Apparatus for Harvesting Tissue Microcolumns
Summary
Here we describe a protocol for producing harvesting needles that can be used to collect full-thickness skin tissue without causing donor site scarring. The needles can be combined with a simple collection system to achieve high-volume harvesting.
Abstract
This manuscript describes the production process for a laboratory apparatus, made from off-the-shelf components, that can be used to collect microcolumns of full-thickness skin tissue. The small size of the microcolumns allows donor sites to heal quickly without causing donor site scarring, while harvesting full-thickness tissue enables the incorporation of all cellular and extracellular components of skin tissue, including those associated with deeper dermal regions and the adnexal skin structures, which have yet to be successfully reproduced using conventional tissue engineering techniques. The microcolumns can be applied directly into skin wounds to augment healing, or they can be used as the autologous cell/tissue source for other tissue engineering approaches. The harvesting needles are made by modifying standard hypodermic needles, and they can be used alone for harvesting small amounts of tissue or coupled with a simple suction-based collection system (also made from commonly available laboratory supplies) for high-volume harvesting to facilitate studies in large animal models.
Introduction
Autologous skin grafting is the mainstay of wound repair, but it is limited by donor site scarcity and morbidity, leading to concerted efforts in recent decades to develop new therapeutic options to replace conventional skin grafting1,2. We recently developed an alternative method of harvesting skin to harness the benefits of full-thickness skin grafting while minimizing donor site morbidity. By collecting full-thickness skin in the form of small (~0.5 mm diameter) "microcolumns", donor sites are able to heal rapidly and without scarring under normal circumstances (for potential exceptions, see the discussion section below)3. Microcolumns can be applied directly into wound beds to accelerate wound closure, reduce contraction3, and restore a diverse range of epidermal and dermal cell types and functional adnexal structures4, many of which are lacking in conventional split-thickness skin grafting or current bioengineered skin substitutes5. The ability of microcolumns to augment healing and of their donor sites to heal without scarring have both been independently validated by other research groups6,7.
We have previously developed a laboratory harvesting system to enable the collection of microcolumns at scale8; however, this system is composed of many customized components that are not widely available. Here, we describe in detail the process for producing harvesting needles, as well as simple collection systems, made from mostly off-the-shelf components, that can be used to achieve high-volume harvesting. The apparatus described in this manuscript is suitable for in vitro and animal work, but not for use in humans. A clinical device with FDA clearance for applying this technique in humans is commercially available but will not be discussed in detail here.
Protocol
All work involving live animals and animal tissue samples have been approved by the Massachusetts General Hospital Institutional Animal Care and Use Committee (IACUC).
1. Production of Harvesting Needles
- Setup of the production stage
- Secure a female luer lock connector onto a post, and mount the post onto a rotation stage so that the luer lock is at the center of the stage (Figure 1A).
- Position this first rotation stage vertically, and mount it perpendicularly onto a second, horizontal rotation stage (Figure 1B).
- Mount the horizontal rotation stage onto a two-axis translation stage (Figure 1B).
- Fasten the combination onto a stable surface, such as an optical breadboard.
- Separately mount a rotary tool onto the same breadboard, with the tool positioned in parallel with the breadboard and at approximately the same height as the luer lock connector on the rotation/translation combination stage (Figure 1C).
- Install two cut-off wheels concentrically onto the rotary tool (a smaller, lower grit diamond cut-off wheel over a larger, higher grit stone cut-off wheel) (Figure 1D).
NOTE: About a 9 mm difference between the diameters of the two wheels is generally sufficient. - Position an overhead light source with an adjustable arm over the rotary tool, with the light aimed at the cutting wheels.
- To enhance visualization, position a dissecting microscope over the production setup so that the eyepiece is focused on the cutting discs (Figure 1C).
NOTE: Alternatively, users can wear magnifying eyewear.
- Reshaping the needle tip
- Wear protective eyewear and a surgical mask to prevent fine metal particles from entering the eyes or airways.
- Choose hypodermic needles of the appropriate gauge size, based on experimental requirements.
