Vascular Reconstruction with the Cuff Technique in Mouse Orthotopic Liver Transplantation
Summary
This protocol provides technical information for vessel reconstruction using the cuff technique in mouse orthotopic liver transplantation.
Abstract
Mouse orthotopic liver transplantation is an effective methodology for investigating the underlying mechanisms of liver ischemia and reperfusion injury. However, the technical challenges pose a barrier to utilizing this valuable experimental model and passing on these skills to the next generation. The most challenging aspect of this procedure is vascular reconstruction, including the portal vein (PV), infrahepatic inferior vena cava (IHIVC), and suprahepatic inferior vena cava. The use of plastic cuffs, rather than sutures, allows for smoother PV and IHIVC reconstruction. Vessels are reconstructed by attaching a cuff made from an intravenous catheter to the tip of the graft vessel and interposing the cuff into the recipient vessel. The two most crucial aspects are properly visualizing the inner lumen of the vessel and avoiding the use of excessive force. Our aim is to provide a technical overview of vascular reconstructions using the cuff technique in recipient surgery. These technical tips for the cuff technique are expected to help microsurgeons facilitate vascular reconstruction and advance their research.
Introduction
Mouse orthotopic liver transplantation (MOLT) is an effective experimental method first reported in 19911. This experimental model, which utilizes genetically modified mice and various research reagents, has played a pivotal role in investigating warm and cold ischemia and reperfusion injuries. However, the model's high technical complexity has hindered the development of basic medicine for liver transplantation2. MOLT involves three major steps: (1) retrieval of the liver from the donor mouse, (2) back table surgery, and (3) implantation of the liver into the recipient. Among these recipient procedures, vascular anastomosis poses the greatest challenge. While the suprahepatic inferior vena cava anastomosis is typically completed by hand stitching2, the infrahepatic inferior vena cava (IHIVC) and portal vein (PV) can be reconstructed more efficiently using plastic cuffs in place of hand-sewn sutures.
The anhepatic period signifies the interval between the removal of the recipient's native liver and the graft implantation. To ensure consistent results, it is imperative to limit the anhepatic time to less than 20 min. Consequently, studies employing this model have been confined to specific institutions3,4,5,6,7,8,9. Among the various stages of MOLT, achieving smooth PV and IHIVC reconstruction is crucial for minimizing anhepatic time and ensuring successful transplantation.
PV and IVC reconstruction is generally carried out using vascular cuffs since the cuff technique simplifies vascular anastomosis compared to hand-sewn sutures2,5,8. The technique involved in vascular cuff preparation and secure attachment of the cuff significantly impacts the complexity of recipient vascular reconstruction. Our objective is to provide detailed visual guidance for numerous technical tips related to the cuff method, thereby reducing the learning curve. These video clips will provide a clear understanding of how to attach the cuff to the vessels and reconstruct the PV and IHIVC during recipient surgery.
Protocol
The experimental protocol was approved by the Animal Experimentation Committee at Kyoto University. The study utilized C57BL/6 mice, aged over 10 weeks and weighing between 25 g and 30 g, obtained from a commercial source (see Table of Materials). All animals were anesthetized with 2.5% isoflurane (following institutionally approved protocols), maintained under specific pathogen-free conditions, and all experimental procedures were conducted in compliance with Kyoto University's Regulations on Animal Experimentation. The appropriate instruments used for the study are listed in the Table of Materials and depicted in Figure 1 and Figure 2.
1. Animal selection
- Use mice with a body weight of 25-30 g.
NOTE: While mouse liver allografts2 are generally accepted across major histocompatibility complex barrier, we opted for the syngeneic MOLT model in this study to focus on the detailed mechanism of cold ischemia reperfusion injury10. Mice weighing less than 25 g are not recommended because it is impossible to insert an inner stent into a thin bile duct. Similarly, mice weighing more than 30 g are not recommended due to the presence of a large amount of intraabdominal fat around the vessels.
2. Cuff making
- Prepare 20 G and 16 G intravenous catheters for the PV and IHIVC, respectively.
- Using a scalpel, cut the catheter to create the cuff. The main body of the cuff should measure 2 mm in length, with an extension handle of 1 mm (Figure 3A).
NOTE: If the handle is too large, it may become challenging to insert the cuff. - On the cuff's surface, create a shallow groove for thread fixation using the back of the scalpel blade.
