Development of a Quantitative Recombinase Polymerase Amplification Assay with an Internal Positive Control
Summary
Provided is a protocol for developing a real-time recombinase polymerase amplification assay to quantify initial concentration of DNA samples using either a thermal cycler or a microscope and stage heater. Also described is the development of an internal positive control. Scripts are provided for processing raw real-time fluorescence data.
Abstract
It was recently demonstrated that recombinase polymerase amplification (RPA), an isothermal amplification platform for pathogen detection, may be used to quantify DNA sample concentration using a standard curve. In this manuscript, a detailed protocol for developing and implementing a real-time quantitative recombinase polymerase amplification assay (qRPA assay) is provided. Using HIV-1 DNA quantification as an example, the assembly of real-time RPA reactions, the design of an internal positive control (IPC) sequence, and co-amplification of the IPC and target of interest are all described. Instructions and data processing scripts for the construction of a standard curve using data from multiple experiments are provided, which may be used to predict the concentration of unknown samples or assess the performance of the assay. Finally, an alternative method for collecting real-time fluorescence data with a microscope and a stage heater as a step towards developing a point-of-care qRPA assay is described. The protocol and scripts provided may be used for the development of a qRPA assay for any DNA target of interest.
Introduction
Quantitative nucleic acid amplification is an important technique for detection of environmental, foodborne, and water-borne pathogens as well as for clinical diagnostics. Real-time quantitative polymerase chain reaction (qPCR) is the gold standard method for sensitive, specific, and quantitative detection of nucleic acids, e.g., for HIV-1 viral load testing, detection of bacterial pathogens, and screening for many other organisms1–3. During real-time qPCR, primers amplify pathogen DNA in cycles, and a fluorescent signal is generated that is proportional to the amount of amplified DNA in the sample at each cycle. A sample containing an unknown concentration of pathogen DNA may be quantified using a standard curve that relates the initial DNA concentration of standard samples and the time at which the fluorescent signal reaches a certain threshold (i.e., the cycle threshold, or CT).
Because real-time qPCR requires expensive thermal cycling equipment and several hours to receive results, alternative isothermal amplification techniques, such as recombinase polymerase amplification (RPA), have been developed4. These platforms generally provide results faster and amplify nucleic acids at a lower, single temperature, which may be accomplished with less expensive, simpler equipment. RPA, which is particularly attractive for point-of-care applications, amplifies DNA in minutes, requires a low amplification temperature (37 °C), and remains active in the presence of impurities5,6. RPA assays have been developed for a wide range of applications, including food analysis, pathogen detection, cancer drug screening, and detection of biothreat agents7–12. However, use of RPA for quantification of nucleic acids has been limited13,14.
In previous work, it was shown that real-time quantitative RPA (qRPA) is feasible15. Here, a more detailed protocol is provided for using real-time quantitative RPA to quantify unknown samples using a standard curve, a method that is analogous to quantification using qPCR. This protocol describes how to perform an RPA reaction on a thermal cycler to detect HIV-1 DNA as a proof-of-concept, as well as how to develop an internal positive control (IPC) to ensure the system is functioning properly. Data collection using a thermal cycler or microscope and data analysis for constructing a standard curve using training data is also detailed. Finally, the method for quantifying unknown samples using the standard curve with a custom script is demonstrated. This qRPA technique enables quantification of samples with unknown concentrations and has many advantages over traditional real-time qPCR.
Protocol
1. Program the Thermal Cycler for Real-time qRPA Reactions
- Create a new protocol in the thermal cycler software.
- Insert a pre-incubation step: 39 °C for 1 min.
- Add a second step: 39 °C for 20 sec followed by a plate read.
- Finally, insert a “GO TO” that repeats the second step 59 more times.
- Save the protocol.
- Create a new plate in the “plate editor” tab of the software. Select wells on the plate corresponding to the locations of the RPA reactions (here, use wells A1, A4-A8, B1, and B4-B8).
NOTE: It is not important whether the sample type of the wells (e.g., “Standard,” “NTC”) is specified, as data analysis is done using a custom script.- For experiments containing HIV-1 DNA only, select the “HEX” fluorophore for all wells. For experiments containing HIV-1 DNA and an internal positive control, select both “HEX” and “FAM” fluorophores for all wells. Save the plate.
2. Prepare for HIV-1 qRPA Experiments
- Assemble RPA reactions in a designated pre-amplification workspace containing designated pipettes, pipette tips, vortexer, and mini-centrifuge, as for qPCR. As RPA may amplify a single copy of DNA by as much as ten orders of magnitude (data not shown), handle and dispose of tubes containing post-amplified DNA products in a designated post-amplification area.
- Prevent contact between all pre-amplification reagents and post-amplification products. Spray pre-amplification workspaces and equipment with 50% bleach before and after all experiments. Use a paper towel to wipe away excess bleach.
- Purchase forward primer sequence 5’-TGG CAG TAT TCA TTC ACA ATT TTA AAA GAA AAG G-3’ and reverse primer sequence 5’-CCC GAA AAT TTT GAA TTT TTG TAA TTT GTT TTT G-3’ from commercial DNA synthesizers. Purchase the probe sequence 5’- TGC TAT TAT GTC TAC TAT TCT TTC CCC [SIMA/HEX] GC [THF] C [dT-BHQ1] GTA CCC CCC AAT CCC C -3’ from commercial sources. Store all primers and probes at 4 °C in aliquots at a concentration of 10 µM prior to use.
NOTE: The qRPA assay amplifies a unique HIV-1 sequence. For this proof-of-concept assay, use the plasmid pHIV-IRES- eYFPΔEnvΔVifΔVpr described by Sutton et al. which contains the target sequence16. For qRPA experiments, the plasmid was stored at 4 °C in aliquots containing 1, 10, 102, 103, and 104 copies per µl in a buffer containing 10 mM Tris, 0.1 mM EDTA at pH 8.0, and 1 ng/µl human genomic carrier DNA (360486). This carrier DNA concentration is equivalent to the DNA concentration obtained from commercial serum DNA extraction kits used for HIV-1 diagnosis17. These HIV-1 dilutions were used as templates in qRPA reactions to build a standard curve.
3. Assemble an HIV-1 qRPA Standard Curve
- Before assembling the qRPA reactions, prepare the pre-amplification workspace as described in step 2.1.1. Place a 96-well cold block in storage at 4 °C.
