Evaluation of Hydration Status by Bioelectrical Impedance Vector Analysis in Patients with Ischemic Heart Disease Undergoing Exercise Stress Test
Summary
Hydration status imbalances can have a short-term effect on direct and indirect determinants of oxygen uptake and pulse, and morbidity and mortality prognostic factors in ischemic heart disease. This protocol describes the technique for assessment of hydration status through bioelectrical impedance vector analysis and cardiopulmonary response during exercise stress test.
Abstract
Ischemic heart disease (IHD) represents a group of clinical syndromes characterized by myocardial ischemia, leading to an impairment in the myocardial blood supply and compromised perfusion. Several clinical variables assessed through a stress test, such as oxygen uptake (VO2) and heart rate oxygen pulse (HR/O2), have been attributed as cardiopulmonary prognostic factors in patients with IHD. However, other factors like hydration status (HS), potentially affecting the cardiopulmonary response, have been barely addressed. Unbalanced HS has a short-term effect on plasma volume and the sympathetic nervous system, which impacts blood volume, and lowers VO2 and HR/O2. Recently, bioelectrical impedance analysis (BIA), a method based on the opposition of body tissues (including fluid volume) to a low electrical current, has been widely used to assess HS by obtaining two components: resistance (R) and reactance (Xc) and using prediction formulas. However, several limitations as chronic illness or abnormal fluid status, may affect the results. In this sense, alternative BIA methods, such as bioelectrical impedance vector analysis (BIVA), have become relevant. R and Xc (adjusted by height) result in a vector plotted on the R/Xc graph, which allows interpreting the HS as normal or abnormal according to the distance of the mean vector. This study aims to describe how to determine HS by BIVA using a single-frequency device and compare the results with the cardiopulmonary response in patients with IHD.
Introduction
Ischemic heart disease (IHD) represents a group of clinical syndromes characterized by myocardial ischemia, a mismatch in the myocardial blood supply and demand. The underlying pathophysiological defect includes inadequate perfusion, mainly due to atherosclerotic disease of epicardial coronary arteries1,2,3. In general, the presence of cardiovascular disease (CVD) is common, showing poor survival worldwide4. Particularly in 2015, IHD contributed to approximately 9 million deaths and more than 160 million disability-adjusted life years, and nowadays, IHD remains one of the main causes of mortality, and it favors the heart disease burden around the world5.
To evaluate both the presence and prognosis of IHD, some non-invasive procedures like the exercise stress test (EST) are routinely used. EST provides an assessment of the overall performance of cardiovascular, muscular, pulmonary, hematopoietic, neurosensory, and skeletal systems when the maximum tolerable stress appears under the EST6.
Under normal conditions, physiological adaptations would be expected while exercising. During exercise, several changes occur, like a dynamic change of fluid in blood within the vascular compartment, the reduction of plasma and blood volume, and the increase in hematocrit and plasma metabolite concentrations. Reduced plasma volume normalizes approximately 1 h after exercise, which may also vary depending on individual training level and water replenishment7.
However, IHD may lead to an acute impaired response to exercise, affecting EST performance in some variables comprising aerobic capacity and exercise tolerance, such as oxygen uptake (VO2) and heart rate/oxygen pulse (HR/O2)8. Recently, hydration status (HS), a measure of the water contained in the body1, has been proposed as a factor linked to plasma volume, able to modify blood flow and viscosity. HS has also been related to systolic volume, heart rate, and arteriovenous oxygen difference, determinants of VO2. Furthermore, some studies describe the relation of HS with a lower cardiopulmonary response (cardiac chronotropic and inotropic, VO2 and HR/O2)9.
In addition, several factors like age, environmental conditions, the level of physical activity/exercise, and dietary factors like fluid intake have been described to participate in HS balance10. Likewise, pathophysiological conditions such as IHD and its progression may influence HS11.
Although HS closely relates to cardiopulmonary, biological-environmental responses or lifestyle factors, the particular association of IHD in population with previous conditions has been scantily addressed; and it represents a significant challenge for clinical research, specifically due to the assessment of early stages, as well as the requirement for reliable and standardized methods to evaluate the HS.
To address this, bioelectrical impedance analysis (BIA), a practical, non-invasive, and cost-effective method, can be used to estimate body composition within a clinical setting but also has been proposed as an alternative method to evaluate HS showing advantages over other methods like biomarkers tests (urinary or plasma osmolality) due to the presence of high variability in the results and even over the gold standard method (isotope dilution) due to the complexity of the technique that requires specific training and highly cost equipment, becoming clinically impractical12,13,14,15.
