Light Spot-Based Assay for Analysis of Drosophila Larval Phototaxis
Summary
This protocol introduces a light-spot assay to investigate Drosophila larval phototactic behavior. In this assay, a light spot is generated as light stimulation, and the process of larval light avoidance is recorded by an infrared light-based imaging system.
Abstract
The larvae of Drosophila melanogaster show obvious light-avoiding behavior during the foraging stage. Drosophila larval phototaxis can be used as a model to study animal avoidance behavior. This protocol introduces a light-spot assay to investigate larval phototactic behavior. The experimental set-up includes two main parts: a visual stimulation system that generates the light spot, and an infrared light-based imaging system that records the process of larval light avoidance. This assay allows tracking of the behavior of larva before entering, during encountering, and after leaving the light spot. Details of larval movement including deceleration, pause, head casting, and turning can be captured and analyzed using this method.
Introduction
The larvae of Drosophila melanogaster show obvious light-avoiding behavior during the foraging stage. Drosophila larval phototaxis has been under investigation, especially in the past 50 years1,2,3,4,5,6,7,8. In recent years, despite the fact that 1) many neurons mediating larval light avoidance have been identified4,5,9,10,11,12 and 2) the complete connectome of larval visual system at the resolution of synapses has been established13, the neural mechanisms underlying larval phototaxis remain largely unclear.
A number of behavioral assays have been used in studying larval phototaxis. They can be largely divided into two classes: one involving spatial light gradients and the other involving temporal light gradients. For spatial light gradient assays, the arena is divided into equal number of sections in light and dark. The arena can be divided into light and dark halves2,4 or light and dark quadrants14,15, or can even be separated into alternate light and dark squares like on a checkerboard7. Usually, agar plates are used for spatial light gradient assay, but tubes that are divided into alternate light and dark sections can also be used10,14.
In older version of assays, light illumination generally originates from below the larvae. However, illumination in newer versions largely originates from above, since larval eyes (e.g., the Bolwig's organs that are sensitive to low or medium light intensities16) are contained in the opaque cephalopharyngeal skeleton with openings towards the upper front. This makes larvae more sensitive to light from upper front directions than from below behind directions7. For temporal light gradient assays, the light intensity is spatially uniform in the arena, but the intensity changes over time. In addition to temporal square wave light (i.e., flashing on/off or strong/weak light3,7), temporally varying light that conforms to a linear ramp in intensity is also used8 to measure the sensitivity of larvae to a temporally changing light stimulus.
A third type of phototaxis assay is the directional light scape navigation, which involves illumination from above at an angle of 45°7. Before the work of Kane et al.7, only coarse parameters such as the number of larvae in light and dark regions, frequency of turning, and trail length were calculated in larval phototaxis assays. Since the work of this same group, with the analysis of high temporal resolution video record for larval phototaxis, detailed dynamics of larval movement during phototaxis (i.e., instant speeds of different parts of larval body, heading direction, turning angle and corresponding angular velocity) have been analyzed7. Thus, more details of larval phototaxis behavior have been able to be discovered. In these assays, larvae are tested in groups so that group effects are not excluded.
This protocol introduces a light-spot assay for the investigation of larval behavioral responses to individual light stimulation. The main experimental set-up consists of a visual stimulation system and infrared light-based imaging system. In the visual stimulation system, an LED light source generates a round 2 cm-diameter light spot on an agar plate, where the larva is tested. The light intensity can be adjusted using an LED driver. The imaging system includes an infrared camera that captures the behavior of the larva in addition to three 850 nm infrared LEDs that provide illumination for the camera. The lens of the camera is covered by an 850 nm band-pass filter to block light from the visual stimulation system from entering the camera, while the infrared light is allowed to enter the camera. Thus, interference of visual stimulation on imaging is prevented. In this assay, the behavioral details of the fast responses of individual larvae within a period including before, during, and after entering light are recorded and analyzed.
Protocol
1. Preparation of Drosophila larvae
- Prepare standard medium consisting of boiled corn meal (73 g), agar (5.6 g), soybean meal (10 g), yeast (17.3 g), syrup (76 mL) and water (1000 mL).