- Prepare reagents for 12 reactions:
- Move the magnesium acetate, the rehydration buffer, and 12 RPA reaction pellets to the pre-amplification workspace.
- Thaw the magnesium acetate and the rehydration buffer at room temperature.
- Aliquot 32.5 µl of magnesium acetate (2.5 µl per reaction) to a microcentrifuge tube.
- Prepare a 13x master mix. Add 383.5 µl of the rehydration buffer (29.5 µl per reaction) to a separate microcentrifuge tube for the master mix. Add 41.6 µl of nuclease free water (3.2 µl per reaction) to the master mix. Vortex the master mix.
- Return the magnesium acetate and rehydration buffer to storage at -20 °C.
- Move the primer and probe aliquots from the refrigerator to the pre-amplification area, avoiding excessive light exposure to minimize photobleaching.
- Add 27.3 µl each of the 10 µM forward and reverse primers (2.1 µl per primer per reaction) to the master mix.
- Add 7.8 µl of the 10 µM HEX-labeled HIV-1 DNA probe (0.6 µl per reaction) to the master mix.
- Return primers and probe to storage.
- Matching the plate layout described in Section 1.2, place 1 enzyme pellet in each of the far left tubes and 1 enzyme pellet in each of the 5 far right tubes of 2 low-rise 8-well PCR tube strips.
- Carefully add 37.5 µl of master mix to each tube containing an enzyme pellet, using a new pipette tip for each tube and gently stirring the master mix and pellet with the tip until the pellet is fully dissolved. Stir gently to prevent bubble formation, and remove the tip carefully to prevent any loss of the reaction volume.
- After all enzyme pellets have been rehydrated with the master mix, retrieve the 96-well cold block from 4 °C storage. Place the PCR tube strips in the cold block to chill the master mix.
- While the master mix is cooling, load the thermal cycler software with the protocol and plate described in Section 1.
- Cut 2 strips of clear plastic micro-seal adhesive to be slightly wider than the PCR tubes, and retrieve 2 flat PCR tube strip lids.
- Retrieve 6 template aliquots from storage (containing 0, 1, 101, 102, 103, or 104 copies of the HIV-1 plasmid per microliter).
- Add 10 µl of each template to the tubes designated for that template concentration. Use a new pipette tip for each tube, gently stirring the solution with the tip to mix the master mix and template. Be sure to add the template to the correct location to match the plate layout described in Section 1.2.
- Ensure that a PCR tube strip adapter for the microcentrifuge is in place.
- Dispense 2.5 µl of magnesium acetate into each tube lid that will be placed on a tube containing a qRPA reaction.
- Gently place the lids on top of the PCR tubes such that they are not fully sealed and may be removed. The magnesium acetate will adhere to the lids and be clearly visible from above.
- Insert each PCR tube strip with lids into each side of the PCR tube holder. Close the lid of the minifuge to spin the magnesium acetate out of the lids and into the RPA reaction, initiating the RPA reaction. Centrifuge until all bubbles are eliminated and all liquid is collected in the bottom of each tube (about 10 sec).
- Quickly remove the PCR tube strips and return them to the cold block to halt the qRPA reactions.
- Carefully remove the lids to prevent aerosol formation and seal each PCR tube strip with the cut pieces of clear micro-seal film.
- Quickly walk to the thermal cycler and load the two PCR tube strips into the thermal cycler in the locations matching the plate layout (wells A1-B8). Close the lid of the thermal cycler, click “Start Run,” and designate a filename for the experiment.
NOTE: Although the manufacturer recommends agitating the sample after 5 min of incubation, this step is not necessary for this particular assay and may be omitted. - After the run is completed, select “Settings,” “Baseline Setting,” “No Baseline Subtraction” to display the raw fluorescence data. Export the data to a spreadsheet program by selecting “Export,” “Export all Data Sheets to Excel.” A file with the addendum “Quantification Amplification Results” will be created containing the raw real-time fluorescence data.
4. Develop an Internal Positive Control
- Select a DNA sequence from a species that is unlikely to be found in the target sample. The sequence needs to be longer than the target amplicon so that generation of the target sequence is favored.
NOTE: For this assay, Cryptosporidium parvum, an intestinal parasite, was chosen to serve as the template for generation of the IPC sequence because this organism is unlikely to be found in blood and was available in the lab from another project. To generate the IPC sequence, extract and purify DNA from C. parvum oocysts as described elsewhere18. - Purchase the forward (5’-TGG CAG TAT TCA TTC ACA ATT TTA AAA GAA AAG G/ ATC TAA GGA AGG CAG CAG GC-3’) and reverse (5’- CCC GAA AAT TTT GAA TTT TTG TAA TTT GTT TTT G/ TGC TGG AGT ATT CAA GGC ATA -3’) primers from commercial sources. The IPC PCR primers used in this assay are shown in Figure 1 and contain one region that is complimentary to the IPC template DNA (e.g., C. parvum, purple in Figure 1) and one region that is complimentary to the target organism (e.g., HIV-1, blue in Figure 1).
- Perform PCR using the primers and IPC template DNA.
- Assemble 50 µl PCR reactions using the Phusion High-Fidelity DNA Polymerase kit reagents according to the manufacturer’s instructions, excluding the polymerase. Use 2 µl of DNA template and Phusion Buffer HF.
- Add the Phusion polymerase to each reaction after a hot start at 98 °C, followed by 60 sec at 98 °C, followed by 40 cycles of 15 sec at 98 °C, 30 sec at 62 °C, and 30 sec at 72 °C with a final extension at 72 °C for 5 min. After thermal cycling, hold samples at 4 °C.
- Analyze samples by gel electrophoresis on a 2% TAE agarose gel, and then extract and purify DNA using the QIAquick Gel Extraction Kit according to the microcentrifuge protocol included with the kit.
- Screen the IPC candidates for their ability to amplify using qRPA.
- Prepare qRPA reactions as described in Section 3, using HIV-1 primers, a FAM-labeled probe specific to the IPC (5’-AGG TAG TGA CAA GAA ATA ACA ATA CAG GAC [FAM] T [THF] T [dT-BHQ1] GGT TTT GTA ATT GGA A -3’), and a total of 0, 10, 102, 103, 104, or 105 copies of each IPC candidate per reaction, testing all concentrations in duplicate.