The conventional BIA method applies an alternating, low electric current intensity (below the perceptual thresholds), entering the human body and crossing internal tissues. Then, based on the principle that body organs may act as electric conductors or dielectrics, we may obtain a register of electric impedance (or bioelectrical impedance [Z]) that reflects the opposition of the organs to the free applied electric flow (EF), depending on their composition (fat or muscle mass, bone, water, etc.)12. Here, Z sources are resistance (R) and reactance (Xc). The former is related to the opposition of the EF throughout cellular ionic solutions (intracellular and extracellular), while the latter is a capacitive component of tissue interfaces, cell membranes, and organelles12.
In addition, bioelectrical impedance vector analysis (BIVA) is an alternative BIA method approach that uses spatial relationships between R and Xc (both adjusted by height) to assess soft tissue hydration. The R and Xc data are plotted on a bivariate resistance-reactance graph, which allows visualizing body composition and HS12,16.
Considering the less explored field of HS balance associated with cardiopulmonary, as well as the growing interest to characterize new applications of methods like BIVA in the evaluation of HS, this study aims to determine HS by BIVA method and to analyze HS relation with VO2 and HR/O2 in ambulatory patients with IHD.
Protocol
The Institutional Research Ethics Committee from Centro Médico Nacional "20 de Noviembre", ISSSTE, approved this protocol (ID 383.2019). All enrolled patients signed written informed consent.
1. Before bioelectrical impedance analysis (BIA) measurement
NOTE: The BIA protocol procedure is measured using a single-frequency bioelectrical impedance device (Table of Materials). This device provides two values (resistance and reactance) at 50 kHz. Also, the BIA protocol described here is specific according to the single-frequency bioelectrical impedance device used.
- Standardization and technical requirements of the device
- Test the correct calibration of the device using the test resistor (500 Ω) provided by the manufacturer. Acceptable values for resistance and reactance are 495.0 to 505.0 Ω and -3.0 to 3.0 Ω, respectively. Ensure that the skin electrodes correspond to the same manufacturer's brand and that the device is not connected to the electric current.
- Clean and disinfect the device and the leads with hydrogen peroxide solution-based water (1:4). Dampen a clean wiping cloth with the solution and rub gently along the wires and the outside of the device case.
- Patient analysis and preparation for the test (pre-test)
NOTE: Patients diagnosed with IHD, male or female, and aged >18, were included. As well as patients with controlled comorbidities such as systemic arterial hypertension, dyslipidemia, and type 2 diabetes mellitus were also included. The mean weight and height were 70 kg and 160 cm, respectively. In addition, patients were excluded if they presented any of the following characteristics: active smoking or smoking cessation <6 months, myocardial infarction or stroke in the last 6 months, anemia, electrolyte imbalance, and chronic terminal diseases such as renal or hepatic failure.- Inform the patient regarding fasting for at least 2-3 h before the measurement.
- Explain the procedure to the patient.
- Collect gender, age, weight (in kg), and height (in cm) information. For measuring height and weight, use a digital portable precision scale, and a portable precision stadiometer, respectively (Table of Materials), and perform the measurements based on the standard method described by Lohman et al.17.
- Instruct the patient to take out any metal objects he/she is wearing (watches, rings, bracelets, necklaces, or any other) to avoid interference with the measurements. Ask the patient to remove their shoes and stocks.
- Place the patient in a supine position on a stretcher bed. Ensure that arms and legs maintain an angular separation of 30° to 45° and the palms of the hands face upwards. If the thighs are too big, use a clean towel/sheet to create a non-conductive barrier and separate them. Also, make sure the material of the stretcher bed is not metal conductive.
NOTE: To achieve stable fluid distribution after the patient acquires a supine position, recommended lying down for at least 5 min before BIA measurements. - Clean the area before connecting skin electrodes using a pad soaked with 70% ethyl alcohol.
- Place four skin electrodes as described below. Place all skin electrodes according to manufacturer recommendations and with a distance between them of at least 5 to 10 cm to avoid electrode-electrode interaction.
- First, place proximal electrodes on the hands as follows: locate one electrode on the wrist, just between the lunate-scaphoid carpal joint and ulna-radius joint. Then, locate another electrode at the middle finger, just behind the metacarpophalangeal joint.
- Then, place the distal electrodes on the feet, as follows: locate one electrode on the ankle, at the joint between the internal and external malleoli and astragalus. Then, locate another electrode between the metatarsophalangeal joints of the third finger.
2. BIA measurements
- Connect the circular outlet of the leads to the back of the device.