- Raise all flies at 25 °C on standard medium in a room with a 12 h/12 h light/dark cycle.
2. Preparation of agar plates
- Prepare 1.0% agar solution. Weigh 3 g of agar in a 500 mL beaker with a balance, then add 300 mL of distilled water. Place a foil paper over the breaker to prevent the water from evaporating. Heat the beaker in a microwave to boil.
- Take out the beaker and stir well with a glass rod, then heat in a microwave oven to boil. Repeat until the liquid is completely transparent and fluid.
NOTE: The final hot agar solution should be free of air bubbles; otherwise, pouring the agar into the plate will result in uneven and dented surface of the agar plate, which will affect subsequent larval behavior testing. The concentration of the agar solution should not be too high or low. If the concentration is too high, the light diffuse reflection on the agar plate will be strong and smear the light/dark boundary. If the concentration is too low, the larvae will leave traces on the plate. Both of these errors will hinder video processing later in the protcol. - Slowly pour the hot agar solution into a Petri dish (diameter 15 cm) until the bottom is evenly covered with a layer of agar (~4 mm in thickness), and cool at room temperature (RT) until the agar solution is solidified.
- Agar plate should be used when it is freshly prepared. If it is not, pour a layer of water onto the surface and store it in the refrigerator at 4 °C for later use.
3. Set-up of visual stimulation system
- Select the LED light source: collimated LED blue light at 470 nm or warm white light.
NOTE: The light source can be replaced with LED light of any other wavelength. In this experiment, blue light LED is used as an example. - Roll a thick piece of aluminum foil or black cardboard (ensure opacity) to form an open cylinder of 12 cm in length with a diameter similar to that of the blue-light LED with a diameter of 3 cm. Let the top end of the cylinder cover the front end of the blue-light LED. Cover the bottom end with a black cardboard with a small round hole (0.5 cm diameter) in the center. This constitutes the light source system set-up.
- Fix the prepared light source onto the iron frame with a clip, making sure the LED light projects down towards the desktop. Tilt the cylinder slightly. The angle between the cylinder plane and vertical plane is about 10° (see Figure 1). Connect the 470 nm blue LED light to the "LED1" plug of the high-power LED driver. Turn on the driver, turn the knob in the upper right corner of the driver to select channel 470 nm, then click LED. Then, when the screen displays "√", a blue light spot will appear on the desktop.
NOTE: If the cylinder leaks light in addition to the small round hole, it is recommended to use black tape on the leaking parts to make sure that only the hole can pass light through. - Click OK and turn the knob to adjust the intensity of the light. Rotate the knob to a higher light intensity of 50 mA. Measure and record the spectrum of the light with a spectrometer.
- Move the position of the light source up and down to adjust the diameter of the light spot to 2 cm. The desktop should be black for a better contrast effect.
- Rotate the knob to choose the light intensity according to experimental needs. Use a compact power meter console with a standard photodiode power sensor to measure the maximal and minimal light power in the spot, record it, measure three times, and take the average value.
NOTE: It is recommended to use a photodiode power sensor to measure the light intensity for light of a specific wavelength and use a thermal power sensor to measure the light intensity for white light. - Calculate the light intensity in the light spot by dividing the measured light power by the area of the sensor.
NOTE: For example, if the measured light power in step 2.6 is 20 pW and area of the sensor is 0.81 mm2, the light intensity is 24.69 pW/mm2 (dividing 20 pW by 0.81 mm2).
4. Set-up of the imaging system
- Clamp a high-resolution web camera with an iron clip, at about 10 cm above the light spot on the desktop (Figure 1).
- Adjust the orientation of the camera lens towards the desktop. Connect the camera to a computer through a USB interface.
- Place an agar plate on the desktop right beneath the camera.
- Open the "Amcap9.22" software on the computer with Windows 7, and the light spot will be automatically shown in the window of AMcap. Move the camera slightly left or right to ensure that the light spot is near the center of the window. Ensure that the camera does not block the light path. The light spot should be complete and round.
