A Simple Pit Assay Protocol to Visualize and Quantify Osteoclastic Resorption In Vitro
Summary
Here, we present a simple and effective assay procedure for resorption pit assays using calcium phosphate coated cell culture plates.
Abstract
Mature osteoclasts are multinucleated cells that can degrade bone through the secretion of acids and enzymes. They play a crucial role in various diseases (e.g., osteoporosis and bone cancer) and are therefore important objects of research. In vitro, their activity can be analyzed by the formation of resorption pits. In this protocol, we describe a simple pit assay method using calcium phosphate (CaP) coated cell culture plates, which can be easily visualized and quantified. Osteoclast precursors derived from human peripheral blood mononuclear cells (PBMCs) were cultured on the coated plates in the presence of osteoclastogenic stimuli. After 9 days of incubation, osteoclasts were fixed and stained for fluorescence imaging while the CaP coating was counterstained by calcein. To quantify the resorbed area, the CaP coating on plates was stained with 5% AgNO3 and visualized by brightfield imaging. The resorption pit area was quantified using ImageJ.
Introduction
Osteoclasts (OCs) are tissue-specific macrophages derived from hematopoietic stem cells (HSCs), playing a pivotal role in bone remodeling together with osteoblasts1. Sex hormone-induced, immunological, and malignant bone disorders that destroy bone systemically or locally are due to excess osteoclastic activity, including menopause-related osteoporosis2, rheumatoid arthritis3, periodontal disease4, myeloma bone disease5, and osteolytic bone metastasis6. In contrast, defects in OC formation and function can also cause osteopetrosis7. HSCs undergo differentiation into OC progenitors under macrophage colony-stimulating factor (M-CSF, gene symbol ACP5) stimulation. In the presence of both M-CSF and receptor activator of NF-κB ligand (RANKL, gene symbol TNFSF11), OC progenitors differentiate further into mononuclear OCs and subsequently fuse to become multinucleated OCs8,9,10. Both cytokines M-CSF and RANKL are indispensable and sufficient for induction of osteoclastic markers such as calcitonin receptor (CT), receptor activator of nuclear factor κ B (RANK), proton pump V-ATPase, chloride channel 7 alpha subunit (CIC-7), integrin β3, tartrate-resistant acid phosphatase (TRAP, gene symbol ACP5), lysosomal cysteine protease cathepsin K (CTSK), and matrix metallopeptidase 9 (MMP9). Activated OCs form a sealing zone on the bone surface through the formation of an actin ring with a ruffled border11,12. Within the sealing zone, OCs mediate resorption through secreting protons via the proton pump V-ATPase12,13, MMP914, and CTSK15, leading to the formation of lacunae.
For in vitro experiments, OC progenitors can be obtained by expansion of bone marrow macrophages from mice's femur and tibia16,17, as well as by isolation of human peripheral blood mononuclear cells (PBMCs) from blood samples and buffy coats18,19,20, or by differentiation of the immortalized murine monocytic cells RAW 264.721,22.
In the present protocol, we describe an osteoclastic resorption assay in CaP coated cell culture plates using OCs derived from primary PBMCs. The CaP coated cell culture plates method used here are adopted and refined from the method described previously by Patntirapong et al.17 and Maria et al.21. To obtain OC precursors, PBMCs are isolated by density gradient centrifugation and expanded as described previously20.
Protocol
The protocol was reviewed and approved by the local ethics committee (approval number 287/2020B02).
1. Preparation of calcium phosphate coated cell culture plates
- Preparation of calcium stock solution (25 mM CaCl2·2H2O, 1.37 M NaCl, 15 mM MgCl2·6H2O in Tris buffer)
- Prepare 50 mM Tris buffer and adjust pH to 7.4 using 1 M HCl.
- Set up a glass beaker on a magnetic stirrer and add 100 mL of 50 mM Tris buffer.
- Weigh out 0.368 g of CaCl2·2H2O, 8.0 g of NaCl, and 0.305 g of MgCl2·6H2O and dissolve in Tris buffer one by one.
- Adjust pH to 7.4 using 1 M HCl. Store at room temperature.
