Microfluidic Fabrication of Core-Shell Microcapsules carrying Human Pluripotent Stem Cell Spheroids
Summary
This article describes encapsulation of human pluripotent stem cells (hPSCs) using a co-axial flow focusing device. We demonstrate that this microfluidic encapsulation technology enables efficient formation of hPSC spheroids.
Abstract
Three-dimensional (3D) or spheroid cultures of human pluripotent stem cells (hPSCs) offer the benefits of improved differentiation outcomes and scalability. In this paper, we describe a strategy for the robust and reproducible formation of hPSC spheroids where a co-axial flow focusing device is utilized to entrap hPSCs inside core-shell microcapsules. The core solution contained single cell suspension of hPSCs and was made viscous by the incorporation of high molecular weight poly(ethylene glycol) (PEG) and density gradient media. The shell stream comprised of PEG-4 arm-maleimide or PEG-4-Mal and flowed alongside the core stream toward two consecutive oil junctions. Droplet formation occurred at the first oil junction with shell solution wrapping itself around the core. Chemical crosslinking of the shell occurred at the second oil junction by introducing a di-thiol crosslinker (1,4-dithiothreitol or DTT) to these droplets. The crosslinker reacts with maleimide functional groups via click chemistry, resulting in the formation of a hydrogel shell around the microcapsules. Our encapsulation technology produced 400 µm diameter capsules at a rate of 10 capsules per second. The resultant capsules had a hydrogel shell and an aqueous core that allowed single cells to rapidly assemble into aggregates and form spheroids. The process of encapsulation did not adversely affect the viability of hPSCs, with >95% viability observed 3 days post-encapsulation. For comparison, hPSCs encapsulated in solid gel microparticles (without an aqueous core) did not form spheroids and had <50% viability 3 days after encapsulation. Spheroid formation of hPSCs inside core-shell microcapsules occurred within 48 h after encapsulation, with the spheroid diameter being a function of cell inoculation density. Overall, the microfluidic encapsulation technology described in this protocol was well-suited for hPSCs encapsulation and spheroid formation.
Introduction
There is considerable interest in 3D cultures of human pluripotent stem cells (hPSCs) due to the improved pluripotency and differentiation potential afforded by this culture format1,2,3. hPSCs are typically formed into spheroids or other 3D culture formats by means of bioreactors, microwells, hydrogels, and polymeric scaffolds4,5,6. Encapsulation offers another means for organizing single hPSCs into spheroids. Once encapsulated hPSC spheroids may be handled with ease and transferred into microtiter plates for differentiation, disease modeling, or drug testing experiments. Encasing hPSCs in a hydrogel layer also protects cells against shear damage and allows to culture spheroids in a bioreactor at high rates of stirring7.
Our methodology for stem cell encapsulation evolved over time. First, we focused on solid hydrogel microparticles and demonstrated successful encapsulation and cultivation of mouse embryonic stem cells (mESCs)8. However, it was noted that human embryonic stem cells (hESCs) had low viability when encapsulated in such hydrogel microparticles, presumably due to the greater need for these cells to re-establish cell-cell contacts after the encapsulation. We reasoned that heterogeneous microcapsule, possessing an aqueous core, may be better suited for encapsulation of cells that rely on rapid re-establishment of cell-cell contacts. The concept of co-axial flow focusing microfluidic device for making aqueous core/hydrogel shell microcapsules was adapted from He et al.9, but instead of alginate employed in the original approach, a PEG-based hydrogel was incorporated into the shell. We first demonstrated successful encapsulation and spheroid formation of primary hepatocyte in core-shell microcapsules10 and most recently described encapsulation of hES and iPS cells7. As outlined in Figure 1A, capsules are fabricated in a flow focusing device where the shell and core flow streams transition from side-to-side to co-axial flow before ejection into the oil phase. The core flow contains cells and additives that increase the viscosity of the solution (non-reactive PEG MW 35kD and iodixanol – commercial name OptiPrep) while the shell stream contains reactive molecules (PEG-4-Mal). Continuous co-axial flow stream is discretized into droplets that retain core-shell architecture. The core-shell structure is made permanent by exposure to di-thiol crosslinker (DTT), which reacts with PEG-4-Mal via click chemistry and results in formation of a thin (~10 µm) hydrogel skin or shell. After the emulsion is broken and capsules are transferred into an aqueous phase, molecules of PEG diffuse from the core and are replaced by water molecules. This results in aqueous core and hydrogel shell microcapsules.
Provided below are step-by-step instructions on how to make microfluidic devices, how to prepare cells, and how to carry out encapsulation of hPSCs.
Protocol
1. Device fabrication
- Make the designs for the microencapsulation device and dissociation device using CAD software10,11.