- First, connect the hand guide leads (proximal)- the red clip on the wrist, and the black clip on the middle finger, then connect the foot guide (distal) – the red clip on the ankle and the black clip on the third finger. Verify that all clips are placed on the skin electrode´s edge.
- Instruct the patient not to move while the measurement is performed.
- Turn on the device by pressing the ON button. Observe the values on the screen immediately and wait for 30 s to 60 s for resistance and reactance data to stabilize, then register those values. Turn off the device by pressing the OFF button.
- Once the measurement is done, remove the red and black clips for the hand and the foot, then remove the skin electrodes carefully and discard them.
3. Analysis and evaluation of hydration status by BIVA
NOTE: Before starting the BIA analysis, it is necessary to download the BIVA software (Table of Materials). Note that the BIVA software is a spreadsheet with data from different populations and reference values of resistance and reactance arranged by sex18.
- The BIVA software contains the following seven sheets: guide, reference populations, point graph, path, subjects, z-score, and z-graph. Click the Reference Population sheet and select the complete line according to the population to be evaluated. This methodology uses rows 9 and 10 (Mexican population)19.
- Copy the selected data and paste them into the second row.
- Click the Disciplinas sheet and fill manually the following information located in the second row- Column 1: Patient ID; Column 2: Seq, should always be 1; Column 3 and Column 4: indicate the Surname and Name, respectively; Column 5: Sex, assign F if female or M if male; Column 6 and Column 7: add values of resistance and reactance (previously registered), respectively; Column 8: Height, indicate value in cm; Column 9: Weight, indicate the value in kg; Column 10: Popl Code, indicate a value between 1 to 13 that appears in the first column of the reference population sheet to choose the population to be evaluated; Column 11: Group Code, add a value between 1 to 10 to select the patient to be evaluated; Column 12: Age, indicate the age in years.
- Click the Complement option in the main menu and click Calculate to obtain the resistance and reactance values adjusted by height (columns 13 and 14).
- Click the Point Graph sheet, and the R/H-Xc/H graph will be displayed for females or males. This graph contains the 50%, 75%, and 95% percentile tolerance ellipses adjusted by specific reference populations (Figure 1).
- When a dialog box named select groups is displayed, select the Group Option according to the last number indicated in the subject's sheet – Column 11. Add the value previously registered at the group code and click OK. A vector will be plotted showing the shape characteristics.
- Interpret the hydration status from the plot as follows: a vector between 50% and 95% percentiles tolerance ellipses indicates euhydration, whereas a vector outside the upper pole of 95% tolerance ellipse indicates hypohydration and a vector outside in the lower pole of 95% tolerance ellipse indicates hyperhydration.
4. Before starting the exercise stress test (EST) protocol
NOTE: The exercise stress test (EST) protocol20,21 is measured using a specialized medical treadmill (Table of Materials). EST is carried out according to the modified Bruce ramp protocol20,21 and supervised by an experienced cardiologist.
- Patient analysis and preparation
- Ensure the patient complies with the following characteristics before the test: avoid any strenuous physical activity at least 1 day before the test, and fast the patient at least 4 h before the test. Ask the patient to wear comfortable clothing for the test.
- Explain to the patient the EST protocol and register the gender, age, weight (in kg), and height (in cm). Enter the gender, age, weight, and height data into the EST system to automatically estimate the METS, VO2, and HR/O2 values during the test.
- Ask the patient about any physical disability (e.g., joint or chest pain, dyspnea, etc.) before EST. Observe the patient´s gait and ensure it does not present a walking disorder.
- EST devices connection to patient and system calibration
- Select and place a size-fit EST mask, then gently fix it to the patient´s face.
- Perform an air leak test instructing the patient to cover the hole in the mask and breathe out. No sound should be heard.
- Connect the pink flow ends, one to the hole of the mask and the other to the wires of the EST equipment.
- Wait for automatic calibration of the EST equipment's gas analysis elements, ensuring the environmental CO2 does not exceed 1,200 ppm.
- Instruct the patient to uncover the chest area and clean the skin with 70% isopropyl alcohol before placing the electrodes. Shave the area if necessary.
- Connect the 12-lead electrocardiographic electrodes on the patient´s chest according to the modified Mason-Likar22 standardization method and AHA scientific statement: exercise standards for testing and training20,21 as described below.
- Place four electrodes on the arms: one on the right arm (RA) over the acromion bone, one on the left arm (LA) over the acromion bone, one on the right costal margin (RCM), and one on the left costal margin (LCM).
