Programming Stem Cells for Therapeutic Angiogenesis Using Biodegradable Polymeric Nanoparticles
Summary
We describe the method of programming stem cells to overexpress therapeutic factors for angiogenesis using biodegradable polymeric nanoparticles. Processes described include polymer synthesis, transfecting adipose-derived stem cells in vitro, and validating the efficacy of programmed stem cells to promote angiogenesis in a murine hindlimb ischemia model.
Abstract
Controlled vascular growth is critical for successful tissue regeneration and wound healing, as well as for treating ischemic diseases such as stroke, heart attack or peripheral arterial diseases. Direct delivery of angiogenic growth factors has the potential to stimulate new blood vessel growth, but is often associated with limitations such as lack of targeting and short half-life in vivo. Gene therapy offers an alternative approach by delivering genes encoding angiogenic factors, but often requires using virus, and is limited by safety concerns. Here we describe a recently developed strategy for stimulating vascular growth by programming stem cells to overexpress angiogenic factors in situ using biodegradable polymeric nanoparticles. Specifically our strategy utilized stem cells as delivery vehicles by taking advantage of their ability to migrate toward ischemic tissues in vivo. Using the optimized polymeric vectors, adipose-derived stem cells were modified to overexpress an angiogenic gene encoding vascular endothelial growth factor (VEGF). We described the processes for polymer synthesis, nanoparticle formation, transfecting stem cells in vitro, as well as methods for validating the efficacy of VEGF-expressing stem cells for promoting angiogenesis in a murine hindlimb ischemia model.
Introduction
The overall goal of this technique is to promote therapeutic angiogenesis using non-virally programmed stem cells overexpressing therapeutic factors at the site of ischemia. Stem cells were modified ex vivo first using biodegradable nanoparticles synthesized in the lab, and then transplanted in a murine model of hindlimb ischemia to validate their potential for enhancing angiogenesis and tissue salvage.
Controlled vascular growth is an important component of successful tissue regeneration, as well as for treating various ischemic diseases such as stroke, limb ischemia, and myocardial infarction. Several strategies have been developed to promote vascular growth, including growth factor delivery and cell-based therapy.1 Despite the efficacy observed in the animal disease models, these methods still face limitations such as the need for supraphysiological doses for growth factor delivery, or insufficient paracrine release by cells alone. One potential strategy to overcome the above limitations is to combine stem cell therapy and gene therapy, whereby stem cells are genetically programmed ex vivo prior to transplantation to overexpress desirable therapeutic factors. This approach has been demonstrated in various disease models including hindlimb ischemia2, heart disease3, bone healing4 and neural injury5, etc. However, most gene therapy techniques rely on viral vectors, which are associated with safety concerns such as potential immunogenicity and insertional mutagenesis. Biomaterials mediated non-viral gene delivery may overcome these limitations, but often suffer from low transfection efficiency. To speed up the discovery of novel biomaterials for efficient non-viral gene delivery, recent studies have employed combinatorial chemistry and high-throughput screening approach. Biodegradable polymer libraries such as poly(β-amino esters) (PBAE) have been developed and screened, which led to the discovery of leading polymers with markedly enhanced transfection efficiency compared to the conventional polymeric vector counterparts.6-7
Herein, we describe the synthesis of PBAE and verification of their ability to transfect adipose-derived stem cells (ADSCs) in vitro, followed by subsequent transplantation of genetically-modified ADSCs overexpressing vascular endothelial growth factor (VEGF) in a murine model of hindlimb ischemia. The outcomes were evaluated by tracking cell fate using bioluminescence imaging, assessing tissue reperfusion using laser Doppler perfusion imaging (LDPI), and determining angiogenesis and tissue salvage by histology.
Protocol
1. Polymer Synthesis
- In a fume hood, weigh out 3,523 mg of butanediol diacrylate (C) and transfer to a glass scintillation vial containing a stir bar.
- Pre-heat 5-amino-1-pentanol (32) to 90 °C to solubilize the salt, then in a fume hood, weigh out 1,533 mg 32 and add to the scintillation vial containing C. This method will result in a molar ratio of C:32 = 1:1.2.
- Immediately place the vial containing both solutions onto a stir plate. Set stir speed at 600 rpm.
- Transfer the scintillation vial to an oven set at 90 °C. Set stirring speed to 1,000 rpm for 4 hr. If the stir bar gets stuck, lower the speed. After 4 hr, lower speed to 300 rpm and maintain at 90 °C for another 12-16 hr.