- Spin-coat the three layers of SU-8 photoresist sequentially on a silicon wafer (Figure 2A) to achieve structures with the desired heights: 60, 100, and 150 µm.
NOTE: The process for the top and bottom molds is identical.- Spin coat a clean 10 cm silicon wafer with SU-8 2025 negative photoresist at 1,100 rpm to create the first 60 µm layer. After a soft bake at 65 °C for 3 min and 95 °C for 10 min on a hot plate, expose the mold using a maskless aligner by uploading the design file of the core channel pattern to the µPG 101 PC. Then, proceed with the post-exposure bake at 65 °C for 3 min and at 95 °C for 10 min.
- Spin coat the second SU-8 2025 layer (40 µm) at 1,500 rpm and expose by uploading the appropriate CAD file for the shell channel pattern.
NOTE: Soft and post-exposure bakes were similar to the previous step. - Spin coat the third SU-8 2025 layer at 1,400 rpm to achieve 50 µm height and repeat the above process but expose the structures for the oil phase.
- Develop the mold with all three layers in a single step by submersion in SU-8 developer until all the unexposed photoresist is removed.
- Hard bake the mold on a hot plate at 160 °C for 10 min to improve adhesion and seal minor cracks that could appear after the development.
- Expose the mold to chlorotrimethylsilane to avoid elastomer bonding in the next step and place it in 15 cm Petri dishes until use.
- Prepare the dissociation device mold in the same manner, as described in Figure 1D and Figure 2B. Spin coat one layer of SU-8 photoresist on a silicon wafer, as described earlier in step 1.2.3, to achieve structures with the desired height: 50 µm. Conduct the soft and post-exposure bake, development, and hard bake similarly to the previous step.
NOTE: An array of triangular-shaped posts covered the whole chamber, with spacing between posts decreasing as chamber width decreased toward the outlet. Triangle posts were 200 µm per side with a pitch ranging from 400 µm (at the inlet) to 30 µm (at the outlet). - Prepare the molds by soft lithography using polydimethylsiloxane (PDMS).
- Mix PDMS elastomer base and elastomer curing agent in a 10:1 w/w ratio in a planetary centrifuge. Pour the mixture onto both microencapsulation master molds and dissociation master mold to create a 3-4 mm layer.
NOTE: A mixture of 50 g of elastomer base with 5 g of curing agent is sufficient to cover a master mold with 3 mm thickness. - Place the Petri dishes containing the molds in a vacuum desiccator for 15 min to de-gas the uncured PDMS.
- Move the Petri dishes to an oven and bake at 80 °C for a minimum of 60 min.
- Remove the master molds from the oven and allow them to cool down. Using a precision knife, carefully cut the PDMS following the shape of the wafer and peel out the PDMS pieces from the master mold.
- Cut out the PDMS slabs by trimming the edges of each device with a scalpel, then punch out inlet/outlet fluidic ports in the dissociation device and only in the top microfluidic device using stainless steel needles. Cover the devices with magic tape to avoid contamination.
NOTE: The needle gauge is 14 G and 15 G for inlet and outlet ports, respectively. - For bonding, treat the patterned sides of the top and bottom PDMS pieces with O2 plasma on a plasma etcher for 2 min.
- Align both PDMS parts under a stereoscope after adding a thin separating layer of distilled water directly in the microchannels (2 µL).
- Place the aligned PDMS device in the oven at 80 °C overnight to complete the bonding and restore the PDMS to its original hydrophobic state. Store the devices until further use.
NOTE: This results in a coaxial flow-focusing device with three different heights of 120 µm for core, 200 µm for shell, and 300 µm for oil channels. - In order to use the device right after the plasma bonding, carefully but firmly inject 30 µL of hydrophobic coating solution into the fluidic channels to achieve hydrophobic coating. Then, immediately purge air into the channels until no residues of the hydrophobic coating solution are observed in the device.
- Bond the dissociation device by first treating the patterned side of the device and a cleaned slide glass with O2 plasma for 2 min, and then assemble them for further curing in the oven at 80 °C overnight.
NOTE: Devices can be stored until further use.
- Mix PDMS elastomer base and elastomer curing agent in a 10:1 w/w ratio in a planetary centrifuge. Pour the mixture onto both microencapsulation master molds and dissociation master mold to create a 3-4 mm layer.
2. Preparation of solutions
- Stock solutions. First, prepare stock solutions (concentrated solutions) that will be diluted to lower concentrations for actual use.
NOTE: Stock solutions are used to save preparation time, conserve materials, reduce storage space, and improve accuracy when preparing a working solution in lower concentrations.- Prepare 30 mL of 2x core solution (16% PEG and 34% density gradient media): mix 4.8 g of 35 kDa PEG, 10.2 mL of density gradient media, and 19.8 mL of DMEM/F12 media. Filter this core solution using a 0.22 μm syringe filter.
