Summary
To investigate simple fabrication approaches for multiple assay needs, we created a fluid-absorbing channel system made of cotton material. This device was used to establish a multiple detection platform, and solve contamination issues that commonly affect lateral flow-based biomedical devices, for clinical urinalysis of nitrite, total protein, and urobilinogen.
Abstract
A robust, low-cost analytical device should be user-friendly, rapid, and affordable. Such devices should also be able to operate with scarce samples and provide information for follow-up treatment. Here, we demonstrate the development of a cotton-based urinalysis (i.e., nitrite, total protein, and urobilinogen assays) analytical device that employs a lateral flow-based format, and is inexpensive, easily fabricated, rapid, and can be used to conduct multiple tests without cross-contamination worries. Cotton is composed of cellulose fibers with natural absorptive properties that can be leveraged for flow-based analysis. The simple but elegant fabrication process of our cotton-based analytical device is described in this study. The arrangement of the cotton structure and test pad takes advantage of the hydrophobicity and absorptive strength of each material. Because of these physical characteristics, colorimetric results can persistently adhere to the test pad. This device enables physicians to receive clinical information in a timely manner and shows great potential as a tool for early intervention.
Introduction
The development of point-of-care (POC) diagnostic devices that are affordable, robust, and easily used is imperative for improving global health1,2. In particular, devices composed of cellulose substrates (e.g., paper, thread, and cotton) provide promising analytical platforms for low-cost analysis because of their ubiquity, affordability, ease of use, robustness, and capacity to provide rapid results3-7.
Here, we unveil the development of a cotton-based analytical device that uses a lateral flow-based format for urinalysis. This cotton-based analytical device provides an alternative detection approach with several key advantages: i) fabrication with minimal human effort; ii) low cost; iii) the capacity to be used to conduct multiple, different assays without cross-contamination concerns; iv) device independence, i.e., the ability to be run without additional equipment and/or electricity; and, v) speed (colorimetric assays can be completed within 10 min).
The structure of this cotton-based analytical device can be divided into four parts: i) cotton that is naturally hydrophobic on its external hydrophobic layer; ii) cotton that is internally hydrophilic and serves as a transportation channel for liquid wicking; iii) lamination film that binds and compresses the cotton being used but contains drilled out holes for the placement of reaction/test pads; and, iv) chromatography paper test pads, which are coated/embedded with reactive reagents, placed on the exterior surface of the cotton (specifically, in the space drilled out of the lamination film) as reaction areas for colorimetric assays (i.e., nitrite, total protein, pH, and urobilinogen assays) and results display.
The underlying mechanism of the test is as follows. The cotton-based analytical device is scored with lines that penetrate half way through the depth of the cotton material to create a flow channel that allows sample fluid to reach the reactive pads being used. The absorptive edge of the analytical device is immersed into the target sample, whereupon the solution is wicked along the fluidic channel from the absorption end to the test pads (Figure 1). Because the absorptive strength of the test pad is greater than that of cotton, solutions absorbed by the test pads are firmly contained inside the test pad paper so that there is no reflow back into the fluidic channel, and the colorimetric results become subsequently fixed on the test pad material. At the end of the reaction, colorimetric results are recorded via a desktop scanner, and analyzed via image analyzing software.
Protocol
CAUTION: Proper laboratory hygiene practice is required. Gloves and universal precautions are required when using this POC device. Contamination of results or infection may occur if adequate sterilization procedures are not carried out properly.
1. Prepare Test Strip Devices
- Determine the hydrophobicity of the exterior layer of the cleansing cotton by contact angle measurement8 (Figure 3).
- Fabricate the cotton-based analytical device by cutting cleansing cotton into 5.5 cm x 1 cm pieces with a paper cutter (Figure 2A).
- Drill holes (ø = 0.5 cm) in a standard sheet of lamination film in rows of three; the distance between two subsequent holes is 1 cm.