- Choose the IPC candidate that amplifies most consistently and has the lowest limit-of-detection.
- Co-amplify HIV-1 and the IPC to determine the optimal concentration of IPC DNA.
- Similar to Section 3, combine the following in each 50 μl reaction: 29.5 μl of rehydration buffer, 2.1 μl of RPA of forward primer [10 μM], 2.1 μl of RPA reverse primer [10 μM], 0.6 μl of HIV-1 HEX-labeled probe [10 μM], 10 μl of HIV-1 DNA at varying concentrations, 2.5 μl of magnesium acetate, and 1 enzyme pellet. Instead of adding 3.2 µl of nuclease free water as was done in step 3.2.4, add 0.6 μl of IPC FAM-labeled probe [10 μM], and 2.6 μl of IPC DNA. Use the IPC concentration that is the limit-of-detection (LOD) when qRPA is performed with IPC DNA alone (LOD defined as the lowest concentration where the amount of fluorescence signal generation is greater than that generated by the fluorescence generated by the samples with no target DNA).
- Incubate the samples using the protocol described in Section 1.1.
- If the IPC does not amplify in every sample, consider repeating the experiment with an IPC concentration one order of magnitude higher. The optimal concentration of IPC DNA for the HIV-1 assay is 10,000 copies/μl. While samples are considered valid if there is either IPC or HIV-1 DNA amplification, ideally the IPC exhibits detectable amplification for every reaction.
5. Building a Standard Curve from Multiple Experiments
- Ensure the data for each experiment has been exported to a spreadsheet program as described in Section 3.13.
- Open and run the script “JoVE_qRPA_standard_curve.m”. All inputs are automatically prompted in the command window. After the script is initiated, the user is automatically prompted to designate the “Number of data files” used to build the standard curve.
- In the command window, type the number of files to be used to build the standard curve and press enter.
- Type in the command window the number of samples and the number of replicates in each training file (pressing enter after each entry).
NOTE: In the plate layout described in Section 1.2, there were 6 samples with different concentrations and 2 replicates of each sample). - Type in the command window the lowest DNA concentration used to build the standard curve (in log10 copies) and press enter (for the plate layout described in Section 1.2, the lowest template concentration is ‘1’ log10 copies).
- Type in the command window the difference in concentration between each standard (in log10 copies) then press enter (for the plate layout described in Section 1.2, the concentration interval is ‘1’ log10 copies).
- After being prompted, type the “Slope threshold” in the command window and press enter.
- In the command window, type the “Number (z) standard deviations above the background for positive threshold,” and then press enter. Typical values for z range from 1 to 5.
- Specify whether the data was collected using the thermal cycler (‘1’), *.tiff microscope images (‘2’), or *.jpg microscope images (‘3’) by typing the number ‘1,’ ‘2,’ or ‘3,’ and pressing enter.
- Choose to set a new IPC threshold or to use a default value for the threshold by typing ‘y’ or ‘n,’ respectively, when prompted.
- Next, select each of the spreadsheet files used to build the standard curve.
NOTE: The script will then automatically import the HEX and FAM fluorescence data; determine whether each reaction was valid (i.e., whether the fluorescence signals exceeded the thresholds for the HEX or FAM channels); determine the r2 coefficient for an exponential, linear, and second-order polynomial fit; plot “log10 copies versus the cycle threshold” for every sample used to build the standard curve; and create a spreadsheet file containing essential variables to rebuild the standard curve for validation experiments without requiring the user to re-enter all of the input parameters again.
6. Assay Validation and Quantification of Unknown Samples Using the Standard Curve
- Use the script “JoVE_qRPA_validation_and_quantification.m” to estimate the precision and accuracy of the assay by using samples with known concentrations or use it to quantify samples with unknown concentrations.
NOTE: Because previously published research showed that an exponential fit yielded the best r-squared coefficient18, this script utilizes an exponential fit for the standard curve. If a different type of fit is desired, the script may be modified accordingly.- Open and run the script “JoVE_qRPA_validation_and_quantification.m”. After the script is initiated, the user is automatically prompted to enter all inputs into the command window.
- Enter ‘1’ to validate an assay using a standard curve with known sample concentrations or enter ‘2’ to quantify unknown samples.
- When prompted, either select a saved standard curve or build a new one by entering the information that is automatically solicited in the command window (the same information that is required for the “JoVE_qRPA_standard_curve.m” script from Section 5).
- Type the number of experiments (i.e., spreadsheet files) to analyze for validation/quantification.
7. Preparation for Data Collection Using a Fluorescence Microscope and a Heated Chip
- Ensure the fluorescence microscope has a stage heater and 1-Channel Precision High Stability temperature controller in place.
- Ensure the fluorescence microscope has a filter cube to excite and collect fluorescence from each fluorophore. Excite and collect the FAM fluorescence generated during IPC DNA amplification through a GFP filter (excitation BP 470/40 nm, emission BP 520/50 nm). Excite and collect the HEX fluorescence generated during HIV-1 DNA amplification through a HEX filter (excitation BP 530/30 nm, emission BP 575/40 nm).
- Use the microscope script editing software to create an automated algorithm to replicate data-collection on the thermal cycler.
- First insert a “Settings” step to close the shutter. Next add an “Exposure” step to set the exposure to 60 sec. Insert a “Snap” to execute the 60 sec delay, which implements the 1 min pre-incubation period. Add a “Setting” step to change the working group to the GFP filter. Insert another “Exposure” step to adjust the exposure to 200 msec. All exposures using the GFP or the HEX filter cubes will be set to 200 msec.
- After the “Exposure” step, add a “Snap” and specify the camera with which the image will be taken. Use another “Setting” step to close the shutter and a “Save Image” step to save the image to a user-defined temporary folder.
- Next switch to the HEX filter cube using another “Setting” step and then add an “Exposure” at 200 msec, a “Snap,” and a “Save Image” step.
- Repeat this process 59 times with an initial delay of 20 sec instead of 60 (close shutter, wait 20 sec, switch to GFP filter cube, set exposure to 200 msec, snap, close shutter, save, switch to HEX filter cube, set exposure to 200 msec, snap).
- Create a chip that will hold qRPA reactions.