- Prepare 50 mL of mineral oil with 3% surfactant: mix 48.5 mL of mineral oil and 1.5 mL of surfactant. Keep in a sterile condition after filtration through a 0.22 µm syringe filter.
- Prepare 50 mL of mineral oil with 0.5% surfactant: mix 49.75 mL of mineral oil and 0.25 mL of surfactant. Keep in a sterile condition.
- Prepare 1 M Dithiotheritol (DTT): dissolve 1.5425 g of DTT in 10 mL distilled water. Keep in a sterile condition.
- Prepare 1 M Triethanolamine (TEA): add 66.4 µL of TEA in 433.6 µL of distilled water. Keep in a sterile condition.
- Prepare 1% Pluronic F127 (PF127): dissolve 100 mg of PF127 in 10 mL of distilled water. Keep in a sterile condition.
- Working solutions
- Prepare a crosslinker emulsion by mixing 4.5 mL of mineral oil with 3% surfactant and 300 µL of 1 M DTT (1:15 ratio). Vortex the solution for 2 min and sonicate for 45-60 min in an ultrasonic bath at 20 °C. Load this solution to a 5 mL syringe with a 27 G needle.
NOTE: Emulsion is generated by sonicating oil/water mixture. - Prepare shielding oil solution by loading the mineral oil with 0.5% surfactant to a 5 mL syringe with a 27 G needle.
- Prepare shell (8% w/v PEG-4-Mal and 15 mM TEA) solution by adding 32 mg of PEG-4-Mal in 400 µL of 1x DPBS to form 8% w/v solution, and vortex the solution for 2 min. Then, add 6 µL of 1 M TEA (final concentration 15 mM TEA). Finally, centrifuge at 13,000 x g for 5 min through a spin-filter and keep it in the dark on the rocker for 30 min. Then, load this solution to a 1 mL syringe with a 27 G needle.
NOTE: Leave the PEG-4-Mal container to sit at room temperature for 10 min before opening the lid, and blow N2 gas to replace the O2 with N2 before closing the lid. Vortex TEA before adding to the solution.
- Prepare a crosslinker emulsion by mixing 4.5 mL of mineral oil with 3% surfactant and 300 µL of 1 M DTT (1:15 ratio). Vortex the solution for 2 min and sonicate for 45-60 min in an ultrasonic bath at 20 °C. Load this solution to a 5 mL syringe with a 27 G needle.
- Prepare the cell suspension in core solution
- Treat the hPSCs with trypsin for 5 to 10 min at 37 °C. Then, collect them from plates. Quench the trypsin with the medium after cell detachment, and then transfer the cells to a 15 mL conical tube.
- Centrifuge the bulk cell suspension (400 x g, 5 min). Carefully aspirate the supernatant and then resuspend the cell pellet using a minimal volume of growth medium. Count the cells using an automated cell counter.
NOTE: Typical cell aliquots contain 12-20 x 106 cells and are enough to make 400 µL of the core solution. - Take 12-20 x 106 cells from the bulk cell suspension and centrifuge the cell/media suspension (400 x g, 5 min).
- Carefully aspirate media from the cell pellet. Then, add 200 µL media and 200 µL of 2x PEG solution (final concentration 30-50 x 106 cell/mL) and mix gently.
NOTE: Low cell concentrations (less than 20 x 106 cells/mL) resulted in many empty capsules. - Add 30 µL of 1% PF127 to the solution.
- Insert a micro stirrer bar (2 mm x 7 mm) into a 1 mL syringe, and then load the cell containing core solution to the syringe with a 27 G needle. Keep the solution cold with ice during the whole encapsulation process.
3. Experimental setup
- Place bonded PDMS droplet device, four pieces of 20 cm long and one piece of 5 cm long inlet micro tubes (0.38 mm I.D. x 1.1 mm O.D.), one piece of 10 cm outlet tube (0.5 mm I.D. x 1.5 mm O.D.), and six pieces of 0.5 cm fitting tubes (1 mm I.D. x 1.8 mm O.D.) beneath a germicidal light source (typically built into biological safety cabinets) and sterilize for 30 min.
- Use forceps to fit the tubing into the fluidic inlet ports and the dissociation ports.
- Use the three pieces of 20 cm long inlet microtubes for the shell, oil, crosslinker emulsion inlets, and one piece for the inlet of the dissociation device. Then, insert the opposite end of the tubes to the needle of the syringe with the corresponding solution. Meanwhile, connect the core inlet of the microfluidic device and the outlet of the dissociation device with one 5 cm microtube (Figure 1C).
NOTE: The inlet microtubes should be tightly secured within the fitting tubes, which were inserted in the last step. - Insert one outlet tube into the fluidic outlet port and place the opposite end into a collection reservoir.
