Observation and Quantification of Mating Behavior in the Pinewood Nematode, Bursaphelenchus xylophilus
Summary
A protocol for investigating the mating behavior of the pinewood nematode, Bursaphelenchus xylophilus, is presented. Behavioral features of B. xylophilus are described in the mating process.
Abstract
A method for observing and quantifying the mating behavior of the pinewood nematode, Bursaphelenchus xylophilus, was established under a stereomicroscope. To improve the mating efficiency of B. Xylophilus and to increase the chances of mating observation, virgin adults were cultured and used for the investigation. Eggs were obtained by keeping the nematodes in water and allowing the females to lay eggs for 10 min. The second-stage juveniles (J2) were synchronized by incubating the eggs for 24 h at 25 °C in the dark, and the early J4 were obtained by culturing the J2 with grey mold, Botrytis cinerea, for another 52 h. At this time point, most J4 nematodes could be clearly distinguished as being male or female using their genital morphology. The male and female J4 were collected and cultured separately in two different Petri dishes for 24 h to get virgin adult nematodes. A virgin male and a virgin female were paired in a drop of water in the well of a concave slide. The mating behavior was filmed with a video recorder under a stereomicroscope. The whole period of the mating process was 82.8 ±3.91 min (mean ±SE) and could be divided into 4 different phases: searching, contacting, copulating, and lingering. The mean minutes of duration were 21.8 ± 2.0, 28.0 ± 1.9, 25.8 ± 0.7 and 7.2 ± 0.5, respectively. Eleven sub-behaviors were described: cruising, approaching, encountering, touching, hooping, locating, attaching, ejaculating, separating, quiescence, and roaming. Interestingly, obvious intra-sexual competition was observed when one female was grouped with 3 males or one male with 3 females. This protocol is useful and valuable, not only in investigating the mating behavior of B. xylophilus, but also in acting as a reference for ethological studies of other nematodes.
Introduction
The pinewood nematode, Bursaphelenchus xylophilus (Steiner and Buhrer) Nickle, is one of the most destructive invasive species, causing the wilting and eventual death of pine trees. This pathogenic nematode is believed to be native to the USA, but it was transported to several countries, including China, Japan, South Korea, and Portugal. Recently, it has also been reported in Spain, killing millions of pine trees at huge economic losses and threatening forest production and ecological stability1-5.
Once a host pine tree is infected by pinewood nematodes, thousands of millions of offspring are propagated rapidly within the trunk. This causes xylem dysfunction, resulting in wilting and eventually in the death of the host tree6. However, there is currently no efficient way to control this disease. Mating behavior might play an important role in the high fecundity of this nematode7. We therefore investigated the mating behavior of B. xylophilus in the laboratory, trying to find an effective way of disturbing its mating and decreasing its fecundity.
This protocol is intended to introduce the detailed methods of how to obtain the virgin adults of B. xylophilus and how to observe and analyze the mating behavior using a video recorder and a stereomicroscope. This protocol can also be used as a reference for behavioral studies of other nematodes.
Protocol
1. Acquisition of Virgin Adult Nematodes
- Pinewood nematode isolate and culture conditions
NOTE: The B. xylophilus isolate Nxy61 was originally extracted from a diseased Pinus massoniana in the Ningbo area of Zhejiang province in China.- Prepare Potato Dextrose Agar (PDA, See Table of Materials) plates for growing Botrytis cinerea.
- For inoculation, punch a piece of fungal mat with diameter of 10 mm from a B. cinerea mat and put it in the middle of a new PDA plate. Culture the B. cinerea for 5 d at 25 °C in the dark.
- Culture the pinewood nematodes in the prepared petri dish containing B. cinerea at 25 °C in the dark.
- Acquisition of B. xylophilus eggs
- Place a clamped rubber tube below a funnel and hold the prepared funnel in a rack to assemble the Baermann funnel apparatus. Place two layers of filter paper instead of gauze in the mouth of the funnel.