- Then, place six electrodes on the chest: V1 on the second intercostal space and over the right border of the sternum, V2 on the second intercostal space and the left border of the sternum, V3 between V2 and V4, V4 on the fourth intercostal space crossing with the left midclavicular line, V5 on the fifth intercostal space at the level of the left anterior axillary line and V6 on the sixth intercostal space at the level of the left middle axillary line.
- Perform a resting spirometry instructing the patient to inhale deeply as much as possible, then indicate to exhale as fast and forcefully as possible, trying to maintain at least 6 s in the breath-out effort.
- Evaluate the reproducibility and quality criteria of the resting spirometry by repeating the breath-out effort 3x.
- Evaluate the forced expiratory volume in 1 s (VEF1) and the forced vital capacity (FVC) parameters provided in the EST system.
- Choose the best parameters the patient achieves according to VEF1/FVC to monitor it during the test.
5. Performing the EST
- Evaluate the cardiac conditions (baseline electrocardiogram [EKG], heart rate, and blood pressure) to ensure the patient does not present limiting factors before starting the EST.
- Explain to the patient that the perceived physical effort during the EST is measured using the Borg scale from 0 to 2020.
- Monitor and evaluate the EST with attention to blood pressure, heart rate, and electrocardiographic trace changes every 3 min. Continue monitoring the perception of any symptoms (chest pain, shortness of breath, dizziness, or extreme fatigue) and the perceived physical effort by the Borg scale. Perform these evaluations as follows.
- Blood pressure: Measure systolic and diastolic blood pressure every 3 min with a pneumatic device to evaluate a rise in systolic blood pressure proportional to the exercise load.
- Heart rate: Record beat by beat, in real-time, using 12-lead telemetry (stress electrocardiogram) to expect a linear increase in this variable with increasing load. Also, analyze the heart rhythm to differentiate it from non-sinus rhythms and detect arrhythmias by telemetry.
- Symptoms and signs: During the procedure, ask the patient if there is any discomfort, such as pain in the chest or extremities, difficulty breathing, altered alertness, decreased visual acuity, dizziness, or gait instability, and evaluate if exists the presence of low heart rate, oxygen saturation, or blood pressure.
- Perception of effort: As the procedure progresses, ask the patient about the degree of effort according to the Borg scale from 0 to 20. The lower numbers indicate light efforts, while the higher ones are heavy.
- Stop the EST if the patient achieves > 85% of their heart rate (calculated according to age) or if the patient experiences any discomfort during the effort or shows significant electrocardiographic disturbances (ischemia or complex arrhythmias).
- After EST is finished, monitor closely the patient´s blood pressure, heart rate, and EKG during the next 1 min, 3 min, 5 min, and 8 min (recovery period), and ensure these parameters return to baseline values.
- Extract and register cardiopulmonary data from the EST software: METS, VO2, and HR/O2.
Representative Results
First, R and Xc (both adjusted by patient´s height) data registered from the single frequency (SF-BIA) device was used to obtain the BIVA R/Xc graph. Second, we classified hydration status as euhydration, hyperhydration, and hypohydration. Representative hydration data from male patients plotted with circle and triangle, aged 66 years and 67 years, weighing 72.2 kg and 72.3 kg, height 169 cm and 163 cm, are shown (Figure 2). In addition, impedance data are shown R/H= 280.7, Xc/H= 23.6 Ω and R/H= 254.3, Xc/H= 61 Ω, for the two patients, classified as hyperhydration and hypohydration status, respectively. EST values for patient 1 were VO2 = 7.7 mL/kg/min, METS= 3.7, and HR/O2= 7.5 O2/beats, and for patient 2, these were VO2 = 25.5 mL/kg/min, METS= 7.3 and HR/O2= 11.3 O2/beats. In comparison, patients with euhydration (plotted with a square, aged 68 years, weighing 83.1 kg, height 174 cm, with bioimpedance results: R/H= 265.5, Xc/H= 28.7 Ω) showed EST values of VO2 = 43.3 mL/kg/min, METS= 12.4 and HR/O2= 27.7 O2/beats. These results suggest that abnormal HS may induce a lower cardiopulmonary response (lower VO2, METS, and HR/O2 values), especially when hyperhydration occurs.
