Techniques for the Analysis of Extracellular Vesicles Using Flow Cytometry
Summary
Many different methods exist for the measurement of extracellular vesicles (EVs) using flow cytometry (FCM). Several aspects should be considered when determining the most appropriate method to use. Two protocols for measuring EVs are presented, using either individual detection or a bead-based approach.
Abstract
Extracellular Vesicles (EVs) are small, membrane-derived vesicles found in bodily fluids that are highly involved in cell-cell communication and help regulate a diverse range of biological processes. Analysis of EVs using flow cytometry (FCM) has been notoriously difficult due to their small size and lack of discrete populations positive for markers of interest. Methods for EV analysis, while considerably improved over the last decade, are still a work in progress. Unfortunately, there is no one-size-fits-all protocol, and several aspects must be considered when determining the most appropriate method to use. Presented here are several different techniques for processing EVs and two protocols for analyzing EVs using either individual detection or a bead-based approach. The methods described here will assist with eliminating the antibody aggregates commonly found in commercial preparations, increasing signal–to-noise ratio, and setting gates in a rational fashion that minimizes detection of background fluorescence. The first protocol uses an individual detection method that is especially well suited for analyzing a high volume of clinical samples, while the second protocol uses a bead-based approach to capture and detect smaller EVs and exosomes.
Introduction
Extracellular Vesicles (EVs) are small, membrane-derived vesicles found in bodily fluids that are highly involved in cell-cell communication and help regulate a diverse range of biological processes. Analysis of EVs using flow cytometry (FCM) has been notoriously difficult due to their small size and lack of discrete populations positive for markers of interest. Methods for EV analysis, while considerably improved over the last decade, are still a work in progress. Unfortunately, there is no one-size-fits-all protocol, and several aspects must be considered when determining the most appropriate method to use. Presented here are several different techniques for processing EVs and two protocols for analyzing EVs using either individual detection or a bead-based approach. The methods described here will assist with eliminating the antibody aggregates commonly found in commercial preparations, increasing signal–to-noise ratio, and setting gates in a rational fashion that minimizes detection of background fluorescence. The first protocol uses an individual detection method that is especially well suited for analyzing a high volume of clinical samples, while the second protocol uses a bead-based approach to capture and detect smaller EVs and exosomes.
EVs, also known as microparticles, are small, membrane-derived vesicles found in bodily fluids that are involved in cell-cell communication and help regulate a diverse range of biological processes1. Through expression of various surface markers and/or direct transfer of biological material, EVs are able to alter the function of recipient cells to play either activating or suppressing roles in intercellular communication2–4. Clinically, platelet-derived EVs are known to have strong anticoagulant activity5, while others have been shown to contribute to a wide range of conditions, from promoting tumor metastasis6 to protecting against disease7. EVs can be classified into smaller categories of cell-derived vesicles such as exosomes and microvesicles (MVs), depending on their size and mechanism of generation8. The nomenclature of cell-derived vesicle subpopulations continues to be a topic of ongoing debate8,9, however, exosomes are generally described as small, 40 to 100 nm particles derived from endosomal fusion with the plasma membrane, while MVs are larger 100 to 1,000 nm particles formed by shedding of the plasma membrane10. Here, the general term “EVs” will be used to refer to all types of extracellular biological vesicles released by cells.
Isolation of EVs from whole blood is a multi-step procedure and many different processing variables have been shown to affect EV content, including storage temperature and duration11,12, anticoagulant/preservative used13 and centrifugation method used14. A need for standardization of these variables has led to recommendations by the International Society on Thrombosis and Haemostasis Scientific and Standardization Committee (ISTH SSC) for proper blood processing and EV isolation procedures15,16, yet there exists no consensus among researchers on the optimal protocol to use 12. Most agree, however, that tightly controlled pre-analytical variables are crucial for accurate and reproducible data.
In order to analyze EVs, researchers have utilized various methods, including transmission electron microscopy17, scanning electron microscopy18,19, atomic force microscopy, dynamic light scattering20,21 and western blotting22,23. While FCM is the method of choice for many researchers9,24–26 due to its high throughput capabilities, analysis of EVs using FCM has been notoriously difficult due to their size and lack of discrete positive populations27–32. Compared to analysis of cells, the small size of the EVs results in 1) less fluorescence emitted due to the fewer number of antigens per particle and 2) limited feasibility of post-stain washing, which is necessary to reduce background fluorescence. Common challenges among researchers include signals arising from immunoglobulin aggregates27,28and self-aggregation of antibodies29. Furthermore, the long processing times and lengthy washing/isolation procedures used by many of the current protocols33,34 require multi-day time commitments to analyze a small number of samples, making them less than ideal for high throughput applications. Some researchers forgo a wash step altogether, rendering traditionally used FCM negative controls such as fluorescence minus one (FMO) and antibody isotypes useless for accurately assessing background fluorescence30.
Our protocols address three common problems that can impede proper FCM analysis of EVs: signals arising from antibody aggregates and other non-vesicles, difficulty in removing unbound antibody, and lack of discernible positive populations. The techniques described here will assist with eliminating the antibody aggregates commonly found in commercial preparations, increasing signal–to-noise ratio, and setting gates in a rational fashion that minimizes detection of background fluorescence. Two different detection methods are presented here: the first protocol uses an individual detection method that is especially well suited for analyzing a high volume of clinical samples, while the second protocol uses a beads-based approach to capture and detect smaller EVs and exosomes.
Protocol
NOTE: The following protocols have been performed in compliance with all institutional, national and international guidelines for human welfare. All human subject samples were tested under an institutional review board (IRB)-approved protocol and with informed consent of the subjects.
1. METHOD A: Individual Detection Method
1.1) Processing of Blood Sample/Isolation of EVs
- Draw blood from donor/patient into two 10 ml glass tubes containing 1.5 ml of ACD-Solution A or other suitable anticoagulant and process immediately (within 30 min max) using the following 2-step differential centrifugation protocol.
NOTE: This protocol will yield approximately 10 ml of platelet poor plasma (PPP) from the combined ~17 ml of blood drawn. If more or less PPP is needed, the number of tubes of blood collected may be adjusted accordingly. - Centrifuge the samples at 1,500 x g for 10 min at RT to separate the plasma from the buffy coat and red cells. Transfer 1.2 ml aliquots of the plasma supernatant to 1.5 ml centrifuge tubes, being careful not to disturb the bottom layers containing the buffy coat and red cells.
- Spin at 13,000 x g for 10 min at RT to remove platelets and large cell fragments. Carefully transfer the PPP, leaving behind 200 µl to avoid disturbing the pellet and add the PPP to a new tube.
- At this point, use PPP immediately for analysis or transfer in 1.0 ml aliquots to new 1.5 ml centrifuge tubes and store at -80 °C for up to two years for later analysis (refer to Figure 1A for overview).
- If purified EVs are needed for functional experiments, transfer 6 ml of the PPP to an ultracentrifuge tube and add 28 ml of 0.2 µm-filtered phosphate buffered saline (PBS). Spin for 60 min at 100,000 x g at RT using an ultracentrifuge equipped with a swinging bucket rotor. Aspirate supernatant and resuspend EV pellet in 1.5 ml media.