- Transfer the fungal cultures to the funnel setup and add water until the fungal mat is immersed. Wait 2 h, and nematodes will come out from the fungal culture, cross the filter paper, and settle down in the lowest part of the Baermann funnel.
- Gather the first couple of drops of water from the bottom of the tube into a 15 mL tube and collect nematodes by slowly releasing the clamp on the tube.
- Centrifuge the above collections at 3,000 x g for 2 min at RT.
- Remove the upper layer of water with a pipette and resuspend it with 1 mL of sterile double distilled (dd) H2O.
- With a pipette, transfer the above resuspension to a new glass Petri dish (6 cm diameter). Add 3 mL of sterile dd H2O to the Petri dish to ensure that the nematodes are able to swim freely.
- Keep the nematodes in the glass Petri dish for 10 min at 25 °C in the dark. Eggs will be laid by pregnant females. Because of their glycoprotein surface, the eggs will stick to the bottom of the glass Petri dish.
- Remove the water and worms carefully, without disturbing the eggs. Add 3 mL of sterile dd H2O to the glass Petri dish immediately to prevent the eggs from drying out.
- Repeat step 1.2.8 3x to remove all the larvae and adults, and obtain the pure eggs. Add an extra 2 mL (5 mL in total) of sterile dd H2O to the Petri dish.
- Acquisition of the fourth-stage juveniles (J4 larvae)
- Hatch the collected eggs for 24 h in the dark at 25 °C to obtain J2 larvae.
- Transfer the J2 larvae with a pipette into a 15 mL tube, centrifuge at 3,000 x g for 2 min, and remove the upper layer of water.
- Resuspend the J2 larvae with 200 µL of sterile dd H2O, and then transfer them to the PDA plate containing B. cinerea.
- Culture the J2 larvae for 52 h at 25 °C in the dark. At this time point, most J2 larvae will have developed into the early stage of J4 larvae.
- Acquisition of virgin adult nematodes
- Prepare a needle for picking up nematodes by drawing a glass capillary after heating it over an alcohol burner.
NOTE: The tip of this capillary needle usually needs to be 50 µm in diameter, roughly the body width of an adult nematode, so that it can be used as a tool for picking up a single nematode. - Assemble a Baermann funnel and extract nematodes from the fungal cultures for 2 h, as described in steps 1.2.1 – 1.2.3.
- Transfer the J4 with water into a clean Petri dish.
- Observe the nematodes under a stereomicroscope and distinguish between males and females by genital morphology, as described by Mamiya and Kiyohara8 (Figure 1). Pick up a nematode of the desired sex with the prepared capillary needle and transfer it to the appropriate fungal-culture plate for males or females.
NOTE: There are more females than males, with the percentage of females at 60 – 70%. The female vulva is obviously different from the male spicule. Compared to the adult, the spicule or vulva in early-stage J4 is smaller, but they can still be easily seen and distinguished. The nematode is always moving in the water, which sometimes increases the difficulty in distinguishing males and females. - Culture the J4 larvae for another 24 h at 25 °C in the dark.
NOTE: All the nematodes should develop into adults, but since they are unmated, they are regarded as virgin adults. - Assemble two Baermann funnels and extract the virgin adults from the fungal cultures for 2 h, as described in steps 1.2.1 – 1.2.3, and transfer the virgin adult males and females into two separate, clean Petri dishes.
- Prepare a needle for picking up nematodes by drawing a glass capillary after heating it over an alcohol burner.
2. Observation and Video Recording of Mating Behavior
- Prepare several concave slides and add 200 µL of sterile dd H2O to each of their wells.
- Pick up the prepared virgin adult male or and female with a capillary needle and put only one nematode into the water of each concave slide.
- Keep the virgin adults inside the well for 1 h to let them adapt to the new aqueous environment.
- Pair a virgin male and female by transferring one of them from one well to another using a pipette.
- Observe the mating behavior of the 1♀ + 1♂ combination in the concave slide under a stereomicroscope.