- Place the device on a microscope stage and attach the tubing to avoid moving. Align and focus the microscope on the device nozzle for flow inspection.
- Place each syringe on different syringe pumps and secure properly (Figure 1B). Program each pump according to manufacturer's protocols to the following flow rates: core (3-5 µL/min), shell (3-5 µL/min), shielding oil (20-80 µL/min), and crosslinking oil (40-60 µL/min). Start the flow in all pumps.
- Wait for each fluid to enter the device and fill up the channels while pushing out the remaining air.
NOTE: If there is a large amount of air in the inlet tubing, temporarily increase each flow rate until the air is completely removed. Be mindful that excessive flow rate could lead to PDMS-to-PDMS bond failure. - When the capsule formation is stabilized (Figure 3), place the opposite end of the collection tube to a new conical tube with 5 mL of mTeSR media.
NOTE: The media contains 10 μM ROCK inhibitor, 100 U/mL penicillin, and 100 μg/mL streptomycin. - After a sufficient number of capsules are collected, incubate at 37 °C with 5% CO2 for 10 min. Capsules residing in the oil phase will be at the top of the tube (see Figure 4A). Gently tapping on the tube causes capsules to sediment to the bottom. After this, most of the oil and media may be aspirated.
- Collect the capsules from the bottom of the conical tube, place them on a cell strainer (100 μm pore size), and wash with copious amounts of media. Then, collect microcapsules using 2 mL media in a 6-well culture plate by running the media through the filter as it is inverted on top of the well-plate (see Figure 4A).
4. hPSC culture and analysis in microcapsules
- Basic culture
- Incubate the stem cell capsules in a 6-well plate culture dish at 37 °C with 5% CO2.
- Change the media every other day by collecting the capsules on a cell strainer filter, washing them with excess media, and resuspending them to a new well in 6-well plates with 2 mL media as was done in step 3.10.
- Culture the capsules for at least 10 days.
- Perform the live/dead assay to determine viability of encapsulated stem cells.
- Thaw live/dead assay solutions (ethidium homodimer-1 and calcein AM).
- Add 4 µL of the 2 μM ethidium homodimer-1 and 1 µL of the 4 μM calcein AM solutions to 2 mL of sterile DPBS. Vortex to ensure complete mixing.
- Collect approximately 100 microcapsules on a cell strainer and rinse them with sterile DPBS to allow diffusion of medium out from the capsules.
- Collect them again to a 12 well culture plate by using 1 mL of the solution prepared in step 4.2.2. Incubate at 37 °C for 20 min.
- Visualize the cells under a fluorescence microscope.
NOTE: Cell viability is quantified by calculating the percentage of live cells, which are visualized as green, among the total number of cells.
- Spheroid size characterization.
- When desired, place the well plate with microcapsules under the microscope and measure the diameter of spheroids shown on the related software with proper scale bars.
- Measure and analyze the spheroid size.
Representative Results
By following the above-mentioned protocol, the reader will be able to fabricate microfluidic devices and produce cell-carrying microcapsules. Figure 3A shows examples of optimal and suboptimal microcapsules fabricated using microfluidic droplet generation. Different formulations of PEG-4-Mal resulted in capsules of varying morphologies – wrinkled capsules were associated with poor gelation, low mechanical integrity, and did not withstand cultivation in a stirred bioreactor. Smooth capsules observed at PEG content of >6% represented the desired capsule morphology and were robust enough for cell cultivation. The core-shell structure was confirmed by incorporation of microbeads into the core and fluorescent moieties into the shell of microcapsules. Aggregation of beads in the center of the capsules as seen in Figure 3B was used as indication that the core was aqueous, and the beads were free to move about. Fluorescent annulus observed in Figure 3C indicated the presence of a hydrogel shell and was used to determine shell thickness. We recommend that users employ microbead incorporation and fluorescent labeling in the early stages of establishing the encapsulation process.
Once the encapsulation process is established, one can move onto encapsulation of hPSCs. While this protocol describes encapsulation of H9 cells, we have used a similar strategy for encapsulating another hESC line (HUES-8) and an iPSC line (1016).7
While encapsulation may be carried out using only the encapsulation device, we noted that hPSCs tended to clump in the system resulting in non-uniform encapsulation or capsule occupancy. To address this challenge, we designed a dissociation or filtration device and positioned it upstream of the encapsulation device. This dissociation device comprised a flow channel with an array of triangle-shaped posts measured 200 µm per side and with pitch ranging from 400 µm at the inlet to 30 µm at the outlet such that clumps are either retained or broken up before entering the encapsulation device (see Figure1D)7. The use of the dissociation device allows to significantly improve the uniformity of spheroids and increase cell occupancy of capsules from 57% to >90%7.