NOTE: The software can be found at http://amcap.en.softonic.com/. - Fix an 850 nm ± 3 nm band-pass filter with a clip at 5-7 mm right below the camera.
NOTE: The diameter of the filter is about 2.5 cm, and the camera lens is less than 1 cm in diameter, so the filter can cover the visual field of the camera. With the filter below the camera, the light spot should not be seen in the window of AMcap. - Place three infrared-light-generating LEDs (central wavelength = 850 nm) evenly around the agar plate. Each LED should be about 5 cm away from the edge of the agar plate, and the lens face of the LED should be at a 70° downwards angle towards the agar plate. Connect the LEDs to the power through the AC-to-DC converter.
NOTE: It is better to fix the positions and angles of the infrared light LEDs to ensure consistency of the brightness of the field in various experimental trials and facilitate later video processing. - Put a black board between the computer and the device. Set down the brightness of the computer screen to prevent the computer screen light from affecting the experiment.
NOTE: Keep the environment dark when measuring wavelength or intensity of the light.
5. Setting parameters of imaging
- On the menu of the AMcap software, choose Options | Video Device | Capture format, and set the pixel size of the captured video to 800 x 600 and frame rate to 60 fps.
- Remove the filter from beneath the camera, put a ruler under the camera and adjust the camera focus to make the scale line clear and parallel to the width of the video field of view.
- Click Capture | Set up | Video capture to select the save path, click Start recording, record the actual distance corresponding to 600 pixels, and calculate the ratio of each pixel to the actual distance.
6. Video recording of light avoidance behavior
- Maintain a temperature of 25.5 °C through all experiments. Control room temperature with an air conditioner if required. Keep the humidity constantly at 60% with a humidifier.
- Take a short video of the light spot position named "lightarea1". Then, move the 850 nm ± 3 nm filter back to cover the camera lens.
NOTE: When recording larval behavior, the camera lens is covered by the 850 nm ± 3 nm filter so that the light spot is not shown in the video. The light spot can be reconstructed in videos with larvae later with Matlab. Do not change the position of the camera, and avoid changing the ratio of each pixel to the actual distance measured in step 4.3. - Turn on a light (i.e., a room light) far away from the experimental device. Turn down the light as low as possible, as long as the larvae can be clearly seen with the eyes. Take the larvae out of the culture medium with a spoon, gently pick a third-instar larva, and wash it clean with distilled water. Be careful to wash larva one at a time to avoid interference from hunger. A single experiment requires at least 20 larvae.
- Transfer the larva to the center of the agar plate placed beneath the camera during step 3.3. Gently remove excess water from the larva with a brush or use blotting paper to remove water from the larva to prevent reflection of light under the lens. Turn off the room light and allow the larva to acclimate for 2 min in the dark environment.
- Turn on the LED light to generate infrared light, and gently brush the larva to the center of the plate. When the larva begins to crawl straight, rotate the plate to make the larva head towards the light spot. Make sure that it crawls straight from the start, or else it may not obtain access to the light spot.
- Click Capture | Set up | Video capture to select the save path, then click Start recording to record. Allow the larva to crawl towards the light spot, enter the light spot, then leave the light spot until it is nearly out of the field of view. Click Stop recording. If the larva turns away from the light spot before getting close, directly click Stop recording.
- Move the filter away from the camera. Take a short video of the position of the light spot named "lightarea2" and compare it to "lightarea1" to ensure that the light spot position is not changed. If an obvious position change is observed, discard the data.
7. Data analysis
- Use SOS17 to extract animal body contour and movement parameters from videos using image processing methods as previously described17.
NOTE: Parameters including headSpeed (velocity of larval head), tailSpeed (velocity of larval tail), midSpeed (velocity of larval midpoint of skeleton line), and cmSpeed (velocity of larval centroid) were used to measure larval movement speed. Parameters including headTheta (the angle between the lines of head-midpoint and midpoint-tail) and headOmega (the changing speed of headTheta) were used to measure larval body bending and the angular speed of bending.
