Proteolytically Degraded Alginate Hydrogels and Hydrophobic Microbioreactors for Porcine Oocyte Encapsulation
Summary
Presented here are two protocols for the encapsulation of porcine oocytes in 3D culture conditions. In the first, cumulus-oocyte complexes (COCs) are encapsulated in fibrin-alginate beads. In the second, they are enclosed with fluorinated ethylene propylene powder particles (microbioreactors). Both systems ensure optimal conditions to maintain their 3D organization.
Abstract
In reproductive biology, the biotechnology revolution that began with artificial insemination and embryo transfer technology led to the development of assisted reproduction techniques such as oocyte in vitro maturation (IVM), in vitro fertilization (IVF) and cloning of domestic animals by nuclear transfer from somatic cell. IVM is the method particularly of significance. It is the platform technology for the supply of mature, good quality oocytes for applications such as reduction of the generation interval in commercially important or endangered species, research concerning in vitro human reproduction, and production of transgenic animals for cell therapies. The term oocyte quality includes its competence to complete maturation, be fertilized, thereby resulting in healthy offspring. This means that oocytes of good quality are paramount for successful fertilization including IVF procedures. This poses many difficulties to develop a reliable culture method that would support growth not only of human oocytes but also of other large mammalian species. The first step in IVM is the in vitro culture of oocytes. This work describes two protocols for the 3D culture of porcine oocytes. In the first, 3D model cumulus-oocyte complexes (COCs) are encapsulated in a fibrin-alginate bead interpenetrating network, in which a mixture of fibrin and alginate are gelled simultaneously. In the second one, COCs are suspended in a drop of medium and encapsulated with fluorinated ethylene propylene (FEP; a copolymer of hexafluoropropylene and tetrafluoroethylene) powder particles to form microbioreactors defined as Liquid Marbles (LM). Both 3D systems maintain the gaseous in vitro culture environment. They also maintain COCs 3D organization by preventing their flattening and consequent disruption of gap junctions, thereby preserving the functional relationship between the oocyte, and surrounding follicular cells.
Introduction
The development of various culture systems, including three-dimensional (3D) ones, aims to provide optimal conditions for the growth and maturation of oocytes isolated from the follicles even at earliest stages of development. This is of great importance for assisted reproductive techniques (ART), especially in view of the increasing number of women who are struggling with infertility after cancer treatment1. Maturation of oocytes in in vitro conditions (IVM) is already a well-established technique mainly used for in vitro embryo generation for the purpose of livestock reproduction2. However, in most mammalian species, even if high rates of maturation of cumulus-oocyte complexes (COCs) can be achieved (range 60 to 90 %)3, their developmental competence is still inadequate to the needs. This is because the development of the zygotes obtained in such a way even up to the blastocyst stage is low and after the transfer into surrogate animals their viability to term is reduced. Consequently, there is a need to increase the developmental competence of embryos obtained from oocytes that were subjected to the IVM procedure4. Therefore, new maturation media5 are being devised and various periods of in vitro culture are tested6,7 along with supplementation of culture media with various growth factors and molecules8,9.
The first step of any complete IVM system is to create optimal conditions for sustainable growth of oocytes during in vitro culture. The oocyte growth is one of the specific indicators of the oocyte's ability to resume meiosis10,11. In addition, an appropriate oocyte in vitro culture system must be capable of supporting its nuclear maturation and cytoplasmic differentiation12. The morphology of the cumulus-oocyte complex is another important indicator used in ART clinics to select the best oocyte for subsequent steps of in vitro fertilization (IVF) procedure in humans and livestock12,13. Among morphological characteristics of COCs considered are: the oocyte diameter, its cytoplasm granulation and the first polar body integrity14,15. Besides, the oocyte developmental potential is correlated to the appearance and compaction of cumulus cells and number of their layers surrounding the oocyte. Very important for the appropriate oocyte in vitro culture system is also the maintenance of oocyte–cumulus cells proper interactions and cytoskeleton stability16,17,18,19. So far, in vitro oocyte growth within human COCs has been demonstrated20. The use of cow COCs also resulted with live births. These were isolated from immature ovarian follicles and then cultured for 14 days until the oocyte was sufficiently large to undergo the IVF procedure21. Similarly, COCs isolated from baboon antral follicles, subjected to IVM after in vitro culture yielded oocytes capable of reinitiating meiosis to the metaphase II stage with a normal appearing spindle structure22. However, in this study the authors did not try to fertilize them. Nevertheless such results indicate that a similar procedure could be applied not only to these particular mammalian species but also to human cumulus-oocyte complexes obtained from follicles what should allow to obtain oocytes of good quality suitable for a successful IVF technique.
