pathogen, which causes pyogenic and systemic infections. S. aureus infections are difficult to eradicate not only due to the emergence of antibiotic-resistant strains but also its ability to form biofilms. Recently, photodynamic therapy (PDT) has been indicated as one of the potential treatments for controlling biofilm infections. However, further studies are required to improve our knowledge of its effect on bacterial biofilms, as well as the underlying mechanisms. This manuscript describes an in vitro model of PDT with 5-aminolevulinic acid (5-ALA), a precursor of the actual photosensitizer, protoporphyrin IX (PpIX). Briefly, mature S. aureus biofilms were incubated with ALA and then exposed to light. Subsequently, the antibacterial effect of ALA-PDT on S. aureus biofilm was quantified by calculating the colony forming units (CFUs) and visualized by viability fluorescent staining via confocal laser scanning microscopy (CLSM). Representative results demonstrated a strong antibacterial effect of ALA-PDT on S. aureus biofilms. This protocol is simple and can be used to develop an in vitro model to study the treatment of S. aureus biofilms with ALA-PDT. In the future, it could also be referenced in PDT studies utilizing other photosensitizers for different bacterial strains with minimal adjustments.
Introduction
S. aureus is an important Gram-positive pathogen that colonizes the skin and mucosa of human hosts. Its ability to form biofilms is considered an important aspect of its pathogenesis1. Bacterial biofilms are a community of bacteria embedded in a self-produced matrix, which is composed of extracellular polymeric substances, including polysaccharide, DNA, and protein. This matrix plays a significant role in the persistence of bacterial infections, contributing to a high degree of resistance to the human immune system and current anti-microbial therapies2. Antibiotics are still the major treatment for biofilm infections, although the effects of antibiotics on biofilms are limited. It has been shown previously that cells in biofilms are 10 – 1,000 times more resistant to antibiotics compared to their planktonic counterparts3. Thus, alternative strategies are needed to conquer this issue.
PDT, an alternative treatment for bacterial infections, uses the light of an appropriate wavelength to activate photosensitizers. This leads to the production of reactive oxygen species (ROS), which are lethal to target cells by disrupting the cell wall, inactivating enzymes, and damaging DNA4. This multi-target characteristic makes it difficult for bacteria to develop resistance to the PDT treatment.
The antimicrobial effect of PDT on bacterial and fungal biofilms, with multiple photosensitizers, such as toluidine blue, malachite green, methylene blue, chlorine e6, and porphyrins, has been studied in previous reports5,6,7,8,9,10,11,12,13. 5-ALA, a prodrug of the actual photosensitizer, PpIX, is characterized by its small molecular weight and rapid clearance12,14. These advantages give ALA-PDT major potential as a therapeutic application. Although the effect of ALA-PDT on planktonic bacteria has been studied by many groups12, the antimicrobial effect of ALA-PDT on bacterial biofilms has not yet been elucidated. Meanwhile, it is difficult to compare the results between previous studies. One of the reasons is that the different protocols are used by diverse groups. Thus, this protocol describes an in vitro model of an ALA-PDT system based on our previous work15. The effect of this model was confirmed by CFU calculation and viability staining with CLSM.
Protocol
1. Biofilm Formation
- Biofilm formation in 96-well microplates
- Retrieve the S. aureus strain USA300 and 3 biofilm-forming clinical strains (C1 – C3) stored at -80 °C.
NOTE: The ability of the clinical strains to form biofilms was determined by the microtiter plate assay described previously15. - Inoculate the bacterium in 5 mL tryptone soya broth (TSB) medium, and cultivate in an incubator with shaking at 37 °C overnight to the stationary phase.
- Centrifuge the overnight bacterial culture at 4,000 x g for 10 min at 25 °C and then discard the supernatant. Resuspend the pellets in phosphate buffered saline (PBS) to a final concentration of 2.0 x 109 CFU/mL.
NOTE: The concentration of the bacteria was estimated by measuring optical density and further determined by plate count16, revealing that 1 OD600 of suspension contained 1.5 x 108 CFU/mL. - Dilute the bacterial suspension to 1:200 (1.0 x 107 CFU/mL) in TSB medium containing 0.5% glucose. Inoculate 200 µL of bacterial suspension into each well of a cell-culture-treated polystyrene 96-well microplate.