Figure 4B,C describes representative results from an encapsulation run. As can be seen from these images, H9 cells have high viability with >95% viability achieved routinely during the encapsulation runs. We compared growth rate (see Figure 4B,C) and differentiation potential of encapsulated vs. unencapsulated spheroids7, and determined that the process of encapsulation has no adverse effects on hPSCs. On the other hand, there are several benefits to encapsulating hPSCs. Encapsulated spheroids are easy to handle and may be dispensed/distributed into microtiter plates for developing differentiation protocols or testing therapies. Our lab is interested in using microcapsules as stem cell carriers for suspension cultivation in stirred bioreactors and has demonstrated that hydrogel shell offers protection against shear damage in such cultures7. This is an additional avenue being pursued by us is in creating bioactive microcapsules where hydrogel shell may be loaded with growth factors for local and continuous delivery of inductive cues during differentiation.
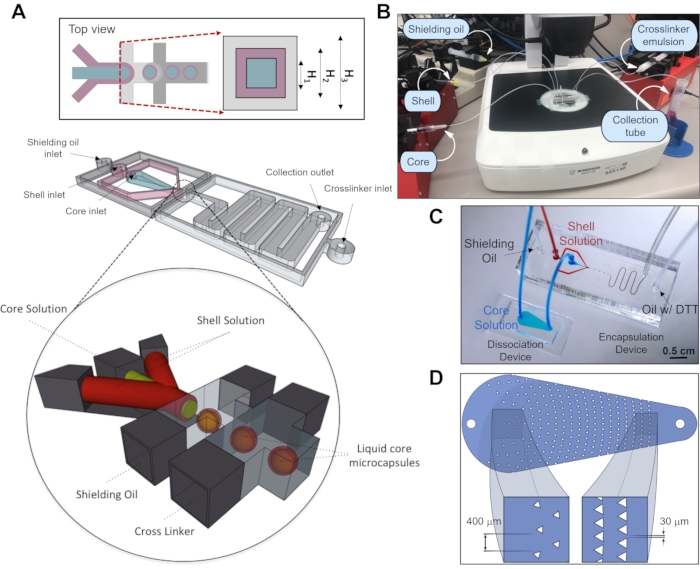
Figure 1: Fabrication of core−shell microcapsules. (A) The microfluidic device for capsule generation consists of four inlet channels (core, shell, oil, and crosslinker oil) and a serpentine channel that leads to a collection tube. The device is fabricated such that core, shell, and oil channels are 120 µm (H1), 200 µm (H2), and 300 µm (H3) in height, respectively. The inset represents a cross-sectional view at the nozzle – the junction between aqueous and oil streams. This cross-sectional view is tilted by 90° to give the reader a better view of coaxial flow channels. A 3D representation of the core-shell microcapsule fabrication process depicts how the coaxial flow is generated upstream of the flow-focusing junction to produce core-shell microcapsule structure. Emulsification is achieved by exposing aqueous droplets to two oil streams, the first of which is designed to stabilize the droplets, and the second to provide the crosslinker DTT, which reacts with PEG-4-Mal. This figure has been modified from reference12. Copyright 2019, American Chemical Society. (B) Experimental setup of the encapsulation system showing locations of syringe pumps, tubing, microfluidic devices and of capsule collection tube. (C) An image of the encapsulation system, which consists of dissociation and encapsulation devices in a sequence. (D) Design of the microfluidic dissociation device used to avoid large cell aggregates entering microencapsulation device. Please click here to view a larger version of this figure.
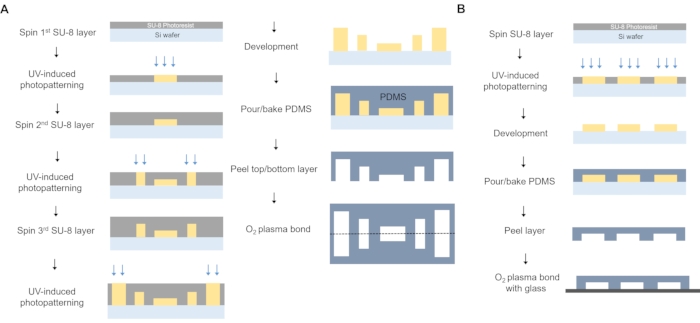
Figure 2: Fabrication of microfluidic devices used for encapsulation. (A) Fabrication of the encapsulation device. Three layers of SU-8 photoresist were spin-coated, and photo patterned to generate core, shell, and oil channels with different heights. Core channels have a width of 220 µm that narrows at the capsule junction to 135 µm. Channels for all phases follow the same principle: shell and oil channels start with a width of 500 µm, narrowing at the junction to 220 µm, and shielding oil starts at 500 µm narrowing to 270 µm. Outlet serpentine has a width of 1.5 mm and a length of ~55 mm. Top and bottom PDMS pieces were then aligned, and plasma bonded. (B) Fabrication of the dissociation device. One layer of SU-8 photoresist was spin-coated, and photo patterned to generate dissociation channels. PDMS piece was then plasma bonded with glass slide. Dissociation device consists of a small chamber with a height of 30 µm, length of 16.5 mm, and width of 5 mm at the inlet and 1.7 mm at the outlet. It has triangle shaped structures (200 µm, equilateral) that are separated from each other by 420 µm of the inlet and become closer on the way to the outlet, with a separation of 50 µm in the last row. Please click here to view a larger version of this figure.