- To analyze the mating efficiency of virgin adults, calculate their mating success rate (R) and compare it with that of randomly selected adult pairs. Calculate R as the percentage of replicates in which copulation was observed compared to the total number of replicates studied.
- To investigate mating choice and intrasexual competition, group and observe virgin nematodes of two different combinations: 1♀ + 3♂ and 3♀ + 1♂.
- In order to collect data for qualitative and quantitative analysis, film the mating behavior using video microscopy. Normally, the whole mating process, from the time when the nematodes are placed in the well of the concave slide to the moment when they move away after copulation, is completed within 2 h. Therefore, record the mating process continuously for at least 2 h as one replicate. Complete at least 30 replicates in total for each combination.
NOTE: Add more sterile dd H2O (usually 50 µL) every 0.5 h to the well to compensate for evaporation during the recording process.
3. Data Collection
- View all videos carefully on a computer screen and characterize the sub-behaviors qualitatively.
- Measure the duration of mating using the frame-by-frame function of the video recorder. Manually subtract the starting time from the end point.
- For the 1♀ + 1♂ combination, analyze each video recording and measure the duration of the following events:
- Measure the duration of searching (i.e., the time from when the female and male are introduced into the well of the concave slide to the moment they encounter and contact each other).
- Measure the duration of contacting (i.e., the time from the first contact to the moment when the male is precisely positioned at the female's vulva for the first copulation).
- Measure the duration of copulating (i.e., the time from when the male is precisely positioned at the female's vulva until he slides down from the female's body after ejaculation). Note: Some nematodes can copulate several times in 2 h. In such cases, measure the duration of copulation from the first copulation to the end of the last one, including the time in between.
- Measure the duration of lingering (i.e., the period after copulation until the male and female swim away from each other and are separated by a distance of more than the nematodes' body length (usually 1 mm)).
- Count the copulation frequency and duration of every copulation and the average duration of all copulations in 2 h for each of the different combinations (i.e., 1♀ + 1♂, 3♀ + 1♂, and 1♀ + 3♂).
- Calculate the mating success rate for the first contact (P) for the different combinations as the success rate of mating after the first contact, which is conducted immediately and without any break. In the contacting phase, several contacts might be observed before actual copulation.
- When the distance between two individuals is more than the body length, consider one contact to be over. For one replicate, record P as 100% when copulation is observed immediately after the first contact, without break, or 0.00% if no copulation was observed or if there was copulation but after two or more contacts.
NOTE: There were 30 replicates for each combination described here.
- When the distance between two individuals is more than the body length, consider one contact to be over. For one replicate, record P as 100% when copulation is observed immediately after the first contact, without break, or 0.00% if no copulation was observed or if there was copulation but after two or more contacts.
Representative Results
The mating success rate (R) of virgin adults was 86.67% on average, which was significantly higher than that of the randomly selected adults: 13.33 ± 4.65% (F = 1301.71, df = 1, P = 0.0001) (Figure 2). This finding suggests that it is easier to observe the mating behavior of B. xylophilus with virgin adults than with randomly selected adults.
When one male was paired with one female, the entire mating process lasted 82.8 min on average. The whole process could be described by four different phases: searching, contacting, copulating, and lingering. The duration for these four phases were 21.8 ± 2.0 min, 28.0 ± 1.9 min, 25.8 ± 0.7 min, and 7.2 ± 0.5 min, respectively (Figure 3). In a recent publication, we described the 11 sub-behaviors as cruising, approaching, encountering, touching, hooping, locating, attaching, ejaculating, separating, quiescence, and roaming9.
When one male met two or more females simultaneously, the male seemed to mate randomly with one of them, without any selection. However, obvious mating choice was observed for female B. xylophilus. When a female contacted a male, the female tended to touch the male's head, body, tail, and genital region gently with her head and appeared to be evaluating her potential partner. If the female did not accept the male, the female left him quickly and swam to another male for the next evaluation. One of the males was then selected by the female for mating. Mating success rates after the first contact (P) of the 1♀ + 1♂, 3♀ + 1♂, and 1♀ + 3♂ combinations were 46.7 ± 9.3%, 80.0 ± 7.4%, and 23.4 ± 7.4%, respectively, and the differences between them were significant (F = 8.21, df = 2, P = 0.0124) (Figure 4).