- Incubate the microplate statically at 37 °C for 24 h under a well oxygenated environment.
NOTE: The incubation time for mature biofilm formation may vary for different bacterial strains; this should be determined before the PDT experiment15. - Discard the media in the wells and wash the microplate wells gently with PBS three times and then discard the supernatant.
NOTE: The step should be carried out very gently to avoid disturbing the formed biofilm.
- Retrieve the S. aureus strain USA300 and 3 biofilm-forming clinical strains (C1 – C3) stored at -80 °C.
- Biofilm formation in dishes
- Inoculate the S. aureus strain USA300 into 5 mL of TSB medium, and cultivate in an incubator with shaking at 37 °C overnight to the stationary phase.
- Centrifuge the overnight bacterial culture at 4,000 x g for 10 min at 25 °C and then discard the supernatant. Resuspend the pellets in PBS to a final concentration of 2.0 x 109 CFU/mL. Then, dilute the bacterial suspension to 1:200 (1.0 x 107 CFU/mL) in TSB medium containing 0.5% glucose. Inoculate 2 mL of bacterial suspension into a 35-mm optical quality glass bottom cell culture dish, and incubate statically at 37 °C for 24 h.
NOTE: The concentration of the bacteria was estimated by measuring the optical density. - Aspirate the media with a pipette, and then rinse the biofilms in the dish gently with PBS three times and then, discard the supernatant carefully.
NOTE: Avoid touching the pipette tip to the bottom of the dish. The step 2.2 should be performed immediately after this step to prevent drying of the formed biofilm.
2. Light Irradiation
- Store 5-ALA in a 4 °C refrigerator. Before the experiment, dilute 5-ALA with PBS to 10 mM.
NOTE: 5-ALA solution should be freshly prepared before the experiment. - In the experimental group, add 200 µL of 10 mM ALA to each well of the microplate or 2 mL to the culture dish. Cover the plate/dish with aluminum foil, and incubate for 1 h. Then, irradiate the plate/dish with a light-emitting diode (LED) with a light intensity of 100 mW/cm2 for 1 h to achieve a light dose of 360 J/cm2 at a major wavelength of 633 ± 10 nm17.
NOTE: In order to let the light energy be effectively and equally delivered to the biofilm in all of the wells/dishes, fix the distance from the peak of the light source to the well/dish at 6.0 cm, and limit the experimental region to the central irradiation area (10 cm x 8 cm). To ensure that the results are reproducible, the experiments should be performed at the same room temperature.
In the LED irradiation step, to avoid direct exposure of the plate to other light sources, such as sunlight, room lighting or lamplight, the LED was turned on before moving the plate/dish to the irradiation area, and the light was bright enough to finish the operation. - Set up the control groups (three control groups were set up in our experiment).
- For the first control group (ALA-LED-), add 200 µL of PBS to each well of the microplate or 2 mL to the culture dish. Cover the plate/dish with aluminum foil, and incubate it for 2 h.
- For the second control group (ALA+LED-), add 200 µL of 10 mM ALA to each well of the microplate or 2 mL to the culture dish. Cover the plate/dish with aluminum foil, and incubate it for 2 h.
- For the third control group (ALA-LED+), add 200 µL of PBS to each well of the microplate or 2 mL to the culture dish. Cover the plate/dish with aluminum foil, and incubate it for 1 h. Then expose the plate to the LED with 360 J/cm2 light irradiation at a major wavelength of 633 ± 10 nm17.
NOTE: In the LED irradiation step, avoid direct exposure of the plate to other light sources, such as sunlight, room lighting, or lamplight.
3. Determination of the Effectiveness of PDT Treatment
NOTE: To confirm the effect of ALA-PDT on the S. aureus biofilms, the viability of the cells with or without ALA-PDT was evaluated by CFU counting as well as by viability staining.
- Determination of the remaining viable bacterial cells
- After ALA-PDT treatment, discard the media in the wells and wash the wells with PBS three times to remove all non-adherent cells for both experimental and control groups.
