Non-Intubated Video-Assisted Thoracoscopic Surgery
Summary
Here, we present a non-intubated protocol for performing video-assisted thoracoscopic surgery with preserved autonomic breathing.
Abstract
Double-lumen intubation under general anesthesia is currently the most commonly performed intubation technique for pneumonectomy, wedge resection of the lung, and lobectomy. However, there is a high incidence of pulmonary complications arising from general anesthesia with tracheal intubation. Non-intubation with the preservation of voluntary breathing is an alternative to anesthesia. Non-intubation procedures minimize the adverse effects of tracheal intubation and general anesthesia, such as intubation-related airway trauma, ventilation-induced lung injury, residual neuromuscular blockade, and post-operative nausea and vomiting. However, the steps for non-intubation procedures are not detailed in many studies. Here, we present a concise non-intubated protocol for the performance of video-assisted thoracoscopic surgery with preserved autonomic breathing. This article identifies the conditions necessary to convert from non-intubated to intubated anesthesia and also discusses the advantages and limitations of non-intubated anesthesia. In this work, this intervention was performed on 58 patients. In addition, the results of a retrospective study are presented. Compared with intubated general anesthesia, patients in the non-intubated video-assisted thoracic surgery group had lower rates of post-operative pulmonary complications, shorter operative times, less intraoperative blood loss, shorter PACU stays, a lower number of days to chest drain removal, less post-operative drainage, and shorter hospital stays.
Introduction
In the past decade, non-intubated video-assisted thoracic surgery (NIVATS) anesthesia has gradually been accepted in clinical practice1,2,3. Although this novel strategy enhances the rapid recovery of patients and avoids the complications of general anesthesia (GA) and one-lung ventilation4, many surgeons regard this approach as less desirable than the traditional lung isolation technique.
Blood oxygen levels decline with age, and some patients may have decreased or borderline pulmonary function. GA may be associated with an increased risk of complications in such patients, including delayed emergence from anesthesia, airway complications, hoarseness, hypoxia, and arytenoid dislocation5,6,7,8,9. In contrast, multiple studies have documented shorter hospital stays among patients managed with NIVATS, as well as a reduction in respiratory complications compared to general anesthesia among low-risk patients10; additionally, successful surgery has even been reported in high-risk patients with very poor pulmonary function11,12,13.
Spontaneous ventilation during surgery is achieved with carefully administered local anesthesia or a regional nerve block supplemented with sedation, but the cough reflex with unexpected lung movement can be problematic during NIVATS. There is little emphasis on and no standard treatment for mediastinal flutter, irritating cough, or tachypnoea, which may disrupt a surgical procedure. In preliminary observations, the results showed that sevoflurane could decrease the respiratory rate and the occurrence of mediastinal flutter during NIVATS while maintaining spontaneous breathing14. Therefore, it could be hypothesized that sevoflurane inhalation may prevent coughing and reduce the requirement for mechanical ventilation, thereby reducing post-operative pulmonary complications (PPCs).
Firstly, this report presents a step-by-step protocol detailing the execution of non-intubated video-assisted thoracoscopic surgery. Secondly, a retrospective study was conducted to investigate the potential benefits of non-intubated anesthesia on post-operative outcomes.
Protocol
The study was approved by the Ethics Committee of the Affiliated Hospital of Ningbo University School of Medicine (KY20181215) on December 10, 2018.
1. Inclusion criteria
- Include all patients (aged >18 years) undergoing pulmonary bullae resection, pulmonary wedge resection, or lobectomy.
2. Exclusion criteria
- Exclude patients based on an American Society of Anesthesiologists (ASA) physical status classification of >315.
- Exclude patients with a body mass index (BMI)16 >30 kg/m2.
- Exclude patients with oesophageal cancer, total pneumonectomy, and open-heart surgery with resection of the ribs.
- Exclude patients with a previous medical history of bronchiectasis, destroyed lung, or chronic obstructive pulmonary disease (COPD).
- Exclude patients with definite multiple pulmonary infections or inflammation prior to surgery, as well as other severe systemic diseases.
- Exclude patients with coagulation disorders and severe psychiatric or neurological disorders.
3. Preparation before the anesthesia
- Fast the patient for 8 h before the surgery.
- Adjust the operating room temperature to be between 24 ° C and 26 ° C.
- Insert a 20 G top-winged infusion needle into the non-surgical dorsal vein of the hand.
- Monitor the electrocardiography, blood pressure, pulse oxygen saturation (SpO2), and respiratory rate of the patient.
- Apply a bispectral index (BIS) quatro sensor to the forehead of each patient.