Representative Results
According to the protocol, the light spot assay was used to investigate light avoidance behavior of third instar larva that were raised at 25 °C on standard medium in a room with a 12 h/12 h light/dark cycle. A single w1118 larva was tested using the light spot assay at 25.5 °C. The average light intensity of the light spot generated by a 460 nm LED was 0.59 µW/cm2. The whole process of larval entering and exiting the light spot was recorded and analyzed using SOS software and custom written scripts12,17. Time curves of tail speed, body bending angle, and angular speed of body bending of a representative larva are shown in Figure 2 and Movie 1.
To investigate the effects of octopaminergic neurons on larval light avoidance, third instar larvae with octopaminergic neurons inhibited by expressing tetanus toxin (UAS-TNTG) with a Tdc2-Gal4 driver were tested with the light-spot assay. As shown in Figure 3, the size of the larval head cast (maximal body bending angle) was significantly reduced compared to the parental controls, indicating that the Tdc2-Gal4 neurons are necessary for a normal larval light response.
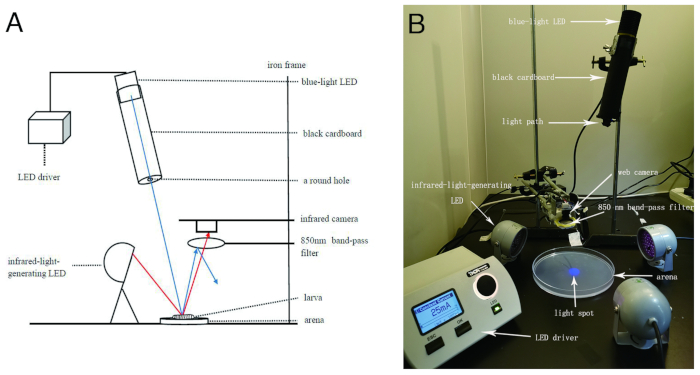
Figure 1: Experimental set-up. (A) Schematic representation of the set-up for the light spot-based larval fast phototaxis assay. The blue lines represent the paths of visible light used as visual stimulation, and the red lines represent the paths of infrared light. Arrows indicate the direction of the light. The 850 nm band-pass filter allows infrared light to pass, but it blocks visible light. (B) An image of the set-up for the light spot assay. It should be noted that the image was taken under light conditions for better visualization. Please click here to view a larger version of this figure.
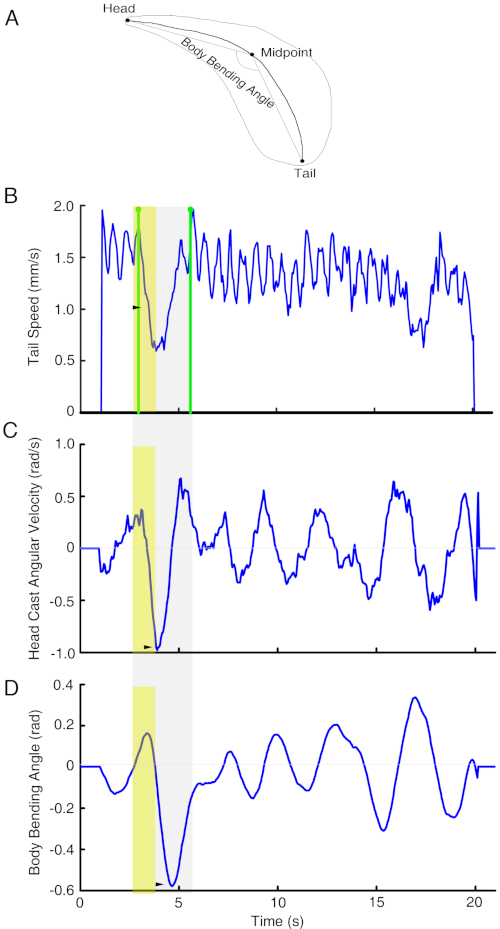
Figure 2: Quantitative description of the reaction of a larva when entering a light spot. (A) A diagram showing the parameters used in measuring larval body movement. The contour of a larva is shown in thin line. The thick line shows the skeleton of thinned larval body contour. The two ends and midpoint of skeleton line are assigned as positions of larval head, midpoint and tail. The angle between the line from the head to midpoint and the line from midpoint to tail is the body bending angle. The speed of the change of body bending angle over time is defined as angular velocity of larval head cast. Represented here are tail speed (B, tailspeed), head cast angular velocity (C, headomega), and body bending angle (D, headtheta) of a w1118 larva that enters and leaves a light spot. Green lines mark the timepoint that the larval head entered and left the light spot. The time window of a strong deceleration period is in yellow. Arrow heads point to deceleration periods and related peaks in head cast angular velocity and body bending angle. The behavioral process is shown in Movie 1. This figure has been modified from Gong et al.12. Please click here to view a larger version of this figure.