- Preparation of phosphate stock solution (11.1 mM Na2HPO4, 42 mM NaHCO3 in Tris buffer)
- Set up a glass beaker on a magnetic stirrer and add 100 mL of 50 mM Tris buffer.
- Weigh out 0.158 g of Na2HPO4 and 0.353 g of NaHCO3 and dissolve in Tris buffer one by one.
- Adjust pH to 7.4 using 1 M HCl. Store at room temperature.
- Pre-calcification of 96-well cell culture plates
- Prepare 30 mL of working solution by mixing 15 mL of 50 mM Tris buffer, 7.5 mL of calcium stock (step 1.1.4) solution, and 7.5 mL of phosphate stock solution (step 1.2.3). Filter the solution with a 0.2 µm filter.
- Prepare a 96-well cell culture plate with flat bottom. Pipette 300 µL of calcium phosphate solution (step 1.3.1.) to each well. Cover the plate with a lid and incubate the plate at 37 °C for 3 days.
- Preparation of calcium phosphate solution (2.25 mM Na2HPO4, 4 mM CaCl2·2H2O, 0.14 M NaCl, 50 mM Tris base in ddH2O)
- Add 2 mL of 1 M HCl to 40 mL of deionized water in a beaker with a magnetic stirring bead and dissolve 0.016 g of Na2HPO4, 0.0295 g of CaCl2·2H2O, 0.409 g of NaCl, and 0.303 g of Tris base one by one.
- Adjust pH to 7.4 using 1 M HCl. Add deionized water to fill the volume to 50 mL. Filter the solution with a 0.2 µm filter.
- Calcification of 96-well cell culture plates
- Aspirate pre-calcification solution from the pre-calcified 96-well cell culture plates and add 300 µL of calcium phosphate solution (step 1.4.2) to each well. Cover the plates with a lid and incubate at 37 °C for 1 day.
- Turn the plate over to pour out the solution and wash the plate three times with deionized water thoroughly. Dry the plate immediately using a hair dryer or compressed CO2 or N2 gas to maintain a uniform surface.
- Sterilize the coated plate by UV radiation in a clean bench for 1 h. Use the coated plate immediately or seal the plate with parafilm and store it at room temperature.
2. Isolation of PBMCs from human peripheral blood
- Draw 15 mL of blood from the vein after obtaining written informed consent from the blood donor (ethical vote: 287/2020B02).
- Dilute 15 mL of fresh blood with an equal volume of PBS and mix by inverting the tube several times or by drawing the mixture in and out of a pipette.
- Prepare a 50 mL conical tube containing 15 mL of density gradient solution (e.g., Ficoll, Table of Materials) per tube. Tilt the tube and carefully layer 30 mL of diluted blood sample onto the 15 mL of density gradient solution (diluted blood sample: density gradient solution, 1:0.5-1 ratio).
NOTE: When overlaying the sample, pay attention not to mix the density gradient solution with the diluted blood sample. - Centrifuge at 810 x g for 20 min at 20 °C in a swinging-bucket rotor without brake.
- Aspirate the upper layer leaving the mononuclear cell layer (lymphocytes, monocytes, and thrombocytes) undisturbed at the interphase.
- Carefully transfer the mononuclear cell layer to a new 50 mL conical tube.
- Fill the tube with PBS, mix, and centrifuge at 300 x g for 10 min at 20 °C (brake can be used from this stage onwards).
- Carefully remove supernatant completely. Resuspend the cell pellet in 50 mL of PBS and centrifuge at 300 x g for 10 min at 20 °C. Carefully remove the supernatant completely.
- For removal of platelets, resuspend the cell pellet in 50 mL of PBS and centrifuge at 200 x g for 10 min at 20 °C. Carefully remove the supernatant completely.
- Transfer resuspended cells to cell culture flasks for expansion of OC progenitors.
3. Expansion of OC progenitors
- Resuspend PBMCs in complete α-MEM (10% FBS, 1% Pen/Strep, 1% amphotericin B) containing 20 ng/mL M-CSF. Seed PBMCs at a density of 2.5 x 105 cells/cm2. Usually, cells isolated from 15-20 mL fresh blood can be seeded in one 75 cm2 flask (1.5-2 x 107 cells).