NOTE: Continuously monitor the radial arterial pressure and central venous pressure, if necessary.
4. Ultrasound-guided thoracic paravertebral blockade
- Place the patient in a lateral decubitus position.
- Place the ultrasound probe directly above the spinous processes of the third thoracic and seventh thoracic vertebrae, and obtain a cross-sectional image of the spinous processes.
NOTE: In the middle of the image is the hyperechoic spinous process with a posterior acoustic shadow, and the bony structures with a posterior acoustic shadow on either side of the spinous process are, in order, the vertebral plate and the transverse process. - Move the ultrasound probe laterally to show the transverse process in its entirety.
- Move the ultrasound probe outward to visualize the transverse process, transverse rib joints, and ribs.
- Move the ultrasound probe caudally until the transverse process, the pleura, and the thoracic paravertebral space between them are detected in the image.
- Locally anesthetize the skin by injecting 2 mL of 1% lidocaine.
- Insert the block needle from lateral to medial with the in-plane approach under ultrasound guidance.
- Carefully increase the aspiration before the injection. Ensure that no blood reflux is observed.
NOTE: The purpose of this step is to prevent systemic toxicity from the local anesthetic. - Inject 2 mL of saline, and then visualize the anterior displacement of the pleura and the widening of the thoracic paravertebral space with the ultrasound.
- Inject 15 mL of 0.375% ropivacaine at the levels of T3 and T7.
5. Anesthetic induction
- Spray 1% levobupivacaine (3 mL) onto the throat.
- Inject 1.5 µg/kg of fentanyl and 1-1.5 mg/kg of propofol.
- For continuous oxygen administration, place a facial mask, or insert a laryngeal mask airway (double-tube mask; #3 for 30-50 kg, #4 for 50-70 kg, #5 for 70-100 kg). Oxygen enters the patient's airway via the facial mask or laryngeal mask.
NOTE: Adjust the injection dose to achieve a BIS value between 40-6016.
6. Maintenance of anesthesia
- Maintain an oxygen flow rate of 0.5-1 L/min.
- Maintain the concentration of sevoflurane at 1.5%-2.0% after the artificial pneumothorax.
NOTE: Inject 0.5 µg/kg of fentanyl if the intraoperative spontaneous respiratory rate is higher than 20 breaths/min or when there is mediastinal flutter or coughing. Sevoflurane enters the patient's airway via the facial mask or laryngeal mask. - Monitor the inhaled oxygen concentration and the end-tidal carbon dioxide (ETCO2 < 60 mmHg).
- Monitor the body temperature. Monitor the level of consciousness, and maintain a BIS value of 40-6016,17.
- Inject 20µg/kg of atropine if the patient develops sinus bradycardia (HR ≤ 50 beats/min).
- Continuously perfuse 2 µg/kg/h of norepinephrine using an infusion pump if the systolic blood pressure is lower than 30% or 90 mmHg.
- Perform arterial blood gas analysis 15 min before the end of the surgery.
- Inject 1 mg/kg of flurbiprofen 30 min before the end of the surgery.
7. Thoracoscopic vagal block techniques and pleural infiltration anesthesia
- Use a 24 G top-winged infusion needle to produce an infiltration of 0.375% ropivacaine (3 mL) near the vagus nerve at the level of the lower trachea when performing right-sided procedures.
- Use a 24 G top-winged infusion needle to produce an infiltration of 0.5% ropivacaine (3 mL) near the vagus nerve at the level of the aortopulmonary window when performing left-sided procedures.
- Spray 10 mL of 2% lidocaine onto the surface of the visceral pleura using a 10 mL syringe.
8. Conversion from non-intubated anesthesia to intubated general anesthesia
- Convert the non-intubated anesthesia to intubated general anesthesia if the patient meets one of the following conditions:
-Severe hypoxemia (pulse oximetry < 80%)
-Severe hypercapnia (PaCO2 > 80 mmHg)
-Hemodynamic instability: intractable arrhythmias and right ventricular failure
-Persistent coughing resulting in surgery becoming difficult or impossible
-Intraoperative bleeding requiring thoracotomy
9. Post-operative care
- After fully waking, ask the patient to take a deep breath and cough to re-expand the collapsed lung.
- Connect a patient-controlled intravenous analgesia (PCA) pump to the intravenous catheter with a 100 mL solution in a PCA reservoir bag (containing 1 µg/kg of sufentanil and a 0.9% sodium chloride injection), and titrate 2 mL/h of the PCA solution.
- Remove the chest drain when there is no air leak on coughing, no obvious fluid pneumothorax on the review of the chest X-ray, and a 24 h drainage of <300 mL.