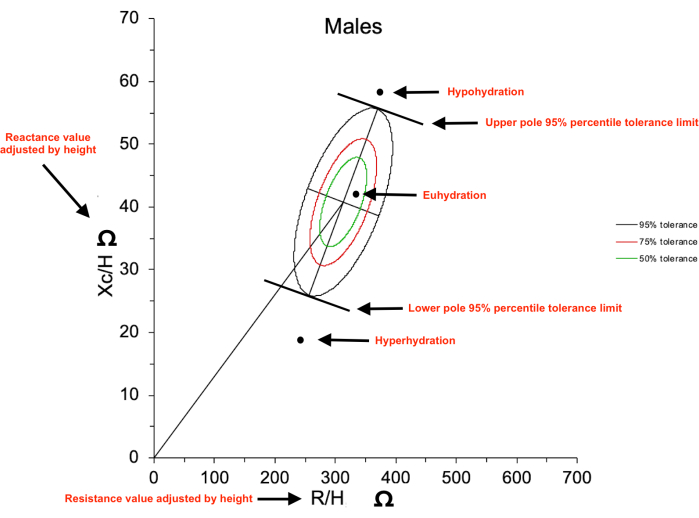
FIGURE 1: Bioelectrical impedance vector analysis (BIVA) resistance/reactance (R/Xc) graph. The axes show the raw resistance (R) and reactance (Xc) values adjusted by height (H) in m, which form a vector plotted on the R/Xc graph. The vector plotted (circle symbol as an example) under the lower pole 95% percentile tolerance limit is considered as hyperhydration status, a vector plotted above the upper pole 95% percentile tolerance limit is considered as hypohydration status, and a vector plotted between the upper and lower poles is considered as euhydration status. Please click here to view a larger version of this figure.
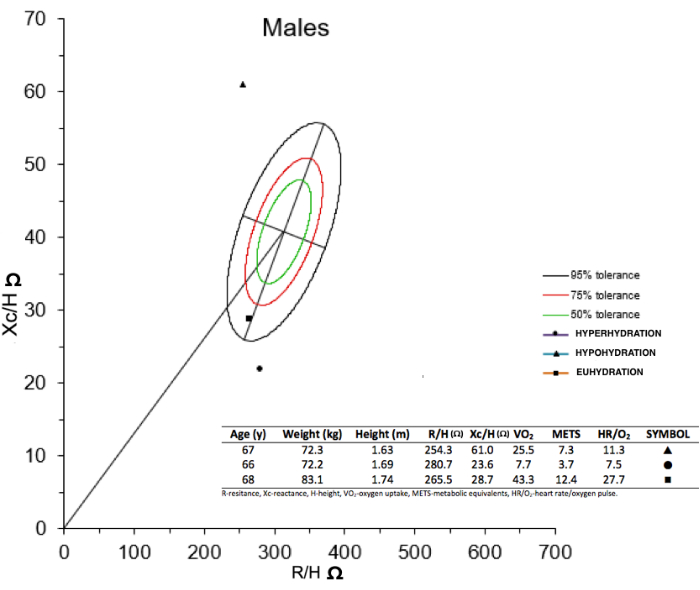
FIGURE 2: BIVA R/Xc graph of three male newly admitted patients to the cardiac rehabilitation service. As a representative result, the circle is a patient classified as hyperhydration status, the triangle is a patient classified as hypohydration status, and the square is a patient classified as euhydration status. In addition, it shows that circle and triangle patients have lower cardiopulmonary values than the square patient. Please click here to view a larger version of this figure.
Discussion
Although BIA is considered a safe, practical, and noninvasive method, which overcomes the limitations of other methods to measure body composition and body water19,23, it is relevant to consider the potential bias occurring regarding the type of bioelectrical impedance (the method described here is specific for a single-frequency bioelectrical impedance device), or the variation in the steps and technique verification methods.
It has been reported in the medical literature that there are different types of BIA; the SF-BIA, based on 50 kHz, and the multifrequency (MF-BIA), which uses a range between 1 to 500 kHz. The differences between them reside in the better capacity of MF-BIA to measure and differentiate total body water (TBW) from extracellular water (ECW) and intracellular water (ICW), in comparison with SF-BIA, both in healthy subjects and in populations with chronic diseases24,25,26,27. Nevertheless, some SF-BIA types of equipment can approximate the ECW values as long as the HS remains stable. Some studies show controversy regarding TBW, ICW, and ECW prediction ability when MF-BIA is used. Therefore, a more accurate method like BIVA may be preferred to monitor and evaluate HS. Some advantages of the BIVA method include its performance in subjects with altered fluid balance, given its determinations without the requirement of prediction formulas. However, it is important to consider variables that affect the measurements of BIVA. Other methods like segmental BIA (SEG-BIA) have been proposed as an alternative method to evaluate both body composition and HS, but some of their results are still under discussion due to percentage variability between the upper and lower limbs24,25,26,27.