- Mark off the intended length on each harvesting needle.
NOTE: For harvesting from swine skin, 8 mm needles are typically sufficient; although, the length may vary based on experimental needs (e.g., thickness of target skin tissue, needle gauge size). Generally, a 19-gauge needle works well for swine skin. - Lower the needle perpendicularly to the rotary tool with the power on, using the edge of the outer cutting disc to cut off the excess length of the needle, at the point marked in step 1.2.3.
- Connect the shortened, blunt needle to the female luer lock connector on the production stage.
- Adjust the horizontal rotation stage so that the needle is at a 12° angle parallel to the cutting discs on the rotary tool (changing the angle will impact the force required for needle insertion).
- Turn on the overhead light and adjust its position while observing the needle under magnification, until the light is reflected off the midline (lengthwise) of the needle.
- Power on the rotary tool, then use the translation stage to advance the needle towards the inner (diamond) cutting disc (Figure 2A).
- Keep advancing the needle slowly against the cutting disc until the cutting disc reaches approximately the midline of the needle (as visualized by the overhead light's reflection along the midline).
- Slowly move the cut needle surface from the inner diamond wheel onto the outer stone wheel to finish the cut needle surface with a finer polish (Figure 2B).
- Retract the needle away from the cutting disc.
- Using the vertical rotation stage, rotate the needle 180°.
- Repeat steps 1.2.9-1.2.10 to reshape the other side of the needle.
NOTE: The needle should now have two cutting tips of approximately equal length (Figure 2C and 2D). - Remove the needle from the production stage.
- Clean the inside bore with a metal wire that is slightly smaller than the needle's inner diameter.
- Using a sharp wooden stick (e.g., by snapping off the end of the small wooden stick on a cotton tip applicator), remove any burrs that may be still attached to the edges of the newly-formed needle.
NOTE: Harvesting needles can be electropolished and sterilized by autoclave if necessary.
2. Skin Tissue Harvesting
- Use the harvesting needles to collect skin microcolumns from ex vivo tissue or live animals.
- For ex vivo skin tissue that is thin (especially from samples where the subcutaneous fat is missing or was trimmed away), hold the target tissue over the opening of a 50 mL centrifuge tube, or stack two pieces of tissue on top of each other, to avoid damaging the needle tips by hitting them against hard surfaces.
NOTE: For in vivo harvesting from live animals, it is recommended that local lidocaine and epinephrine be administered by intradermal injection for analgesia and to reduce bleeding. - Assemble the harvesting apparatus according to the amount of microcolumns needed, as described below.
- Low-medium volume option:
- To harvest small to medium amounts of microcolumns, simply fill a standard syringe (10-20 mL syringes usually work well) with normal saline and connect it to a harvesting needle.
- Completely insert the harvesting needle into the donor skin, then retract it.
- Push on the piston of the syringe to flush saline through the harvesting needle and expel the microcolumn that is lodged in the needle bore.
- To speed up the harvesting process, repeat step 2.4.2 3 to 5 times before expelling the microcolumns in step 2.4.3.
NOTE: It is usually convenient to expel the microcolumns into a standard cell strainer to ease the subsequent collection of microcolumns. - If needles become clogged, increase the pressure on the piston to expel the stuck tissue and remove the clog. If simply increasing pressure is insufficient, insert a metal wire through the needle tip opening to clear the clog in the needle bore.
- Keep the microcolumns submerged in saline or medium until use to prevent desiccation .
NOTE: With an experienced operator, the method described above can be used to harvest microcolumns at a rate of approximately 1 microcolumn per second.
- High volume option:
- Create a simple suction-assisted device that can be constructed to facilitate collection of large amounts of microcolumns.
NOTE: The device consists of a harvesting needle, 20 mL syringe with luer lock nozzle, suction adapter typically used for liposuction (Figure 3A), and sterile suction canister. - Remove the plunger from the 20 mL syringe.
- Attach the syringe to a suction adapter.
- Complete the assembly by attaching a harvesting needle to the syringe (Figure 3B).