NOTE: As many as 36 individual test strips with three test areas each may be made using a standard sheet of lamination film (5.5 cm x 2 cm) (Figure 2B). - Sandwich the cleansing cotton (characterized by exterior hydrophobicity and interior hydrophilicity) between two pieces of lamination film, i.e., the hole-drilled sheet of lamination film on one side, and an undrilled sheet of lamination film (no holes) on the other side. Place the assembly into a laminator to package the device. This enhances the device's mechanical strength, and provides an external, impervious layer.
NOTE: The diameters of holes will shrink from 0.5 cm to ~ 0.47 cm after lamination. Some slight gaps will remain between the cleansing cotton and lamination film around the perimeter of each hole. These slight gaps facilitate the placement of test pad circles made in a subsequent step. - Use a pair of scissor to cut individual 5.5 cm x 1 cm test strip devices from the assembly, i.e., the drilled lamination sheet/cotton pad/undrilled lamination sheet "sandwich" (Figure 2C). Make 48 devices total to create standard curves as described later (18 devices for the nitrite assay, 15 devices for the BSA assay, and 15 devices for the urobilinogen assay).
- Use a sharp knife or razor to cut a slit (as a small fluidic channel) from the center of each test area (not covered by lamination film), across and through the hydrophobic layer of test area to the hydrophilic layer of the longitudinal device, being cautious to cut halfway through the depth of the sandwiched device (Figure 2D).
- Apply paper test pads (ø = 0.5 cm) by sliding them into the gaps at the perimeter of each hole in the lamination (Figure 2E).
2. Prepare Standards and Indicator Solutions for Urinalysis
- Prepare 10.0 mM nitrite stock solution by dissolving 69.0 mg sodium nitrite in 100 ml deionized (DI) water. Dilute the nitrite stock solution in DI water to create a series of nitrite concentration standard solutions (2,500, 1,250, 625, 312, 156, and 78 µM) (Figure 4A).
- Prepare 60 µM bovine serum albumin (BSA) stock solution by dissolving 415 mg BSA in 100 ml PBS solution (10 M phosphate buffer, 0.0027 M potassium chloride and 0.137 M sodium chloride; pH 7.0). Dilute the BSA stock solution in DI water to create a series of BSA concentration standard solutions (30, 15, 7.5, 3.75, and 1.875 µM) (Figure 4B).
- Prepare 25 g/L urobilinogen stock solution by dissolving 2.5 g urobilinogen in 100 ml DI water. Dilute stock urobilinogen solution in DI water to create a series of urobilinogen concentration standard solutions (7.8, 15.6, 31.2, 62.5, and 125 mg/L) (Figure 4C).
- Prepare nitrite indicator solution containing 50 mM sulfanilamide, 330 mM citric acid, and 10 mM N-(1-naphthyl) ethylenediamine dihydrochloride.
- Prepare BSA indicator solution containing 250 mM citrate buffer solution (pH 1.8) and 3.3 mM solution of tetrabromophenol blue in 95% ethanol.
- Prepare urobilinogen indicator solution containing 0.1 M 4-Dimethylamine benzaldehyde.
3. Prepare Indicator Pads
- Cut three circular shaped pads or disks (ø = 0.5 cm) from chromatography paper and insert each of them (one for each assay on the three-assay strip) onto each test strip device to create a solution standard curve (section 4).
- At each of the three lamination film holes of the assembly, observe a slight gap between the lamination film and the cotton test pad at the very edges of the cut hole. Note, the hole in the lamination film is slightly smaller in diameter than the paper test pad.
NOTE: Together, these features allow the paper test pad to be applied against the cotton test pad portion of the device so that the edges of the paper test pad slip into the slim gap mentioned, i.e., under the edges of the lamination film hole. This holds the paper test pads in place. - Prepare the Nitrite Assay Portion of the Device.
- Coat the nitrite assay pad with 4 µl of nitrite indicator solution (prepared in step 2.4) and allow it to dry completely at ambient temperature.