Representative Results
Clinical data were retrospectively collected on 58 consecutive patients undergoing non-intubated video-assisted thoracoscopic surgery from January 2016 to December 2022. The patients were given a preoperative visit by the anesthesiologist and provided with a detailed explanation of the contents of the anesthesia informed consent form prior to the anesthesia. The patients were allowed to choose one of the two groups (the NIVATS group or GA group) of anesthesia, and they signed an informed consent form.
The patients in the GA group were induced with 0.04 mg/kg of midazolam, 2.5 mg/kg of propofol, 0.3 mg/kg of etomidate, 0.5 µg/kg of sufentanil, and 1.2 mg/kg of rocuronium. After the inotropic drugs had taken full effect, a visual laryngoscope was used to guide the transoral insertion of a double-lumen bronchial tube or a single-lumen tracheal tube with a bronchial occluder. Preoperative radiography of the chest was performed, and the tracheal internal diameter was measured at the level of the sternoclavicular joint. A 41 Fr was selected if the tracheal internal diameter was ≥19 mm, a 39 Fr was selected for a tracheal inner diameter of ≥17 mm, a 37 Fr was selected for a tracheal inner diameter of ≥15 mm, a 35 Fr was selected for a tracheal inner diameter of ≥13 mm, and a 32 Fr was selected for a tracheal inner diameter of ≥11 mm. After the insertion of the double-lumen tube, the position of the tube was assessed and adjusted with a fibreoptic bronchoscope in the horizontal and lateral positions. Intraoperative anesthesia was maintained with a continuous infusion of 2-6 mg/kg/h of propofol and 0.25 µg/kg/min of remifentanil, all of which were delivered as intravenous anesthesia. The surgeons and patients undergoing non-intubated video-assisted thoracoscopic surgery are seen in Figure 1, Figure 2, and Figure 3.
Two research assistants (G.B. and L.W.J.) collected perioperative information from the patients, including on their age, BMI, anesthesia, duration of surgery, intraoperative bleeding, length of stay in the hospital, length of stay in the ICU, medical history, post-operative chest radiograph results, and their temperature and other laboratory tests performed on each post-operative day until discharge.
Measures of post-operative pulmonary complications (PPCs)
Referring to the guidelines published by the European Joint taskforce for perioperative clinical outcome (EPCO) definitions and taking into account the characteristics of this study18, the PPCs diagnostic tool was as follows: (i) pneumothorax: air inside the pleural space; (ii) pleural effusion: a chest radiograph with blunting of the costophrenic angle and loss of the sharp silhouette of the same side of the upright hemidiaphragm; (iii) pneumonia: use of new antibiotics, radiographic changes, fever, or a white cell count >12,000 µL−1; (iv) atelectasis: pulmonary opacification with mediastinum, hilum, or hemidiaphragm shifting toward the affected area; (v) pulmonary embolism: not defined; and (vi) acute respiratory distress syndrome (ARDS): PaO2:FIO2 ≤ 300 in the ventilated state and bilateral infiltrates on the chest radiograph.
Statistical analysis
For data analysis, 95% confidence intervals were used. A value of P < 0.05 was considered statistically significant. The lost data was adjusted for by using two-way interpolation. Continuous variables were represented as the average (standard deviation [SD]) or medium number (quartid digits range), and an independent-samples t-test or Mann-Whitney U-test was used for comparison. Categorical variables were presented as numbers and were compared by a Pearson's chi-squared test, a Fisher's exact test, or a continuous-corrected chi-square test. The above data analysis was conducted and completed by two data analysts independently. All the statistical analyses were performed with the SPSS26.0 software.
Resultados
Overall, 58 patients were eligible for analysis, including 31 patients in the GA group and 27 patients in the NIVATS group. The clinical characteristics of the two groups are shown in Table 1. There were no significant differences between the two groups in terms of gender, age, BMI, and ASA scores (P > 0.05).
Primary outcomes
The pulmonary complication rate was significantly lower in the NIVATS group (3.7%; one patient) compared with the GA group (25.8%; eight patients) (P = 0.051). Firstly, six patients in the GA group developed post-operative pneumothorax; however, no patients in the NIVATS group developed pneumothorax. The difference between the two groups was statistically significant (P = 0.026). Secondly, three patients in the GA group developed pleural effusion compared to one patient in the NIVATS group, although the difference between the two groups was not statistically significant (P = 0.707). Moreover, seven patients in the GA group developed pneumonia compared to none in the NIVATS group, and the difference between the two groups was statistically significant (P = 0.012).