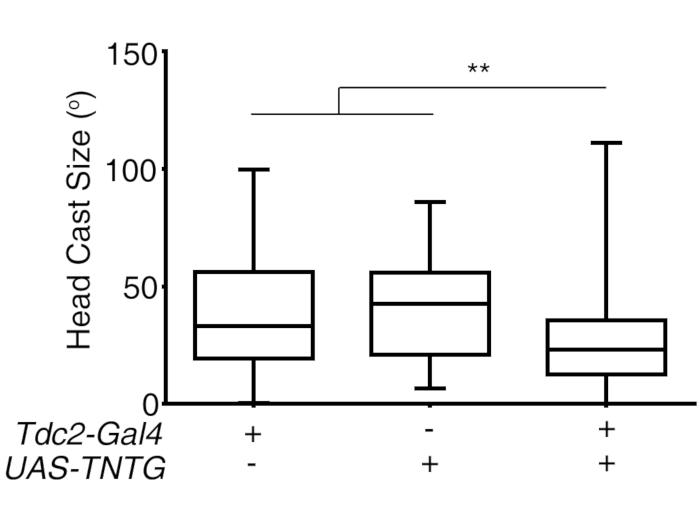
Figure 3: Inhibiting Tdc2-Gal4 labeled neurons using tetanus toxin TNTG reduces the size of larval head cast in response to light spot entrance. **, P < 0.01, n = 81, 52, 92; Kruskal-Wallis test followed by post hoc Dunn's multiple comparison test was used. This figure has been modified from Gong et al.12. Please click here to view a larger version of this figure.

Movie 1: A w1118 larva enters and leaves a light spot in the light spot assay. Light spot with edge smoothed is in white. The track of larval head is shown. Corresponding curves of larval tailspeed, headtheta, and headomega are played simultaneously. This movie has been modified from Gong et al.12. Please click here to view this video. (Right-click to download.)
Discussion
This protocol presents the light spot assay to test the ability of Drosophila larvae to escape from light. This assay allows tracking of the behavior of larvae before entering, during encountering, and after leaving a light spot. Details of larval movement can be captured and analyzed. The light spot assay is very simple and possesses strong practicability. The cost of the whole device is not high. In the experiment, LED light is used as the light source. It can be replaced with light sources of different wavelengths, if required. The light intensity can also be adjusted by the LED drive. The lowest light intensity in the spot can reach 1.80 pW/mm2 (cold white light). Even at such a low light intensity, larvae still can sense the light and show light-avoiding behaviors11.
It should be noted that the concentration of the agar plate is controlled between 0.8% and 1.0%. If the concentration is too high, scattering of light on the agar plate can be serious, and the size of light spot recognized in the video is exaggerated. Therefore, the brightness of the spot should not be too high. Since larvae in a light spot are hardly visible, if visible light is used for illumination, it is necessary to use infrared light to illuminate the larva and add an 850 nm band-pass filter on the camera to prevent the light spot signals from entering the camera. The video of larval response to light spots can be synthesized later based on the larva-only and light-spot-only videos.
The light spot assay possesses three main virtues: 1) the process of larval light avoidance can be monitored and analyzed in detail; 2) larval light response is tested only once, so that the possible involvement of light adaptation can be excluded; and 3) possible effects on light response from other larvae can be excluded. One obvious disadvantage of this assay is that it is low throughput, since only one larva is tested at a time. Although this assay is used here mainly at low intensities of light11,12, it can also apply to larval avoidance in strong light that can excite class IV DA neurons that tile the surface of body walls16.