- Prepare the Total Protein Assay Portion of the Detection Device.
- Coat the total protein assay pad with 4 µl of BSA indicator solution (prepared in step 2.5) and allow it to dry completely at ambient temperature.
- Prepare the Uroblinogen Assay Portion of the Detection Device.
- Coat the uroblinogen assay pad with 4 µl of uroblinogen indicator (prepared in step 2.6) and allow it to dry completely at ambient temperature.
4. Create Standard Curves for the Cotton-based Analytical Device
- For each standard prepared, dip the absorptive end of a prepared device (preloaded with assay indicators (see section 3.4)) into the standard solution for 10 sec (the absorption volume ~ 200 – 300 µl) (Figure 2F).
- Place the cotton-based device in an ambient environment for 10 min to complete colorimetric reactions (Figure 2F). Repeat each experiment in triplicate.
- Scan the device to analyze the colorimetric change occurring at each test pad (Figure 2G). Place resulting test pads on the glass of a desktop scanner and scan test pads in RGB mode with 600 dpi of image resolution. Save each image in jpeg format.
- Analyze the scanned image on a computer using image analysis software to determine respective color intensities (Figure 2H).
- Download ImageJ software at http://imagej.nih.gov/ij/. Open the scanned image files. Select the Image in the menu commands, and select “Type” to convert RGB images to 8-bit grayscale.
- Use the "Oval" tool in the toolbar to select the test pad analytical area. Select "Analyze" in the menu commands, and select "Measure" to analyze color intensity. Record mean intensity results.
- Establish the standard curves of nitrite, total protein, and urobilinogen assays (Figure 2I).
- Indicate different concentrations of standard nitrite, total protein, and urobilinogen solutions and correlated mean intensity results on a two-dimensional coordinate system using stastical software.
- Fit the standard curve for a nitrite assay by a quadratic model and the standard curves for total protein and urobilinogen assays by linear regression models.
5. Determine Unknown Sample Concentrations Using Established Standard Curve Values
- To determine nitrite, total protein, and urobilinogen concentrations in unknown solutions, use the device as described for measuring standard solution concentrations and substitute measured intensity results for nitrite, total protein, and urobilinogen assays into the equations of the models built in step 2.
Representative Results
We successfully demonstrated the development of cotton-based analytical devices by using commercially available cleansing cotton characterized by hydrophilic (inner portion) and hydrophobic (exterior portion) properties (Figure 1A). Figure 3 shows the results of contact angle measurement. The hydrophobic interface of exterior cotton was 127.35° ± 4.73°. From a user-friendly perspective, colorimetric assays employed here could be directly observed by the naked eye, and further quantified through color intensity analyses (Figure 2F). Urine total protein in a normal individual should be less than 150 mg/dl (4 µM). The value of urine nitrite ions is associated with urinary tract infections or bacterial infections, so the analytical sensitivity of urine nitrite detection should be as low as possible when it comes to the development of analytical devices for a nitrite assay. Urobilinogen is excreted by the feces and recovered by the enterohepatic circulation, but some levels of urobilinogen, approximately 1 – 4 mg/daily, can be found in urine. Figure 4A–C illustrates the results from our efforts to create standard curves for each assay target. These standard curves were established by examining mean colorimetric intensity results and comparing them to the concentrations of standard concentrations that we established. This allowed us to evaluate the limit of detections (LODs) for each assay. The LODs for nitrite, BSA, and urobilinogen in buffer systems were, 0.147 mM, 3.672 µM, and 4.861 mg/L, respectively. This also provided us with a mathematical framework for determining the concentrations of nitrite, BSA, and urobilinogen in unknown solutions.