- Use a piece of sterile suction tubing to connect the suction adapter to a sterile suction canister. Make sure the harvesting apparatus is connected to a canister input that allows fluid to flow into the canister unimpeded (which may require connection to a canister port that is marked for outflow rather than inflow).
- Connect the apparatus to a negative pressure source.
NOTE: The pressure required depends on needle diameter and length, as previously described8. For the apparatus described in this manuscript, the suction system found in typical operating rooms is generally sufficient. - Connect a harvesting needle to the syringe.
- Harvest the microcolumns by inserting the harvesting needle into the skin.
NOTE: The microcolumns will be drawn into the syringe by suction, then flushed into the suction canister. - Intermittently dip the harvesting needle into a container of sterile saline during the harvesting procedure to flush the system.
NOTE: This saline flush facilitates transport of the microcolumn and ensures they stay hydrated. Alternatively, an MFF luer adaptor can be connected to the luer lock of the syringe, and through a connection tubing, also to a hanging saline drip bag. - Keep on hand a metal wire, or smaller gauge needle, that is slightly smaller than the harvesting needle's inner diameter. If the needle becomes clogged, it can be cleared by inserting the metal wire into the needle bore.
- When the desired amount of microcolumns have accumulated in the canister, disconnect the device from suction, then pour the contents of the suction canister out through a filter to collect the microcolumns.
- Create a simple suction-assisted device that can be constructed to facilitate collection of large amounts of microcolumns.
- After harvesting, apply a topical antibiotic ointment over the donor sites.
NOTE: Additional dressings are generally not required for donor sites.
Representative Results
The harvesting needles should be able to collect microcolumns of full-thickness skin tissue with approximately a 80-90% success rate, and each microcolumn should contain epidermis, dermis, and some subcutaneous fat (Figure 4). If the success rate of harvesting is low, or if it becomes difficult to insert a needle into tissue, then a new needle is likely needed. If the success rate for harvesting is consistently low, even with new needles, then the needles are probably too short.
If used in vivo, donor sites should heal quickly, as re-epithelialization typically occurs within a few days3. Microcolumns can be applied directly to wound beds to augment wound healing3,4, or they may be combined with different matrix materials to produce combination constructs. Microcolumns can also be maintained in culture for in vitro studies9.

Figure 1: Needle making apparatus. (A) A male luer lock connector secured via a mounting post onto a vertically placed rotation stage, so that the luer lock is at the center of the stage. (B) The vertical rotation stage is mounted perpendicularly onto a second, horizontal rotation stage (black arrow). The horizontal rotation stage is secured to a two-axis translation stage (white arrow). (C) Positioning of the rotary tool parallel to the breadboard (white arrow), and the dissecting microscope over the needle-making apparatus (red arrow). (D) Concentric cut-off wheels mounted onto rotary tool, with a diamond wheel on the inside (black arrow) and stone wheel on the outside (white arrow). Please click here to view a larger version of this figure.

Figure 2: After cutting the needle to the desired length, the rotary tool is used to grind new needle tips. (A) First, the diamond cut-off wheel is used to make "rough" cuts to form the new cutting tips and surfaces. (B) After the new cutting tips are formed with the diamond wheel, the needle is moved to the stone wheel for fine polishing. (C) Finished harvesting needle viewed from the front and (D) from the side. Please click here to view a larger version of this figure.
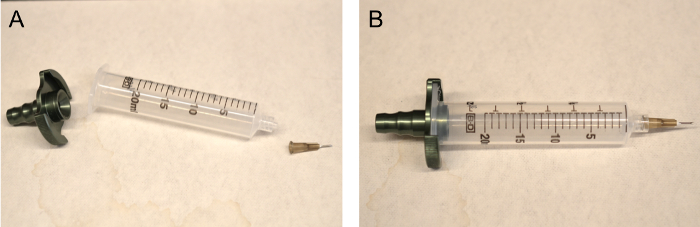
Figure 3: Assembly for high-volume harvesting. (A) Individual components of the assembly, including (left to right) the suction adapter, 20 mL syringe with luer lock nozzle, and harvesting needle. (B) Shown is the completed assembly, ready to connect to negative pressure source. Please click here to view a larger version of this figure.