NOTE: This step should be carried out very gently. - Scrape the adherent bacteria cells thoroughly from the wells with the pipette tip and collect the cells in conical tubes.
- Centrifuge the bacterial suspension at 4,000 x g for 10 min at 4 °C, then discard the supernatant.
- Resuspend the bacteria in 1 mL of 0.25% pancreatin enzyme in PBS and incubate at 37 °C for 1.5 h.
- Centrifuge at 4,000 x g for 10 min; discard the supernatant, then resuspend the pellet in 200 µL of PBS.
- Make 1:10 serial dilutions of the cell solution with PBS; then, add 5 µL of each serial dilution sample onto the tryptone soya agar (TSA) plate. Incubate the TSA plate at 37 °C for 16 h; then, count (by naked eye) the number of bacterial colonies (CFU/mL).
- After ALA-PDT treatment, discard the media in the wells and wash the wells with PBS three times to remove all non-adherent cells for both experimental and control groups.
- Observation of S. aureus biofilms by CLSM
- After light irradiation, wash the biofilms in the culture dish with PBS three times.
NOTE: This step should be carried out very gently. - Add 1 mL of 1 µM green-fluorescent nuclear and chromosome stain that is permeable to the prokaryotic cell membranes (e.g., SYTO9) and 1 mL of 1 µM propidium iodide (PI) for 20 min to stain the biofilm as well as dead cells.
- Observe viable cells (green fluorescence, Ex/Em 485 nm/530 nm) and dead cells (red fluorescence, Ex/Em 485 nm/630nm) under a CLSM with a 63X 1.4-NA oil-immersion objective lens.
- Generate images using microscopy software.
- After light irradiation, wash the biofilms in the culture dish with PBS three times.
Representative Results
The viability of the bacteria in the biofilms was decreased after ALA-PDT treatment when compared to the controls (ALA-LED-, ALA+LED-, and ALA-LED+) in both USA300 and the three clinical strains (Figure 1).
To confirm the results from the CFU assay and observe the antibacterial effect of ALA-PDT on the S. aureus biofilm in situ, the USA300 biofilms were visualized by CLSM with viability staining. The viable and dead cells were stained with green and red fluorescence, respectively. The image showed that most of the bacteria in the biofilms were killed by ALA-PDT, which was consistent with the results of the CFU assay (Figure 2).
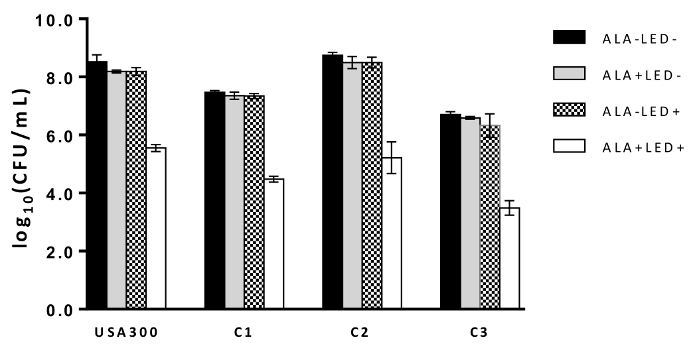
Figure 1: The effect of ALA-PDT on biofilms. CFU/mL was log-transformed and shown as the mean ± standard deviation in USA300 and three clinical strains (C1 – C3) treated with ALA-PDT (ALA+LED+) or under the control conditions (ALA-LED-, ALA+LED-, ALA-LED+). Please click here to view a larger version of this figure.