In addition, three patients in the GA group developed pulmonary atelectasis compared to none in the NIVATS group. In the GA group, two patients developed pulmonary embolisms compared to none in the NIVATS group. No significant differences were found between the two groups in terms of pulmonary atelectasis or pulmonary embolism (P = 0.240 and P = 0.494, respectively). In the two groups, no patients developed ARDS.
Secondary outcomes
The use of non-intubated thoracoscopic anesthesia significantly reduced the intraoperative blood loss (100 mL [50-200] vs. 20 mL [5-50]; P < 0.001). Additionally, the NIVATS group had a shorter mean surgery duration (P = 0.024) and PACU stay time (P = 0.004). Furthermore, the difference in the day when the chest drain was removed between the NIVATS group and the GA group was significant (day 3 [2-4] vs. day 2 [1-3], respectively; P < 0.001). Moreover, the difference in the amount of post-operative chest drainage between the NIVATS group (260 mL [100-380]) and the GA group (672 mL [452.5-1,197.5]) was significant (P = 0.001). In the GA group, there were three patients who had a prolonged stay (>48 h) in the ICU, compared to no patients in the NIVATS group (P = 0.240). In the GA group, four patients had post-operative chest tightness and shortness of breath compared to one patient in the NIVATS group (P = 0.483). Finally, compared with the GA group, the NIVATS group had significantly shorter hospital stay lengths (6 days [5-7] vs. 5 days [4-6]; P < 0.001).
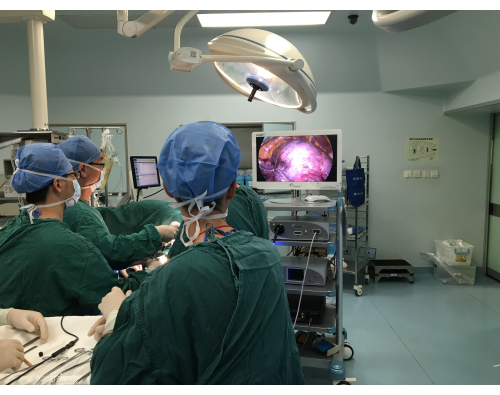
Figure 1: The surgeon performing non-intubated video-assisted thoracoscopic surgery. Please click here to view a larger version of this figure.
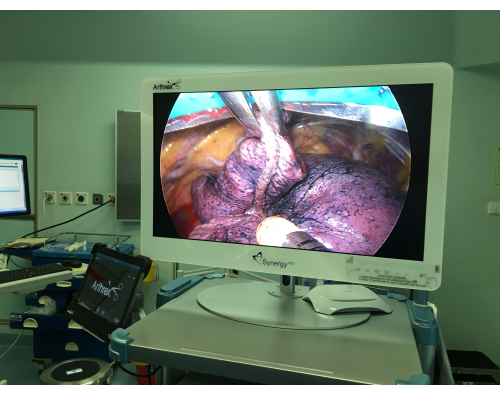
Figure 2: The affected lung under non-intubated video-assisted thoracoscopic surgery. Please click here to view a larger version of this figure.
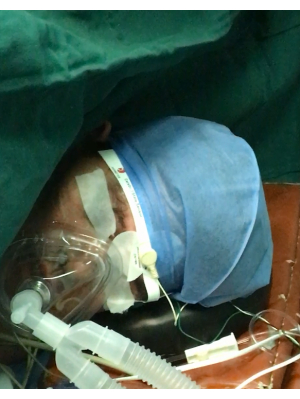
Figure 3: A patient with a facial mask undergoing non-intubated video-assisted thoracoscopic surgery. Please click here to view a larger version of this figure.
Table 1: Patient demographics and post-operative results in each group. Please click here to download this Table.
Discussion
The advantages of this protocol are as follows: (i) providing a sevoflurane inhalation anesthesia regimen to reduce the cough reflex during thoracoscopic surgery; (ii) minimizing over-sedation while providing a safe and pain-free operating environment for patients undergoing thoracic surgery; (iii) minimizing the patient's spontaneous breathing and mediastinal oscillations during the procedure, taking into account the associated technical challenges. This was achieved by providing pre-emptive intraoperative local anesthesia.