- Feed the cells with fresh complete α-MEM containing 20 ng/mL M-CSF every third day until the attached cells reach a desired confluency. Typical yields are 1.5-2 x 106 cells/flask when cells are 95% confluent. Usually, the expansion period lasts 6 days.
NOTE: The osteoclastogenic potential of the precursors can decrease with prolonged cultivation time.
4. Induction of osteoclastogenesis in CaP coated plates
- To facilitate cell adhesion, incubate the CaP coated plates with 50 µL of FBS for 1 h in a 37 °C incubator.
- To detach the OC precursors, wash the flask with PBS twice to remove dead or non-adherent cells. Add 4 mL of trypsin (Table of Materials) per 75 cm2 flask, for 30 min.
- Add 4 mL of complete α-MEM to stop the digestion, carefully detach the cells using a cell scraper and transfer the cells into a 50 mL tube.
- Determine the total cell number using a Neubauer chamber or similar.
- Pellet the cells by centrifugation for 7 min at 350 x g. Resuspend the pellet in sufficient complete α-MEM containing 20 ng/mL M-CSF and 20 ng/mL RANKL to get a concentration of 1 x 106 cells/mL.
- Aspirate FBS solution from the CaP coated 96-well plate and pipette 200 µL of cell suspension per well (2 x 105 cells/well).
- Incubate the OC precursors for the desired time period or with the desired reagents relevant to the experimental design. Usually, high numbers of large and multinucleated OCs can be observed after 6 days and when resorption pits are forming. Feed cells with fresh complete α-MEM medium containing 20 ng/mL M-CSF and 20 ng/mL RANKL every third day.
- At the end of incubation, wash cells with PBS twice, fix with 4% paraformaldehyde for 10 min, and wash with PBS again.
- Use the fixed OCs directly for fluorescence staining or store at 4 °C.
5. Fluorescence staining of OCs and CaP coating
- Incubate the fixed cells with permeabilization buffer (0.1% Triton in PBS) for 5 min.
- Stain actin filaments with 100 µL of AlexaFluor 546 labeled phalloidin solution in PBS for 30 min and aspirate the staining solution. Add 100 µL of Hoechst 33342 staining solution (10 µg/mL in PBS) for 10 min to stain nuclei. DAPI can also be used.
- Stain CaP coating with 100 µL of 10 µM calcein in PBS for 10 min. Wash three times with PBS and take images.
- After fluorescence imaging the same plates can be used to quantify resorption pit area by Von Kossa staining. To do so, wash the plates with deionized water twice.
6. Quantification of total resorption pit area
- To stain CaP coating with Von Kossa staining, incubate with 50 µL of 5% AgNO3 in deionized water per well under UV radiation for 1 h, until the coating on the bottom of the wells has turned brown.
- Wash the plate three times with deionized water and take brightfield images. An objective with little magnification, such as a 1.25x objective, can be used to capture the whole well in one image. This facilitates subsequent image analysis. Alternative methods to capture the whole area of the well can be applied.
- Open an image file with ImageJ: File | Open | Image | Type | 8-bit. Verify the scale unit in the lower right corner of the image using Straight Line | Analyze | Set Scale. Enter the length in the "known distance" blanket, enter new scale unit in Unit of length, and check the box Global.
- List measurement parameters to be analyzed: Analyze | Set Measurements. In the Set Measurements window, check the Area and Limit to threshold boxes.
- Measure the area of pits: Image | Adjust | Threshold. In the Threshold window, check box Dark background and click Auto. The area of pits turns red. Close the Threshold window and select Analyze | Measure. Save the result file: File | Save As.
7. Quantification of OC number and size, and normalized resorption pit area
- Open a fluorescence image of osteoclasts with ImageJ and verify the scale unit (see step 6.3).
- List measurement parameters to be analyzed: Analyze | Set Measurements. In the Set Measurements window, check the Area box.
- Outline the OCs using ROI Manager and Polygon selections: Analyze | Tools | ROI Manager. Click Polygon selections in toolset, outline one OC (cells with actin ring and ≥ three nuclei) and click Add [t] in ROI Manager window. Repeat outlining until all OCs are included.
- Measure number and size of OCs: Select all items in ROI Manager and click Measure. Save the result file: File | Save As (Figure 3E).