NOTE: For highest reproducibility, blood samples should be processed as consistently as possible from donor to donor. Any variation in EV isolation method could significantly impact the number and type of EVs detected.
1.2) Preparing Samples for Analysis
NOTE: From this point on, the steps explain a high throughput protocol for analyzing 12 samples for 14 markers in 3 panels. However, other combinations of antibodies can be used here; the protocol can be adapted to study other EV populations by substituting the suggested markers for those of interest.
- Remove 12 samples from freezer (if stored at -80 °C) and thaw at 37 °C.
- Pipette contents up and down several times to mix. Remove 320 µl from each sample and add to the top row of a 96 well plate.
NOTE: A width-adjustable multi-well pipet is extremely helpful for this and many other steps throughout the assay, particularly when analyzing multiple samples at once.
1.3) Staining EV Samples
- Prior to staining, filter all antibodies (Abs) to remove aggregates, which can cause positive signals.
- Combine antibodies to be used in each of the 3 panels into separate 0.22 µm centrifugal filter tubes and centrifuge using a fixed angle single speed centrifuge (~750 x g) at RT for 2 min, or until all of the Ab mixture has passed through the filter and no antibody liquid remains on the surface of the filter. Store Ab cocktails in the fridge for up to two weeks but re-filter each time before use.
- Add the appropriate amount of filtered Ab mixture to each well in row 2 (e.g., samples in Panel I are stained with 2 µl of each Ab, so a total of 12 µl of the filtered Ab cocktail is added per well to row 2). Refer to Figure 1B for an outline of the suggested plate map. Repeat these additions to the rows beneath if more panels are run (here, add 8 µl/well of the Panel II cocktail to row 3 and 11 µl/well of the Panel III cocktail to row 4; refer to Materials List for specific panel information).
- Using the multichannel pipet, mix the PPP samples in row 1 up and down and transfer 100 µl from the wells in row 1 to the wells in row 2. Mix up and down. Change tips and repeat, transferring 100 µl from row 1 to rows 3 and 4. Incubate at 4 °C for 30 min.
1.4) Washing MV Samples
- Remove the 96-well plate from 4 °C and transfer to biological safety cabinet. Using a multichannel pipette, add 220 µl of PBS/well to rows 6-8 (to be used for rinsing/washing the wells containing stained PPP).
- Transfer the contents of each well to pre-labeled centrifugal filter tubes using the width-adjustable multichannel pipet (For 12 samples, with 3 panels of antibodies, 12 x 3 = 36 filter tubes will be needed). Using the same tips, remove 200 µl of PBS from the wash rows and add to the corresponding wells from which PPP was just removed.
- Mix up and down to rinse the wells and transfer the rinse solution to the same filters to which the PPP was previously added. Close tops of centrifugal filters. Change tips.
- Repeat this process with the remaining stained samples until all stained PPP samples have been transferred along with their rinse solutions to centrifugal filters.
- Transfer the centrifugal filters to a fixed rotor centrifuge and spin at 850 x g for 3 min at RT.
NOTE: Ensure that no liquid remains on the filter tops. After centrifugation, the filter should appear to be “dry” with no visible fluid layer remaining on top. While unlikely, certain PPP samples may require a longer centrifugation time to effectively move through the filter. - Remove the centrifugal filter tubes and return to the biological safety cabinet. Using the multichannel pipet, resuspend the tops of the filters in 300 µl of PBS. Transfer the resuspended contents to pre-labeled tubes for immediate FCM analysis.
NOTE: It is very important to keep the force and number of pipette plunger depressions consistent among samples to avoid sample-to-sample variation. This should ideally be done using an electronic pipet that has been programmed to pipet up and down a specific volume (e.g., 280 µl) an exact number of times (e.g., 8 times) for each sample.
1.5) Cytometer Setup
- Open the FCM software. Prior to experiment setup, perform daily instrument calibration and setup using instrument setup beads (following manufacturer’s instructions).
- If EV samples have been stained with more than one antibody and multiple fluorochromes are to be measured at once, calculate compensation values as follows:
- Add 2 drops of compensation beads to pre-labeled tubes (1 tube for each fluorochrome-conjugated antibody) and add the recommended amount of antibody. Add 2 drops of negative compensation beads to another tube to use as the unstained compensation control.
- Incubate at 4 °C for 30 min, wash with PBS and resuspend in 400 µl of PBS.
- Using the flow cytometer software included with the instrument, run each compensation tube and adjust fluorescent voltages to place each peak at approximately 104 on a 5-decade log scale. Ensure that the fluorescence peak is highest (brightest) in its own fluorescent channel compared to all other channels, and adjust voltages of fluorescent parameters again if necessary. Run each individually-stained comp tube and capture at least 5,000 events per tube.
- Select the tab “Experiment,” then select “Compensation,” then select “Calculate Compensation” to apply compensation values to all samples.
- Set the forward scatter (FSC) and side scatter (SSC) voltage parameters to log scale and select the lowest thresholds allowed by the cytometer (FSC=200 and SSC=200) for each.
- While running a tube of 0.22 µm-filtered PBS, adjust the FSC & SSC voltages to the highest values that exclude the majority of background noise (i.e., just below the voltage threshold at which event rate surpasses 5 events/sec).
- Next, run a tube containing 0.2 µm – 1.0 µm beads, diluted in PBS if necessary. In an FCS vs. SSC plot, draw a gate around the bead population to capture events between 0.2 µm and 1.0 µm. Alternatively, in an SSC-H histogram, draw a gate to include all events smaller than the 1.0 µm beads.
- Set the cytometer’s flow rate to “Lo” (approximately 8-12 µl/min). Using the beads tube (or other tube containing a known concentration of beads) adjust the flow rate dial on the cytometer until the event rate reaches approximately 200 events/sec. Read all sample tubes at the same flow rate and use the same bead concentration in future experiments to ensure that flow rates remain consistent between runs.
- Run a tube of rainbow fluorescent particles diluted in PBS. Acquire 5,000 events. Record the mean intensity values for FSC, SSC, and each color channel. Use these values to adjust voltages in future experiments to ensure that fluorescence intensities remain consistent between experiments.
1.6) Sample Reading
- Set the cytometer’s flow rate to “Lo” (approximately 8-12 µl/min) and run each sample for exactly 1 or 2 min.
- After the first reading, add 20 µl of 10% NP-40 to each sample, pipette up and down, and re-read for the same amount of time (either 1 or 2 min) to allow for the subtraction of positive events detected in the lysed sample over an equal time frame.
NOTE: It is extremely important that samples be mixed using a pipet rather than a vortex. In our experience, vortexing can cause self-aggregation of some antibodies, leading to EV-mimicking positive events. - Once all samples have been read, export all .fcs files into a separate file to be used for further analysis using FCM analysis software.
1.7) Data Analysis
- Open the FCM analysis software. Import all of the .fcs files into a new experiment file.