- For experiments using the microscope, use the file JoVE_qRPA_base.ai to cut a 40 x 15 mm rectangular base from a sheet of 1/8” thick black acrylic using a laser cutter set to 100% power, 10% speed.
- Use the file JoVE_qRPA_well.ai to cut reaction well consisting of a 10 x 10 mm square with a 5 mm diameter circular hole cut from a sheet of 1.5 mm thick clear acrylic.
- Attach up to three wells to a single base using superglue gel.
- Dry overnight.
8. Data Collection and Analysis Using a Fluorescence Microscope
- Prepare the microscope for data collection by loading the automatic data collection script JoVE_AxioVision_Script.ziscript.
- Set the stage heater to 48 °C and preheat the reaction chip on the stage heater for about 20 min. A temperature setting of 48 °C maintains a temperature of 39 °C in the reaction well (data not shown).
- Using a 10X, 0.45 NA objective, focus on the top of the inner edge of the reaction well with the GFP filter in place. The edges of acrylic are autofluorescent after laser cutting and may be easily visualized. After focusing, do not adjust the height of the microscope stage until all data are collected.
- Assemble the qRPA master mix in a designated pre-amplification workspace as described in step 4.5.1. For each sample, mix an enzyme pellet, master mix, and HIV-1 DNA template in a microcentrifuge tube. Add 2.5 µl of magnesium acetate to the first reaction and briefly vortex.
- Quickly transport the microcentrifuge tube containing the qRPA sample to the fluorescence microscope.
- Carefully pipette 30 µl of the RPA reaction into the reaction well without creating bubbles. Place a drop of PCR-grade mineral oil over the RPA reaction to prevent evaporation.
- Position the well directly beneath the microscope objective, taking care to image only the center of the well, not any of the auto-fluorescent acrylic edges. After the well is positioned, run the automated script 7. The script generates one JPEG image using the FAM filter cube and one JPEG image using the HEX filter cube every 20 sec.
- After the script is completed, transfer these images out of the temporary storage folder before running additional samples.
- Create 2 folders: 1 for each fluorophore (i.e., ‘HEX’ and ‘FAM’). Store both folders in the same parent folder. In each fluorophore folder, create a folder labeled with the number of DNA copies present in the sample (e.g., 0, 10, etc.).
- Transfer all files with the prefix ‘HEX’ into the corresponding subfolder in the ‘HEX’ folder. Transfer all files with the prefix ‘GFP’ into the corresponding subfolder in the ‘FAM’ folder.
- Repeat Sections 8.2-8.4 for qRPA reactions with 0, 10, 102, 103, 104, and 105 HIV-1 DNA copies.
- After collecting the data for all qRPA samples in an experiment, load the script “JoVE_real_time_intensity_to_excel.m.” When prompted by the script, select the parent folder containing the 2 fluorophore folders, which in turn contain the images for each concentration in subfolders.
NOTE: The script will automatically generate a spreadsheet file in the parent directory in which the HEX fluorescence data will be saved in a sheet named ‘HEX’, and the FAM fluorescence data will be saved in a sheet named ‘FAM’. In each sheet, the cycle number is listed in column B, and the real-time fluorescence data for increasing concentrations of template are given in columns C, D, etc. Each real-time fluorescence datum is the average fluorescence intensity of the image captured at that time point. - Use the default scatter plot function in the spreadsheet program to plot cells B2:H61 of the ‘HEX’ and ‘FAM’ sheets to visualize real-time fluorescence and generate plots similar to those in Figures 2A, 2B, 3A, and 3B.
NOTE: The spreadsheet generated in step 8.6 may also be used in the scripts “JoVE_qRPA_standard_curve.m” and “JoVE_qRPA_validation_and_quantification.m”.
Representative Results
Before selecting a sequence to serve as the IPC in qRPA experiments with target (HIV-1) DNA, internal positive control (IPC) candidates are generated and screened for their ability to amplify in qRPA reactions without HIV-1 DNA present. IPC candidates are longer than the target (HIV-1) DNA to prevent IPC formation from out-competing HIV-1 amplicon formation. As shown in Figure 2A, the generation of two C. parvum IPC candidates was verified by the presence of 415 and 435 bp bands using gel electrophoresis. In qRPA reactions, the shorter IPC candidate exhibited little amplification (Figure 2B), while the longer candidate consistently amplified when a total of 104 and 105 copies were present (Figure 2C). Thus, the longer candidate was chosen to be the IPC for HIV-1 qRPA experiments.
Real-time qRPA may be performed using the target of interest alone or using both the target and the IPC. Figure 3 shows an experiment on the thermal cycler using both HIV-1 DNA and the C. parvum IPC. In this experiment, 2.6 x 104 copies of IPC DNA were added to each reaction, a concentration close to the IPC limit of detection, to avoid affecting the limit of detection of HIV-1 target DNA. As demonstrated in Figure 3A, which displays HEX fluorescence data corresponding to real-time HIV-1 DNA generation, the time at which detectable amplification begins is inversely proportional to the initial target concentration. Thus, amplification is apparent earlier for high concentrations of HIV-1 DNA and later for low concentrations of HIV-1 DNA. In contrast, detectable amplification of IPC DNA begins at approximately the same time because the starting IPC concentration is the same in all samples, as shown in Figure 3B, which displays FAM fluorescence data corresponding to IPC DNA generation during the same experiment. The rate of fluorescence generation from the IPC is inversely proportional to the concentration of HIV-1 DNA due to competition during amplification.
With the goal of developing a field-operable fluorescence reader with qRPA reactions, qRPA experiments may be performed on an upright fluorescence microscope with a stage heater and 1-Channel Precision High Stability temperature controller. Figure 4A and 4B display HEX and FAM fluorescence data collected on a microscope. Data collected on the microscope using laser-cut chips demonstrate slight variability in baseline fluorescence and crests and troughs that may be due to photobleaching. However, the script determines the cycle threshold using the rate of change of fluorescence during the initial amplification period, which is unaffected by baseline fluorescence or variability in fluorescence after the initial amplification has occurred. As seen in Figure 4C, the standard curve built from this experiment has an r2 coefficient of 0.990. Notably, the IPC is amplified only for low concentrations of HIV-1 DNA. Although this behavior differs from experiments performed on the thermal cycler, all samples are still classified as valid using this method.