The position of the body and electrode locations are also relevant. Some BIA types of equipment are easy to use when the subject is in a vertical position, and usually, the electrodes are placed according to the technique hand-hand or foot-foot23,25,26. However, the R and Xc data are not available. On the other hand, some other BIA models require the subject to be in a supine position and the location of four electrodes (two electrodes on one hand and two electrodes on one foot, all positioned either on the right or left side). Such techniques often require lying down for 5 to 15 min before BIA measurement to balance fluid shift23,25,26. Likewise, the previously mentioned SEG-BIA (tetrapolar BIA equipment) method has been recently proposed as a possible alternative. Although, standardization techniques related to both electrodes and the subject position may represent limitations for their use24,26.
Nevertheless, other technical considerations about the patient's characteristics should be considered before BIA is performed. For example, in patients with skin areas affected, the electrode placement could be difficult, as well as in patients with altered fluid and electrolyte balance. Also, both biological factors (age, sex, or ethnicity) and external factors (eating, liquid intake, especially alcoholic beverages), extreme physical activity or exercise, edema presence, menstrual period, or even menopausal stages should be considered before BIA evaluations in order to reduce bias in measurements or interpretation. Likewise, the correct type and size of BIA electrodes (possibly specific BIA device brand), the electrode placement, and the handling device should be checked23,25,26. The last consideration is mentioned in detail in step 1.1 and step 1.2.
Comparing the BIA with other methods reported to evaluate HS, the isotope dilution method (considered the gold standard to measure body fluids) remains impractical due to its complex characteristics like the need for tracers, expensive equipment, dependent operator, and trained personnel, mainly12,13,14,15. Other methods, like the evaluation of body weight changes, are simple and cost-effective and especially useful to evaluate dehydration, but with limited application given the reference, measurements are required and, in most cases, may include changes in fluid-independent body components and not allow to evaluate the HS for a long period12, 25,26,27. Other methods, such as urinary osmolality, are also cheap and non-invasive, and considered sensible for small changes in HS12,13,14,15. Nevertheless, it has been reported that high intra- and inter-individual variations of 28.3% and 57.9%, respectively, are observed. Likewise, plasma osmolality is highly reproducible with lower intra- and inter-individual variations (1.3% and 1.5%, respectively) but with very limited use for research. BIA (and also BIVA) is considered a safe, practical, and non-invasive method that overcomes the limitations of the previously commented methods12,13,14,15.
Specifically, the BIVA method has been widely investigated for body composition assessment with potential use to measure HS by determining body water and its cell divisions when a multi-frequency device is used 14,15,28. In addition, BIVA provides some other advantages, such as Z-vector length has been validated as a reliable indicator of fluid volume during dynamic changes of total body water and has been shown to be effectively used in clinical conditions (renal disease, hemodynamic disturbance, and vascular surgery, among others); but also, some challenges that remain to be addressed are low sensibility and high specificity to detect depletion or the difficulty to identify acute changes in HS14, 15, 28.
It is important to emphasize that although in this protocol, we used an SF-BIA device to obtain the raw R and Xc values (at 50 kHz) in patients with IHD, these data were not used to evaluate the HS directly. Instead, we adjust the values according to variables of interest (subject´s height, sex, and specific population), plotted using the BIVA method, and finally evaluate the HS. In this context, some studies have described the usefulness of BIVA in heart disease, as mentioned below.
Di Somma et al.29 commented in a literature review that the utility of BIVA in conjunction with blood BNP value (a biomarker associated with fluid overload) could improve a better prescription of diuretic therapy in patients with acute heart failure (AHF). Although the easy use of BIVA makes it suitable for clinical settings, this method could be a good parameter to evaluate HS in conjunction with biomarkers related to diagnosis and prognosis in patients with AHF29. Other literature review by Thanapholsart et al.30 describe interesting data about the use of BIA-Xc and BIA-Xc adjusted by height, indicating both as associated factors for AHF diagnosis but also the conjunction of BIA-BNP acting as a better predictor in patients with decompensated heart failure. However, the BIVA method seems more reliable for enhancing the diagnosis of AHF accompanied by BNP, as well as for identifying fluid overload more accurately30. Likewise, Sugizaki et al.31 demonstrated that in patients under hemodialysis therapy, the BIVA method contributes to achieving dehydration status in up to 30% of the studied population with chronic kidney disease, indicating the importance of considering the evaluation of both altered HS (hypohydration or hyperhydration)31. The last seems to be a crucial point to study further in patients with IHD, especially because dehydration status is scantily addressed.