- Add 5 g of C32 to 10 ml of anhydrous tetrahydrofuran (THF) in a glass scintillation vial containing a stir bar. Wrap in foil and vortex on high until fully dissolved.
- In a separate glass scintillation vial containing a stir bar, add 10 mM of Tetraethyleneglycoldiamine (122). Add 40 ml of THF.
- Place both vials (C32 and 122) on a stir plate for 5 min then combine together. Cover in foil and leave at room temperature stirring at 400 rpm for 24 hr. The final product is termed C32-122.
- Weigh out 5 x 50 ml Falcon tubes for each 5 g batch of C32-122. Note the mass of each tube in a notebook.
- Transfer 30 ml of anhydrous diethyl ether to each 50 ml Falcon tube.
- Transfer 10 ml of C32-122 to each 50 ml Falcon tube.
- Vortex Falcon tubes vigorously then centrifuge at 2,500 rpm for 2 min. Note: The extracted polymer will collect at the base of the tube.
- Discard the upper solution and repeat steps 1.9 and 1.11 two more times.
- Place the open tubes containing the extracted C32-122 in the desiccator and vacuum overnight. Ensure all tubes are protected from light.
- Weigh all tubes containing C32-122 to determine the final mass of the extracted polymer.
- Dissolve the extracted polymer in anhydrous DMSO at a concentration of 100 mg/ml.
- Store the dissolved polymer at -20 °C protected from light and moisture.
2. Cell Seeding
- Pre-coat the base of a T-150 tissue culture flask with 0.1% (wt/vol) gelatin. The gelatin should remain in the flask for 30-45 min. The gelatin coating helps adhesion of ADSCs.
- Aspirate gelatin from the pre-coated T-150 flask.
- Add 4 x 106 ADSCs (≤ passage 3) to the flask in 20 ml of DMEM containing 10% FBS, 1% Penicillin/Streptomycin and 10 ng/ml bFGF.
- Place the T-150 flask in the incubator at 37 °C, 5% CO2 overnight.
- Begin transfection procedure the following day.
3. Nanoparticle Preparation and Transfection
- The following procedure is for the preparation of nanoparticles containing 16.1 μg plasmid DNA per 1.0 x 106 cells at a polymer to plasmid weight ratio of 30:1.
- Dilute plasmid DNA (1 mg/ml, pVEGF165, Aldevron, ND, USA) to a final concentration of 120 μg/ml in sodium acetate (25 mM) in a 15 ml Falcon tube.
- Dilute C32-122 (100 mg/ml) to a final concentration of 3.6 mg/ml in sodium acetate (25 mM) in a second 15 ml Falcon tube.
- Combine the contents of each Falcon tubes into a single 15 ml falcon tube and vortex immediately on high for 10 sec.
- Let the tube sit at room temperature for 10 min for nanoparticle formation to occur.
- While incubating, replace medium in tissue culture flask with 7.8 ml fully supplemented DMEM per 1.0 x 106 cells transfected.
- Transfer the nanoparticle solution to the tissue culture flask and tilt forward and backwards to spread evenly.
- Place the tissue culture flask in the incubator at 37 °C, 5% CO2 for 4 hr.
- After 2 hr, replace the nanoparticle containing media with DMEM.
- Following a further 2 hr incubation at 37 °C, 5% CO2, the cells are ready for in vivo injection.
4. Hindlimb Ischemia Procedure
- All experiments were performed in accordance with the Stanford University Animal Care and Use Committee Guidelines and approved APLAC protocols. Generating the murine model of hindlimb ischemia is performed as previously described.8 Please refer to the reference for detailed instructions. A short description is provided below.
- Place the mouse under anesthesia with 1-3% dosage isoflurane by inhalation and oxygen flow rate of 1 L/min. Ensure anesthetic depth by toe pinch, reduction in respiratory rate, and lethargy.
- Remove hair from the abdominal region and both hindlimb areas of the mouse with shaving cream.
- Make incision at center of medial thigh towards midline and then cut through overlying fat pad to expose the femoral artery.
- Ligate the artery at two sites: distal site (close to the knee) and proximal site (just distal to the inguinal ligament).
- To create the ligations, separate the artery from the vein and thread a 5-0 silk suture around the artery. Tie off the artery with two knots to make the ligation.
- Remove the artery between the two ligation points.