- Open the beads-only tube. In the FSC-A vs. SSC-A plot, draw a gate around all beads sized between 0.2 µm and 1.0 µm. This is the EV gate. Drag to add to all samples.
- Using the lysed samples as negative controls, draw gates at the edge of background fluorescence in each fluorescence channel used. Drag fluorescent gates into the EV gate of each corresponding non-lysed sample.
NOTE: At this point it is also useful to examine dual fluorescent bi-parameter plots. Rocket shapes in the double-positive quadrant (see Figure 8), particularly if the markers are known to reside on unrelated cell types, may be indicative of artifact from aggregation or other vesicle-mimicking events. - For each fluorescent marker, subtract the number of events in the lysed sample from the number of events in the non-lysed sample. Optionally, divide this number by the total number of EVs within the non-lysed EV gate to get % positive values.
2. METHOD B: Beads Method
2.1) Processing of Blood Sample/Isolation of EVs
- Refer to the blood processing method described in Method A (Section 1.1).
2.2) Preparing Samples for Analysis
- If desired, fractionate PPP or ultracentrifuged EVs into exosomes and microvesicles. Add 250 µl of PPP or ultracentrifuged EVs to 0.22 μm centrifugal filters and transfer to a fixed rotor centrifuge and spin at 750 x g for 2 min at RT.
NOTE: Ensure that no liquid remains on the filter tops. After centrifugation, the filter should appear to be “dry” with no visible fluid layer remaining on top. While unlikely, certain samples may require a slightly longer centrifugation time for the fluid to effectively move through the filter. - Wash uncoated 6 µm polystyrene beads (e.g., negative AbC beads) 2x with RPMI media, and resuspend in 2 ml. Add 6,000 beads to each FACS tube. To the negative control tube, add 400 μl of RPMI media alone to the beads. To all other tubes, add 200 μl of PPP or ultracentrifuged EVs (or their fractions) and 200 μl of RPMI media.
- Adjust the final volume of all tubes to 400 μl with media and incubate overnight at 4 °C on a shaker.
- The next morning, wash beads with 2 ml of media. Aspirate off the supernatant.
- Block with 5% bovine serum albumin (BSA) in media (400 μl) for 3 hr at 4 °C on a shaker.
- Wash beads with 2 ml of media. Aspirate and resuspend pellet in 100 μl of medium.
2.3) Staining EV Samples
- Filter all antibodies. Use the same antibody panels used in Method A, or if desired, create a different combination of antibodies as long as their fluorochromes are compatible with one another.
- Combine all antibodies to be used in a single panel into a 0.22 µm centrifugal filter tube. Centrifuge using a fixed angle single speed centrifuge for 2 min or until all of the Ab mixture has passed through the filter and no antibody liquid remains on the surface of the filter.
- Add appropriate volume of filtered antibody cocktail to all tubes and incubate for 30 min at 4 °C.
- Wash beads with 2 ml of media, resuspend in 400 μl of media and run immediately (or within the same day) on flow cytometer.
2.4) Cytometer Setup and Sample Reading
- Open the FCM software. Prior to experiment setup, perform daily instrument calibration and setup using instrument setup beads (following manufacturer’s instructions).
- If EV samples have been stained with more than one antibody and multiple fluorochromes are to be measured at once, calculate compensation values as follows:
- Add 2 drops of compensation beads to pre-labeled tubes (1 tube for each fluorochrome-conjugated antibody) and add the recommended amount of antibody. Add 2 drops of negative compensation beads to another tube to use as the unstained compensation control. Incubate at 4 °C for 30 min, wash with PBS and resuspend in 400 µl of PBS.
- Using the FCM software, run each compensation tube and adjust fluorescent voltages to place each peak at approximately 104 on a 5-decade log scale. Ensure that the fluorescence peak is highest (brightest) in its own fluorescent channel compared to all other channels, and adjust voltages of fluorescent parameters again if necessary. Run each individually-stained comp tube and capture at least 5,000 events per tube.
- Select the tab “Experiment,” then “Compensation,” then “Calculate Compensation” to apply compensation values to all samples.
- Change the FSC and SSC voltage parameters to log scale and select the lowest thresholds allowed by the cytometer (FSC = 200 and SSC = 200) for each.
- Run samples, gate on the singlet beads population and acquire 2,000 events in this gate. Export .fcs files.
- Use FCM analysis software to analyze .fcs files. Gate on singlet beads. Calculate the geometric mean fluorescent intensity (MFI) for each fluorochrome and compare with the MFI of the negative control.
Representative Results
Figure 1 outlines the overall processing scheme for the isolation and detection of EVs using either the bead-based method or individual detection method. Individual detection of EVs using FCM works well for analyzing larger EVs but most cytometers are not capable of individually detecting particles as small as exosomes. A bead-based approach allows small EVs to be detected, however, there are drawbacks associated with using this method, as outlined in Table 1. Generally, isolation of EVs using ultracentrifugation (with or without the addition of a sucrose gradient fractionation procedure) is recommended when EVs are needed for functional assays. Ultracentrifugation removes impurities including serum proteins and other soluble contaminants from the plasma, which can affect functional experimental outcomes. However, ultracentrifugation is time consuming and may alter EV quantity and quality12.
Expected results for the two detection assays are depicted in Figures 2-3. For the individual detection assay, the lysed control (bottom row, Figure 2) is used to set gates for the corresponding non-lysed sample (top row, Figure 2). The majority of events should fall within the EV gate. Quadrant gates should not reveal double positive events when the two markers in comparison are not normally found on the same cell. The right biparameter plots in Figure 2 show the markers CD108a and CD235a, which are two red blood cell markers known to coexist on cells. Here, on EVs, over half of the positive events are positive for both markers, as expected. In the same way, cell surface markers known to reside on the same cell should show similar patterns of double positivity on EVs. The center biparameter plots show EV expression of two markers that are known not to coexist on cells. In this analysis of CD235a (a red blood cell marker) and CD41a (a platelet marker), the EVs show distinct, separate, positive populations, which is expected since they come from different cell types. When lysed, positive events should disappear. In general, any positive events remaining after lysis indicate the presence of signal coming from non-vesicle particles, aggregates, and/or detergent-resistant EVs. Figure 3 shows expected results using the bead-based detection method. Unlike the individual detection method, these data cannot/should not be viewed in bi-parameter plots. In the upper dot plots, no separation between the positive and negative populations exist, and events appear in the double positive quadrants even though they aren’t normally found on the same cell types due to the fact that both types of EVs will bind to a single bead. For the bead-based detection method, data are best analyzed using histogram overlays with the negative control (depicted underneath the dot plots). Positivity is measured using a marker’s MFI (mean fluorescence intensity) and compared directly with that of the negative control. If a sample is positive for the marker in question, its MFI will be higher than the negative control. The negative control for the bead method is simply beads blocked with BSA (no EVs added), which have been stained with the same antibodies and washed alongside the EV-coated beads. A comparison of expected results using the two methods can be seen in Figure 4.