- Measure the pit area of fluorescence images. Open the fluorescent image of coating correlated with the image in step 7.3 and verify the scale unit (see step 6.3).
- List measurement parameters to be analyzed (see step 6.4). Select Image | Adjust | Threshold. In the Threshold window, uncheck all boxes and click Auto. The area of the pits turns red. Close the Threshold window and select Analyze | Measure. Save the result file.
- Calculate normalized pit area by OC number.
Representative Results
The calcium phosphate coating on the bottom of cell culture plates was performed in two coating steps comprising a 3-day pre-calcification and a 1-day calcification step. As shown in Figure 1, uniformly distributed calcium phosphate was obtained on the bottom of the 96-well plates. The coating adhered very well to the bottom after the performed washing steps.
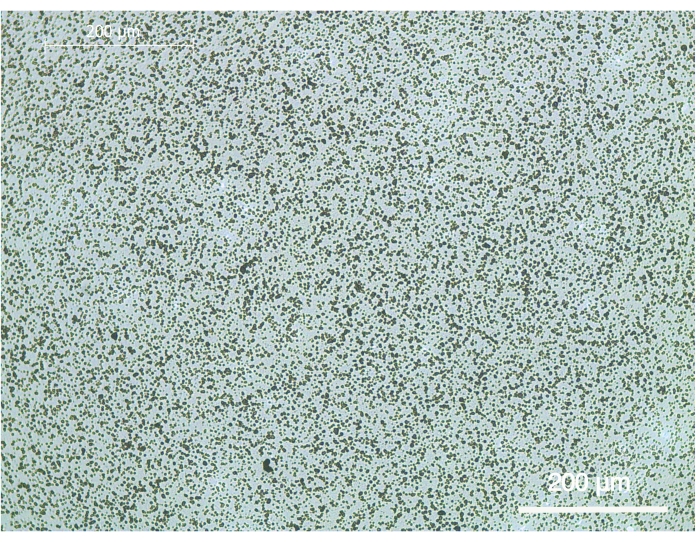
Figure 1: Representative brightfield image of the calcium phosphate coating on 96-well cell culture plates. The coating was carried out in two coating steps comprising a 3-day pre-calcification step and a 1-day calcification step. The scale bar represents 200 µm. Please click here to view a larger version of this figure.
OC precursors derived from human PBMCs were cultured on the CaP coated plates. Resorption pits and large OCs (blank area) were formed in the presence of M-CFS and RANKL after 9 days of culture (Figure 2A). The multinucleated OCs located in the pits of the CaP coating expressed high levels of TRAP (Figure 2B).
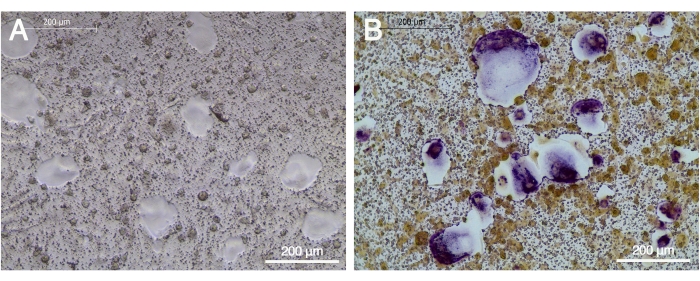
Figure 2: TRAP expression by OCs after maturation on the CaP coated cell culture plates. OC precursors were cultured on the CaP coated plates in the presence of 20 ng/mL M-CSF and 20 ng/mL RANKL for 9 days. (A) Blank area represented resorption pits. (B) Several multinucleated TRAP-positive (purple) OCs were observed within the resorption pits. The scale bar represents 200 µm. Please click here to view a larger version of this figure.
To further characterize the maturation of OCs on the CaP coated plates, cells were stained for actin (red fluorescence). Cell nuclei were stained with Hoechst (blue) and calcein was used for calcium visualization in green. Resorption pits are visible as black areas in the green CaP coating (Figure 3B). Functional mature OCs showed three or more nuclei and the characteristic actin ring which is essential for the osteoclastogenic resorption (Figure 3A,C). The pit area of fluorescence images can be measured using the thresholding tool of ImageJ (Figure 3D). The number and size of OCs can be calculated using the ROI Manager and Polygon selection tool of ImageJ (Figure 3E).