In 2004, Pompeo et al. reported, for the first time, the completion of thoracoscopic pulmonary nodule resection without tracheal intubation and preserving spontaneous breathing anesthesia19. Non-intubated anesthesia with preserved voluntary breathing has also become a hot topic of research in the field of thoracic anesthesia in recent years. Compared to conventional tracheal intubation, this technique avoids the potential for airway damage caused by intubation and better preserves the endotracheal and bronchial mucosal epithelial ciliary clearance. It also reduces the amount of intraoperative anesthesia, avoids the residual effects of muscle relaxants20,21, and reduces the risk of post-operative hypoxia22. In addition, laryngeal masks have a low risk of pharyngeal and laryngeal injury, lower invasiveness, and easier insertion compared to intubation23,24,25. On the other hand, the use of a laryngeal mask better ensures respiratory stabilization compared to face masks26. The laryngeal mask relieves the obstruction of the upper airway and prevents hyperventilation, thus providing better maintenance of respiratory stability26,27. However, laryngeal masks reduce the tension of the lower esophageal sphincter by 15%, thus increasing the risk of gastric reflux28. There is evidence that NIVATS has less impact on post-operative defense mechanisms, such as inflammatory cytokine or lymphocyte responses29,30, and blood stress hormone levels31, which may contribute to the reduced incidence of post-operative respiratory complications with this method.
Cough reflexes and unexpected lung movements are inevitable during NIVATS lung manipulation32. Coughs are caused by the activation of the mechanically and chemically sensitive vagal afferent nerves that innervate the airways. The current and available cough suppression techniques include vagal nerve blocks and intravenous or nebulized local anesthetics16,33. However, the latter two are not as effective as direct vagal nerve blocks. Moreover, nerve blocks carry the risk of systemic toxicity of local anesthesia, nerve damage, or aspiration34,35,36. In recent years, sevoflurane anesthesia has come to our attention. Sevoflurane anesthesia inhibits the pulmonary irritant receptors and attenuates the cough reflex37. The inhalation of high-concentration sevoflurane weakens the bronchoconstriction reflex caused by mechanical stimulation of the airway and inhibits multiple ion channels of the bronchial smooth muscle38,39.
The results of this observation study suggested that NIVATS shortened the duration of chest drainage, reduced blood loss, and reduced PPCs. Hung et al. found that patients in the non-intubated group could have their chest drain removed earlier40. Mechanical ventilation causes pressure-related damage to the lungs, causes lung hyperextension, and promotes the release of various pro-inflammatory mediators41. A meta-analysis42 concluded that reduced levels of inflammatory cytokines, less impairment of lymphocyte activity, and a diminished stress response were the reasons for fewer post-operative complications, such as pneumothorax, in relation to non-intubated thoracoscopic surgery29,31,40. Additionally, non-intubated surgeries have only a slight effect on the patient's normal respiratory physiology, and the patient recovers more quickly19.
Hung et al. recruited 238 patients with lung cancer to undergo non-intubated thoracoscopic lobectomy, and the study found less blood loss in the non-intubated group40. There are probably two reasons for the lower intraoperative blood loss in the non-intubated group: (i) there may be better analgesia in the non-intubated group43, resulting in stable intraoperative blood pressure control and, thus, less intraoperative bleeding; (ii) according to our observations, the non-intubated group had a better state of lung collapse, and the surgeon was able to identify key anatomical areas more quickly and complete the operation quickly, so there may be relatively less blood loss with this technique.
In addition, our study also found that non-intubated anesthesia shortened the operation time, reduced hospital stays, reduced patient time in the PACU, and reduced the probability of patient transfer to the ICU postoperatively compared to tracheal intubation anesthesia. Similar to Wu et al.44, the non-intubated group had a shorter anesthesia induction time. A meta-analysis also found that the duration of the procedure was shorter in non-intubated groups than in intubated groups42. This shorter duration of non-intubated compared to intubated procedures worldwide could be explained by the fact that non-intubated procedures under local anesthesia do not require tracheal intubation and subsequent bronchoscopy42.
Studies have also shown that hospital stays are significantly reduced with non-intubated compared to intubated procedures30,45. It is well known that general anesthesia requires the use of drugs such as muscle relaxants and intravenous analgesics. They are associated with important post-operative complications, which reduce patient comfort, increase the need for post-operative analgesia, and prolong the post-operative stay14,46. Bevilacqua Filho et al. found that post-operative pulmonary complications were associated with an increased prevalence of prolonged hospital stays47. In our study, patients in the non-intubated group had fewer PPCs than those in the GA group, which we believe is one of the reasons for the lower hospital stay length in the non-intubated group.
The relatively good results observed for the non-intubated group may be biased in part due to the retrospective evaluations of these non-intubated patients. Indeed, this is a retrospective study from a single center, so we acknowledge some limitations. Due to the retrospective nature of this study, there was no randomization and no elimination of selection bias. To address this issue, we aim to design a randomized comparative study in the future to elucidate the safety and benefits of this non-intubated video-assisted thoracoscopic surgery. The small sample size is also a major limitation of this study. As surgeons and anesthetists become proficient in NIVATS, the technique may become more commonly used for surgery in patients who do not want general anesthesia and tracheal intubation.