Figure 4: Representative skin microcolumns harvested using the apparatus described in this manuscript. Each microcolumn contains the epidermis (1), full dermis (2), and some subcutaneous fat (3). Checkmarks in the figure represent 1 mm. This figure has been modified from Tam et al.4 in accordance with the terms of the corresponding creative commons license. Please click here to view a larger version of this figure.
Discussion
The methods described here are intended to enable the collection of tissue microcolumns in sufficient quantities for in vivo large animal studies, using tools made from commercially available laboratory supplies. This apparatus has been used previously in harvesting tissue from excised human skin4,9 as well as live swine skin3. The specific parameters described are those that were found to be most suited for use in swine. It is expected that the same apparatus can be modified and adapted for collecting tissue from rodents and other small animals, but this has not been tested in our laboratory.
Critical steps in this protocol include ensuring the harvesting needles are of sufficient length (inefficient harvesting is usually due to needles being too short), keeping the microcolumns submerged in liquid throughout the harvesting process to prevent desiccation, and flushing the system with saline at least intermittently (otherwise there is a higher likelihood of microcolumns clogging the needle bore). The main limitation of this technique is speed; for example, in our demonstration, the suction-assisted apparatus can generally harvest 120 mg of tissue per minute, which is sufficient for smaller wound sizes typically used in animal experiments. It would likely be logistically challenging to use this approach for very large wounds (e.g., in major burn injury models). Needle gauge is another limitation – the smaller the needle gauge, the more susceptible it is to buckling, which is the main failure mode of this technique (in contrast, the technique is relatively insensitive to needle dulling during the procedure). For swine skin, we normally use 19-gauge needles, which are mechanically robust enough that they rarely buckle. Futhermore, each animal experiment (typically involving about 3,000-5,000 microcolumns) usually requires only 2 to 3 needles. 25-gauge is the smallest needle size we have used with this technique.
The ability of small skin wounds to stimulate tissue regeneration is the underlying principle behind clinical procedures such as fractional laser resurfacing12 and microneedling13. These treatments are known to improve the cosmetic appearance of photoaged skin, and more recently, shown to induce scar remodeling and improve the function and cosmesis of skin scars14,15. The extensive clinical experience with these techniques also provides further validation that skin is able to heal without scarring after these microinjuries in the vast majority of cases, with certain exceptions (fractional laser resurfacing reportedly has a 3.8% incidence of scarring, almost always as a result of infection16, highlighting the importance of post-procedural skin care). In addition, people with a history of keloids or hypertrophic scarring may be susceptible to scarring even with these smaller injuries; thus, treatments involving the production of microinjuries may be counterindicated.
While our previous investigations have focused on directly applying microcolumns into skin wounds to enhance healing, the ability to collect significant amounts of tissue without causing other long-term donor site morbidities (scarring, contracture, etc.) may be useful for a broad range of other applications. Skin microcolumns may serve as the tissue source for approaches involving culture expansion or dissociation and dispersion of autologous skin cells10. Furthermore, microcolumns provide the additional benefits of minimizing donor site morbidity and including dermal cell types, such as those associated with adnexal structures and the various stem/progenitor cell populations that reside in deeper parts of the dermis11 (which are not available with conventional methods that utilize split-thickness skin as the starting material). Autologous microcolumns may also be used in ex vivo assays to study tissue response to various stimuli such as drugs or cosmetic products in which, unlike conventional cell culture-based assays, the cellular and extracellular structures in each microcolumn are maintained in their respective natural organization formats. More generally, the microcolumn harvesting approach may also be broadly applicable to providing autologous cells and tissues for various tissue engineering/regenerative medicine purposes (for skin and other organs), as the underlying principle of small donor wounds undergoing complete and scarless healing is likely to be generalizable to other tissue types.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by the Army, Navy, NIH, Air Force, VA and Health Affairs to support the AFIRM II effort, under Award No. W81XWH-13-2-0054. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick, MD 21702-5014 is the awarding and administering acquisition office. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense.