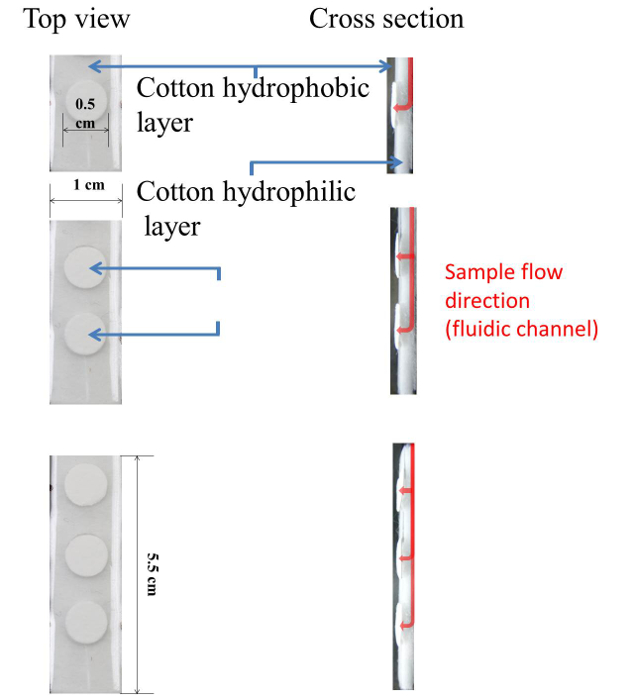
Figure 1. Schematic Diagram of Cotton-based Analytical Device. Top and cross-section view images display the dimensions of each test pad (0.5 cm diameter) and cotton-based device (1 cm width x 5.5 cm length). Please click here to view a larger version of this figure.
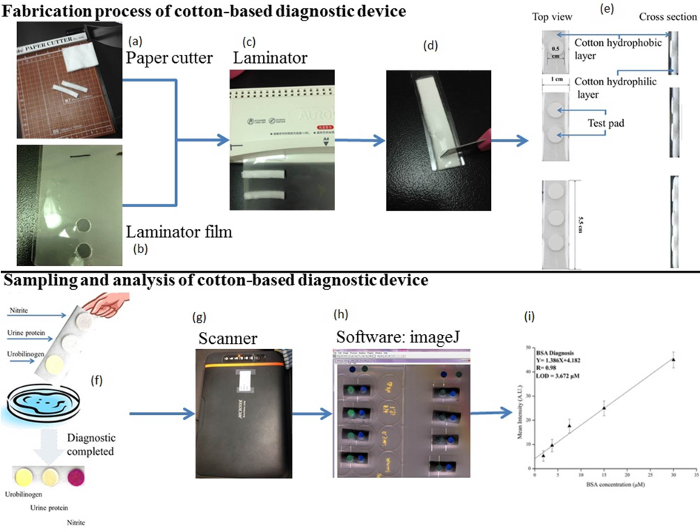
Figure 2. Fabrication Process of Cotton-based Analytical Device, Sampling, and Results Analysis. (A) A paper cutter was used to cut pieces of cleansing cotton to the specified size. (B) A pen was used to mark the locations for lamination film holes in the assembly. (C) A laminator was used to sandwich the assembly between two layers of laminating film by heating. (D) The laminated pieces of the completed device were separately cut. (E) The test pads were installed onto the cotton device. (F) Urobilinogen, BSA, and nitrite assays were performed with our cotton-based analytical device. (G) Images of the results were made and imported into a computer using a scanner. (H) Image analysis software was used to analyze the images by gray-scale contrast state analysis. (I) The obtained intensity results were fitted into a standard curve. Please click here to view a larger version of this figure.
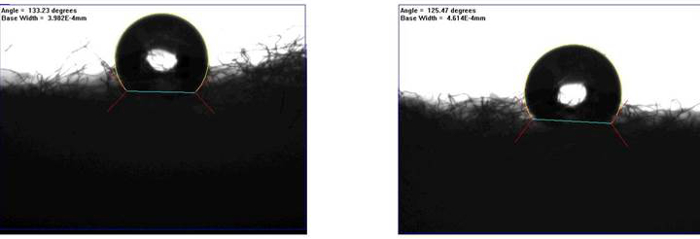
Figure 3. Contact Angle Results for the Cleansing Cotton. DI water (1 µl) was dropped onto the cotton's external, hydrophobic layer to determine contact angle.