Figure 3: Characterization of microcapsules. (A) Differences in capsule morphology as a function of PEG-4-Mal content in the shell. Smooth capsules with bright edges are associated with desired mechanical integrity. (B) Confirmation of aqueous core of the microcapsules. Entrapped microbeads are free to move in the aqueous core and aggregate in the center of microcapsules. (C) Confirmation of core-shell structure by incorporating Rhodamine B-labeled PEG into the shell of microcapsules. Please click here to view a larger version of this figure.
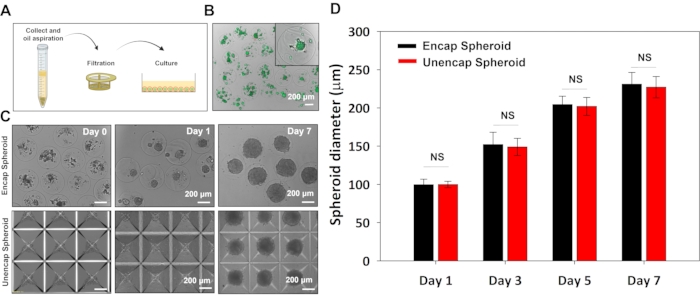
Figure 4: Encapsulation of hPSCs. (A) Microcapsules in the oil phase are collected into a conical tube filled with media. After microcapsules are made to settle to the bottom of the tube, oil and media are aspirated, microcapsules are washed and then transferred to a 6-well plate for cultivation. (B) Live/dead staining 6 h after encapsulating H9 cells. Upper image was taken at 10x, and lower image at 20x. (C) Images comparing spheroid sizes that change over time for H9 cells in capsules and in commercial 3D culture plates (bottom images). (D) Quantification of spheroid diameter that increases for hPSCs in microcapsules and in standard 3D culture plates during 7 days in H9 media. Statistical analysis –t-test, p < 0.05, n = 20. Scale bar: 200 µm. Please click here to view a larger version of this figure.
Discussion
The encapsulation process described here results in reproducible formation of hPSC spheroids. The microcapsule format makes it easy to dispense spheroids into wells of a microtiter plate for experiments aimed at improving/optimizing differentiation protocols or testing therapies. Encapsulated stem cell spheroids may also be used in suspensions cultures where hydrogel shell protects cells against shear-induced damage7.
There are several critical steps within the protocol. It is important to keep the flow rates of core, shell, and oil streams within the range described in the Protocol section. If the shell flow rate is too low, the flow becomes unstable, resulting in distorted and mechanically unstable capsules. A successful cell encapsulation should look similar to the microcapsules shown in Figure 4C. Microcapsules should be of similar diameter and have a comparable number of cells trapped in the core. Aggregation of single cells into spheroids typically occurs within 48 h and lack of spheroid formation at 72 h is a warning sign. One reason for lack of spheroid formation may be that viscous molecules in the core are not leaching out rapidly enough through the hydrogel shell. We encountered this scenario when adjusting viscosity of the core solution using high molecular weight polymers such as carboxymethyl cellulose (MW 250 kD). One can encapsulate microbeads (see Figure 3B) to test whether these cell-sized objects are free to move about the core and how rapidly components of the core leach out. Lack of spheroid formation was also encountered by us when using PEG-4-Mal concentrations of >8% w/v. In this scenario, porosity and diffusivity of the hydrogel shell are decreased and high viscosity elements in the core do not leach out rapidly enough to allow cells to form into aggregates. It is conceivable that the quality of PEG-4-Mal may vary from batch to batch or manufacturer. Therefore, some adjustment of polymer concentration in the shell may be needed. Once again, bead incorporation method should reveal how rapidly viscous components in the core are displaced by water molecules. In our experience, this should occur within 3 h of transferring microcapsules into the aqueous environment.