The ability of the individual detection assay to properly assess EV phenotypes relies heavily on correct gating to separate Ab-positive events from background fluorescence. Therefore, it is critical to choose a negative control that most appropriately mimics/predicts background fluorescence for a given sample. When stained EVs are not washed before reading, commonly used negative controls (e.g., isotypes) fail to accurately predict background fluorescence for all markers (Figure 5A). In these cases, if washing is not an option, lysed samples tend to work better for predicting background fluorescence. However, when stained EVs are washed before reading (using centrifugal filtration, in this case), both negative controls (isotypes and lysed samples) work well for predicting background fluorescence of a sample (Figure 5A). It should be noted, however, that while all negative controls “work,” lysed controls are preferred because they provide additional information about a sample (e.g., the presence of detergent-resistant, non-vesicle-related events and/or aggregates) that can result in non-EV positive signals and improperly inflate Ab counts. Furthermore, isotype controls can sometimes be unreliable, even in washed samples, as shown in Figure 5B, where the stained sample has fewer positive events than the same sample stained with matched isotype control antibodies.
Without thorough removal of unbound antibody, FCM dot plots of some EV markers are nearly impossible to interpret, appearing as clouds of dimly fluorescent particles indistinguishable from their highly fluorescent backgrounds (Figure 6, top plot). Washing stained samples using centrifugal filters enhances the separation between background and positive marker signals (Figure 6, bottom plot); however, small EVs and exosomes may be lost through the pores of the filter.
The use of a detergent lysis step reveals positive, vesicle-mimicking events from immune complexes and protein aggregates21. When PPP is analyzed using individual detection, encountering positive events that do not disappear with lysis is a fairly often occurrence. These detergent-resistant events often appear as suspicious, highly fluorescent diagonal signals in both single parameter and biparameter plots (Figure 7). Clinically, these protein complexes and/or insoluble immune complexes are more prevalent in patients afflicted with various diseases21, such as rheumatoid arthritis28, nephrotic syndrome19, and systemic lupus erythematosus29. Therefore, depending on the objective of the research, one may wish to include or remove them from the analysis.Another way diagonal signals can form is by vortexing the samples, particularly after the addition of the lysis reagent (Figure 8). Samples should always be mixed up and down by pipet to prevent the formation of aggregates.
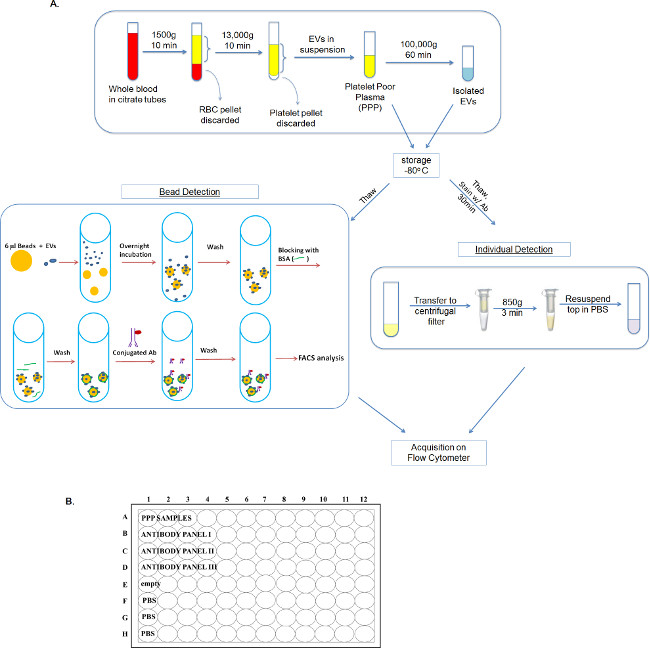
Figure 1: Overall processing scheme for the isolation and detection of EVs using either the bead-based method or individual detection method. (A) Whole blood is first processed into PPP. From there, PPP can either be processed further using ultracentrifugation to yield isolated EVs or used as-is in the individual detection or bead-based assays. (B) Outline of suggested plate map for high throughput sample analysis using the individual detection method. Please click here to view a larger version of this figure.
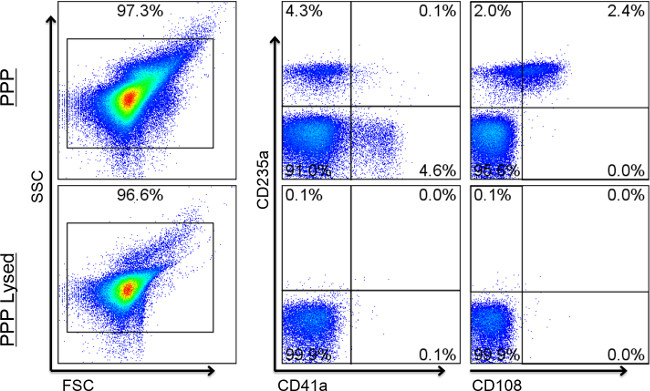
Figure 2: Expected results for Individual Detection. Flow cytometry dot plots show representative staining of lysed and unlysed EV samples. Values show percentages of positive events. The majority of events fall within the EV gate. Events shown in the right biparameter plots are within the FSC/SSC EV gates on the left. The lysed sample (bottom row) is used to set fluorescent-based gates for each corresponding (non-lysed) sample. Quad gates should not reveal double positive events when the two markers in comparison are not normally found on the same cell. Here, the biparameter CD235a and CD41a plot shows a distinct separation between the EVs expressing red blood cell markers and those expressing platelet cell markers. Likewise, cell surface markers known to reside on the same cell should show similar patterns of double positivity on EVs. The right biparameter plot shows that over half of CD235a-positive EVs are also positive for the secondary red blood cell (RBC) marker, CD108a. When lysed, positive events should disappear. Positive events remaining after lysis indicate the presence of signal coming from non-vesicle or detergent resistant particles and/or aggregates.
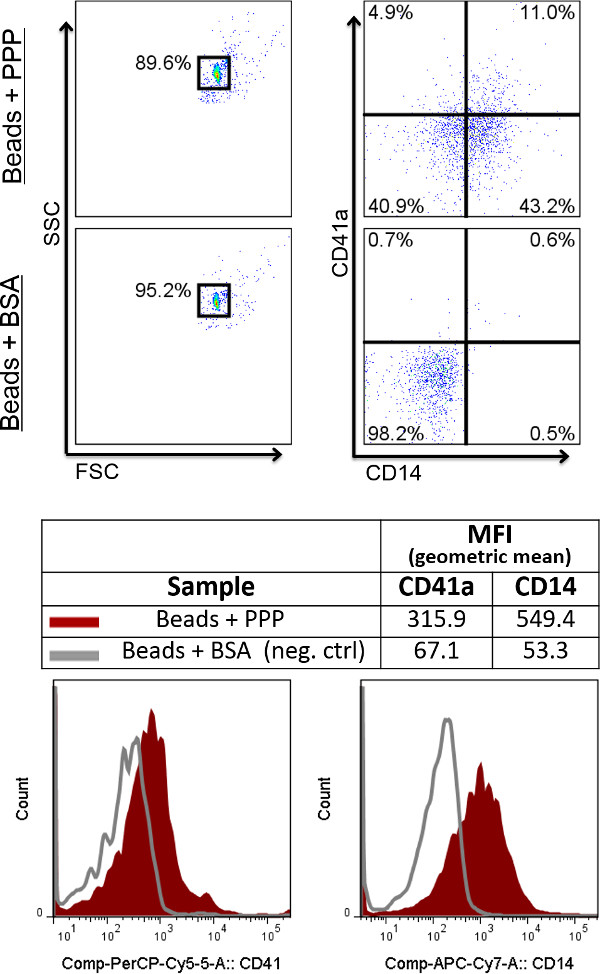
Figure 3: Expected results using the bead-based detection method. Flow cytometry dot plots show representative staining of EVs coupled to beads, as compared to beads blocked with BSA, which serves as the negative control. Values show percentages of positive events. Events shown in the right biparameter plots are within the FSC/SSC beads gates on the left. For the beads-based detection method, data are best analyzed using histogram overlays with the negative control (depicted underneath the dot plots). Positivity is measured using a marker’s MFI (mean fluorescence intensity) and compared directly with that of the negative control.