Data from multiple experiments using known target concentrations may be compiled to construct a standard curve, which may then be used to assess assay performance or quantify samples with unknown concentrations. Figure 5 shows the data from five experiments and an exponential standard curve generated using the script “JoVE_qRPA_standard_curve.m”, relating the initial HIV-1 target concentration to the time at which detectable amplification began. Figure 5 used 5 separate experiments, each containing 2 qRPA reactions at each template concentration to build a standard curve. Note that the exponential fit has a high r2 coefficient of 0.959. The standard curve was then used to predict the concentrations of additional samples with known concentrations for assay validation using the script “JoVE_qRPA_validation_and_quantification.m” script. Table 1 shows the quantification results for 5 additional experiments using the standard curve in Figure 5. Note that the algorithm correctly classified all samples containing HIV-1 DNA as positive and 9 out of 10 of the no-target-control samples as negative. In addition, the average predicted concentration was within one order of magnitude of the correct concentration and the standard deviation for predicted concentrations was less than 0.5 log10 (copies per reaction).
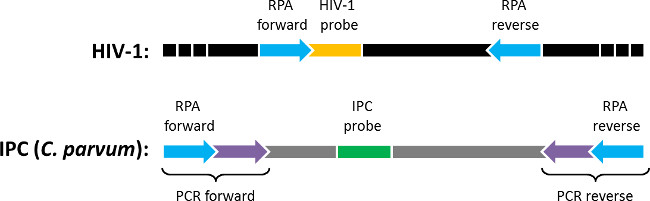
Figure 1: Block diagram of quantitative RPA DNA targets. The qRPA assay amplifies two DNA sequences flanked by identical sequences to which primers bind (“RPA forward” and “RPA reverse”). The amplified sequences are 1) a sequence within the HIV-1 genome (“HIV-1”) that is detected with the “HIV-1 probe” and 2) an internal positive control sequence (“IPC (C. parvum)”) included in each reaction that is detected with the “IPC probe.” The IPC was generated via PCR using the Cryptosporidium parvum genome as a template and primers (“PCR forward” and “PCR reverse”) that contain a region complementary to C. parvum (purple) and a region complementary to HIV-1 (blue). Figure originally published in Analytical Chemistry 2014, reprinted here with permission9.
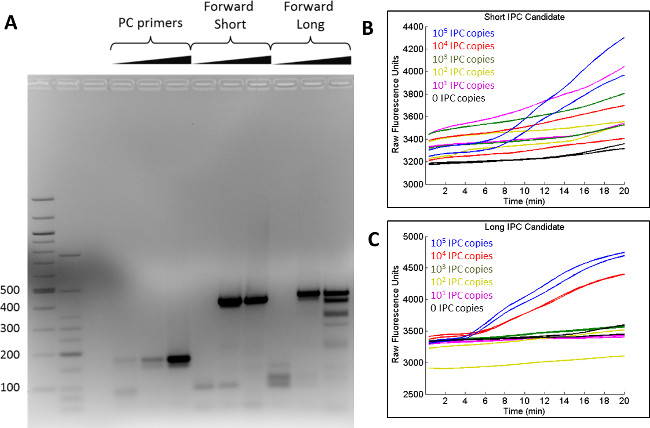
Figure 2: Screening of IPC candidates. (A) Two IPC candidates (‘Forward Short’ and ‘Forward Long’) and a known PCR positive control sequence were generated using PCR and visualized on an agarose gel, where 415 bp and 435 bp bands were visible. (B) The short IPC candidate exhibited little amplification during real-time RPA, while (C) the longer IPC candidate amplified consistently when a total of 104 and 105 copies were present. Figure originally published in Analytical Chemistry 2014, reprinted here with permission9. Please click here to view a larger version of this figure.
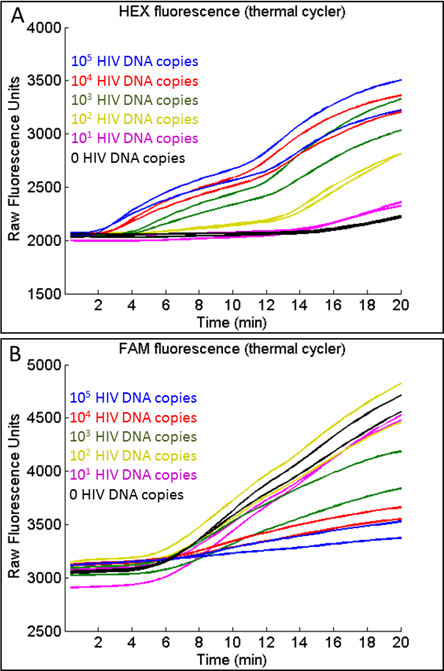
Figure 3: Typical raw fluorescence data generated during co-amplification of HIV-1 DNA and IPC on a thermal cycler. (A) For HIV-1, the onset of detectable amplification, shown by an apparent increase in HEX fluorescence, occurs earlier for high concentrations of HIV-1 DNA. (B) For the IPC, the onset of detectable amplification, shown by an apparent increase in FAM fluorescence, occurs at approximately the same time regardless of the HIV-1 DNA concentration. However, the rate of FAM fluorescence generation is inversely proportional to the amount of HIV-1 DNA present due to competition. Figure originally published in Analytical Chemistry 2014, reprinted here with permission9.
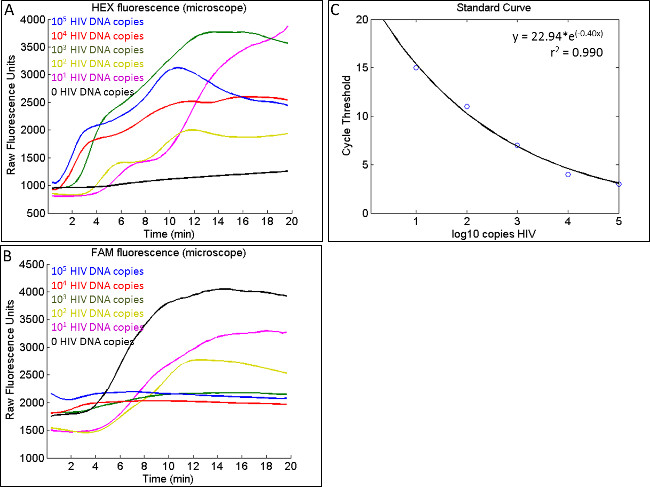
Figure 4: Typical raw fluorescence data generated during co-amplification of HIV-1 DNA and IPC on a microscope. (A) Similar to data generated on the thermal cycler, the onset of detectable HIV-1 amplification, shown by an apparent increase in HEX fluorescence, occurs earlier for high concentrations of HIV-1 DNA. (B) IPC amplification on the microscope, shown by FAM fluorescence, is apparent for low HIV-1 DNA concentrations but not always apparent in samples with high HIV-1 DNA concentrations. (C) A standard curve can be built using the JoVE_standard_curve.m scripts that yields a high r2 coefficient.