The encapsulation protocol described here has worked well for several hPSC lines7, primary hepatocytes10, and multiple cancer cell lines (unpublished results). However, we did encounter difficulties forming spheroids after encapsulation of stem cell-derived (sc)-hepatocytes and sc-β-cells. Troubleshooting with encapsulated microbeads revealed that viscous components were rapidly eluted from the core and that cells were free to move and form cell-cell contacts. Additional troubleshooting involved titrating concentration of di-thiol crosslinker DTT (present in the oil phase) down from 66 mM to 10 mM. Spheroid formation for sc-hepatocytes and β-cells did occur at this lower concentration of DTT. Therefore, the user may want to consider optimizing crosslinker concentration but only if other troubleshooting steps listed above have not been successful. We find that DTT concentration is critical for maintaining mechanical integrity of microcapsules with 66 mM DTT resulting in mechanically robust microcapsules and being non-toxic to vast majority of cell types used to date.
A number of strategies for creating 3D stem cell constructs have been described in the literature. The benefits of forming stem cell constructs within core-shell microcapsules are multiple. Once encapsulated, spheroids may be handled with ease with hydrogel capsules providing protection against shear damage. Encapsulated spheroids may be cultured in suspension with vigorous agitation without damaging stem cells.
There are multiple avenues for expanding the capabilities of microcapsules. Our team has previously described the use of heparin-containing hydrogels for encapsulation of hepatocytes and stem cells8,12, and is presently developing heparin-containing bioactive core-shell microcapsules that serve as depots for storage and local release of inductive cues (GFs) to stem cells. Such microcapsules may help decrease the usage of expensive GFs while improving differentiation outcomes. Microcapsules may be further enhanced by incorporating ECM proteins into the core thus modulating stem cell microenvironment. Core-shell microcapsules may be made degradable to make it easier to retrieve hPSC spheroids13,14. Overall, core-shell microcapsules represent a platform technology that can be modified and enhanced to address the needs of a specific stem cell differentiation protocol.
Declarações
The authors have nothing to disclose.
Acknowledgements
This study was supported in part by the grants from the Mayo Clinic Center for Regenerative Medicine, J. W. Kieckhefer Foundation, Al Nahyan Foundation, Regenerative Medicine Minnesota (RMM 101617 TR 004), and NIH (DK107255).
Materials
| 0.22 µm Syringe Filters | Genesee Scientific | 25-244 | |
| 1 ml syringe luer-lock tip | BD | 309628 | |
| 1x DPBS | Corning | 23220003 | |
| 4-arm PEG maleimide, 10kDa | Laysan Inc. | 164-68 | |
| 5 ml syringe luer-lock tip | BD | 309646 | |
| 6-WELL NON-TREATED PLATE | USA Scientific | CC7672-7506 | |
| Aquapel Applicator Pack | Aquapel Glass Treatment | 47100 | |
| CAD software | Autodesk | AutoCAD v2020 | |
| CELL STRAINER 100 µm pore size | cardinal | 335583 | |
| Chlorotrimethylsilane | Aldrich | 386529-100mL | |
| Countess II FL Automated Cell Counter | Life technology | A27974 | |
| Digital hot plate | Dataplate | ||
| Digital vortex mixer | Fisher Scientific | 215370 | |
| Distilled water | Gibco | 15230-162 | |
| Dithiotheritol (DTT) | Sigma | D0632-10G | |
| DMEM/F12 media | gibco | 11320-033 | |
| Falcon 15 mL Conical Centrifuge Tubes | Fisher scientific | 14-959-53A | |
| Fisherbrand accuSpin Micro 17 Microcentrifuge | live | 13-100-675 | |
| HERACELL VIOS 160i CO2 Incubator | Thermo Scientific | 50144906 | |
| Inverted Fluorescence Motorized Microscope | Olympus | Olympus IX83 | |
| Laurell Spin Coaters | Laurell Technologies | WS-650MZ-23NPPB | |
| Live/Dead mammalian staining kit | Fisher | L3224 | |
| Magic tape | Staples | 483535 | |
| Micro Medical Tubing (0.015" I.D. x 0.043" O.D.) | Scientific Commodities, Inc | BB31695-PE/2 | |
| Micro stir bar | Daigger Scientific | EF3288E | |
| MilliporeSigma Filter Forceps | Fisher scientific | XX6200006P | |
| Mineral oil | Sigma | M8410-1L | |
| mTeSR 1 Basal Medium | STEMCELL TECHNOLOGY | 85850 | |