NOTE: When applying the back of the scalpel blade to the cuff, ensure that the tip of the blade is at an angle of approximately 60 degrees (as shown in Figure 3B). After making the first groove, secure the scalpel blade and rotate the cuff. Ideally, two grooves are recommended, but a single groove can suffice without issues.
3. Cuff attachment
- In a plastic rectangular-shaped container, place small ice cubes and position a small metal cup (6 cm in diameter) on top. Fill the cup with organ preservation fluid (see Table of Materials) and place the syngeneic graft within.
- Gently roll the syngeneic liver graft (extracted from another donor mouse) using a cotton swab, ensuring that the porta hepatis faces upwards. Thoroughly remove any blood stored in the lumen of the IVC with organ preservation fluid.
- Thread the PV through the cuff using a thread attached, for example, to the splenic vein (see Figure 4A).
- With the cuff handle at the 12 o'clock position, secure both the handle and cuff with a bulldog clamp (see Table of Materials), positioning it 2 to 3 mm from the PV edge.
- Secure the bulldog clamp further using large vascular forceps and a fixation mold to establish the operative field (see Figure 4B).
- Carefully inspect the PV lumen at approximately 15 to 20x magnification. If necessary, dripping water can enhance visibility of the lumen.
- Gently grasp the PV lumen and externalize it through the cuff.
- Once fully externalized, secure it with a double ligation using 8-0 silk thread along the cuff's groove (Figure 4C,D).
NOTE: Ensure that the ligature is not loose. Also, take care that the ligature point is not too large, as it may interfere with cuff insertion. The ligature point can be positioned in any direction. - Attach the cuff to the IHIVC using a procedure similar to that used for the PV (Figure 4E).
NOTE: When clamping the IHIVC with the cuff, avoid biting into the liver parenchyma. Since the neck is shorter than the PV, carefully determine the clamping position. - Create a slipknot by passing the thread around the base of the IHIVC to temporarily ligate the IHIVC (Figure 5). This thread will be removed after reperfusion.
4. Portal vein reconstruction
- Anesthetize the recipient mouse by inducing anesthesia with isoflurane at 2.5% and reduce it to 0.6% before extracting the native liver as described in published literature2. Dinsinfect the surgical area atleast three times with alternating rounds of iodophor and 70% ethanol before laparotomy.
- Ensure that the hepatic lobe is properly positioned and that the cuffed PV is free from kinking and torsion.
- Gently grasp the recipient's PV edge using Pean forceps and secure it with a vise.
NOTE: In this article, a vise refers to a mechanical apparatus used to secure Pean forceps on the table. For more information on vise, see Table of Materials. - Apply a vascular clamp to the recipient's PV and pass an 8-0 silk thread around it.
- Create a 1/4 circumferential section approximately 0.5 mm from the PV edge (Figure 6A). Enlarge the hole while passing saline solution through the lumen using a specialized instrument (a 27 G needle with the tip cut off and bent into an L-shape).
- Use straight forceps to grip the cuff handle and insert it into the lumen.
- Once fully inserted, secure the cuff with an 8-0 silk thread. A single ligation is sufficient for fixation.
- Remove the vascular clamp and reperfuse the liver.
- Carefully release the Pean forceps and complete the portal vein reconstruction. The entire PV reconstruction process should take approximately 5 min.
5. Infrahepatic inferior vena cava reconstruction
- Position the liver lobe appropriately and move the Pean forceps attached to the recipient IHIVC dorsal to the graft's right lower lobe, securing it with a vise.
- Eliminate any remaining liver tissue around the recipient's IVIHC by gently rubbing with a cotton swab.
- Pass an 8-0 silk thread around the recipient's IVIHC.
- Create a 1/4 circumferential section at approximately 0.5 mm from the recipient's IVIHC edge (Figure 6B).
- After introducing normal saline solution into the lumen, insert the cuff into the lumen.
NOTE: Due to the short neck, the cuff is often inserted more shallowly than in the PV. - Carefully secure the cuff with an 8-0 silk thread and then remove the vascular clamp and Pean forceps.
6. Postoperative care
- Utilize a heating pad and warming lamp to facilitate postoperative recovery.
- Continuously monitor blood pressure, heart rate, respiratory rate, body temperature, as well as food and water consumption.