- Wash the surgical area with PBS and then suture the incision closed.
- The anesthesia can now be removed. Keep the mouse warm until re-awakening. For post-operative care, 2-4 mg/kg Lidocaine can be injected subcutaneously at the incision site prior to re-awakening. Further pain management may include subcutaneous delivery of 0.05-0.1 mg/kg Buprenorphine at 12 and 24 hr. Monitor the health status of the mice once a day for seven days by observing changes in breathing pattern, overt pain or distress, and skin color.
- Confirm hindlimb ischemia by laser Doppler perfusion imaging.
5. Cell Injections
- When the transfected cells and the mice are ready, wash transfected cells with PBS, trypsinize, centrifuge, and then count.
- Resuspend cells at a density of 10 x 106 cells per ml in PBS.
- Cell suspensions should be separated into separate Eppendorf tubes at a volume of 110 μl. This will ensure that each mouse receives an equal amount of cells.
- Place each mouse under anesthesia (2.5% isoflurane, 1 L/min oxygen flow rate).
- Clean the injection site with alcohol wipes.
- Utilizing a 27 G tuberculin syringe, draw the cells up and down into the syringe to ensure a homogeneous suspension is achieved. Draw up 100 μl of the cell suspension volume into the syringe.
- Inject half the cell suspension into the adductor muscle region and then inject the other half into the calf muscle region. Upon injecting, leave the syringe in place for at least 15-30 sec to prevent leakage of the cell suspension.
- Proper controls for the animal study include: 1) cells transfected with a non-coding plasmid (e.g. plasmid with no biological activity); 2) cells alone; and 3) PBS.
- When injections are complete, remove the mouse from anesthesia and keep warm until the mouse re-awakes.
6. Bioluminescence Imaging
- To begin bioluminescence imaging (BLI), anesthetize mice as described above.
- Clean the injection site with alcohol wipes.
- Utilizing insulin syringe, load 100 μl of 15 mg/ml D-luciferin (the substrate for firefly luciferase). Keep substrate away from light.
- To perform intraperitoneal injections, hold mice by the skin of their back to pull their anterior skin taut. Insert needle intraperitoneally into the abdominal region and inject 100 μl of substrate.
- Place the mouse in the IVIS luciferase imaging chamber under anesthesia.
- Image the mice with an exposure time of 1.0 min. When parameters are set, begin to acquire images.
- When the image is acquired, mark the region of interest to measure the luciferase signal. In this case, mark the ischemic limb at the cell-injection site.
- Continue to measure the luciferase signal every 3-5 min until the signal reaches a peak and begins to decline. This is the value utilized for comparison across mice and over several time points.
- When complete, remove the mice from the chamber and anesthesia. Keep mice warm until re-awakening.
7. Laser Doppler Perfusion Imaging
- Laser Doppler perfusion imaging is performed as previously described.8 The procedure is briefly described here.
- Prior to Doppler imaging the mouse should be cleanly shaven at the areas overlying both hindlimbs and the abdominal region to prevent artifact due to hair.
- When prepared to Doppler image, the mouse should be anesthetized as described above.
- Warm the mouse to 36 °C on a hot plate while monitoring body temperature by rectal thermometer.
- Place the mouse on a non-reflective black surface and keep anesthetized by nose cone. The mouse should be placed on its dorsal surface with its limbs exposed evenly.
- Position the laser Doppler sensor roughly 15 cm above the mouse and directing the laser pointer from the sensor towards the groin region of the mouse.
- When the mouse and the Doppler sensor are in position, images can be taken utilizing the LDPIwin software by pressing the Start button.
- Relative perfusion measurements could be taken by indicating both hindlimbs (ischemic and non-ischemic) as region of interests (ROI). The ratio of mean perfusion of the ischemic to the non-ischemic hindlimb will indicate relative perfusion.
8. Tissue Harvest Procedure
- When prepared to harvest mouse tissue for histology and/or biochemical processing, the mouse should be euthanized. Here, the mouse is euthanized by exposing to 5% isoflurane for 3-5 min, then euthanizing the mouse by cervical dislocation.
- Cut the overlying skin surrounding the hindlimb circumferentially at the distal abdominal region and proximal to the hindlimb. The skin is cut from the ventral to the dorsal surfaces, such that the skin can be pulled back over the hindlimb to the foot.
- Upon pulling the skin back, the limb could be amputated by utilizing blunt dissection scissors to cut at the pelvic and femur joint.