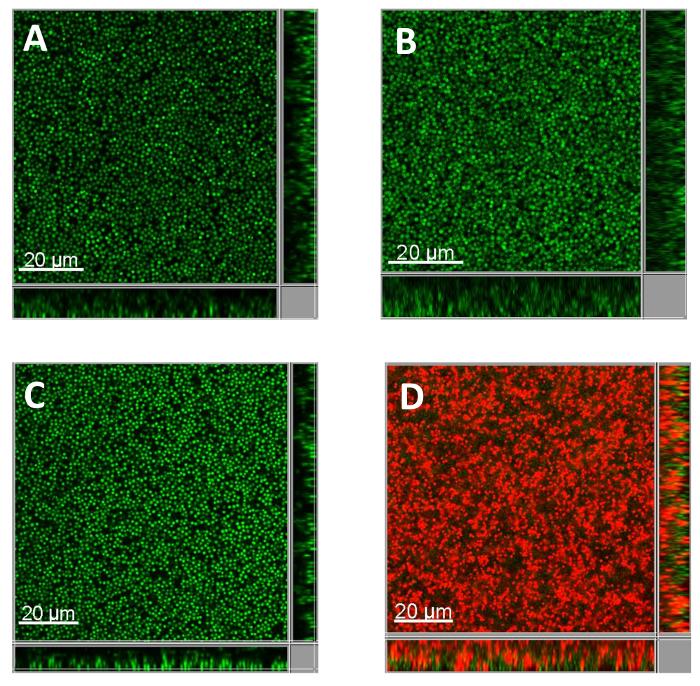
Figure 2: Representative CLSM images of S. aureus biofilms with LIVE/DEAD staining. Biofilms formed by USA300 bacteria were treated with or without ALA-PDT (panel A: ALA-LED-, panel B: ALA+LED-, panel C: ALA-LED+, panel D: ALA+LED+), and then stained with SYTO9 (green fluorescence) and PI (red fluorescence) to represent the live and dead bacteria independently. A 63X 1.4-NA oil immersion objective was used. Please click here to view a larger version of this figure.
Discussion
PDT has been a well-studied therapy for the treatment of cancer since it was invented more than 100 years ago18. Over the last decade, PDT has been applied as an antimicrobial strategy and has shown effectiveness against some antibiotic-resistant pathogenic bacteria12. Compared to the planktonic state, bacterial biofilms appear to be more resistant to antibiotic treatment3, while the effect of ALA-PDT on biofilms has not been fully investigated yet.
In this article, an in vitro ALA-PDT system was described, and the antibacterial effect of this model on S. aureus biofilms was demonstrated. Two methods were used to test the effect of ALA-PDT on S. aureus biofilms in this protocol. While the CFU test demonstrated the antimicrobial effect by calculating the viable cells after treatment, fluorescent viability staining with CLSM not only confirmed the results of the CFU test but also detected the morphological character of the live and dead bacteria in situ. Using both analytical techniques together is an ideal approach for determining the effect of ALA-PDT on biofilms. Based on the CLSM results, the dead bacterial cells were predominantly distributed in the upper layer, while some of the bacteria in the bottom layer remained alive15. The latter may be the source of bacterial colonies in the CFU assay. A similar result has been observed in a study conducted by O'Neill et al., which was explained by the low accumulation of photosensitizers in the inner layer or the inability of the light to penetrate these regions19.
In this protocol, the mature biofilm was incubated with 10 mM of ALA for 1 h before exposure to PDT. These parameters were chosen based on the results of two pre-experiments. First, the antimicrobial effect of ALA was tested against biofilms without light irradiation using different concentrations of ALA and different amounts of time for incubation with S. aureus biofilms. The groups without any bactericidal effect were chosen as the candidates. Second, the PDT effect was detected in these candidate groups, and the group with the most potent bactericidal effect was finally chosen in this protocol. Thus, the parameters used in this protocol ensured that there was no bactericidal effect of ALA alone. A 1-h incubation time is significantly shorter than those used in previous studies17,20, making it convenient for in vitro studies and for potential application in future clinical treatments.
There are several critical points for successful use of this model. First, the whole process involving the manipulation of ALA treated bacteria should be performed in darkness. Second, the manipulation of the mature biofilms should be gentle to avoid disturbing the formed biofilm. Third, in the CFU test, scraping the bacteria from the bottom of the plates should be thorough. Finally, the freshly prepared ALA should be used; therefore, it is better to prepare ALA right before the experiment.