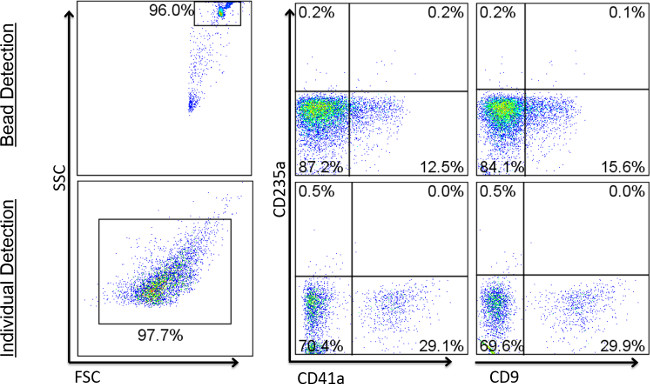
Figure 4: Comparison of expected results using beads vs. individual detection. Flow cytometry dot plots show representative staining of EVs coupled to beads (top row), compared to EVs analyzing using individual detection (bottom row). Events shown in the right biparameter plots are within the FSC/SSC beads gates on the left.
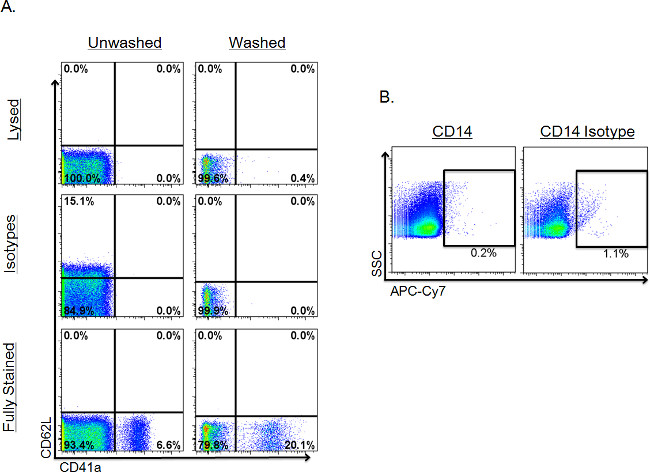
Figure 5: Comparison of different negative controls in individual detection analysis. Values show percentages of positive events. Events shown are within the FSC/SSC EV gate. (A) Comparison of negative controls in unwashed vs. washed samples. Isotype or lysed controls were evaluated for their ability to provide appropriate indications of background fluorescence across two different markers in a fully stained sample (bottom row). Gates for each marker were made using the lysed sample (top row) and then copied to the rows beneath. Washed samples were washed using post-stain filtration. (B) Example of a sample stained with the isotype control having more positive events than the sample stained with actual antibody.
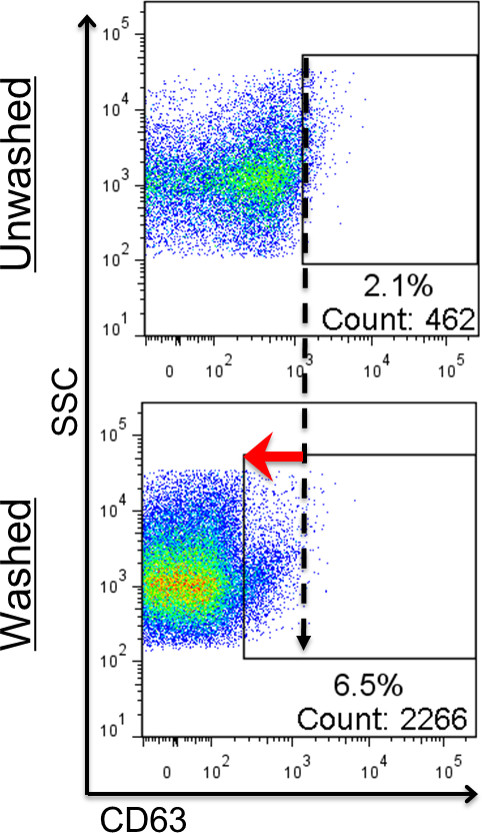
Figure 6: Effect of post-stain washing in individual detection. Values show percentages and numbers of positive events. Events shown are within the FSC/SSC EV gate. Gates for each sample were made using each sample’s lysed counterpart (not shown; refer to previous figure for gate-setting in unwashed vs washed samples). High background fluorescence makes distinguishing positive from negative events difficult (top plot). When washed, however, the positive population is revealed as unbound fluorescent antibodies are removed and background fluorescence is reduced (bottom plot).
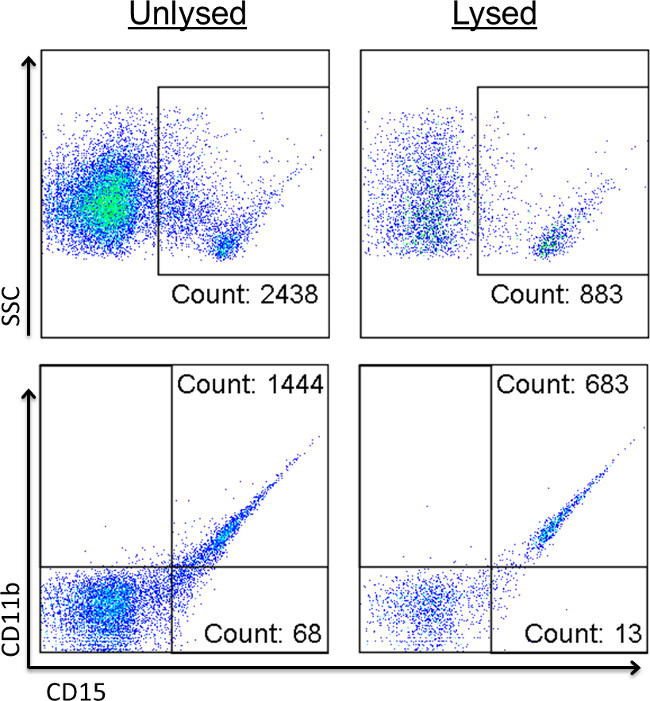
Figure 7: Detergent lysis confirms the presence of non-EV signals. Events shown are within the FSC/SCC EV gate. Values show percentages of positive events. Stained EV samples were read before (left column) and after addition of detergent (right column) to identify positive signals caused by immune complexes and other non-EV-related events.