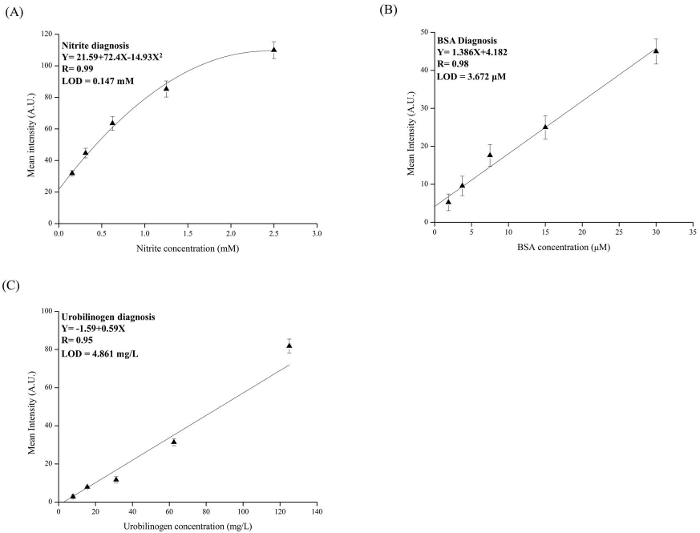
Figure 4. Analysis Results for Nitrite, Urobilinogen, and BSA on Cotton-based Analytical Device. The standard curves for (A) nitrite (0.156-2.5 mM), (B) BSA (1.875-30 µM), (C) urobilinogen (7.8 – 125 µM). For each concentration, tests were performed in triplicate and the R2 values for the fitting curve were nitrite: 0.99, BSA: 0.98, and urobilinogen: 0.95. Please click here to view a larger version of this figure.
Discussion
Critical steps in this protocol included determining the appropriate combination of cotton material (with varying hydrophobicity/hydrophilicity) and filter paper (chromatography filter paper or quantitative filter paper). A well-planned and executed device design renders the best performance attributes for colorimetric assays. From our colorimetric assay results, the cotton-based analytical device presented herein demonstrates great potential as a platform for multiple disease detection.
Most current lateral flow-base products (e.g., pregnancy test) provide no more than a single assay functon14. While some dipstick tests perform multiple-biomarker assays15, their use runs the risk of collateral sample contamination because assay reagents directly contact test samples. Our device overcomes this obstacle and can be used to implement multiple colorimetric assays in a single lateral flow-based channel device.
This method is only limited by target analyte molecular size. The R 2 and LOD values of nitrite are much greater than those of the other assays (BSA and urobilinogen), suggesting that our devices are more suitable for low molecular analysis5 (Figure 4).
Some further optimization may still be required in order to enhance the practicality of this device for clinical practice. This optimization includes improving the reliability and stability of assay reagents when exposed to the outside environment for long periods of time. Nevertheless, we believe that this invention creates a decisive and valuable inroad into the development of low-cost analytical devices that can be reliably used in clinical practice, especially in regards to fulfilling needs for rapid and accurate analysis and suitable follow-up analysis of communicable and infectious diseases.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by grants from Taiwan's Ministry of Science and Technology (MOST 104-2628-E-007-001-MY3 (CMC)), and Taichung Veterans General Hospital (TCVGH-1056904C (MYH)).