- Two main tissues are taken for analysis: the medial adductor muscles and the gastrocnemius muscles.
- For histology, embed one or both tissues in optimal cutting temperature (OCT) for cryosectioning.
- For biochemical analysis, collect one or both tissues into an 1.5 ml Eppendorf and submerge into a bath of liquid nitrogen for at least 2 min. The samples can be stored at -80 °C for future processing.
Representative Results
Upon mixing together, the positively-charged polymer (C32-122) and negatively-charged DNA plasmid self-assembles into nanoparticles. Nanoparticle formation may be confirmed through electrophoresis analysis i.e. the complexation between C32-122 and plasmid DNA will prevent mobilization of the DNA during electrophoresis. The polymer serves as a transfection reagent to facilitate enhanced uptake of DNA into the target cells and the subsequent expression of encoding proteins (Figure 2). Cells can be transfected with any therapeutic genes or reporter DNA such as green fluorescent protein (GFP) to facilitate rapid optimization of polymeric vector design and transfection conditions with high efficiency using fluorescence-activated cell sorting (FACS) and fluorescence microscopy (Figure 3). For ADSCs, an efficiency above 20% is deemed suitable.
To facilitate tracking of the cell fate post-transplantation, cells can be stably transduced with luciferase, which allows real-time monitoring of cell viability and distribution in vivo using non-invasive bioluminescence imaging (BLI). BLI imaging showed high intensity luminescence signal from the hind limb (Figure 4, left panel), indicating that implanted cells remained at the injection site over several days, and we do not observe noticeable cell migration towards other tissues or organs over the course of 21 days. Cell signal in general decreased overtime and lasted up to 14 days (Figure 4, right panel), suggesting that most transplanted cells are available for overexpressing therapeutic factors for up to 2 weeks.
Doppler imaging is a useful tool that enables real-time monitoring of blood reperfusion to the ischemic limb. The left panel of Figure 5 is a representative image of blood flow following induction of ischemia. The dark area indicates successful blocking of blood flow after the surgery. The right panel of Figure 5 illustrates complete reperfusion of the limb 14 days after treatment with transfected cells.
The techniques described above allow real-time quantification, and should be coupled with additional end-point analyses to thoroughly examine the therapeutic efficacy. Muscle tissues that have received transplanted cells can be harvested at different time points for gene expression and histological analyses. RT-PCR can be used to quantify gene expression in the hind limb to confirm genetic up-regulation in transfected cells several days post-transplantation. Figure 6 shows VEGF expression in the un-treated limb, injected with PBS or injected with VEGF expressing ADSCs. The results confirm VEGF upregulation in non-viral transfected cells four days post-transplantation, further providing evidence of transplanted cell survival.
Histological staining allows direct visualization of tissue morphology and degree of tissue regeneration. Histological analysis for blood vessel density can be coupled with Doppler imaging data to help evaluate the efficacy of blood reperfusion (Figure 7). Tissue morphology staining such as H&E and Masson's Trichrome stainings are useful for evaluating the degree of tissue regeneration or necrosis. Successfully newly-regenerated muscle tissue is characterized by muscle cells with centrally located nuclei, whereas necrotic tissues often show substantial tissue fibrosis or increased number of inflammatory cells such as macrophages.
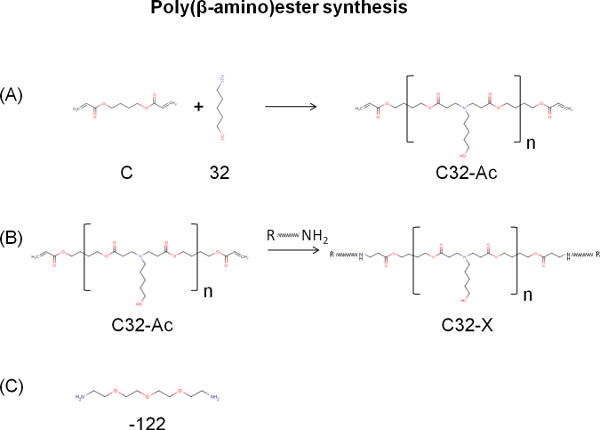
Figure 1. Schematic illustrating the synthesis of poly(β-amino)ester (PBAE)-based vector C32-122. (A) Acrylated terminated C32-Ac was formed by a Michael addition reaction between a monomer with diacrylate end groups (C) and a monomer with primary amine end group (32). (B) End-modified PBAE polymers can be formed by adding amine terminated monomers for enhanced transfection efficiency. (C) Tetraethyleneglycoldiamine (122) was chosen as the terminal amine monomer. Click here to view larger figure.