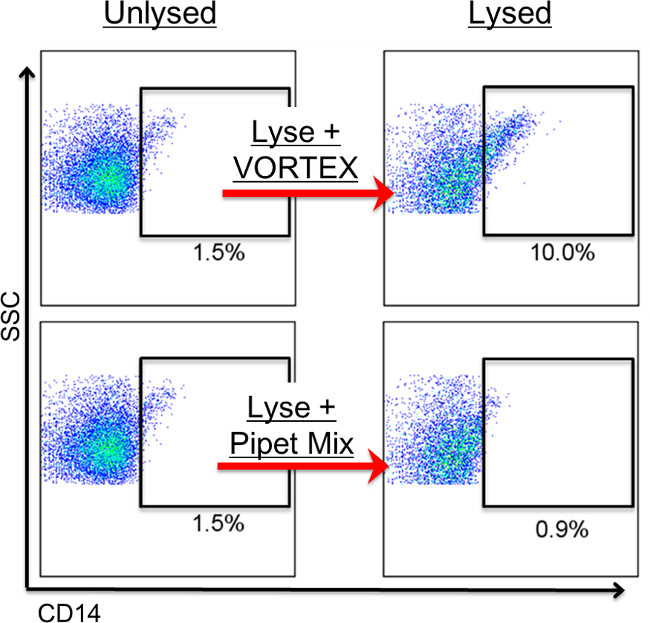
Figure 8: Vortexing causes the appearance of non-EV signals. Events shown are within the FSC/SCC EV gate. Values show percentages of positive events. Stained EV samples were read before (left column) and after addition of detergent (right column). Using a vortex to mix samples can cause EV-mimicking, diagonal populations to form (top row). When mixed gently up and down using a pipet, however, formation of these populations can be avoided (bottom row). Vortexing is not recommended for mixing, as it can cause aggregation in some samples, leading to diagonal-appearing, positive populations. In general, event number within a positive gate should not increase after lysing.
| Bead-Based Detection | Individual Detection | |
| EV Sizes | recommended for < 100 nm only | recommended for > 100 nm only |
| Time | requires overnight incubation | can complete in < 1 day |
| Sensitivity | cluster detection | single particle detection |
| Results | qualitative | quantitative |
| Washing | simple, standard centrifugation | requires centrifugal filters |
| Negative control | BSA-coated beads | lysed samples |
Table 1: Pros and cons of both detection methods.
Discussion
Two different protocols for the isolation, treatment and analysis of EVs were presented, using either an individual detection or bead-based approach. Selecting the most appropriate method to use is not always straightforward and requires an understanding of the sample being tested as well as the individual subpopulations of interest. Furthermore, the sensitivity of the cytometer used for acquisition must be considered when choosing the most appropriate method. Oftentimes there is no single best protocol to use, rather, a combination of methods provides more information about a sample than any one method alone. Ideally, several different isolation and detection techniques should be evaluated first in order to develop a tailored protocol that takes into consideration individual cytometer performance with respect to the specific EV population being studied. Alternative isolation techniques include ultracentrifugation, sucrose density fractionation, immunomagnetic bead separation, chromatography, and affinity purification12, while alternative detection methods include scanning electron microscopy, transmission electron microscopy, atomic force microscopy, dynamic light scattering, and western blotting8. By combining different techniques, the methods presented here can be adapted in order to create protocols best suited for studying various EV populations of interest.
In general, individual detection of EVs using FCM works well for analyzing larger EVs but loses sensitivity as EVs get smaller. While individual detection is more consistent in detecting larger EVs, bead-based detection is less sensitive in detecting larger EVs and more sensitive for exosomes. Larger EVs can be washed easily via post-stain filtration and detected singly via FCM. Smaller EVs and exosomes, on the other hand, are not detected well individually using FCM and are much more difficult to wash post-staining. The bead-capture protocol resolves both of these issues, allowing EVs to be easily washed and multiple EVs to be measured together to create larger positive signals detectable by FCM. However, there are drawbacks associated with using this method, as outlined in Table 1.
When working with a less sensitive cytometer, the capacity for individual detection is limited. Prior to EV analysis, the sensitivity of the cytometer should be determined using a mixture of bead sizes ranging from 0.1-1.0 µm. Failure of the cytometer to detect a majority of particles below 1.0 µm would necessitate the use of the bead-based protocol. Highly expressed markers are easily detected using either protocol. Rarer populations are sometimes easier to detect using the single particle detection protocol rather than the bead capture protocol, however, this can vary depending on such variables as: the brightness of the fluorochrome, the sample’s EV:bead ratio, and the size of the EV bearing the rare cell surface marker. Detection of multiple markers on a single particle necessitates the use of the individual detection method. The bead-based method is not capable of individual EV detection. Therefore, the bead-based protocol will yield data that are more qualitative in nature, while the individual detection method will give more quantitative data.
Additional isolation techniques must be utilized whenever EVs are needed for downstream applications. EVs used in functional assays should be ultracentrifuged using the 3-step differential centrifugation protocol, since the soluble serum proteins in plasma can affect functional experimental outcomes. For characterization of EVs, however, ultracentrifugation is not recommended, since this added step may affect EV quality and quantity due to the high forces imparted on the particles12.
The individual detection protocol contains several key steps optimized for high-throughput testing, including: 1) the implementation of centrifugal filters for the quick and effective removal of positive events caused by Ab aggregates, 2) the use of filters as a more practical alternative to ultracentrifugation or sucrose gradient fractionation for washing unbound Ab from EV samples post-staining, and 3) utilization of detergent lysis as a negative control, which not only reveals positive events caused by non-EVs but provides a good approximation of background fluorescence to distinguish positive from negative populations for drawing gates. The individual detection protocol is recommended whenever a large number of samples needs testing as it can be performed in a single day, whereas the bead-based method requires an overnight incubation.
The negative controls in each protocol have different advantages and disadvantages depending on which detection method is used. One benefit of using the bead-based assay is that the same monoclonal antibodies can be used for negative and positive tubes and the same negative control can be used for all samples. The individual detection method, on the other hand, requires separate controls to be read for each sample tested. The negative control used by the individual detection protocol uses lysed samples, which do not require the use/consumption of additional antibodies but do require that each tube be read a second time after addition of the lysing agent. The lysed controls have the added benefit of being able to identify the proportion of positive signal that can be attributed to non-vesicle-related events such as immune complexes 21. The bead-based assay does not have this ability todistinguish between positive signals arising from true EVs and those arising from non-vesicles.