The protocol of NIVATS is based on multimodal analgesia, including ultrasound-guided thoracic paravertebral blockade, a thoracoscopic vagal block, and pleural infiltration anesthesia. Multimodal analgesia effectively relieves intraoperative pain and inhibits the cough reflex. In addition, it is critical that level of sedation is set to reach a bispectral index value of 40 to 60. Finally, it is important to always pay attention to the alignment of the patient's laryngeal mask airway when swinging the position.
The technique has some limitations. The procedure is not suitable for specific patient groups, such as patients with severely impaired lung function and patients with extensive pleural adhesions, persistent hypoxia, or bleeding requiring conversion to an open chest. There is a lack of clarity regarding the specific inclusion criteria and contraindications for the technique. Further protocols are needed to set appropriate criteria for conversion from non-intubated local anesthesia to intubated general anesthesia42.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by the third batch of the Ningbo Health Youth Technical Cadre program (Dr. Binbin Zhu), and the Zhejiang Medical Association Clinical Research Fund Project (Dr. Bin Gao) (2018ZYC-A66).
Materials
| 20-G top-winged infusion needle | BD Intima II | 383012 | Puncture with a 20-G top-winged infusion needle into the dorsal vein of the non-operative side of the hand. |
| 24-G top-winged infusion needle | BD Intima II | 383033 | Thoracoscopic vagal block techniques |
| Anesthesia machine | Drager | A300 | Maintenance of respiratory function; Inhalation anesthesia; Monitor for electrocardiography, blood pressure, pulse oxygen saturation (SpO2), end-tidal carbon dioxide and respiratory rate |
| Atropine | Jiuquan Dadeli Pharma | H62020772 | Control of heart rate |
| BIS | COVIDIEN | B277243 | Monitor the level of consciousness |
| Disposable nerve block needle | Tuoren Medical Device | 202303007 | Nerve block |
| Facial mask | Emedica | EM01-105S | Provides an effective non-invasive breathing circuit |
| Fentanyl. | Renfu Pharma | 21D04021 | Analgesia |
| Flurbiprofen | Daan Pharma | H20183054 | Analgesia |
| Laryngeal mask | Ambu Aura-i | 2012-2664652 | Airway management to preserve voluntary breathing |
| Levobupivacaine | Rundu Pharma | H20050403 | Local Anaesthesia |
| Lidocaine | Kelun Pharma | F221129C | Local skin infiltration |
| Norepinephrine | Lijun Pharma | H61021666 | Control of blood pressure |
| Portable color doppler ultrasound | SonoSite | M-Turbo | Guided nerve block |
| Propofol | Guorui Pharma | H20030114 | Sedation and hypnosis |
| Ropivacaine | Aspen Pharma | 6091403219940 | Paravertebral nerve block |
| Saline | Kelun Pharma | c221201E1 | Assisted subsonic localisation |
| Sevoflurane | Shanghai Hengrui Pharmaceutical Co.,Ltd | 9081931 | Anesthesia induction and maintenance |
| Sufentanil | Jiangsu Enhua Pharmaceutical Co., Ltd | H20203650 | Postoperative analgesia |
Referências
- Sedrakyan, A., vander Meulen, J., Lewsey, J., Treasure, T. Video assisted thoracic surgery for treatment of pneumothorax and lung resections: Systematic review of randomised clinical trials. British Medical Journal. 329 (7473), 1008 (2004).
- Luh, S. P., Liu, H. P. Video-assisted thoracic surgery–The past, present status and the future. Journal of Zhejiang University Science B. 7 (2), 118-128 (2006).
- Hung, M. H., Hsu, H. H., Cheng, Y. J., Chen, J. S. Nonintubated thoracoscopic surgery: State of the art and future directions. Journal of Thoracic Disease. 6 (1), 2-9 (2014).
- Kelkar, K. V. Post-operative pulmonary complications after non-cardiothoracic surgery. Indian Journal of Anaesthesia. 59 (9), 599-605 (2015).
- Knoll, H., et al. Airway injuries after one-lung ventilation: A comparison between double-lumen tube and endobronchial blocker: a randomized, prospective, controlled trial. Anesthesiology. 105 (3), 471-477 (2006).
- Zhong, T., Wang, W., Chen, J., Ran, L., Story, D. A. Sore throat or hoarse voice with bronchial blockers or double-lumen tubes for lung isolation: a randomised, prospective trial. Anaesthesia and Intensive. 37 (3), 441-446 (2009).