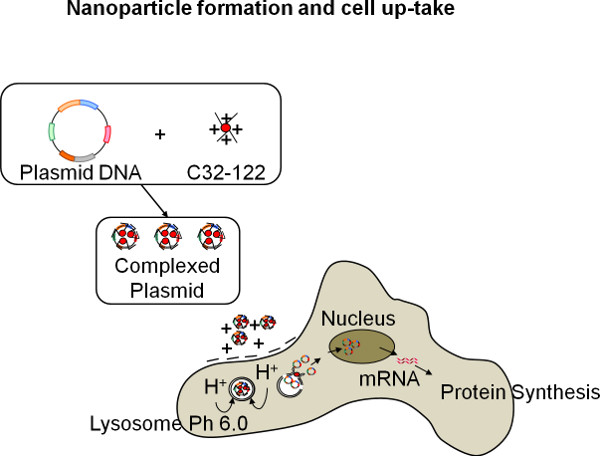
Figure 2. Schematic of formation of biodegradable polymeric nanoparticles, and their subsequent cell up-take process for protein expression.
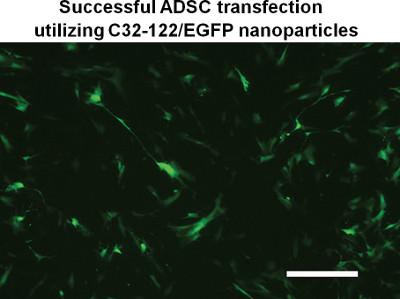
Figure 3. Human adipose-derived stem cells overexpressing green fluorescent protein (GFP). Transfection was performed using polymeric nanoparticles formed using C32-122 and GFP DNA plasmid. Scale bar: 200 μm.
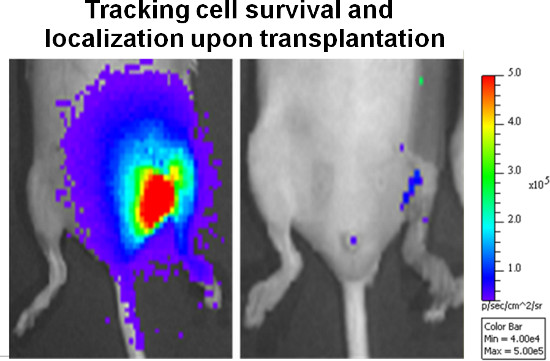
Figure 4. Representative bioluminescence imaging (BLI) data of the mouse limbs at day 0 (left panel) and day 14 (right panel) after injecting GFP-luciferase positive adipose-derived stem cells into the mouse hindlimb.
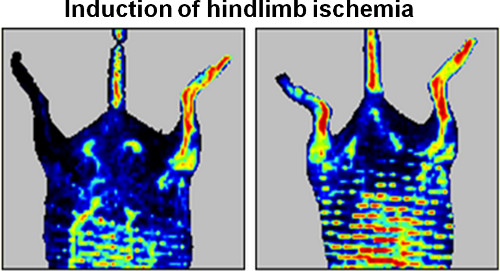
Figure 5. Representative Doppler images demonstrating induction of ischemia in one side of murine hindlimb at day 0 (left panel), and successful blood reperfusion 14 days after the injection of VEGF-overexpressing adipose-derived stem cells (right panel).
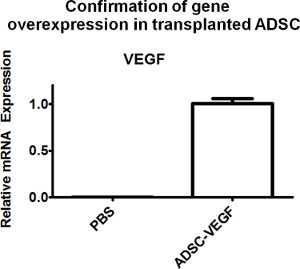
Figure 6. To confirm cell survival and overexpression of encoded therapeutic factors in situ, tissues can be harvested from the injection site for gene expression analyses. RT-PCR confirmed successful up-regulation of VEGF, the encoded therapeutic protein, in the treated group (ADSC-VEGF) 4 days after the cell injection, whereas no expression was detected in the PBS control.