Limitations of the technique
While there is no standardized method for the isolation of EVs, differential centrifugation is a widely used technique among EV researchers. The differential centrifugation method described here is based on common protocols for isolating PPP, which typically require an initial centrifugation between 1,200-1,500 x g for 10-20 min to remove cells, followed by a second centrifugation between 10,000-13,000 x g for 10-30 min to remove platelets 35. The protocol described herein uses a centrifugation at 1,500 x g for 10 min followed by a centrifugation at 13,000 x g for 10 min. While higher forces of 25,000-100,000 x g are typically required to pellet EVs, some of the larger EVs may be removed with the differential centrifugation protocol we have presented.
Up to 90% of EVs detected by FCM are lost with one hour ultracentrifugation at 100,000 x g (data not shown). Longer centrifugation times should be considered, albeit cautiously, as this may adversely impact the sample’s composition. If additional processing is needed for characterization studies, filtration can be performed after the 2-step centrifugation (before staining) to further fractionate samples based on particle size. Similar to ultra-centrifugation, filtration can result in a loss of up to 50% of positive marker events and up to 90% of total particles detected by FCM (data not shown). While the increase in signal-to-noise ratio is of obvious benefit, the loss of smaller EVs represents a significant limitation when considering any washing or isolation method.
Finally, the anticoagulant used (e.g., heparin, ACD, ethylenediaminetetraacetic acid (EDTA), etc.) during blood collection may impact the quality and quantity of EV content. While ACD has proven to be a good and reliable anticoagulant for our studies, testing multiple solutions is recommended to ensure that the most suitable anticoagulant for the application is chosen. This is especially important when EVs will be used in downstream assays where the anticoagulant used can affect the outcome. For example, some anticoagulants (e.g., EDTA and heparin) are known to interfere with PCR reactions while others (e.g., theophylline, adenosine and dipyridamole) have been shown to inhibit EV release from platelets12.
Methods for EV analysis, while considerably improved over the last decade, are still a work in progress. Ultimately, the best methods for analyzing EVs will depend on the research being conducted and tools available to the researcher.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Dale Hirschkorn from Blood Systems Research Institute for his help with flow cytometer instrument settings. This work was supported by NIH grants HL095470 and U01 HL072268 and DoD contracts W81XWH-10-1-0023 and W81XWH-2-0028.
Materials
| LSR II benchtop flow cytometer | BD Biosciences | 3-laser (20 mW Coherent Sapphire 488 nm blue, 25 mW Coherent Vioflame 405 nm violet, and 17 mW JDS Uniphase HeNe 633nm red) | |
| FACS Diva software | BD Biosciences | PC version 6.0 | |
| FlowJo software | Treestar US | Mac version 9.6.1 or PC version 7.6.5 | |
| Sphero Rainbow fluorescent particles | BD Biosciences | 556298 | used to adjust all channel voltages to maintain fluorescence intensity consistency |
| Ultra Rainbow fluorescent particles | Spherotech | URFP-10-5 | used in addition to Megamix-Plus SSC beads to ensure EV gating consistency from batch to batch |
| Megmix-Plus SSC beads | Biocytex | 7803 | used to adjust FSC and SSC voltages to maintain consistency between runs. Can also used to monitor flow rate and ajust flow rate dial in order to ensure that same flow rate is used in all runs |
| AbC Anti-Mouse Bead Kit | Life Technologies | A-10344 | used for compensation controls & negative AbC beads used for beads-based method |
| Nonidet P-40 Alternative (NP-40) (CAS 9016-45-9) | Santa Cruz | sc-281108 | used in the individual detection method only to lyse samples after initial reading for use as negative controls. Stock may be diluted to 1:10 in PBS and stored in fridge for up to 1 month. |
| BD TruCOUNT Tubes | BD Biosciences | 340334 | used whenver absolute EV concentrations are needed |
| Ultrafree-MC, GV 0.22 µm Centrifugal Filter Units | Millipore | UFC30GVNB | used to post-stain wash Evs and/or fractionate EVs based on size |
| Vacutainer glass whole blood tubes ACD-A | BD Biosciences | 364606 | |
| Facs tubes 12×75 polystrene | BD Biosciences | 352058 | |
| 50mL Reservoirs individually wrapped | Phenix | RR-50-1s | |
| Green-Pak pipet tips – 10µL | Rainin | GP-L10S | |
| Green-Pak pipet tips -200µL | Rainin | GP-L250S | |
| Green -Pak pipet tips – 1000µL | Rainin | GP-L1000S | |
| Stable Stack L300 tips presterilized | Rainin | SS-L300S | |
| Pipet-Lite XLS 8 Channel LTS Adjustable Spacer | Rainin | LA8-300XLS | |
| 96 well tissue culture plates | E&K Scientific | EK-20180 | |
| RPMI 1640 Media (without Hepes) | UCSF Cell Culture Facility | CCFAE001 | media used for bead-based detection method |
| Dulbeccos PBS D-PBS, CaMg-free, 0.2µm filtered | UCSF Cell Culture Facility | CCFAL003 | |
| Ultracentrifuge Tube, Thinwall, Ultra-Clear | BECKMAN COULTER INC | 344058 | |
| PANEL I | |||
| CD3 PerCP-Cy5.5 | Biolegend | 344808 | 2 µl |
| CD14 APC-Cy7 | Biolegend | 301820 | 2 µl |
| CD16 V450 | BD Biosciences | 560474 | 2 µl |
| CD28 FITC | biolegend | 302906 | 2 µl |
| CD152 APC | BD Biosciences | 555855 | 2 µl |
| CD19 A700 | Biolegend | 302226 | 2 µl |
| PANEL II | |||
| CD41a PerCP-Cy5.5 | BD Biosciences | 340930 | 2 µl |
| CD62L APC | Biolegend | 304810 | 2 µl |
| CD108 PE | BD Biosciences | 552830 | 2 µl |
| CD235a FITC | biolegend | 349104 | 2 µl |
| PANEL III | |||
| CD11b PE-Cy7 | Biolegend | 301322 | 2 µl |
| CD62p APC | Biolegend | 304910 | 2 µl |
| CD66b PE | Biolegend | 305106 | 2 µl |
| CD15 FITC | exalpha | X1496M | 5 µl |
| CD9 PE | Biolegend | 555372 | |
| CD63 APC | Biolegend | 353008 | |
| APC-Cy7 Ms IgG2a, κ | Biolegend | 400230 |
References
- Andaloussi, S., Mäger, I., Breakefield, X. O., Wood, M. J. A. Extracellular vesicles: biology and emerging therapeutic opportunities. Nature Reviews Drug Discovery. 12 (5), 347-357 (2013).
- Sugawara, A., Nollet, K. E., Yajima, K., Saito, S., Ohto, H. Preventing platelet-derived microparticle formation–and possible side effects-with prestorage leukofiltration of whole blood. Archives of Pathology & Laboratory Medicine. 134 (5), 771-775 (2010).
- Théry, C., Ostrowski, M., Segura, E. Membrane vesicles as conveyors of immune responses. Nature Reviews Immunology. 9 (8), 581-593 (2009).
- Mause, S. F., Weber, C. Microparticles Protagonists of a Novel Communication Network for Intercellular Information Exchange. Circulation Research. 107 (9), 1047-1057 (2010).