Our experimental device can also be used for optogenetics. The 850 nm band-pass filter can block the excitation light, as it does for the light spot signal, so that the camera can record larval behaviors before, during, and after red light stimulation clearly. Specifically, when 620 nm red light is used in combination with Chrimson for optogenetic stimulation, the low halves of infrared light LEDs need to be masked, and the direction of red light should be well-controlled to image the larvae clearly. Meanwhile, moderate levels of noisy signals originating from red light in the image can be used to judge the timing of on/off stimulation. In short, the light spot assay provides an addition method to monitor and analyze detailed spatial and temporal properties of the rapid larval light avoidance behavior.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work is supported by natural science foundation of China (31671074) and Fundamental Research Funds for the Zhejiang Provincial Universities (2019XZZX003-12).
Materials
| 850 nm ± 3 nm infrared-light-generating LED | Thorlabs, USA | PM100A | Compatible Sensors: Photodiode and Thermal Optical Power Rangea: 100 pW to 200 W Available Sensor Wavelength Rangea: 185 nm-25 μm Display Refresh Rate: 20 Hz Bandwidtha: DC-100 kHz Photodiode Sensor Rangeb: 50 nA-5 mA Thermopile Sensor Rangeb: 1 mV-1 V |
| AC to DC converter | Thorlabs, USA | S120VC | Aperture Size: Ø9.5 mm Wavelength Range: 200-1100 nm Power Range: 50 nW-50 mW Detector Type: Si Photodiode (UV Extended) Linearity: ±0.5% Measurement Uncertaintyc: ±3% (440-980 nm), ±5% (280-439 nm), ±7% (200-279 nm, 981-1100 nm) |
| band-pass filter | Thorlabs, USA | DC2100 | LED Current Range: 0-2 A LED Current Resolution: 1 mA LED Current Accuracy: ±20 mA LED Forward Voltage: 24 V Modulation Frequency Range: 0-100 kHz Sine Wave Modulation: Arbitrary |
| Collimated LED blue light | ELP, China | USBFHD01M | Max. Resolution: 1920X1080 F6.0 mm Sensor: 1/2.7" CMOS OV2710 |
| Compact power meter console | Ocean Optics, USA | USB2000+(RAD) | Dimensions: 89.1 mm x 63.3 mm x 34.4 mm Weight: 190 g Detector: Sony ILX511B (2048-element linear silicon CCD array) Wavelength range: 200-850 nm Integration time: 1 ms – 65 seconds (20 seconds typical) Dynamic range: 8.5 x 10^7 (system); 1300:1 for a single acquisition Signal-to-noise ratio: 250:1 (full signal) Dark noise: 50 RMS counts Grating: 2 (250 – 800 nm) Slit: SLIT-50 Detector collection lens: L2 Order-sorting: OFLV-200-850 Optical resolution: ~2.0 nm FWHM Stray light: <0.05% at 600 nm; <0.10% at 435 nm Fiber optic connector: SMA 905 to 0.22 numerical aperture single-strand fiber |
| High-Power LED Driver | Minhongshi, China | MHS-48XY | Working voltage: DC12V Central wavelength: 850nm |
| high-resolution web camera | Thorlabs, USA | MWWHL4 | Color: Warm White Correlated Color Temperature: 3000 K Test Current for Typical LED Power: 1000 mA Maximum Current (CW): 1000 mA Bandwidth (FWHM): N/A Electrical Power: 3000 mW Viewing Angle (Full Angle): 120˚ Emitter Size: 1 mm x 1 mm Typical Lifetime: >50 000 h Operating Temperature (Non-Condensing): 0 to 40 °C Storage Temperature: -40 to 70 °C Risk Groupa: RG1 – Low Risk Group |
| LED Warm White | Mega-9, China | BP850/22K | Ø25.4(+0~-0.1) mm Bandwidth: 22±3nm Peak transmittance:80% Central wavelength: 850nm±3nm |
| Spectrometer | Noel Danjou | Amcap9.22 | AMCap is a still and video capture application with advanced preview and recording features. It is a Desktop application designed for computers running Windows 7 SP1 or later. Most Video-for-Windowsand DirectShow-compatible devices are supported whether they are cheap webcams or advanced video capture cards. |