- Mikuni, I., et al. Arytenoid cartilage dislocation caused by a double-lumen endobronchial tube. British Journal of Anaesthesia. 96 (1), 136-138 (2006).
- Kurihara, N., et al. Hoarseness caused by arytenoid dislocation after surgery for lung cancer. General Thoracic and Cardiovascular Surgery. 62 (12), 730-733 (2014).
- Ceylan, K. C., et al. Intraoperative management of tracheobronchial rupture after double-lumen tube intubation. Surgery Today. 43 (7), 757-762 (2013).
- Yu, M. G., et al. Non-intubated anesthesia in patients undergoing video-assisted thoracoscopic surgery: A systematic review and meta-analysis. PloS One. 14 (11), (2019).
- Ambrogi, V., Sellitri, F., Perroni, G., Schillaci, O., Mineo, T. C. Uniportal video-assisted thoracic surgery colorectal lung metastasectomy in non-intubated anesthesia. Journal of Thoracic Disease. 9 (2), 254-261 (2017).
- Guo, Z., et al. Video-assisted thoracoscopic surgery segmentectomy by non-intubated or intubated anesthesia: A comparative analysis of short-term outcome. Journal of Thoracic Disease. 8 (3), 359-368 (2016).
- Liu, J., et al. The impact of non-intubated versus intubated anaesthesia on early outcomes of video-assisted thoracoscopic anatomical resection in non-small-cell lung cancer: A propensity score matching analysis. European Journal of Cardio-Thoracic Surgery. 50 (5), 920-925 (2016).
- Hausman, M. S., Jewell, E. S., Engoren, M. Regional versus general anesthesia in surgical patients with chronic obstructive pulmonary disease: Does avoiding general anesthesia reduce the risk of postoperative complications. Anesthesia and Analgesia. 120 (6), 1405-1412 (2015).
- Grott, M., et al. Thoracic surgery in the non-intubated spontaneously breathing patient. Respiratory Research. 23 (1), (2022).
- Hung, M. H., et al. Non-intubated thoracoscopic surgery using internal intercostal nerve block, vagal block and targeted sedation. European Journal of Cardio-Thoracic Surgery. 46 (4), 620-625 (2014).
- Guo, Z., et al. Analysis of feasibility and safety of complete video-assisted thoracoscopic resection of anatomic pulmonary segments under non-intubated anesthesia. Journal of Thoracic Disease. 6 (1), 37-44 (2014).
- Jammer, I., et al. Standards for definitions and use of outcome measures for clinical effectiveness research in perioperative medicine: European Perioperative Clinical Outcome (EPCO) definitions: a statement from the ESA-ESICM joint taskforce on perioperative outcome measures. European Journal of Anaesthesiology. 32 (2), 88-105 (2015).
- Pompeo, E., Mineo, D., Rogliani, P., Sabato, A. F., Mineo, T. C. Feasibility and results of awake thoracoscopic resection of solitary pulmonary nodules. The Annals of Thoracic Surgery. 78 (5), 1761-1768 (2004).
- Ali, J. M., Volpi, S., Kaul, P., Aresu, G. Does the ‘non-intubated’ anaesthetic technique offer any advantage for patients undergoing pulmonary lobectomy. Interactive Cardiovascular and Thoracic Surgery. 28 (4), 555-558 (2019).
- Okuda, K., Nakanishi, R. The non-intubated anesthesia for airway surgery. Journal of Thoracic Disease. 8 (11), 3414-3419 (2016).
- Prince, J., Goertzen, C., Zanjir, M., Wong, M., Azarpazhooh, A. Airway complications in intubated versus laryngeal mask airway-managed dentistry: A meta-analysis. Anesthesia Progress. 68 (4), 193-205 (2021).
- Amer, G. F., Abdeldayem, O. T., Lahloub, F. M. F. Effect of local anesthesia and general anesthesia using I-gel laryngeal mask airway in diabetic patients undergoing cataract surgery: Comparative study. Anesthesia, Essays and Researches. 13 (2), 209-213 (2019).
- Sorbello, M., Afshari, A., De Hert, S. Device or target? A paradigm shift in airway management: Implications for guidelines, clinical practice and teaching. European Journal of Anaesthesiology. 35 (11), 811-814 (2018).
- Yamaguchi, T., et al. Feasibility of total intravenous anesthesia by cardiologists with the support of anesthesiologists during catheter ablation of atrial fibrillation. Journal of Cardiology. 72 (1), 19-25 (2018).
- Koyama, T., et al. Laryngeal mask versus facemask in the respiratory management during catheter ablation. BMC Anesthesiology. 20 (1), (2020).