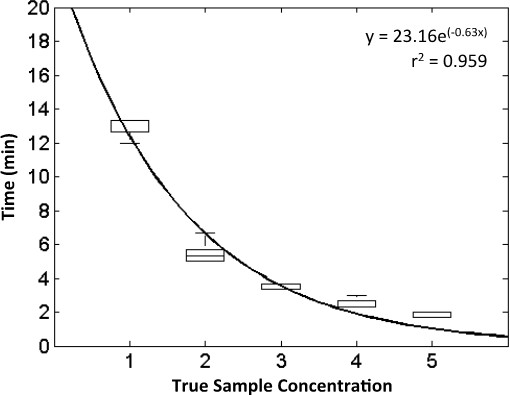
Figure 5: Typical standard curve for HIV-1. Using the JoVE_standard_curve.m script, raw HEX fluorescence data generated during HIV-1 amplification is processed to build a standard curve, which may be used to predict the HIV-1 DNA concentration of unknown samples. This standard curve (z = 1) was generated using data from 5 experiments, in which 6 concentrations were tested using 2 replicates at each concentration. All concentrations are given in log10 copies. Figure originally published in Analytical Chemistry 2014, reprinted here with permission9.
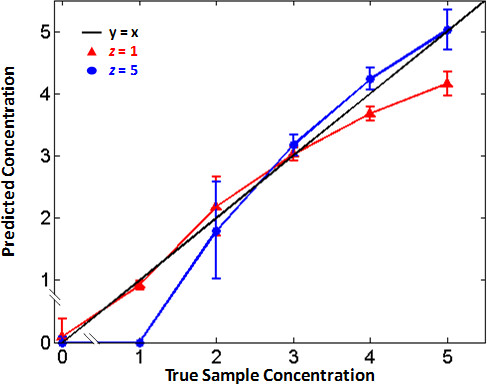
Figure 6: Algorithm tunability. Adjusting the algorithm parameters changes the standard curve used to predict the concentration of unknown samples. The JoVE_validation_and_quantification.m script was used to predict the concentration of unknown samples using z = 1 (red) and z = 5 (blue). All concentrations are given in log10 copies. Predictions are more accurate for low DNA concentrations when z = 1; predictions are more accurate for high DNA concentrations when z = 5. By adjusting the algorithm parameters, the qRPA standard curve may be tuned according to clinical needs. Figure originally published in Analytical Chemistry 2014, reprinted here with permission9.
| Sample Concentration | Average Predicted Concentration | Standard Deviation | Percent of Samples Identified as Positive |
| No target controls | 0.1 | 0.3 | 10% |
| 1 | 0.9 | 0.1 | 100% |
| 2 | 2.2 | 0.5 | 100% |
| 3 | 3 | 0.1 | 100% |
| 4 | 3.7 | 0.1 | 100% |
| 5 | 4.2 | 0.2 | 100% |
Table 1: HIV-1 qRPA assay validation. Using the standard curve in Figure 4, the JoVE_validation_and_quantification.m script was used to predict the concentrations (in log10 copies) of samples for 5 additional experiments. Table originally published in Analytical Chemistry 2014, reprinted here with permission9.
Discussion
In order to obtain meaningful quantification data using the MATLAB algorithm, the user must select appropriate input values when prompted. After initiating each script in Sections 5 and 6, all input variables are automatically solicited in the command window and outputs are automatically generated. In Section 5.7 the user is prompted to select a slope threshold. The value of the slope threshold affects the square of the correlation coefficient (r2) of the fit. When using raw fluorescence data exported from a thermal cycler, values between 2.0 and 5.0 tend to yield a high r2 coefficient. In Section 5.8 the user must designate the number of standard deviations above the background to set the positive threshold. To score a sample as positive or negative, the script automatically determines the difference Δsample between the maximum and minimum fluorescence for each sample using the raw fluorescence data exported from the thermal cycler. It calculates the average difference Δbackground and standard deviation σbackground for all no-target control samples. A sample is considered positive if Δsample is more than z × σbackground above Δbackground. In Section 5.10 the user decides whether to use the default threshold or set a new threshold. If the user wishes to set a new threshold, determine the new threshold experimentally by performing 3 experiments each containing 12 RPA reactions without any HIV-1 DNA present. Set the threshold at the average increase in fluorescence intensity from these experiments plus 3 standard deviations. After completing Section 6, the script JoVE_qRPA_validation_and_quantification.m automatically returns the estimated DNA concentration for each qRPA reaction (in log10 copies). If the script determines that no HIV-1 DNA was present in the sample, the estimated concentration is listed as either “Negative for HIV” or “Invalid,” depending on whether the fluorescent signal for the internal positive control exceeded the threshold (z × σbackground + Δbackground). If the user is validating the standard curve with known sample concentrations, the script will also return an additional table similar to that of Table 1.
In order to develop a real-time RPA assay that provides accurate quantification, experimental consistency is crucial. For example, use the same primer and probe aliquots for both the standard curve and validation experiments. Also avoid freeze-thaw cycles by storing the primer and probe aliquots at 4 °C between experiments rather than -20 °C. The template aliquots used for the standard curve and validation experiments are stored in the same manner. RPA enzyme pellets and reagents from the same lot are used according to the manufacturer recommendations. Lastly, because RPA lacks true ‘cycles’ to precisely control the rate of amplification, standardization of user steps is absolutely imperative. When assembling reactions, the user must always follow the same steps in the same order, spending approximately the same amount of time on each step. Reactions must always be mixed gently with a new pipette tip, and bubbles must always be eliminated. Before amplification, reactions must be held at a consistent temperature, and the thermal cycler or microscope software must always be prepared before loading reactions to avoid any amplification at sub-optimal temperatures that could influence quantification. Any variation in the initial reaction conditions may lead to inconsistency in experimental outcomes.