Intrasexual competition was observed in both males and females. When one virgin female was mixed with 3 virgin males, the males tried to approach and contact the female as much as possible to get the chance of mating with her. When the vulva was occupied by one male, the other males tried to disturb the mating male by using violent body swings or dragging to separate them. Female intrasexual competition was seen to be more or less similar to male intrasexual competition.
Mating success mainly depended on the female. When a female accepted a male, the male moved more slowly and curled or even formed a hoop, and the female moved forward actively to make it easier for the male to locate her vulva and to avoid interference from other males. If a female did not accept a male, the female tried to shake him off, even if the male had mounted and positioned himself by her vulva. Because of the intrasexual competition, copulation frequencies of the 1♀ + 1♂, 3♀ + 1♂, and 1♀ + 3♂ combinations were 1.43 ± 0.091, 1.17 ± 0.074 and 1.83 ± 0.074, respectively, and the differences between them were significant (F = 14.50, df = 2, P = 0.0001) (Figure 5).
In our investigation, the copulation duration varied greatly, with a minimum of 20 s and a maximum of 42 min. According to our data, the duration of copulation could be obviously differentiated into two groups: short-term copulations, which lasted less than 5 min, and long-term copulations, which lasted longer than 10 min. Obvious differences were observed between the different groups for the average duration of short-term copulation (F = 3.81, df = 2, P = 0.0296), long-term copulation (F = 179.52, df = 2, P = 0.0001), and all copulations (F = 2.93, df = 2, P = 0.0367) (Figure 6).
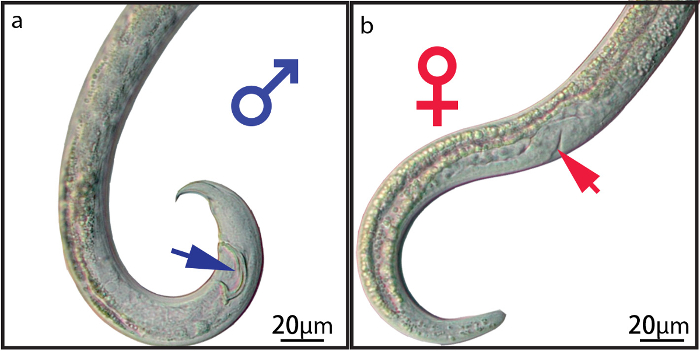
Figure 1: Genitalia of Male and Female B. Xylophilus. (a) spicule of male (blue arrow). (b) vulva of female (red arrow). Please click here to view a larger version of this figure.
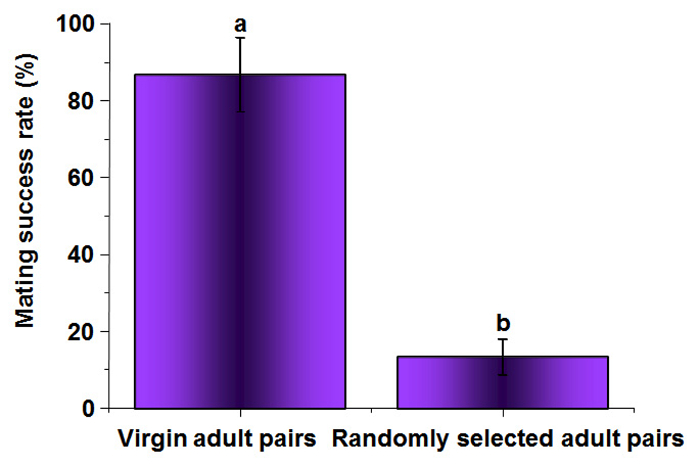
Figure 2: Mating Success Rate of Virgin Adult Couples and Randomly Selected Adult Couples of B. xylophilus. The calculated mean values are from 30 replicates. The vertical bar shows the standard error. The different lowercase letters indicate significant differences at P <0.05 when compared by Tukey's test. Please click here to view a larger version of this figure.