Although this protocol can be used to test the effect of ALA-PDT on S. aureus biofilms in vitro, it is still different from the clinical situation in vivo. For example, biofilms in the human body are usually formed by multiple bacterial strains21,22, and the environment in vivo is more complex than that in vitro, which might influence the effect of PDT. Therefore, future in vivo experiments are needed for a full evaluation of the antibacterial effects of ALA-PDT on S. aureus biofilms. However, because of its advantages of convenience and the ethical issues of conducting in vivo studies, this in vitro platform will be useful and practical to improve the study of the effects of ALA-PDT on S. aureus biofilms. It should also be noted that although ALA, a precursor of the photosensitizer PpIX, has favorable characteristics, including rapid clearance, less and shorter-lasting cutaneous photosensitivity, limited light penetration restricted to superficial lesions and especially its non-cumulative toxicity14, the light dose and photosensitizer concentration required to achieve bacteria killing may still have a bystander effect on host cell viability. Thus, studying the modification of ALA23 for targeted therapy of selected pathogenic bacterial strains like S. aureus is valuable for future antimicrobial therapy.
This protocol can not only be used for the study of the effect of ALA-PDT on S. aureus strains in the future but can also be referenced to study its effect on biofilms formed by other bacteria. The parameters, such as the concentration of ALA and the duration of the bacteria incubation with ALA, may vary among different bacterial strains, but the principles discussed above are commonly shared.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was funded by National Nature Science Foundation of China for Young Scholars (No. 81300810), Shanghai Young Doctor Training Program (No. 20141057), and National Natural Science Foundation of China (81671982, 81271791 and 81571955). We would like to thank LetPub (www.letpub.com) for providing linguistic assistance during the preparation of this manuscript.
Materials
| Tryptone Soya Broth (TSB) | OXOID | CM0129B | |
| Tryptone Soya Agar (TSA) | OXOID | CM0131 | |
| SYTO9 | Thermo Fisher Scientific | L7012 | The LIVE/DEAD BacLight Bacterial Viability Kits |
| Propidium iodide (PI) | Thermo Fisher Scientific | L7012 | The LIVE/DEAD BacLight Bacterial Viability Kits |
| Pancreatin | Sigma-Aldrich | P3292 | |
| 5-aminolevulinic acid (ALA) | Fudan Zhangjiang Bio-Pharm | 3.1 | |
| Staphylococcus aureus strain USA300 | / | / | The source of USA 300 references “Tenover FC, Goering RV. J Antimicrob Chemother. 2009 Sep; 64(3):441-6”. |
| Staphylococcus aureus clinical strains (C1-C3) | / | / | All clinical strains were isolated from patients with chronic rhinosinusitis in the Department of Otorhinolaryngology-Head and Neck Surgery, Eye and ENT Hospital of Fudan University [Zhang QZ, Zhao KQ, Wu Y, et al. PLoS One. 2017 Mar; 12(3): e0174627]. |
| 96-well microplate | Corning Inc | 3599 | Clear Flat Bottom Polystyrene TC-Treated Microplates, Individually Wrapped, with Lid, Sterile |
| Fluorodish | NEST Biotechnology | 801001 | Glass bottom, Non-pyrogenic |
| Eppendorf Safe-Lock Tubes, 1.5 mL | Eppendorf | 0030120086 | |
| Eppendorf microcentrifuge 5417 | Eppendorf | Z365998 | SIGMA | |
| Incubator | Thermo Fisher Scientific | SHKE4000 | MaxQ 4000 Benchtop Orbital Shakers |
| Light emitting diode (LED) | Wuhan Yage Optic and Electronic Technique CO | LED-IB | |
| Leica TCS SP8 confocal laser-scanning microscope | Leica Microsystems | ||
| Leica LAS AF software | Leica Microsystems | ||
| IMARIS software | Bitplane |
Referências
- Lewis, K. Riddle of biofilm resistance. Antimicrob Agents Chemother. 45 (4), 999-1007 (2001).
- Rabin, N., et al. Biofilm formation mechanisms and targets for developing antibiofilm agents. Future Med Chem. 7 (4), 493-512 (2015).
- Mah, T. F., O’Toole, G. A. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 9 (1), 34-39 (2001).
- Sharma, M., et al. Toluidine blue-mediated photodynamic effects on staphylococcal biofilms. Antimicrob Agents Chemother. 52 (1), 299-305 (2008).