When using a microscope to collect data, additional variables must be controlled to minimize variation in fluorescence intensity. All reactions must be placed in the same region on the stage warmer, and the microscope must be focused on the same region of the well for every sample. Even if these practices are followed, fluorescence data collected on a microscope may exhibit variability due to local bright spots that naturally form during RPA reactions, bubble formation within the reaction chambers, or photobleaching resulting from repeated exposure to excitation light. The influence of these variables is evident in fluorescence data collected on the microscope (Figures 4A and 4B), which demonstrate baseline variability, peaks, and troughs. These features are absent from the fluorescence data collected on the thermal cycler (Figures 3A and 3B). Ultimately, data collection on the microscope is for proof-of-principle purposes only and the final assay will be implemented on a field-operable fluorescence reader with more precise geometry and software control that minimizes these variables.
Another important aspect of the qRPA assay development process is consistency in data processing. The protocol described in the methods section uses scripts to process raw fluorescence data (stored in a spreadsheet file) collected from a thermal cycler or microscope. All experiments used to build the standard curve must be formatted identically. When using a thermal cycler to collect data, the same plate layout must be used, and data from wells that do not contain RPA reactions must not be exported. When using a microscope to collect data, the format of the data must match the format of the data automatically exported from the thermal cycler. For example, the no-target-control data must be in cells C2:C61, and data for increasing template concentrations must be in cells D2:D61, E2:E61, etc. If there are multiple replicates of each concentration in an experiment, the 2nd replicate dilution series must be ordered left to right from no target control (NTC) to highest concentration and saved in the columns immediately to the right of the 1st replicate dilution series. For example, in the plate layout used in Section 1.2 with 2 replicates for each sample, fluorescence data for the 1st replicate of each sample in the dilution series must be saved in cells C2:H61 and fluorescence data for the 2nd replicate of each sample in the dilution series must be saved in cells I2:N61. For the plate layout used in Section 1.2, this is the default formatting when exporting data from the thermal cycler software to a spreadsheet.
Representative data provided from HIV-1 qRPA experiments demonstrate proof-of-concept support that RPA may be used for quantification of nucleic acid concentration in unknown samples. Clinically useful HIV-1 viral load tests have a clinical range of at least 4 orders of magnitude, a precision of 0.5 log10 copies, and a limit-of-detection of at least 200 copies19,20. The HIV-1 DNA assay described meets these criteria and is most accurate at low concentrations, as shown in Table 1. Therefore, with the inclusion of a reverse transcriptase step, these results suggest that an HIV-1 RT-RPA assay may have the potential to measure HIV-1 viral load in clinical samples. When developing a qRPA assay, adjusting the algorithm parameters may tune the sensitivity and linear dynamic range depending on clinical needs. Figure 6 shows that adjusting z (a parameter that determines the threshold for positive samples) can influence the sensitivity and accuracy at low and high target concentrations. Furthermore, it may be possible to increase the resolution and accuracy of quantification by incubating reactions at a lower temperature or using less magnesium acetate, thereby decreasing the rate of amplification.
This proof of concept qRPA assay can be used to quantify the concentration of samples containing HIV-1 DNA. The qRPA assay described in this manuscript includes detailed instructions on how to assemble real-time RPA reactions, develop and screen an IPC, and process raw fluorescence data to build a standard curve that can be used to quantify unknown samples. With the detailed instructions included, this protocol may be adapted to quantify DNA concentration in a wide variety of samples.
Declarações
The authors have nothing to disclose.
Acknowledgements
This research was funded by a grant from the Bill & Melinda Gates Foundation through the Grand Challenges in Global Health Initiative.
Materials
| qRPA Assay | |||
| Supply | Vendor | Part number | Comments/Description |
| HIV-1 forward primer | Integrated DNA Technologies | custom DNA oligos | 5’-TGG CAG TAT TCA TTC ACA ATT TTA AAA GAA AAG G-3’ |
| HIV-1 reverse primer | Integrated DNA Technologies | custom DNA oligos | 5’-CCC GAA AAT TTT GAA TTT TTG TAA TTT GTT TTT G-3’ |
| HIV-1 probe | BioSearch Technologies | custom DNA oligos | 5’- TGC TAT TAT GTC TAC TAT TCT TTC CCC [SIMA/HEX] GC [THF] C [dT-BHQ1] GTA CCC CCC AAT CCC C -3’ |
| IPC probe | BioSearch Technologies | custom DNA oligos | 5’-AGG TAG TGA CAA GAA ATA ACA ATA CAG GAC [FAM] T [THF] T [dT-BHQ1] GGT TTT GTA ATT GGA A -3’ |
| RPA exo reagents (pellets, rehydration buffer, magnesium acetate | TwistDx | TwistAmp exo | |
| PCR tube strips | BioRad | TLS0801 | |
| PCR flat cap tube strips | BioRad | TCS0803 | |
| Micro-seal adhesive | BioRad | 558/MJ | |
| HIV-1 target (pHIV-IRES- eYFPΔEnvΔVifΔVpr) | custom plasmid, see: Segall, H. I., Yoo, E. & Sutton, R. E. Characterization and detection of artificial replication-competent lentivirus of altered host range. Molecular Therapy 8, 118–129, doi:10.1016/S1525-0016(03)00134-5 (2003). | ||
| Human male genomic DNA | Applied Biosystems | 360486 | |
| 96 well cold-block | Cole Parmer | EW-36700-48 | |