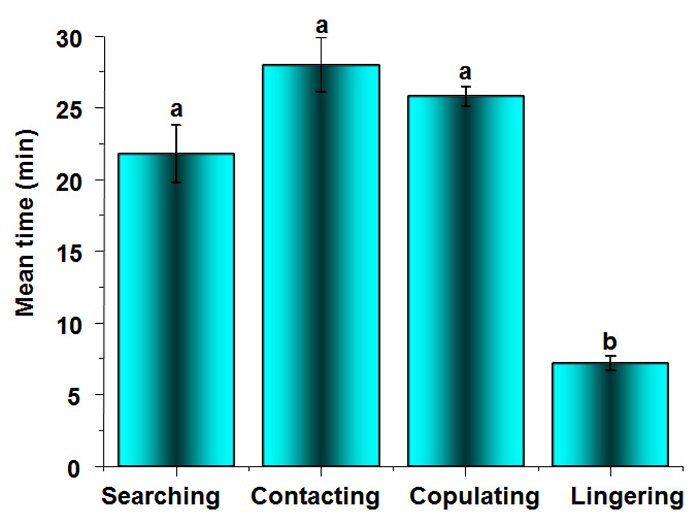
Figure 3: Duration of the 4 Phases in the Mating Process of B. xylophilus. The calculated mean values are from 30 replicates. The vertical bar shows the standard error. The different lowercase letters indicate significant differences at P <0.05 when compared by Tukey's test. The average duration of the copulating phase is 25.80 ± 0.71 min, which is 31.17% of the whole mating process. Please click here to view a larger version of this figure.
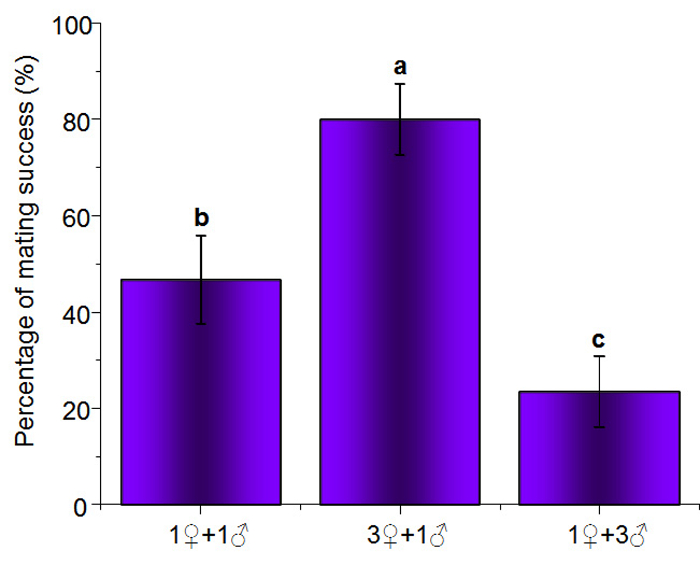
Figure 4: Mating Success for the First Contact of B. xylophilus. The calculated mean values are from 30 replicates ±SE. Means with the same lowercase letter within each column are not significantly different at P <0.05 when compared by Tukey's Honest Significant Difference test. Please click here to view a larger version of this figure.