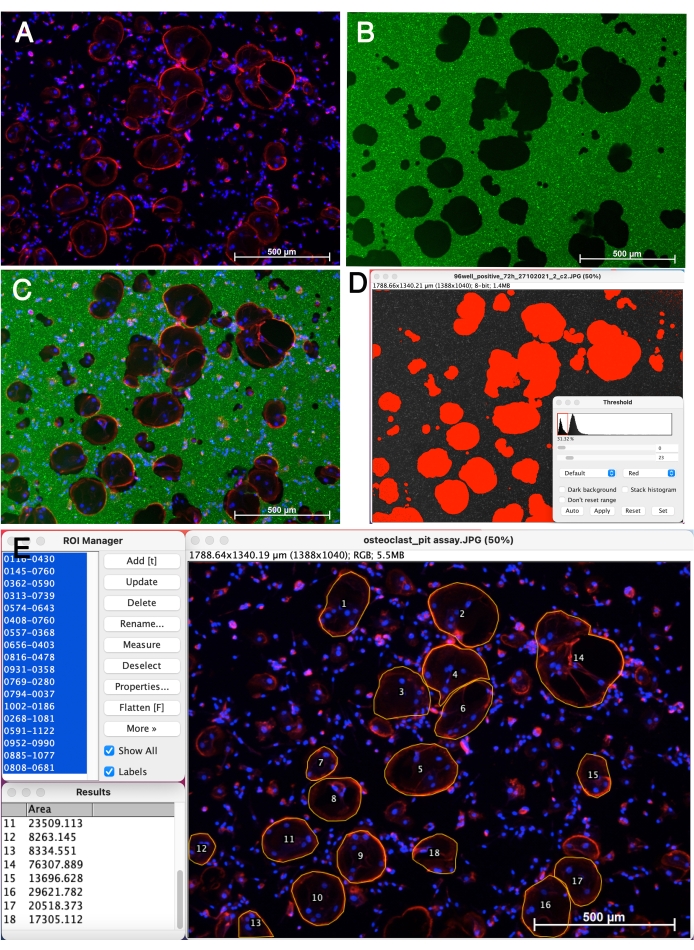
Figure 3: Morphology of mature OCs on the CaP coating. OC precursors were cultured on the CaP coated plates for 9 days in the presence of 20 ng/mL M-CSF and 20 ng/mL RANKL. (A) OCs were stained for actin and nuclei by phalloidin-Alexa Fluor 546 and Hoechst 33342. (B) CaP coating was stained by calcein. Black areas represent resorption pits. (C) Merged image. (D) Quantification of pit area (red area) using the thresholding tool of ImageJ. (E) OC outlining and counting using the ROI Manager and Polygon selection tool of ImageJ. The scale bar represents 500 µm. Please click here to view a larger version of this figure.
For the quantification of the resorption pits on the CaP coated cell culture plates, calcium was stained by incubation with 5% AgNO3 (Von Kossa staining). As shown in Figure 4A, OC precursors did not reach full functionality in the absence of RANKL and were not able to form resorption pits. Osteoclastogenic pits were observed only in the presence of both factors M-CSF and RANKL (Figure 4B). The resorption pit area was quantified with the thresholding tool of ImageJ (Figure 4C).
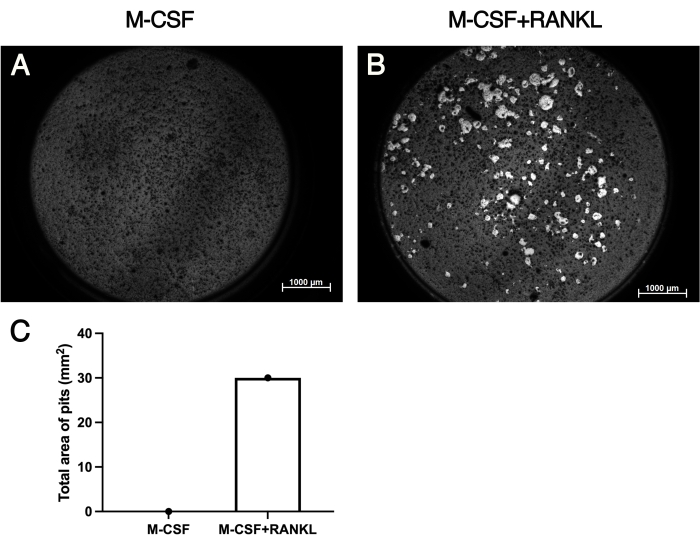
Figure 4: Visualization and quantification of resorption pits. OC precursors were cultured on the CaP coated 96-well cell culture plates in the absence of RANKL (A) and in the presence of both factors M-CSF and RANKL (B). (C) Numbers of formed resorption pits were quantified using the thresholding tool of ImageJ. Scale bar represents 1,000 µm. Please click here to view a larger version of this figure.