| Thermal cycler | BioRad | CFX96 | |
| MiniFuge | VWR | 93000-196 | |
| Vortex | VWR | 58816-121 | |
| Tris buffer 1.0 M, pH 8.0 | EMD Millipore | 648314 | |
| EDTA 0.5 M, pH 8.0 | Promega | V4321 | |
| Nuclease free water | Ambion | AM9937 | |
| IPC Development | |||
| Supply | Vendor | Part number | Comments/Description |
| Cryptosporidium parvum IPC template | Waterborne Inc | P102C | It is also possible to order a double stranded synthetic target from IDT if the user is unequipped to work with C. parvum (a BSL-2 infectious agent). PCR and RPA primers for C. parvum were designed using GenBank accession number AF115377.1 |
| PCR long forward primer | Integrated DNA Technologies | custom DNA oligos | 5’-TGG CAG TAT TCA TTC ACA ATT TTA AAA GAA AAG G/ ATC TAA GGA AGG CAG CAG GC-3’ |
| PCR long reverse primer | Integrated DNA Technologies | custom DNA oligos | 5’- CCC GAA AAT TTT GAA TTT TTG TAA TTT GTT TTT G/ TGC TGG AGT ATT CAA GGC ATA -3’ |
| Phusion High-Fidelty DNA Polymerase | New England Biolabs | M0530S | |
| Qiaquick Gel Extraction Kit | Qiagen | 28704 | |
| TAE 10X buffer | EMD Millipore | 574797 | |
| Agarose | Sigma Aldrich | A9539 | |
| Microscope Experiments | |||
| Supply | Vendor | Part number | Comments/Description |
| Upright fluorescence microscope | Zeiss | Zeiss Imager.J1 | |
| Stage heater | Bioscience Tools | TC-GSH | |
| 1-Channel Precision High Stability Temperature Controller | Bioscience Tools | TC-1100S | |
| FAM/GFP filter cube | Zeiss | filter set 38 (000000-1031-346) | excitation BP 470/40 nm, emission BP 520/50 nm |
| HEX filter cube | Chroma | 49014 | excitation BP 530/30 nm, emission BP 575/40 nm |
| Laser cutter | Engraver's Network | VLS3.60 | |
| 1/8" black acrylic | McMaster Carr | 8505K113 | |
| 1.5 mm clear acrylic | McMaster Carr | PD-72268940 | |
| Super glue | Office Depot | Duro super glue | |
| PCR grade mineral oil | Sigma Aldrich | M8662-5VL | |
| Data Analysis | |||
| Software | Vendor | ||
| Microsoft Excel | Microsoft | ||
| MATLAB | MATLAB | ||
| MATLAB script: "JoVE_qRPA_standard_curve.m” | Included in SI | ||
| MATLAB script: "JoVE_qRPA_validation_and_quantification.m” | Included in SI | ||
| MATLAB script: "JoVE_real_time_intensity_to_excel.m” | Included in SI | ||
| Adobe Illustrator | Adobe | ||
| JoVE_qRPA_well.ai | Included in SI | ||
| JoVE_qRPA_base.ai | Included in SI | ||
| AxioVision software | Zeiss | ||
| JoVE_AxioVision_Script.ziscript | Included in SI |
Referências
- Yoon, J. -. Y., Kim, B. Lab-on-a-Chip Pathogen Sensors for Food Safety. Sensors. 12 (8), 10713-10741 (2012).
- Quintero-Betancourt, W., Peele, E. R., Rose, J. B. Cryptosporidium parvum and Cyclospora cayetanensis: A review of laboratory methods for detection of these waterborne parasites. Journal of Microbiological Methods. 49 (3), 209-224 (2002).
- Sedlak, R. H., Jerome, K. R. Viral diagnostics in the era of digital polymerase chain reaction. Diagnostic Microbiology and Infectious Disease. 75 (1), 1-4 (2013).
- Asiello, P. J., Baeumner, A. J. Miniaturized isothermal nucleic acid amplification, a review. Lab on a Chip. 11 (8), 1420-1430 (2011).
- Piepenburg, O., Williams, C. H., Stemple, D. L., Armes, N. A. DNA detection using recombination proteins. PLoS Biology. 4 (7), 1115-1121 (2006).
- Krõlov, K., Frolova, J., et al. Sensitive and Rapid Detection of Chlamydia trachomatis by Recombinase Polymerase Amplification Directly from Urine Samples. The Journal of Molecular Diagnostics JMD. 16 (1), 127-135 (2014).
- Santiago-Felipe, S., Tortajada-Genaro, L. a., Puchades, R., Maquieira, A. Recombinase polymerase and enzyme-linked immunosorbent assay as a DNA amplification-detection strategy for food analysis. Analytica Chimica Acta. 811 (1), 81-87 (2014).
- Kersting, S., Rausch, V., Bier, F. F., von Nickisch-Rosenegk, M. Rapid detection of Plasmodium falciparum with isothermal recombinase polymerase amplification and lateral flow analysis. Malaria Journal. 13 (1), 99 (2014).
- Crannell, Z. A., et al. Nucleic Acid test to diagnose cryptosporidiosis: lab assessment in animal and patient specimens. Analytical Chemistry. 86 (5), 2565-2571 (2014).
- Rohrman, B. A., Richards-Kortum, R. R. A paper and plastic device for performing recombinase polymerase amplification of HIV DNA. Lab on a Chip. 12 (17), 3082 (2012).
- Loo, J. F. C., Lau, P. M., Ho, H. P., Kong, S. K. An aptamer-based bio-barcode assay with isothermal recombinase polymerase amplification for cytochrome-c detection and anti-cancer drug screening. Talanta. 115 (1), 159-165 (2013).
- Euler, M., et al. Development of a panel of recombinase polymerase amplification assays for detection of biothreat agents. Journal of Clinical Microbiology. 51 (4), 1110-1117 (2013).
- Abd El Wahed, A., et al. A portable reverse transcription recombinase polymerase amplification assay for rapid detection of foot-and-mouth disease virus. PloS One. 8 (8), e71642 (2013).
- Escadafal, C., et al. Rapid Molecular Assays for the Detection of Yellow Fever Virus in Low-Resource Settings. PLoS Neglected Tropical Diseases. 8 (3), e2730 (2014).
- Hutson, S. L., et al. T. gondii RP Promoters & Knockdown Reveal Molecular Pathways Associated with Proliferation and Cell-Cycle Arrest. PLoS ONE. 5 (11), 15 (2010).
- Segall, H. I., Yoo, E., Sutton, R. E. Characterization and detection of artificial replication-competent lentivirus of altered host range. Molecular Therapy. 8 (1), 118-129 (2003).
- Crannell, Z. A., Rohrman, B. A., Richards-Kortum, R. Quantification of HIV-1 DNA using Real-Time Recombinase Polymerase Amplification. Analytical Chemistry. 86 (12), (2014).
- Sollis, K. a., et al. Systematic review of the performance of HIV viral load technologies on plasma samples. PloS one. 9 (2), e85869 (2014).
- . . Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. , 1-166 (2011).