According to our results, the relevance of understanding the complex role of altered HS (hypohydration or hyperhydration) related to an impaired cardiopulmonary response and physical performance could compromise disease prognosis (such as IHD). Therefore, the emphasis on having accurate, reliable, and reproducible methods to assess HS becomes relevant. Even though a wide variety of techniques and methods are available for assessment in impaired HS, until now, none of these have universal acceptance12,26,27. Alike, the possible clinical applications of BIVA related to the cardiopulmonary variables allow us to illustrate how the patients with non-altered HS showed a better response in VO2, METS, and HR/O2 values in comparison with the patients classified as altered HS. In this sense, Marawan et al.32 demonstrated the edema index (an indicator of extracellular fluid volume) measured by SF-BIA device significantly correlates inversely proportional and as an independent factor (adjusted by confounding variables) with VO2 and exercise time in patients with heart failure with reduced ejection fraction and type-2 diabetes mellitus32.
Some limitations like alcohol intake, fasting within the previous 2-3 h, vigorous physical activity/exercise 1 day before, or no data about menstrual cycle were not controlled during the BIA measurements due to patients being new admission in the cardiac rehabilitation service. However, in order to control clinical and environmental variables which possibly impact BIA and BIVA protocols in patients with IHD, the BIA-ESPEN guidelines suggest a detailed set of recommendations in the context of their clinical use24, as well as a scientific statement of the American heart association suggest BIVA method as a strategy to evaluate fluid status in subjects at risk or diagnosis of cardiorenal syndrome33.
Finally, it is important to recognize that HS is highly dynamic during the day and is usually returned to baseline conditions within 24 h; some other factors like lifestyles and environment are implicated in their regulation, as well as pathophysiological conditions such as IHD. The last one represents a major acute challenge to keep in mind during the study of water balance homeostasis, and therefore, a quick and highly reproducible method is ideal.
Declarações
The authors have nothing to disclose.
Acknowledgements
To Consejo Nacional de Ciencia y Tecnología (CONACyT) that sponsored the scholarship CVU 1004551 for Dulce María Navarrete de la O during her MSc degree.
Materials
| BIVA Tolerance | BIVA SOFTWARE 2002 | Piccoli A, Pastori G: BIVA software. Department of Medical and Surgical Sciences, University of Padova, Padova, Italy, 2002 (available at E-mail:apiccoli@unipd.it). | |
| Cardiopoint ECG C600 | BTL | 407-80MANEN03100 | ELECTROCARDIOGRAPH |
| Cardiopoint Trolley | BTL | 40700B000240 | TROLLEY |
| Portable Digital Flat Scale | SECA | 813 | DIGITAL FLAT SCALE |
| Portable Stadiometer | SECA | 213 | STADIOMETER |
| Quantum IV | RJL SYSTEMS | Q4B-2405 | BIOELECTRIC IMPEDANCE ANALYZER |
| Treadmill Clinical | BTL | 216A18 | TREADMILL |
Referências
- Alcalá, J. E., Maicas, C., Hernández, P., Rodríguez, L. Ischemic heart disease: concept, classification, epidemiology, risk factors, prognosis and prevention. Medicina. 12 (36), 2145-2152 (2017).
- Steenbergen, C., Frangogiannis, N. . Muscle: Fundamental Biology and Mechanisms of Disease. , (2012).
- Moreno, P. R., Portillo, J. H. Myocardial ischemia: basic concepts, diagnosis and clinical implications. Revista Colombiana de Cardiología. 23 (5), 403-409 (2016).
- Roth, G. A., et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990-2019: Update From the GBD 2019 Study. Journal of the American College of Cardiology. 76 (25), 2982-3021 (2020).
- Zhang, G., et al. Burden of Ischemic heart disease and attributable risk factors in China from the global burden of disease 2015 study. BMC Cardiovascular Disorders. 18, 1-13 (2018).
- Lomelí, H. I., et al. . Ejercicio, Dieta y Corazón. , (2013).
- García, M., Nuñez, J. P. . Rehabilitación Cardiovascular: Prevención y Deporte. , 35-49 (2019).
- Coeckelberghs, E., Buys, R., Goetschalckx, K., Cornelissen, V. A., Vanhees, L. Prognostic value of the oxygen uptake efficiency slope and other exercise variables in patients with coronary artery disease. European Journal of Preventive Cardiology. 23 (3), 237-244 (2015).
- Lundby, C., Montero, D., Joyner, M. Biology of VO2 max: looking under the physiology lamp. Acta Physiologica. 220 (2), 218-228 (2017).