- Qamarul Hoda, ., Samad, M., Ullah, K., H, ProSeal versus Classic laryngeal mask airway (LMA) for positive pressure ventilation in adults undergoing elective surgery. The Cochrane Database of Systematic Reviews. 7 (7), (2017).
- van Esch, B. F., Stegeman, I., Smit, A. L. Comparison of laryngeal mask airway vs tracheal intubation: A systematic review on airway complications. Journal of Clinical Anesthesia. 36, 142-150 (2017).
- Vanni, G., et al. Impact of awake videothoracoscopic surgery on postoperative lymphocyte responses. The Annals of Thoracic Surgery. 90 (3), 973-978 (2010).
- Liu, J., et al. Nonintubated video-assisted thoracoscopic surgery under epidural anesthesia compared with conventional anesthetic option: a randomized control study. Surgical Innovation. 22 (2), 123-130 (2015).
- Tacconi, F., Pompeo, E., Sellitri, F., Mineo, T. C. Surgical stress hormones response is reduced after awake videothoracoscopy. Interactive Cardiovascular and Thoracic Surgery. 10 (5), 666-671 (2010).
- Lai, H. C., et al. Sevoflurane is an effective adjuvant to propofol-based total intravenous anesthesia for attenuating cough reflex in nonintubated video-assisted thoracoscopic surgery. Medicina. 97 (42), (2018).
- Navarro-Martínez, J., et al. Intraoperative crisis resource management during a non-intubated video-assisted thoracoscopic surgery. Annals of Translational Medicine. 3 (8), 111 (2015).
- Melnyk, V., Ibinson, J. W., Kentor, M. L., Orebaugh, S. L. Updated retrospective single-center comparative analysis of peripheral nerve block complications using landmark peripheral nerve stimulation versus ultrasound guidance as a primary means of nerve localization. Journal of Ultrasound in Medicine. 37 (11), 2477-2488 (2018).
- Reynolds, R. P., Effer, G. W., Bendeck, M. P. The upper esophageal sphincter in the cat: The role of central innervation assessed by transient vagal blockade. Canadian Journal of Physiology and Pharmacology. 65 (1), 96-99 (1987).
- Neville, A. L., et al. Esophageal dysfunction in cervical spinal cord injury: a potentially important mechanism of aspiration. The Journal of Trauma. 59 (4), 905-911 (2005).
- Nishino, T., Kochi, T., Ishii, M. Differences in respiratory reflex responses from the larynx, trachea, and bronchi in anesthetized female subjects. Anesthesiology. 84 (1), 70-74 (1996).
- Regli, A., von Ungern-Sternberg, B. S. Anesthesia and ventilation strategies in children with asthma: part I – preoperative assessment. Current Opinion in Anaesthesiology. 27 (3), 288-294 (2014).
- Regli, A., von Ungern-Sternberg, B. S. Anesthesia and ventilation strategies in children with asthma: part II – intraoperative management. Current Opinion in Anaesthesiology. 27 (3), 295-302 (2014).
- Hung, M. H., et al. Nonintubated thoracoscopic lobectomy for lung cancer using epidural anesthesia and intercostal blockade: A retrospective cohort study of 238 cases. Medicina. 94 (13), 727 (2015).
- Solli, P., Brandolini, J., Bertolaccini, L. Tubeless thoracic surgery: Ready for prime time. Journal of Thoracic Disease. 11 (3), 652-656 (2019).
- Deng, H. Y., et al. Non-intubated video-assisted thoracoscopic surgery under loco-regional anaesthesia for thoracic surgery: A meta-analysis. Interactive Cardiovascular and Thoracic Surgery. 23 (1), 31-40 (2016).
- Liu, C. Y., et al. Tubeless single-port thoracoscopic sublobar resection: Indication and safety. Journal of Thoracic Disease. 10 (6), 3729-3737 (2018).
- Wu, C. Y., et al. Feasibility and safety of nonintubated thoracoscopic lobectomy for geriatric lung cancer patients. The Annals of Thoracic Surgery. 95 (2), 405-411 (2013).
- Pompeo, E., et al. Randomized comparison of awake nonresectional versus nonawake resectional lung volume reduction surgery. The Journal of Thoracic and Cardiovascular Surgery. 143 (1), 47-54 (2012).
- Murphy, G. S., et al. Postoperative residual neuromuscular blockade is associated with impaired clinical recovery. Anesthesia and Analgesia. 117 (1), 133-141 (2013).
- Bevilacqua Filho, ., T, C., et al. Risk factors for postoperative pulmonary complications and prolonged hospital stay in pulmonary resection patients: A retrospective study. Brazilian Journal of Anesthesiology. 71 (4), 333-338 (2021).