| Standard photodiode power sensor | Super Dragon, China | YGY-122000 | Input: AC 100-240V~50/60Hz 0.8A Output: DC 12V 2A |
| Thermal power sensor | Thorlabs, USA | M470L3-C1 | Color: Blue Nominal Wavelengtha: 470 nm Bandwidth (FWHM): 25 nm Maximum Current (CW): 1000 mA Forward Voltage: 3.2 V Electrical Power (Max): 3200 mW Emitter Size: 1 mm x 1 mm Typical Lifetime: 100 000 h Operating Temperature (Non-Condensing): 0 to 40 °C Storage Temperature: -40 to 70 °C Risk Groupb: RG2 – Moderate Risk Group |
| Thermal power sensor | Thorlabs, USA | S401C | Wavelength range: 190 nm-20 μm Optical power range:10 μW-1 W(3 Wb) Input aperture size: Ø10 mm Active detector area: 10 mm x 10 mm Max optical power density: 500 W/cm2 (Avg.) Linearity: ±0.5% |
References
- Grossfield, J. Geographic distribution and light-dependent behavior in Drosophila. Proceedings of the National Academy of Sciences of the United States of America. 68, 2669 (1971).
- Godoy-Herrera, R. C. L. D. The behaviour of Drosophila melanogaster larvae during pupation. Animal Behaviour. 37, (1989).
- Busto, M., Iyengar, B., Campos, A. R. Genetic dissection of behavior: modulation of locomotion by light in the Drosophila melanogaster larva requires genetically distinct visual system functions. Journal of Neuroscience. 19, 3337 (1999).
- Mazzoni, E. O., Desplan, C., Blau, J. Circadian pacemaker neurons transmit and modulate visual information to control a rapid behavioral response. Neuron. 45, 293 (2005).
- Keene, A. C., et al. Distinct visual pathways mediate Drosophila larval light avoidance and circadian clock entrainment. Journal of Neuroscience. 31, 6527 (2011).
- Keene, A. C., Sprecher, S. G. Seeing the light: photobehavior in fruit fly larvae. Trends in Neurosciences. 35, (2012).
- Kane, E. A., et al. Sensorimotor structure of Drosophila larva phototaxis. Proceedings of the National Academy of Sciences of the United States of America. 110, E3868 (2013).
- Humberg, T. H., et al. Dedicated photoreceptor pathways in Drosophila larvae mediate navigation by processing either spatial or temporal cues. Nature Communications. 9. 1260, (2018).
- Gong, Z., et al. Two pairs of neurons in the central brain control Drosophila innate light preference. Science. 330, (2010).
- Yamanaka, N., et al. Neuroendocrine Control of Drosophila Larval Light Preference. Science. 341, 1113 (2013).
- Zhao, W., et al. A disinhibitory mechanism biases Drosophila innate light preference. Nature Communications. 10, (2019).
- Gong, C., et al. A Neuronal Pathway that Commands Deceleration in Drosophila Larval Light-Avoidance. Neuroscience Bulletin. Feb. 27, (2019).
- Larderet, I., et al. Organization of the Drosophila larval visual circuit. eLife. 6, (2017).
- Sawin-McCormack, E. P., Sokolowski, M. B., Campos, A. R. Characterization and genetic analysis of Drosophila melanogaster photobehavior during larval development. Journal of Neurogenetics. 10, (1995).
- Farca, L. A., von Essen, A. M., Widmer, Y. F., Sprecher, S. G. Light preference assay to study innate and circadian regulated photobehavior in Drosophila larvae. Journal of Visualized Experiments. 74 (74), e50237 (2013).
- Xiang, Y., et al. Light-avoidance-mediating photoreceptors tile the Drosophila larval body wall. Nature. 468, 921 (2010).
- Gomez-Marin, A., Partoune, N., Stephens, G. J., Louis, M. Automated tracking of animal posture and movement during exploration and sensory orientation behaviors. PLoS ONE. 7, e41642 (2012).