- Adminsiter subcutaneous injection of carprofen (5 mg/kg, every 6 h or until needed) to alleviate postoperative pain. Follow local institutional guidelines.
NOTE: If the condition of mice deteriorates, the experiment will be stopped immediately and mice were euthanized by inhalation of carbon dioxide. - To collect the tissue sample, re-anesthetize the mice and euthanize them by an IACUC approved method.
Representative Results
PV reconstruction is successful when, upon unclamping the portal vein, there is no tortuosity, and the liver is uniformly perfused. Anhepatic time should be under 20 min, as anhepatic times exceeding 25 min increase the risk of mouse mortality. IHIVC reconstruction is deemed successful if there is no blood regurgitation from the graft.
Storing the graft at cold temperatures for 1 h using organ preservation solution results in a serum alanine aminotransferase level of approximately 2,000 U/L at 6 h after reperfusion (Figure 7A). The survival rate is 100% at 7 days after transplantation (Figure 7B). However, in our model without hepatic artery anastomosis, recipient mice may succumb to intrahepatic biloma-related issues approximately 30 days post-transplantation
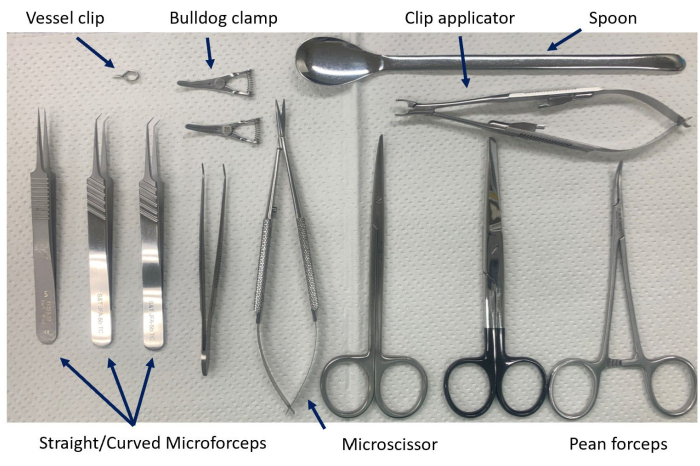
Figure 1: Instruments for vascular reconstruction. Straight or curved forceps are used as needed for the situation and the operative field. Please click here to view a larger version of this figure.

Figure 2: The microscope. The microscope is equipped with an objective lens with 10x magnification and an eyepiece lens with at least 0.8x magnification. Please click here to view a larger version of this figure.
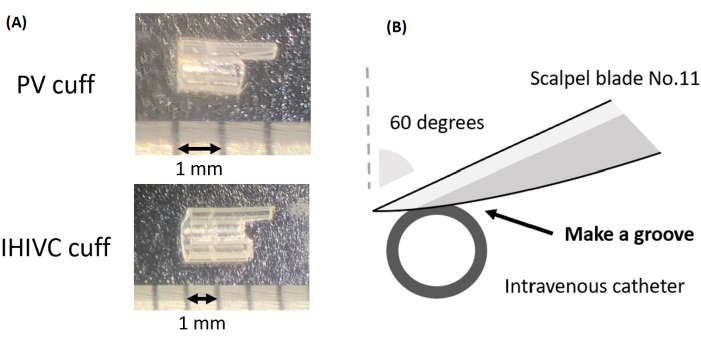
Figure 3: Preparation of the cuff. (A) The cuff for the PV and IHIVC. (B) A schematic representation of the process of creating a groove on the cuff. Please click here to view a larger version of this figure.
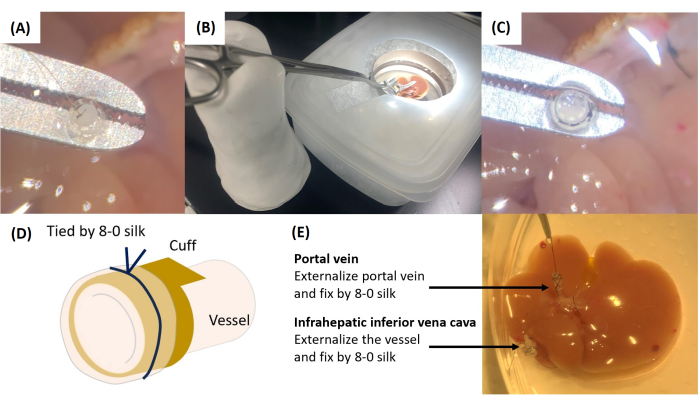
Figure 4: Cuff attachment. (A) The thread attached to the PV is passed into the cuff. (B) Instrument setup for cuff attachment. (C) Securing the cuff with 8-0 silk ligations. (D) Schematic illustration of vascular externalization and fixation to the cuff. (E) Completed cuff attachment. Please click here to view a larger version of this figure.