| Needles-Stainless Steel 14 Gauge | CML supply | 901-14-025 | |
| Needles-Stainless Steel 15 Gauge | CML supply | 901-15-050 | |
| OptiPrep | STEMCELL TECHNOLOGY | 7820 | |
| Oven | Thermo Scientific | HERA THERM Oven | |
| Penicillin:Streptomycin (10,000 U/mL Penicillin G, 10mg/mL Streptomycin) | Gemini | 400-109 | |
| Petri Dish 150X20 Sterile Vent | Sarstedt, Inc. | 82.1184.500 | |
| Plasma Cleaning System | Yield Engineering System, Inc. | YES-G500 | |
| Pluronic F-127 | Sigma | P2443-250G | |
| Poly(ethylene glycol) 35kDa | Sigma | 94646-250G-F | |
| PrecisionGlide Needle 27G | BD | 305109 | |
| Rock inhibitor Y-27632 dihydrocloride | SELLECK CHEM | S1049-10mg | |
| Silicon wafer 100mm | University Wafer | 452 | |
| Slide glass (75mm ´ 25mm) | CardinalHealth | M6146 | |
| Span 80 | Sigma | S6760-250ML | |
| SpeedMixer | Thinky | ARE-310 | |
| Spin-X Centrifuge Tube Filter (0.22 µm) | Costar | 8160 | |
| SU-8 2025 | Kayaku Advanced Materials | Y111069 0500L1GL | |
| SU-8 developer | Kayaku Advanced Materials | Y020100 4000L1PE | |
| Surgical Design Royaltek Stainless Steel Surgical Scalpel Blades | fisher scientific | 22-079-684 | |
| SYLGARD TM 184 Silicone Elastomer Kit (PDMS) | Dow Corning | 2065622 | |
| Syringe pump | New Era Pump System, Inc | NE-4000 | |
| Triethanolamine | Sigma-aldrich | T58300-25G | |
| TrypLE Express | Gibco | 12604-013 | |
| Tygon Tubing (0.02" I.D. x 0.06" O.D.) | Cole-Parmer | 06419-01 | |
| Tygon Tubing (0.04" I.D. x 0.07" O.D.) | Cole-Parmer | 06419-04 | |
| Ultrasonic cleaner FS20D | Fisher Scientific | CPN-962-152R | |
| Vacuum desiccator | Bel-Art | F42025-0000 | |
| Zeiss Stemi DV4 Stereo Microscope 8x-32x | ZEISS | 435421-0000-000 | |
| μPG 101 laser writer | Heidelberg Instruments | HI 1128 |
Referências
- Zhu, Z., Huangfu, D. Human pluripotent stem cells: an emerging model in developmental biology. Development. 140 (4), 705-717 (2013).
- Liu, G., David, B. T., Trawczynski, M., Fessler, R. G. Advances in pluripotent stem cells: history, mechanisms, technologies, and applications. Stem Cell Reviews and Reports. 16 (1), 3-32 (2020).
- Chan, S. W., Rizwan, M., Yim, E. K. Emerging methods for enhancing pluripotent stem cell expansion. Frontiers in Cell and Developmental Biology. 8, 70 (2020).
- Lei, Y., Schaffer, D. V. A fully defined and scalable 3D culture system for human pluripotent stem cell expansion and differentiation. Proceedings of the National Academy of Sciences of the United States of America. 110 (52), 5039-5048 (2013).
- Olmer, R., et al. Suspension culture of human pluripotent stem cells in controlled, stirred bioreactors. Tissue Engineering Part C: Methods. 18 (10), 772-784 (2012).
- Kraehenbuehl, T. P., Langer, R., Ferreira, L. S. Three-dimensional biomaterials for the study of human pluripotent stem cells. Nature Methods. 8 (9), 731-736 (2011).
- Fattahi, P., et al. Core-shell hydrogel microcapsules enable formation of human pluripotent stem cell spheroids and their cultivation in a stirred bioreactor. Scientific Reports. 11 (1), 1-13 (2021).
- Siltanen, C., et al. Microfluidic fabrication of bioactive microgels for rapid formation and enhanced differentiation of stem cell spheroids. Acta Biomaterialia. 34, 125-132 (2016).
- Agarwal, P., et al. One-step microfluidic generation of pre-hatching embryo-like core-shell microcapsules for miniaturized 3D culture of pluripotent stem cells. Lab on a Chip. 13 (23), 4525-4533 (2013).
- Siltanen, C., et al. One step fabrication of hydrogel microcapsules with hollow core for assembly and cultivation of hepatocyte spheroids. Acta Biomaterialia. 50, 428-436 (2017).
- Rahimian, A., Siltanen, C., Feyzizarnagh, H., Escalante, P., Revzin, A. Microencapsulated immunoassays for detection of cytokines in human blood. ACS Sensors. 4 (3), 578-585 (2019).
- Kim, M., Lee, J., Jones, C. N., Revzin, A., Tae, G. Heparin-based hydrogel as a matrix for encapsulation and cultivation of primary hepatocytes. Biomaterials. 31, 3596-3603 (2010).
- Shin, D. S., et al. Photodegradable hydrogels for capture, detection, and release of live cells. Angewandte Chemie International Edition. , (2014).
- You, J., et al. Bioactive photodegradable hydrogel for cultivation and retrieval of embryonic stem cells. Advanced Functional Materials. , (2015).