Discussion
Here we describe a simple and reliable method for an osteoclastic resorption assay using OCs derived and expanded in vitro from PBMCs. The used CaP coated cell culture plates can be easily prepared and visualized using lab-available materials. In addition to unsorted PBMCs adopted in this protocol, OCs generated from murine monocytic cells21 and bone marrow macrophage cells17 have also been cultured on similar synthetic substrates for pit assay, thus these cell sources can be moved to this approach referring to the corresponding literature.
For this protocol, we used a simple in-lab fabricated CaP coating instead of expensive commercially available assay plates. Although bone and dentin slices are two common and affordable resorbable materials for resorption pit assay, the availability and complicated preparation are major drawbacks. As an easily prepared synthetic substrate, CaP coating is more convenient and readily available than the two original materials. Another advantage of this pit assay approach is that cells can be visualized under a bright field microscope during culture, whereas cells growing on opaque bone and dentin slices cannot be observed.
This CaP coating method can be flexibly adapted according to the required experimental needs to different cell culture plate sizes.
Unlike previously reported17,21, we incubate the plates with coating solution at 37 °C instead of at room temperature, resulting in the spontaneous growth of apatite nuclei on cell culture plates at body temperature. CaP coated plates are available for experiments immediately after being dried and sterilized by UV radiation, which saves time compared to previously reported methods17,21.
Filtration of the coating solution (Step 1.3.1 and 1.4.2) is a crucial step, which helps to obtain a uniform particle size and reduce uncoated spots within the coating caused by air bubbles and impurities in the solution. Drying the plate immediately (step 1.5.2) is another critical step, which helps to make a uniform CaP coating. Otherwise, it is prone to form a thicker coating in the middle of the well.
It is worth pointing out that the thickness of the coating and the density of calcium phosphate deposits depend on the volume of coating solution within a certain range. Therefore, it is possible to control coating thickness by changing the volume of coating solution in the well. However, it is suggested to add as much volume of coating solution as possible when the 96-well cell culture plate is used because of the small total volume for this culture plate format. The inner wall of the well is also coated by CaP during both coating steps, therefore caution is required to not touch the bottom nor the wall with the pipette in order to protect the coating from damage.
According to our observation, most of OC precursors expanded from PBMCs are able to attach to the CaP coating without any problems. However, soaking the coated plate with FBS before cell seeding improves the cell adhesion efficiency, as previously reported21.
Detaching the OC precursors gently using a cell scraper (step 4.2) is also important because most of the OC precursors were still adherent, and minimizing the mechanical injury is favorable for cell viability.
A limitation of this method is that the bulk PBMCs that we used as a source of OC precursors is a highly mixed cellular source, of which only a small fraction of cells (CD14+ cells) are actual OC precursors. Since other cells capable of affecting osteoclastogenesis (such as stromal cells and lymphocytes) are present amongst isolated PBMCs, this may impact assay results in ways that may be hard to predict, and might confound assay interpretation. To better investigate direct effects of growth factors, cytokines or glucocorticoids on resorptive activity of OCs, methods of OC precursor purification (e.g., fluorescence-activated cell sorting (FACS) and magnetic beads purification20 based on specific cell-surface markers23,24,25,26) are recommended.