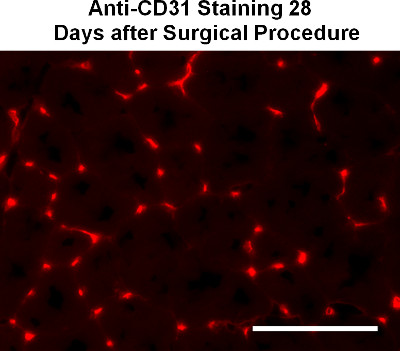
Figure 7. Representative immunohistochemical image demonstrating capillary density by anti-CD31 staining at 28 days after the surgical procedure. Scale bar: 100 μm.
Discussion
Here we report a method to program adult stem cells to overexpress therapeutic factors using non-viral, biodegradable nanoparticles. This platform is particularly useful for treating diseases where stem cells can naturally home, such as ischemia and cancer.9-10 Furthermore, the non-viral gene delivery platform allows for transient overexpression of therapeutic factors, which is suitable for most tissue regeneration and wound healing processes. The transfection process depends upon efficient DNA entry into cells, and in general works well in actively-dividing cell types, but not as well in non-dividing cells. The transfection efficiency of PBAE polymers may vary from cell type to cell type, and should be optimized individually, and further chemical structure modifications may be explored to achieve optimal transfection efficiency in specific targeted cell populations.11-12 Cells transfected with the method described above typically results in transient gene expression and protein secretion for two weeks, with peak gene expression achieved around day 2-4. For animal experiments, ADSCs with transfection efficiency above 20% are considered suitable. It is recommended to perform FACS analysis every time a process parameter is changed in order maintain consistency (e.g. new batch of polymers, plasmids or cells).
Compared to the commercially available transfection reagents such as PEI and Lipofectamine 2000, which are non-degradable, the PBAE polymers used in the reported platform above are biodegradable and more suitable for clinical translation. Given that such polymeric vectors are hydrolytically degradable13 and light sensitive, caution should be taken to store PBAE polymers properly (-20 °C, in the dark, with desiccant) to prevent unwanted hydrolysis and degradation. The resulting polymers have a distribution of molecular weight, and size exclusion chromatography (SEC) may be performed to further purify PBAEs with increased transfection efficiency.14 It is also recommended to perform nuclear magnetic resonance (NMR) at each step in the polymer synthesis protocol. The NMR should be performed to confirm formation of C32 from its individual components and again to confirm successful addition of end capping groups i.e. 122. Note: after addition of end capping groups, the acrylate groups should disappear from the NMR spectrum. The presence of excess amine terminated monomers in the final polymer can lead to increased cell toxicity, therefore it is important to adequately wash final polymer in diethyl ether to ensure removal of excess monomers. As mentioned above, SEC may also be used to further purity the final product.
Unlike many viral-based gene delivery platforms, the polymer-based gene delivery system used here does not integrate into the host genome, thereby avoiding the potential risk for insertional mutagenesis and immunogenicity.15-16
In the described procedure, cells are transplanted without sorting directly after the transfection process, which contains a mixture of cells overexpressing encoding proteins and un-transfected cells. This procedure allows minimal ex vivo manipulation of the cells, and is likely to be more clinically translatable given reduced time, cost and chances of contamination. Cells may also be purified further to select transfected cells only to further enhance the level of overexpression of therapeutic factors in situ.
The efficacy of programmed stem cells for angiogenesis should be examined using a combination of assays to thoroughly asses the outcomes at multiple levels including cellular, morphological and physiological. Bioluminescence imaging enables real-time monitoring of the survival and distribution of transplanted cells over time. Gene expression analyses from harvested tissues allow verification of cell survival and genetic up-regulation of encoded factors in situ. Histological staining enables direct visualization of blood vessel density, inflammation and tissue regeneration. It should be noted that increased blood vessel density does not always lead to successful blood reperfusion, as the newly formed vessels can be immature and non-functional. Therefore it is important to assess the physiological function of newly generated vessels using laser Doppler perfusion imaging. While the methods above focus on using adipose-derived stem cells to promote angiogenesis in a hindlimb ischemia model, the concept of programming cells as drug delivery vehicles is broadly applicable. The platform may be easily adapted to program other cell types for overexpressing therapeutic factors that are relevant for treating other diseases such as cancer and musculoskeletal diseases.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge American Heart Association National Scientist Development Grant (10SDG2600001), Stanford Bio-X Interdisciplinary Initiative Program, and Stanford Medical Scholars Research Program for funding.