- Rosa, L. P., da Silva, F. C., Nader, S. A., Meira, G. A., Viana, M. S. In vitro effectiveness of antimicrobial photodynamic therapy (APDT) using a 660 nm laser and malachite green dye in Staphylococcus aureus biofilms arranged on compact and cancellous bone specimens. Lasers Med Sci. 29 (6), 1959-1965 (2014).
- Rosa, L. P., Silva, F. C., Nader, S. A., Meira, G. A., Viana, M. S. Effectiveness of antimicrobial photodynamic therapy using a 660 nm laser and methyline blue dye for inactivating Staphylococcus aureus biofilms in compact and cancellous bones: An in vitro study. Photodiagnosis Photodyn Ther. 12 (2), 276-281 (2015).
- Mai, B., et al. The antibacterial effect of sinoporphyrin sodium photodynamic therapy on Staphylococcus aureus planktonic and biofilm cultures. Lasers Surg Med. 48 (4), 400-408 (2016).
- Gandara, L., Mamone, L., Bohm, G. C., Buzzola, F., Casas, A. Enhancement of photodynamic inactivation of Staphylococcus aureus biofilms by disruptive strategies. Lasers Med Sci. 32 (8), 1757-1767 (2017).
- Baltazar, L. M., et al. Antimicrobial photodynamic therapy: an effective alternative approach to control fungal infections. Front Microbiol. 6, 202 (2015).
- Fernandes, T., Bhavsar, C., Sawarkar, S., D’Souza, A. Current and novel approaches for control of dental biofilm. Int J Pharm. 536 (1), 199-210 (2017).
- De Sordi, L., et al. Development of Photodynamic Antimicrobial Chemotherapy (PACT) for Clostridium difficile. PLoS One. 10 (8), e0135039 (2015).
- Harris, F., Pierpoint, L. Photodynamic therapy based on 5-aminolevulinic acid and its use as an antimicrobial agent. Med Res Rev. 32 (6), 1292-1327 (2012).
- Donnelly, R. F., McCarron, P. A., Tunney, M. M. Antifungal photodynamic therapy. Microbiol Res. 163 (1), 1-12 (2008).
- Shi, H., Li, J., Zhang, H., Zhang, J., Sun, H. Effect of 5-aminolevulinic acid photodynamic therapy on Candida albicans biofilms: An in vitro study. Photodiagnosis Photodyn Ther. 15, 40-45 (2016).
- Zhang, Q. Z., et al. 5-aminolevulinic acid-mediated photodynamic therapy and its strain-dependent combined effect with antibiotics on Staphylococcus aureus biofilm. PLoS One. 12 (3), 0174627 (2017).
- Chang, Y. C., et al. Rapid single cell detection of Staphylococcus aureus by aptamer-conjugated gold nanoparticles. Sci Rep. 3, 1863 (2013).
- Barra, F., et al. Photodynamic and Antibiotic Therapy in Combination to Fight Biofilms and Resistant Surface Bacterial Infections. Int J Mol Sci. 16 (9), 20417-20430 (2015).
- St Denis, T. G., et al. All you need is light: antimicrobial photoinactivation as an evolving and emerging discovery strategy against infectious disease. Virulence. 2 (6), 509-520 (2011).
- O’Neill, J. F., Hope, C. K., Wilson, M. Oral bacteria in multi-species biofilms can be killed by red light in the presence of toluidine blue. Lasers Surg Med. 31 (2), 86-90 (2002).
- Li, X., et al. Effects of 5-aminolevulinic acid-mediated photodynamic therapy on antibiotic-resistant staphylococcal biofilm: an in vitro study. J Surg Res. 184 (2), 1013-1021 (2013).
- Hall-Stoodley, L., Costerton, J. W., Stoodley, P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2 (2), 95-108 (2004).
- Elias, S., Banin, E. Multi-species biofilms: living with friendly neighbors. FEMS Microbiol Rev. 36 (5), 990-1004 (2012).
- Wu, J., et al. Design and Proof of Programmed 5-Aminolevulinic Acid Prodrug Nanocarriers for Targeted Photodynamic Cancer Therapy. ACS Appl Mater Interfaces. 9 (17), 14596-14605 (2017).