- Bouvy, C., Gheldof, D., Chatelain, C., Mullier, F., Dogne, J. -. M. Contributing role of extracellular vesicles on vascular endothelium haemostatic balance in cancer. Journal of Extracellular Vesicles. 3, (2014).
- Hood, J. L., San, R. S., Wickline, S. A. Exosomes Released by Melanoma Cells Prepare Sentinel Lymph Nodes for Tumor Metastasis. Cancer Research. 71 (11), 3792-3801 (2011).
- Gatti, S., Bruno, S., et al. Microvesicles derived from human adult mesenchymal stem cells protect against ischaemia-reperfusion-induced acute and chronic kidney injury. Nephrology Dialysis Transplantation. 26 (5), 1474-1483 (2011).
- Barteneva, N. S., Fasler-Kan, E., et al. Circulating microparticles: square the circle. BMC Cell Biology. 14 (1), 23 (2013).
- Van der Pol, E., Böing, A. N., Harrison, P., Sturk, A., Nieuwland, R. Classification functions, and clinical relevance of extracellular vesicles. Pharmacological Reviews. 64 (3), 676-705 (2012).
- Raposo, G., Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. The Journal of Cell Biology. 200 (4), 373-383 (2013).
- Dinkla, S., Brock, R., Joosten, I., Bosman, G. J. C. G. M. Gateway to understanding microparticles: standardized isolation and identification of plasma membrane-derived vesicles. Nanomedicine (London, England). 8 (10), (2013).
- Witwer, K. W., Buzas, E. I., et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. Journal of Extracellular Vesicles. 2, (2013).
- Shah, M. D., Bergeron, A. L., Dong, J. -. F., López, J. A. Flow cytometric measurement of microparticles: Pitfalls and protocol modifications. Platelets. 19 (5), 365-372 (2008).
- Dey-Hazra, E., Hertel, B., et al. Detection of circulating microparticles by flow cytometry: influence of centrifugation, filtration of buffer, and freezing. Vascular Health and Risk Management. 6, 1125-1133 (2010).
- Lacroix, R., Judicone, C., et al. Standardization of pre-analytical variables in plasma microparticle determination: results of the International Society on Thrombosis and Haemostasis SSC Collaborative workshop. Journal of Thrombosis and Haemostasis. 11 (6), 1190-1193 (2013).
- Mullier, F., Bailly, N., Chatelain, C., Chatelain, B., Dogné, J. -. M. Pre-analytical issues in the measurement of circulating microparticles: current recommendations and pending questions. Journal of Thrombosis and Haemostasis. 11 (4), 693-696 (2013).
- Peramo, P., et al. Physical Characterization of Mouse Deep Vein Thrombosis Derived Microparticles by Differential Filtration with Nanopore Filters. Membranes. 2 (1), 1-15 (2012).
- Tilley, R. E., Holscher, T., Belani, R., Nieva, J., Mackman, N. Tissue Factor Activity is Increased in a Combined Platelet and Microparticle Sample from Cancer Patients. Thrombosis research. 122 (5), 604-609 (2008).
- Rood, I. M., Deegens, J. K. J., et al. Comparison of three methods for isolation of urinary microvesicles to identify biomarkers of nephrotic syndrome. Kidney international. 78 (8), 810-816 (2010).
- Van der Pol, E., Hoekstra, A. G., Sturk, A., Otto, C., van Leeuwen, T. G., Nieuwland, R. Optical and non-optical methods for detection and characterization of microparticles and exosomes. Journal of Thrombosis And Haemostasis: JTH. 8 (12), 2596-2607 (2010).
- György, B., Szabó, T. G., et al. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cellular and Molecular Life Sciences. 68 (16), 2667-2688 (2011).
- Miguet, L., Béchade, G., et al. Proteomic Analysis of Malignant B-Cell Derived Microparticles Reveals CD148 as a Potentially Useful Antigenic Biomarker for Mantle Cell Lymphoma Diagnosis. Journal of Proteome Research. 8 (7), 3346-3354 (2009).
- Smalley, D. M., Root, K. E., Cho, H., Ross, M. M., Ley, K. Proteomic discovery of 21 proteins expressed in human plasma-derived but not platelet-derived microparticles. Thrombosis and Haemostasis. 97 (1), 67-80 (2007).
- Lacroix, R., Robert, S., Poncelet, P., Dignat-George, F. Overcoming Limitations of Microparticle Measurement by Flow Cytometry. Seminars in Thrombosis and Hemostasis. 36 (08), 807-818 (2010).
- Mobarrez, F., Antovic, J., et al. A multicolor flow cytometric assay for measurement of platelet-derived microparticles. Thrombosis Research. 125 (3), e110-e116 (2010).
- Van der Pol, E., van Gemert, M. J. C., Sturk, A., Nieuwland, R., van Leeuwen, T. G. Single vs. swarm detection of microparticles and exosomes by flow cytometry. Journal of Thrombosis And Haemostasis: JTH. 10 (5), 919-930 (2012).
- György, B., Szabó, T. G., et al. Improved Flow Cytometric Assessment Reveals Distinct Microvesicle (Cell-Derived Microparticle) Signatures in Joint Diseases. PLoS ONE. 7 (11), e49726 (2012).
- György, B., Módos, K., et al. Detection and isolation of cell-derived microparticles are compromised by protein complexes resulting from shared biophysical parameters. Blood. 117 (4), e39-e48 (2011).
- György, B., Pasztoi, M., Buzas, E. I. Response: systematic use of Triton lysis as a control for microvesicle labeling. Blood. 119 (9), 2175-2176 (2012).
- Trummer, A., De Rop, C., Tiede, A., Ganser, A., Eisert, R. Isotype controls in phenotyping and quantification of microparticles: a major source of error and how to evade it. Thrombosis Research. 122 (5), 691-700 (2008).
- Shet, A. S., Aras, O., et al. Sickle blood contains tissue factor-positive microparticles derived from endothelial cells and monocytes. Blood. 102 (7), 2678-2683 (2003).
- Connor, D. E., Exner, T., Ma, D. D. F., Joseph, J. E. The majority of circulating platelet-derived microparticles fail to bind annexin V, lack phospholipid-dependent procoagulant activity and demonstrate greater expression of glycoprotein Ib. Thrombosis and Haemostasis. 103 (5), 1044-1052 (2010).
- Nolte-’t Hoen, E. N. M., van der Vlist, E. J., et al. Quantitative and qualitative flow cytometric analysis of nanosized cell-derived membrane vesicles. Nanomedicine: Nanotechnology, Biology, And Medicine. 8 (5), 712-720 (2012).
- Van der Vlist, E. J., Nolte-’t Hoen, E. N. M., Stoorvogel, W., Arkesteijn, G. J. A., Wauben, M. H. M. Fluorescent labeling of nano-sized vesicles released by cells and subsequent quantitative and qualitative analysis by high-resolution flow cytometry. Nature Protocols. 7 (7), 1311-1326 (2012).
- Orozco, A. F., Lewis, D. E. Flow cytometric analysis of circulating microparticles in plasma. Cytometry Part A. 77 A. 77A (6), 502-514 (2010).