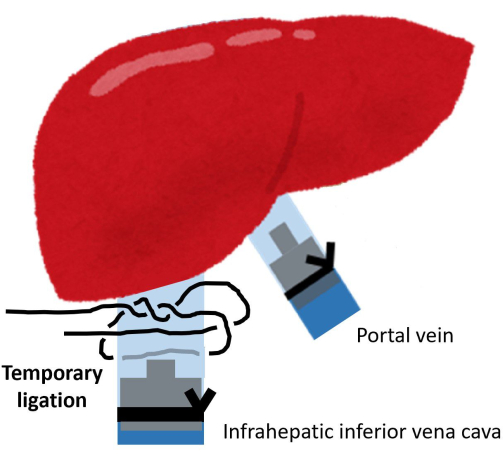
Figure 5: Temporary ligation of the IHIVC root. Please click here to view a larger version of this figure.
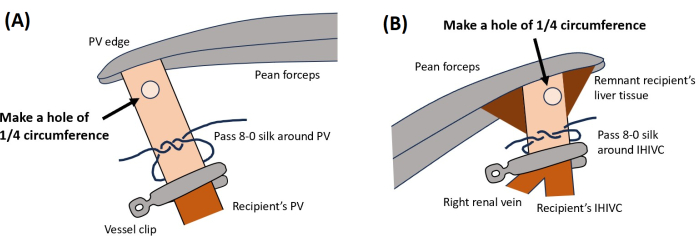
Figure 6: Vascular reconstruction. This illustration shows the locations for creating holes for (A) PV and (B) IHIVC. Please click here to view a larger version of this figure.
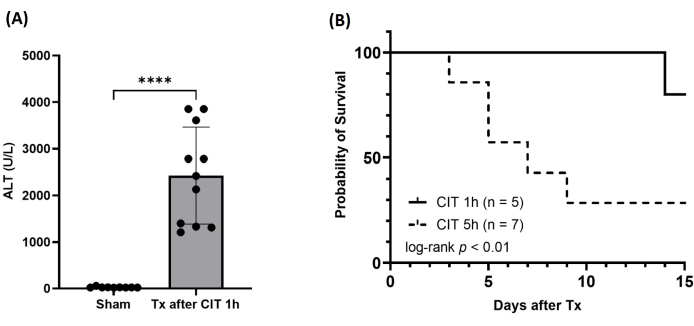
Figure 7: Postoperative results. (A) Serum levels of alanine aminotransferase (ALT) at 6 h after reperfusion. CIT, cold ischemic time; Tx, transplantation. ****p < 0.0001. (B) Survival of the recipient after MOLT. Please click here to view a larger version of this figure.
Discussion
Learning vascular reconstruction is the most challenging aspect of achieving successful MOLT. The cuff’s quality significantly influences the reconstruction’s difficulty, given the small size of mice5. This article provides a detailed protocol for cuff preparation, attachment, and reconstruction.
While there are no major differences from previous reports regarding cuff preparation and connection2,5, some minor points should be considered. First, the cuff handle’s thickness should be limited to approximately 1/6 of the total circumference. If it’s too thin, the handle can easily bend and become hard to grasp. If it’s too thick, it may be challenging to handle in the recipient’s abdomen. Identifying your personal preference early on is essential.
Second, visibility of the lumen at the back table is crucial. A 10-0 nylon thread is used, attached to the donor’s splenic vein as a landmark to confirm the PV lumen. The nylon thread is placed at 8 o’clock to prevent excessive twisting. The vessel wall is externalized by grasping it at 3 o’clock and 9 o’clock with two forceps and pressing it against the cuff wall at 6 o’clock. Fixation with 8-0 silk thread requires careful movements. Adequate fixation is achieved by securing it in the groove. After attachment, lift the cuff to confirm it doesn’t come off.