- Baron, S., Courbebaisse, M., Lepicard, E. M., Friedlander, G. Assessment of hydration status in a large population. British Journal of Nutrition. 113 (1), 147-158 (2015).
- Kemp, C. D., Conte, J. V. The pathophysiology of heart failure. Cardiovascular Pathology. 21 (5), 365-371 (2012).
- Lukaski, H. C., Piccoli, A. Bioelectrical Impedance Vector Analysis for Assessment of Hydration in Physiological States and Clinical Conditions. Handbook of Anthropometry: Physical Measures of Human Form in Health and Disease. , 287-305 (2012).
- Roubenoff, R., Heymsfield, S. B., Kehayias, J. J., Cannon, J. J., Rosenberg, I. H. Standardization of nomenclature of body composition in weight loss. The American Journal of Clinical Nutrition. 66 (1), 192-196 (1997).
- Armstrong, L. E. Assessing Hydration Status: The Elusive Gold Standard Assessing Hydration Status: The Elusive Gold Standard. Journal of the American College of Nutrition. 26 (5 Suppl), 37-41 (2007).
- Armstrong, L. E. Hydration assessment techniques. Nutrition Reviews. 63 (6), S40-S54 (2005).
- Picolli, A., Nescolarde, D., Rosell, J. Análisis convencional y vectorial de bioimpedancia en la práctica clínica. Nefrología. 22 (3), 228-238 (2002).
- Lohman, T. G., Roche, A. F., Martorell, R. . Anthropometric standardization reference manual. , (1991).
- Piccoli, A., Pastori, G. . BIVA software. , (2002).
- Espinosa, M. A., et al. Vectores de impedancia bioeléctrica para la composición corporal en población mexicana. Revista de Investigación Clínica. 59 (1), 15-24 (2007).
- Fletcher, G. F., et al. Exercise Standards for Testing and Training. A Scientific Statement From the American Heart Association. Circulation. 128, 873-934 (2013).
- Thompson, W. R., Gordon, N. F., Pescatello, L. S. . ACSM’S Guidelines for Exercise Testing and Prescription. , (2010).
- Mason, R. E., Likar, I. A new system of multi-lead exercise electrocardiography. American Heart Journal. 71 (2), 196-205 (1966).
- González, C. H., Caicedo, J. C. Bioelectrical impedance analysis (BIA): a proposal for standardization of the classical method in adults. Journal of Physics: Conference Series. 407 (012018), 1-13 (2012).
- Kyle, U. G., et al. Bioelectrical impedance analysis-part I: review of principles and methods. Clinical Nutrition. 23, 1226-1243 (2004).
- Khalil, S. F., Mohktar, M. S., Ibrahim, F. The Theory and Fundamentals of Bioimpedance Analysis in Clinical Status Monitoring and Diagnosis of Diseases. Sensors. 14, 10895-10928 (2014).
- Savegnago, M., Faccioli, J. M., Jordao, A. A. Analysis of Body Composition: A Critical Review of the Use of Bioelectrical Impedance Analysis. International Journal of Clinical Nutrition. 2 (1), 1-10 (2014).
- Kyle, U. G., et al. Bioelectrical impedance analysis-part II: utilization in clinical practice. Clinical Nutrition. 23, 1430-1453 (2004).
- Armstrong, L. E., et al. Human hydration indices:acute and longitudinal reference values. International Journal of Sport Nutrition and Exercise Metabolism. 20 (2), 145-153 (2010).
- Di Somma, S., et al. The emerging role of biomarkers and bio-impedance in evaluating hydration status in patients with acute heart failure. Clinical Chemistry and Laboratory. 50 (12), 2093-2105 (2012).
- Thanapholsart, J., Khan, E., Lee, G. A. A Current Review of the Uses of Bioelectrical Impedance Analysis and Bioelectrical Impedance Vector Analysis in Acute and Chronic Heart Failure Patients: An Under-valued Resource. Biological Research For Nursing. 25 (2), 240-249 (2023).
- Sugizaki, C. S. A., et al. Comparison of Bioelectrical Impedance Vector Analysis (BIVA) to 7-point Subjective Global Assessment for the diagnosis of malnutrition. Jornal Brasileiro de Nefrologia. 44 (2), 171-178 (2021).
- Marawan, A., et al. Edema Index Predicts Cardiorespiratory Fitness in Patients With Heart Failure With Reduced Ejection Fraction and Type 2 Diabetes Mellitus. Journal of the American Heart Association. 10 (8), e018631 (2021).
- Rangaswami, J., et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies. A Scientific Statement From the American Heart Association. Circulation. 139 (16), e840-e878 (2019).