Materials
| bovine serum albumin | Sigma-Aldrich, US | No. 9048468 | ≥ 99% |
| nitrite | Sigma-Aldrich, US | No. 7632000 | ≥ 99% |
| urobilinogen | Santa Cruz Bio, US | No. SC-296690 | |
| citrate | Sigma-Aldrich U.S | No. 6132043 | ≥ 99% |
| tetrabromophenol blue | Sigma-Aldrich U.S | No. 4430255 | ≥ 99% |
| sulfanilamide | Sigma-Aldrich U.S | No. 63741 | ≥ 99% |
| citric acid | Sigma-Aldrich U.S | No. 77929 | ≥ 99.5% |
| N-(1-naphthyl) ethylenediamine dihydrochloride | Sigma-Aldrich U.S | No. 1465254 | ≥ 98% |
| 4-(Dimethylamine)benzaldehyde | AlfaAesar, U.S | No. A11712 | ≥ 98% |
| Methyl Red sodium salt | sigma, U.S | No. 114502 | ≥95% |
| Bromothyle blue | sigma, U.S | No. 114413 | ≥95% |
| Shiseido Cleansing Cotton | Shiseido, Japan | No. 79014 | |
| chromatography paper | GE Healthcare Whatman, Springfield Mill, UK | No. 30306132 | |
| plastic substrate | lamination film, MAS | A4 | 216 mm × 303 mm |
| scanner | microtek scanmaker | i2400 | |
| paper cutter | Life paper cutter | No.306 | |
| laminator | AURORA | LM4231H | |
| laminator film | UNI LAMI | 4A |
References
- Yager, P., Domingo, G. J., Gerdes, J. Point-of-care diagnostics for global health. Annu. Rev. Biomed. Eng. 10, 107-144 (2008).
- Chin, C. D., Linder, V., Sia, S. K. Commercialization of microfluidic point-of-care diagnostic devices. Lab Chip. 12, 2118-2134 (2012).
- Lu, Y., Shi, W., Jiang, L., Qin, J., Lin, B. Rapid prototyping of paper-based microfluidics with wax for low-cost, portable bioassay. Electrophoresis. 30 (9), 1497-1500 (2009).
- Ballerini, D. R., Li, X., Shen, W. Flow control concepts for thread-based microfluidic devices. Biomicrofluidics. 5 (1), 014105 (2011).
- Lin, S., et al. Cotton-based Diagnostic Devices. Scientific reports. 4, 6976-6976 (2013).
- Hsu, M. Y., et al. Monitoring the VEGF level in aqueous humor of patients with ophthalmologically relevant diseases via ultrahigh sensitive paper-based ELISA. Biomaterials. 35 (12), 3729-3735 (2014).
- Hsu, M. Y., et al. Monitoring VEGF levels with low-volume sampling in major vision-threatening diseases: age-related macular degeneration and diabetic retinopathy. Lab Chip. 15 (11), 2357-2363 (2015).
- Kwok, D., Neumann, A. Contact angle measurement and contact angle interpretation. Adv. Colloid Interface Sci. 81 (3), 167-249 (1999).
- Martinez, A. W., Phillips, S. T., Butte, M. J., Whitesides, G. M. Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew. Chem. Int. Ed. Engl. 46 (8), 1318-1320 (2007).
- Li, X., Tian, J. F., Shen, W. Quantitative biomarker assay with microfluidic paper-based analytical devices. Anal. Bioanal. Chem. 396 (1), 495-501 (2010).
- Coad, S., Friedman, B., Geoffrion, R. Understanding Urinalysis. medscape. 7 (3), 269-279 (2012).
- Kupka, T., et al. Accuracy of urine urobilinogen and bilirubin assays in predicting liver function test abnormalities. Ann. Emerg. Med. 16 (11), 1231-1235 (1987).
- Binder, L., et al. Failure of prediction of liver function test abnormalities with the urine urobilinogen and urine bilirubin assays. Arch. Pathol. Lab. Med. 113 (1), 73-76 (1989).
- Gubala, V., et al. Point of care diagnostics: status and future. Anal. chem. 84 (2), 487-515 (2011).
- Vaidya, V. S., et al. Urinary biomarkers for sensitive and specific detection of acute kidney injury in humans. Clin. Transl. Sci. 1 (3), 200-208 (2008).