The limitations of the CaP substrate in obtaining brightfield images of both cells and underlying pits for the same area is caused by the incompatibility of TRAP and Von Kossa staining, so that normalized resorption area of the entire well is not available. But instead, fluorescence imaging can be used to visualize resorption area and cell number to obtain normalized resorption data. These data may provide important insight into whether the increase in total resorption area under experimental conditions is due to increased osteoclast number alone or increased resorption capacity of individual cells. In addition, it allows the study of the relative effects of osteoclast number, size, nucleus number, and resorption capacity.
In summary, we describe a useful and simple protocol for a resorption pit assay using a two-step calcium phosphate coating and OCs derived from primary human PBMCs. This protocol provides an easy method to establish an in vitro bone resorption model for studies related to osteoclastic resorption, and could be applied to study treatments for bone disorder diseases.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was partially funded by the China Scholarship Council [CSC No. 201808440394]. W.C. was financed by CSC.
Materials
| AgNO3 | SERVA Electrophoresis GmbH | 35110 | Silver nitrate |
| a-MEM | Gibco | 32561-029 | MEM alpha, GlutaMAX, no nucleosides |
| amphotericin B | Biochrom | 03-028-1B | Amphotericin B Solution |
| CaCl2 | Sigma-Aldrich | 21097-50G | Calcium chloride Dihydrate |
| Calcein | Sigma-Aldrich | C0875 | Calcein |
| FBS | Sigma-Aldrich | F7524 | fetal bovine serum |
| Ficoll | Cytiva | 17144002 | Ficoll Paque Plus |
| Fixation buffer | Biolegend | 420801 | Paraformaldehyde |
| HCl | Merk | 1.09057.1000 | Hydrochloric acid |
| Hoechst 33342 | Promokine | PK-CA707-40046 | Hoechst 33342 |
| M-CSF | PeproTech | 300-25 | Recombinant Human M-CSF |
| MgCl2 | Sigma-Aldrich | 7791-18-6 | Magnesium chloride |
| Na2HPO4 | AppliChem GmbH | A2943,0250 | di- Sodium hydrogen phosphate anhydrous |
| NaCl | Merk | S7653-250G | Sodium chloride |
| NaHCO3 | Merk | K15322429 | Bicarbonate of Soda |
| PBS | Lonza | 17-512F | Dulbecco's Phosphate Buffered Saline (1X), DBPS without Calcium and Magnesium |
| Pen-Strep | Lonza | DE17-602E | Penicillin-Streptomycin Mixture |
| Phalloidin-Alexa Fluor 546 | Invitrogen | A22283 | Alexa Fluor 546 Phalloidin |
| RANKL | PeproTech | 310-01 | Recombinant Human sRANK Ligand (E.coli derived) |
| Tris | Sigma-Aldrich | 93362 | Tris(hydroxymethyl)aminomethan |
| Triton X-100 | Sigma-Aldrich | T8787 | Alkyl Phenyl Polyethylene Glycol |
| TrypLE Express | Gibco | 12605010 | Recombinant cell-dissociation enzymes |
Referências
- Jacome-Galarza, C. E., et al. Developmental origin, functional maintenance and genetic rescue of osteoclasts. Nature. 568 (7753), 541-545 (2019).
- Moller, A. M. J., et al. Aging and menopause reprogram osteoclast precursors for aggressive bone resorption. Bone Research. 8 (1), 1-11 (2020).
- Yokota, K., et al. Characterization and function of tumor necrosis factor and interleukin-6-induced osteoclasts in rheumatoid arthritis. Arthritis Rheumatology. 73 (7), 1145-1154 (2021).
- Teng, Y. T., et al. Functional human T-cell immunity and osteoprotegerin ligand control alveolar bone destruction in periodontal infection. Journal of Clinical Investigation. 106 (6), 59-67 (2000).
- Terpos, E., et al. Soluble receptor activator of nuclear factor kappaB ligand-osteoprotegerin ratio predicts survival in multiple myeloma: proposal for a novel prognostic index. Blood. 102 (3), 1064-1069 (2003).
- Morony, S., et al. Osteoprotegerin inhibits osteolysis and decreases skeletal tumor burden in syngeneic and nude mouse models of experimental bone metastasis. Pesquisa do Câncer. 61 (11), 4432-4436 (2001).