Materials
| Name of the Reagent | Company | Catalogue Number | Comments (optional) |
| DMEM | Invitrogen | 11965 | |
| Fetal Bovine Serum | Invitrogen | 10082 | |
| Penicillin/Streptomycin | Invitrogen | 15070 | |
| Basic Fibroblast Growth Factor | Peprotech | 100-18B | |
| 1,4-Butanediol Diacrylate (90%) | Sigma Aldrich | 411744 | Acronym: C |
| 5-amino-1-pentanol (97%) | Alfa Aesar | 2508-29-4 | Acronym: 32 |
| Tetraethyleneglycoldiamine >99%) | Molecular Biosciences | 17774 | Acronym: 122 |
| Sodium Acetate | G-Biosciences | R010 | |
| Phosphate Buffered Saline | Invitrogen | 14190-144 | |
| Tetrahyofuran Anhydrous (>99.9%) | Sigma Aldrich | 401757 | |
| Diethyl Ether Anhydrous (>99%) | Fisher Scientific | E138-4 | |
| DMSO Anhydrous (>99.9%) | Sigma Aldrich | 276855 | |
| Gelatin | Sigma Aldrich | G9391 | |
| Trypsin-EDTA | Invitrogen | 25200 | |
| D-luciferin | GoldBio | ||
| Optimal Cutting Temperature (O.C.T) | Tissue-Tek | 4583 | |
| Rat anti-Mouse CD31 | BD Pharmingen | 550274 | |
| Alexa Fluor 594 anti-rat IgG | Invitrogen | A11007 |
References
- Deveza, L., Choi, J., Yang, F. Therapeutic angiogenesis for treating cardiovascular diseases. Theranostics. 2, 801-814 (2012).
- Yang, F., et al. Genetic engineering of human stem cells for enhanced angiogenesis using biodegradable polymeric nanoparticles. Proc. Natl. Acad. Sci. U.S.A. 107, 3317-3322 (2010).
- Mangi, A. A., et al. Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med. 9, 1195-1201 (2003).
- Lee, J. Y., et al. Enhancement of bone healing based on ex vivo gene therapy using human muscle-derived cells expressing bone morphogenetic protein 2. Hum. Gene Ther. 13, 1201-1211 (2002).
- Park, K. I., et al. Neural stem cells may be uniquely suited for combined gene therapy and cell replacement: Evidence from engraftment of Neurotrophin-3-expressing stem cells in hypoxic-ischemic brain injury. Exp. Neurol. 199, 179-190 (2006).
- Green, J. J., Langer, R., Anderson, D. G. A combinatorial polymer library approach yields insight into nonviral gene delivery. Acc Chem Res. 41, 749-759 (2008).
- Yang, F., et al. Gene delivery to human adult and embryonic cell-derived stem cells using biodegradable nanoparticulate polymeric vectors. Gene Ther. 16, 533-546 (2009).
- Niiyama, H., Huang, N. F., Rollins, M. D., Cooke, J. P. Murine model of hindlimb ischemia. J. Vis. Exp. , e1035 (2009).
- Ceradini, D. J., et al. Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat. Med. 10, 858-864 (2004).
- Kidd, S., et al. Direct evidence of mesenchymal stem cell tropism for tumor and wounding microenvironments using in vivo bioluminescent imaging. Stem Cells. 27, 2614-2623 (2009).
- Sunshine, J., et al. Small-molecule end-groups of linear polymer determine cell-type gene-delivery efficacy. Adv. Mater. 21, 4947-4951 (2009).
- Sunshine, J. C., Akanda, M. I., Li, D., Kozielski, K. L., Green, J. J. Effects of base polymer hydrophobicity and end-group modification on polymeric gene delivery. Biomacromolecules. 12, 3592-3600 (2011).
- Lynn, D. M., Langer, R. Degradable poly(β-amino esters): Synthesis, characterization, and self-assembly with plasmid DNA. J. Am. Chem. Soc. 122, 10761-10768 (2000).
- Eltoukhy, A. A., et al. Effect of molecular weight of amine end-modified poly(beta-amino ester)s on gene delivery efficiency and toxicity. Biomaterials. 33, 3594-3603 (2012).
- Glover, D. J., Lipps, H. J., Jans, D. A. Towards safe, non-viral therapeutic gene expression in humans. Nat. Rev. Genet. 6, 299-310 (2005).
- Dave, U. P., Jenkins, N. A., Copeland, N. G. Gene therapy insertional mutagenesis insights. Science. 303, 333 (2004).