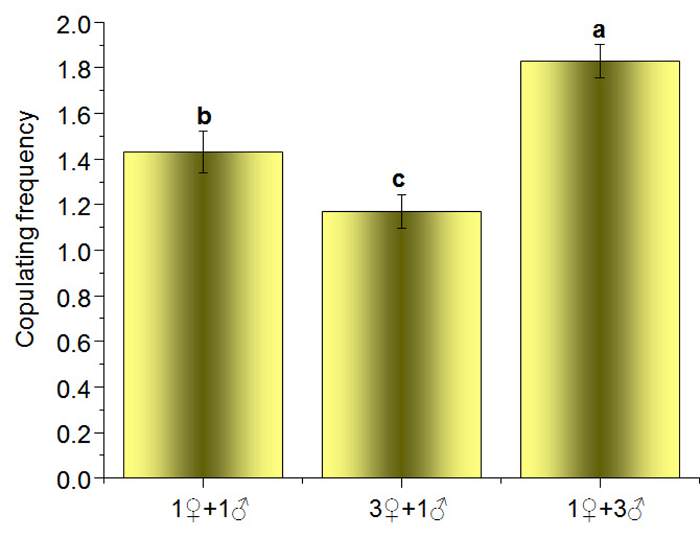
Figure 5: Copulation Frequency of B. xylophilus in Different Groups. The calculated mean values are from 30 replicates ±SE. Means with the same lowercase letter are not significantly different at P <0.05 when compared by Tukey's Honest Significant Difference test. Please click here to view a larger version of this figure.
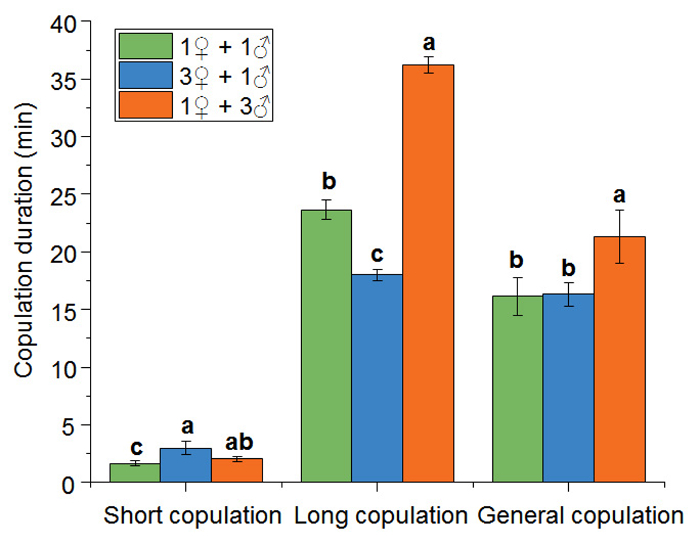
Figure 6: Copulation Duration of B. xylophilus. Means with the same lowercase letter within each set of columns are not significantly different at P <0.05 when compared by Tukey's Honest Significant Difference test. Please click here to view a larger version of this figure.
Discussion
Behavioral tests are a fundamental aspect of chemical ecology, neurobiology, molecular biology, and genetics. Nematodes, and particularly C. elegans, are extensively used for studies in these areas. The mating behavior of C. elegans has been investigated previously10-11. However, B. xylophilus is gonochoristic and different from the hermaphroditic C. elegans, and its mating process has different behavioral features9. Recently, the mating behavior of Bursaphelenchus okinawaensis, a hermaphroditic sister species of B. xylophilus, was reported12. Though different experimental methods were used, this hermaphroditic species showed similar sex attraction to B. xylophilus, and inter-species attraction between them was observed. This hermaphroditic sister species might be used as a model to conduct further investigation on the biology of Bursaphelenchus.
This protocol provides a guideline on how to observe and investigate the behavioral features of B. xylophilus during mating. Our findings showed that using virgin adult nematodes is critical for increasing the chance of successfully observing the mating process (Figure 1). Obtaining J4 larvae and precisely distinguishing their sexes are two important fundamental steps for the acquisition of virgin adults. Under our experimental conditions (i.e., at 25 °C in the dark), the period of 52 h is the proper time to obtain J4 from J2 larvae. However, distinguishing sex at the early J4 stage mostly depends on personal experience. To ensure accuracy, it is necessary for different people to double-check the sex allocation.