- Sobacchi, C., Schulz, A., Coxon, F. P., Villa, A., Helfrich, M. H. Osteopetrosis: genetics, treatment and new insights into osteoclast function. Nature Reviews Endocrinology. 9 (9), 522-536 (2013).
- Amarasekara, D. S., et al. Regulation of osteoclast differentiation by cytokine networks. Immune Network. 18 (1), 8 (2018).
- Kim, J. M., Lin, C., Stavre, Z., Greenblatt, M. B., Shim, J. H. Osteoblast-osteoclast communication and bone homeostasis. Cells. 9 (9), (2020).
- Teitelbaum, S. L. Bone resorption by osteoclasts. Science. 289 (5484), 1504-1508 (2000).
- Boyle, W. J., Simonet, W. S., Lacey, D. L. Osteoclast differentiation and activation. Nature. 423 (6937), 337-342 (2003).
- Baron, R., Neff, L., Louvard, D., Courtoy, P. J. Cell-mediated extracellular acidification and bone resorption: evidence for a low pH in resorbing lacunae and localization of a 100-kD lysosomal membrane protein at the osteoclast ruffled border. Journal of Cell Biology. 101 (6), 2210-2222 (1985).
- Blair, H. C., Teitelbaum, S. L., Ghiselli, R., Gluck, S. Osteoclastic bone resorption by a polarized vacuolar proton pump. Science. 245 (4920), 855-857 (1989).
- Zhu, L., et al. Osteoclast-mediated bone resorption is controlled by a compensatory network of secreted and membrane-tethered metalloproteinases. Science Translational Medicine. 12 (529), 6143 (2020).
- Gowen, M., et al. Cathepsin K knockout mice develop osteopetrosis due to a deficit in matrix degradation but not demineralization. The Journal of Bone and Mineral Research. 14 (10), 1654-1663 (1999).
- Abu-Amer, Y. IL-4 abrogates osteoclastogenesis through STAT6-dependent inhibition of NF-kappaB. Journal of Clinical Investigation. 107 (11), 1375-1385 (2001).
- Patntirapong, S., Habibovic, P., Hauschka, P. V. Effects of soluble cobalt and cobalt incorporated into calcium phosphate layers on osteoclast differentiation and activation. Biomaterials. 30 (4), 548-555 (2009).
- Sorensen, M. G., et al. Characterization of osteoclasts derived from CD14+ monocytes isolated from peripheral blood. The Journal of Bone and Mineral Metabolism. 25 (1), 36-45 (2007).
- Kumar, A., et al. Synergistic effect of biphasic calcium phosphate and platelet-rich fibrin attenuate markers for inflammation and osteoclast differentiation by suppressing NF-kappaB/MAPK signaling pathway in chronic periodontitis. Molecules. 26 (21), 6578 (2021).
- Henriksen, K., Karsdal, M. A., Taylor, A., Tosh, D., Coxon, F. P. Generation of human osteoclasts from peripheral blood. Methods in Molecular Biology. 816, 159-175 (2012).
- Maria, S. M., et al. Reproducible quantification of osteoclastic activity: characterization of a biomimetic calcium phosphate assay. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 102 (5), 903-912 (2014).
- Kong, L., Smith, W., Hao, D. Overview of RAW264.7 for osteoclastogensis study: Phenotype and stimuli. Journal of Cellular and Molecular Medicine. 23 (5), 3077-3087 (2019).
- Li, P., et al. Systemic tumor necrosis factor alpha mediates an increase in peripheral CD11bhigh osteoclast precursors in tumor necrosis factor alpha-transgenic mice. Arthritis & Rheumatology. 50 (1), 265-276 (2004).
- Arai, F., et al. Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-Fms and receptor activator of nuclear factor kappaB (RANK) receptors. Journal of Experimental Medicine. 190 (12), 1741-1754 (1999).
- Xing, L., et al. NF-kappaB p50 and p52 expression is not required for RANK-expressing osteoclast progenitor formation but is essential for RANK- and cytokine-mediated osteoclastogenesis. The Journal of Bone and Mineral Research. 17 (7), 1200-1210 (2002).
- Miyamoto, T., et al. Bifurcation of osteoclasts and dendritic cells from common progenitors. Blood. 98 (8), 2544-2554 (2001).