Materials
| Diamond wheel | Dremel | 545 | |
| Hypodermic needle (19G) | Fisher Scientific | 14-840-98 | Other needle sizes could be used, depending on experimental needs |
| Stome wheel | Dremel | 540 | |
| Syringe (20mL with luer lock) | Fisher Scientific | 22-124-967 | |
| Suction adapter | Tulip Medical | PA20BD | Optional, for high volume harvesting |
| Suction canister | Fisher Scientific | 19-898-212 | Optional, for high volume harvesting. Sterilize before use. |
| Suction tubing | Medline | DYND50216H | Optional, for high volume harvesting |
Referências
- Sun, B. K., Siprashvili, Z., Khavari, P. A. Advances in skin grafting and treatment of cutaneous wounds. Science. 346 (6212), 941-945 (2014).
- Singh, M., et al. Challenging the Conventional Therapy: Emerging Skin Graft Techniques for Wound Healing. Plastic and Reconstructive Surgery. 136 (4), 524-530 (2015).
- Tam, J., et al. Fractional Skin Harvesting: Autologous Skin Grafting without Donor-site Morbidity. Plastic and Reconstructive Surgery. Global Open. 1 (6), 47 (2013).
- Tam, J., et al. Reconstitution of full-thickness skin by microcolumn grafting. Journal of Tissue Engineering and Regenerative Medicine. 11 (10), 2796-2805 (2017).
- Huang, C., et al. Regeneration of hair and other skin appendages: A microenvironment-centric view. Wound Repair and Regeneration. 24 (5), 759-766 (2016).
- Fernandes, J. R., et al. Micro-mechanical fractional skin rejuvenation. Plastic and Reconstructive Surgery. 131 (2), 216-223 (2013).
- Rettinger, C. L., Fletcher, J. L., Carlsson, A. H., Chan, R. K. Accelerated epithelialization and improved wound healing metrics in porcine full-thickness wounds transplanted with full-thickness skin micrografts. Wound Repair and Regeneration. 25 (5), 816-827 (2017).
- Franco, W., et al. Fractional skin harvesting: device operational principles and deployment evaluation. Journal of Medical Devices. 8 (4), 041005 (2014).
- Rasmussen, C. A., et al. Chimeric autologous/allogeneic constructs for skin regeneration. Military Medicine. 179, 71-78 (2014).
- Ter Horst, B., Chouhan, G., Moiemen, N. S., Grover, L. M. Advances in keratinocyte delivery in burn wound care. Advanced Drug Delivery Reviews. 123, 18-32 (2018).
- Wong, V. W., Levi, B., Rajadas, J., Longaker, M. T., Gurtner, G. C. Stem cell niches for skin regeneration. International Journal of Biomaterials. 2012, 926059 (2012).
- Manstein, D., Herron, G. S., Sink, R. K., Tanner, H., Anderson, R. R. Fractional photothermolysis: a new concept for cutaneous remodeling using microscopic patterns of thermal injury. Lasers in Surgery and Medicine. 34 (5), 426-438 (2004).
- Iriarte, C., Awosika, O., Rengifo-Pardo, M., Ehrlich, A. Review of applications of microneedling in dermatology. Clinical, Cosmetic and Investigational Dermatology. 10, 289-298 (2017).
- Anderson, R. R., et al. Laser treatment of traumatic scars with an emphasis on ablative fractional laser resurfacing: consensus report. Journal of the American Medical Association Dermatology. 150 (2), 187-193 (2014).
- Hogan, S., Velez, M. W., Ibrahim, O. Microneedling: a new approach for treating textural abnormalities and scars. Seminars in Cutaneous Medicine and Surgery. 36 (4), 155-163 (2017).
- Manuskiatti, W., Fitzpatrick, R. E., Goldman, M. P. Long-term effectiveness and side effects of carbon dioxide laser resurfacing for photoaged facial skin. Journal of the American Academy of Dermatology. 40 (3), 401-411 (1999).