The above described results were obtained with the application of conventional IVM protocols during which oocytes were cultured in two-dimensional (2D) systems. The routine procedure in 2D culture systems is covering oocytes, immersed in a drop of an appropriate culture media, with mineral oil23,24. It is assumed that an oil overlay during in vitro oocyte culture serves to prevent liquid evaporation, thus ensuring the maintenance of proper pH and osmotic pressure in the culture. Although such a 2D culture system allows to obtain, even up to 87% of mature pig oocytes25, it has been proven that the mineral oil overlay causes substantial diffusion of lipid soluble materials which are necessary for proper oocytes development26. Additionally, because of steroids (progesterone and estrogens) diffusion into the mineral oil during oocyte culture, a delay of nuclear maturation and reduction in developmental competence achievement of pig oocytes was observed. This may result in obtaining a small number of zygotes, which additionally are characterized by low developmental capacity to the stage of the blastocyst and by poor viability after transfer into recipient animals27. Therefore, attempts are being made to increase the developmental competence of embryos derived from oocytes received after IVM procedure, by creating optimal conditions for achieving both cytoplasmic and nuclear maturity of oocytes cultured together with CCs as complexes, especially using three-dimensional (3D) systems. Various innovative 3D in vitro culture systems have been developed in the last two decades28,29. These were designed to maintain the natural spatial organization of cells and to avoid their flattening in culture dishes what cannot be achieved in the traditional 2D cultures. The structural and functional activity of cultured COCs can be ensured by the maintenance of their proper architecture and undisturbed communication through gap-junctions between various compartments30. The suitability of bio-scaffolds for 3D in vitro culture of cumulus–oocyte complexes has been evaluated using natural biomaterials such as various components of the extra-cellular matrix (ECM; collagen and hyaluronic acid)31 or inert polymers (alginate)32. These attempts tested in several species brought promising results in terms of oocyte meiosis resumption and achievement of their full competence33,34,35. However so far, no 3D system suitable for COCs maturation isolated from large domestic animals, including pigs, has been developed.
This work describes two protocols that can be used for the 3D culture of porcine COCs. The first protocol describes encapsulation in fibrin-alginate beads (FAB). FAB can be formed by simultaneous mixing an alginate and fibrin solution, which undergo a synchronous gelation process. This combination provides a dynamic mechanical environment because both components contribute to matrix rigidity. A similar solution has been used previously for mouse ovarian follicle culture and maturation36. In the case of the presented protocol, to avoid premature degradation of the alginate-fibrin network, appropriately higher concentrations of calcium chloride solution are used, ensuring a fast and stable gelation process. The dynamic mechanical environment creates conditions similar to these in the natural intra-follicular environment in which COCs reside and increase in size. Additionally, the work shows representative results of COCs 3D culture systems, in which these are suspended in a drop of medium and encapsulated with fluorinated ethylene propylene (FEP; a copolymer of hexafluoropropylene and tetrafluoroethylene) powder particles, to form microbioreactors (Liquid Marbles, LM). LM are a form of 3D bioreactor that have been previously shown to support, among others, growth of living microorganisms37, tumor spheroids38 and embryonic stem cells39. LMs have been also successfully used for sheep oocyte culture40. In most experiments using LMs, bioreactors were prepared using polytetrafluoroethylene (PTFE) powder bed with particle size of 1 μm41. The presented protocol uses FEP, which is very similar in composition and properties to the fluoropolymers PTFE. But FEP is more easily formable and softer than PTFE and what is especially important, it is highly transparent.
Both 3D systems maintain the gaseous in vitro culture environment. They also maintain COCs 3D organization by preventing their flattening and consequent disruption of gap junctions, preserving their functional relationship between the oocyte and surrounding follicular cells.
Protocol
The following procedures were approved by the Animal Welfare Committee at the Institute of Zoology and Biomedical Research at Jagiellonian University.
1. Isolation of porcine cumulus-oocyte complexes
- To collect material for the isolation of ovarian follicles, excise porcine ovaries from prepubertal gilts (approximately 6-7 months of age, weighing 70 to 80 kg) at a local slaughterhouse. Choose approximately 20 pig ovaries from 10 animals for COCs isolation in each experiment.