Third, during reconstruction, it’s important not to create too large a hole in the recipient vessel because the hole naturally enlarges as the cuff is inserted. Making a large hole can lead to vessel splitting, the most common cause of failure. Particularly in IHIVC reconstruction, smaller holes should be made since the shorter length of IHIVC makes it easier to pull out than a PV. When passing saline solution through the lumen, support cuff insertion by applying upward force rather than downward.
Common causes of failure and precautions have been discussed in detail. The most crucial points include ensuring clear visualization of the inner vessel lumen and avoiding unnecessary force during cuff insertion to prevent vessel splitting. These technical tips for the cuff method are believed to elevate skills and advance research.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by 2022 JST basic research (The Japanese Society for Transplantation).
Materials
| 16 G intravenous catheter | TERUMO | SR-FF2032 | IHIVC cuff |
| 20 G intravenous catheter | TERUMO | SR-FF1651 | PV cuff |
| 8-0 braid silk | Natsume Seisakusho | CR9-80B2 | 8-0 silk |
| Belzer UW Cold Storage Solution | Astellas | Organ preservation fluid | |
| Bulldog clamp | B BRAUN | FB329R | Bulldog clamp |
| C57BL/6 mice | Oriental Bio Service | ||
| Isoflurane inhalation solution | Viatris | Anesthesic | |
| Micro Blunted Tips 0.1 mm x 0.06 mm | F.S.T | 11253-20 | Straight microforceps |
| Micro Serrefine Clamp Applicator with Lock | F.S.T | 18056-14 | Vessel clip applicator |
| Micro Serrefines | F.S.T | 18055-4 | Vessel clip |
| No.11 Spare Blades | FEATHER Safety Razor | 11 | Blades |
| Ophthalmic scissor, round handle | B BRAUN | FD103R | Microscissor |
| Plastic rectangular-shaped container | Daiso | 10 cm long, 15 cm wide and 6 cm high | |
| SuperGrip Tips | F.S.T | 00649-11 | Curved microforceps |
| SZX7 | Olympus | SZX7 | Microscope |
References
- Qian, S. G., Fung, J. J., Demetris, A. V., Ildstad, S. T., Starzl, T. E. Orthotopic liver transplantation in the mouse. Transplantation. 52 (3), 562-564 (1991).
- Yokota, S., et al. Orthotopic mouse liver transplantation to study liver biology and allograft tolerance. Nat Protoc. 11 (7), 1163-1174 (2016).
- Shen, X. D., et al. Inflammatory responses in a new mouse model of prolonged hepatic cold ischemia followed by arterialized orthotopic liver transplantation. Liver Transpl. 11 (10), 1273-1281 (2005).
- Kageyama, S., et al. Ischemia-reperfusion injury in allogeneic liver transplantation: A role of CD4 T cells in early allograft injury. Transplantation. 105 (9), 1989-1997 (2021).
- Li, T., Hu, Z., Wang, L., Lv, G. Y. Details determining the success in establishing a mouse orthotopic liver transplantation model. World J Gastroenterol. 26 (27), 3889-3898 (2020).
- Tian, Y., Rüdiger, H. A., Jochum, W., Clavien, P. A. Comparison of arterialized and nonarterialized orthotopic liver transplantation in mice: Prowess or relevant model. Transplantation. 74 (9), 1242-1246 (2002).
- Steger, U., Sawitzki, B., Gassel, A. M., Gassel, H. J., Wood, K. J. Impact of hepatic rearterialization on reperfusion injury and outcome after mouse liver transplantation. Transplantation. 76 (2), 327-332 (2003).
- Chen, J., et al. A review of various techniques of mouse liver transplantation. Transplant Proc. 45 (6), 2517-2521 (2013).
- Kojima, H., et al. T cell carcinoembryonic antigen-related cell adhesion molecule 1-T cell immunoglobulin domain and mucin domain-containing protein 3 cross talk alleviates liver transplant injury in mice and humans. Gastroenterology. 165 (5), 1233-1248 (2023).
- Yokota, S., Yoshida, O., Ono, Y., Geller, D. A., Thomson, A. W. Liver transplantation in the mouse: Insights into liver immunobiology, tissue injury, and allograft tolerance. Liver Transpl. 22 (4), 536-546 (2016).