The behavioral features of B. xylophilus during the mating process can be described qualitatively and analyzed quantitatively by recording the durations of the four different phases, the percentage of mating success after first contact, the copulation frequency, and the copulation duration of different combinations. Interestingly, for the first time, we observed mating choice by females and strong intra-sexual competition in both males and females. No promiscuity was observed; however, this could be due to the limited number of nematodes used in our investigation. This protocol can not only be used for investigating the mating behavior of B. xylophilus, but it can also be useful and valuable as a reference for other nematodes.
Declarações
The authors have nothing to disclose.
Acknowledgements
Najie Zhu and Liqun Bai contributed equally to this work. This work was financially supported by the Special Fund for Forestry Scientific Research in the Public Welfare (201204501) and the National Natural Science Foundation of China (31170604, 31270688, and 31570638). We thank Dr. Holighaus Gerrit and Dr. Danielle Hickford for their helpful suggestions on English writing.
Materials
| B. xylophilus isolate Nxy61 | Extracted from Pinus massoniana in Ningbo area of China | ||
| Grey mold strain | Obtained from Forestry Academy of China | ||
| Baermann funnels | Sengong | ||
| falcon tubes | Sengong | ||
| centrifuge | Sengong | ||
| pipette | Sengong | ||
| sterile dd H2O | Sterilized by high pressure of steam at 121°C for 30 min. | ||
| Petri dishes | Sengong | ||
| PDA medium | Huankai Bio. | 021050 | |
| incubator | Sengong | ||
| Laminar flow | Sengong | ||
| glass capillary | Sengong | ||
| Stereoscope | Leica | LED5000 RL | |
| inverted stereo microscope | Zeiss | A1 | |
| concave slides | Sengong |
Referências
- Tokushige, Y., Kiyohara, T. Bursaphelenchus sp. in the wood of dead pine trees. J. Jap. For. Soc. 51, 193-195 (1969).
- Sun, Y. C. Bursaphelenchus xylophilus was discovered in Sun Yet-sen’s mausoleum in Nanjing. J. Jiangsu Fore. Sci. Tech. (in Chinese). 4, 47 (1982).
- Mota, M. M., et al. First report of Bursaphelenchus xylophilus in Portugal and in Europe. Nematology. 1, 727-734 (1999).
- Robertson, L., et al. Incidence of the pine wood nematode Bursaphelenchus xylophlius Steiner & Buhrer, 1934 (Nickle, 1970) in Spain. Nematology. 13, 755-757 (2011).
- Vicente, C., Espada, M., Vieira, P., Mota, M. Pine wilt disease: a threat to European forestry. Euro. J. Plant Path. 133, 89-99 (2012).
- Jones, J. T., Moens, M., Mota, M., Li, H., Kikuchi, T. Bursaphelenchus xylophilus: opportunities in comparative genomics and molecular host-parasite interactions. Mol. Plant. Path. 9, 357-368 (2008).
- Mamiya, Y., Furukawa, M. Fecundity and reproduction of Bursaphelenchus lignicolus. Jap. J. Nemat. 7, 6-9 (1977).
- Mamiya, Y., Kiyohara, T. Description of Bursaphelenchus lignicolus. n. sp. (Nematoda: Aphelenchoididae) from pine wood and histophathology of nematode-infested trees. Nematologica. 18, 120-124 (1972).
- Liu, B. J., et al. Behavioural features of Bursaphelenchus xylophilus in the mating process. Nematology. 16, 895-902 (2014).
- Simon, J. M., Sternberg, P. W. Evidence of a mate finding cue in the hermaphrodite nematode Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA. 99, 1598-1603 (2002).
- Barr, M. M., Garcia, L. R. Male mating behaviour. The C. elegans Research Community Wormbook. , (2006).
- Shinya, R., Chen, A., Sternberg, P. W. Sex attraction and mating in Bursaphelenchus okinawaensis and B. xylophilus. J. Nematol. 47 (3), 176-183 (2015).