NOTE: Assuming each ovary yields 3–5 follicles, the total number of follicles varies from 60 to 100. - Place ovaries in a thermos with sterile phosphate-buffered saline (PBS; pH 7.4; 38 °C) containing 1% AAS. Ensure that the experimental material is transported to the laboratory within 1 h where it is rinsed twice with sterile PBS containing antibiotics.
- After rinsing the ovaries, transfer them to the beaker filled with HM medium and store in an incubator at 38 °C for the time of all manipulations.
NOTE: For optimal results during oocyte handling, carry out all procedures in DMEM/F12 medium (handling medium, HM) with the addition of 2.5% antibiotic/antimycotic solution (AAS) and 10% fetal bovine serum (FBS) and control the pH at the CO2 level in the environment, on a heating table with temperature control (38 °C) and under the laminar flow hood to minimize bacterial contamination. - From large porcine follicles (6-8 mm in diameter) collect follicular fluid (FF) by aspiration and centrifugation at 100 x g for 10 min at room temperature. After that, filter the supernatant using sterile syringe attached to 0.2 μm membrane pore filters and snap freeze at -80 ° C.
NOTE: Use an insulin syringe (u- 40) to aspiration of follicular fluid. - Ensure that, only healthy, medium-sized follicles (4-6 mm in diameter) are selected for COCs isolation42.
NOTE: COCs of grade I, possessing an intact and round oocyte with homogeneous ooplasm and multilayered (at least 3-4) compact cumulus are considered suitable for further 3D IVM procedure43. - To isolate COCs, transfer 2-3 ovaries to a sterile 10 cm diameter Petri dish filled with HM. Isolate COCs from medium-sized follicles by following one of the procedures below.
- Gently cut the surface of the protruding ovarian follicles with a sterile surgical blade 15C. This will cause the follicular fluid along with COCs to flow out into the Petri dish.
- Aspirate the follicular content using 28 G needle having a size of 5/8” attached to a disposable syringe and transfer it into the Petri dish.
NOTE: Discard the remains of ovarian tissue. Subsequent stages of isolation are carried out under a stereomicroscope.
- Prepare 3–5 60 mm IVF Petri dishes.
- Add 1 mL of HM to the central wells and place a 3-4 drops of HM (50 µL per drop) in their outer rings.
- Then, using a polycarbonate micropipette, move the undamaged COCs to drops of HM in the outer rings to rinse them briefly for 3-4 times.
- At the end, individually transfer them into the central well. Store this IVF plates in an incubator for further procedures.
- After COCs are collected, perform the final selection step by assigning them randomly into two different IVM encapsulation protocols listed below.
2. Encapsulation in fibrin-alginate hydrogel beads
- Prepare 1 mL of 50 IU/mL thrombin in Tris-buffered saline (TBS; pH 7.4) with 100 mM CaCl2 in a 2.0 mL sterile microcentrifuge tube.
- Prepare fibrinogen stock solution (50 mg/mL in TBS) and keep it frozen.
NOTE: To avoid clumping when dissolving fibrinogen in TBS, heat TBS to 37 °C.- On the day of the procedure, thaw it slowly on ice and right before the use bring to room temperature.
- Just before use mix at a 1:1 ratio 0.5% alginate solution and 50 mg/mL fibrinogen solution to get finally 2 mL (FA). In a sterile microcentrifuge tube gently vortex the mixture. Avoid making bubbles.
- To prepare "incubation chambers", apply thin strips of paraffin films to the glass microscope slides.
NOTE: Wipe the slides with 70 % EtOH before use. One incubation chamber is formed with two slides. To separate them, place 3 mm spacers on one of them. - Pipette 7.5 μL drops of the FA mixture onto this paraffin film coated glass slide, which also has separating spacers. On one slide place 8-10 drops, arranged in two rows.
- Transfer, using a micropipette, 3-5 COCs along with a minimum volume (up to 5 µL) of maturation medium (MM), and place them precisely in the center of the FA drop. This procedure requires good manual skills and precision.
NOTE: The maturation medium (MM) consists of DMEM/F12, supplemented with 10 IU/mL PMSG, 10 IU/mL hCG, 10% FBS and 50% porcine follicular fluid (FF) collected in step 1.4. - Add 7.5 μL of thrombin/Ca2+ solution on each FA drop, just to cover them. There is no need to mix them because the gel forms almost instantaneously.
- Cover the incubation chamber by applying the second, previously prepared glass slide to the FA capsules. With a very careful but firm movement, turn the chamber upside down and place it in a 100 mm Petri dish lined with moist filter paper to avoid drying.
- Then, transfer the Petri dish to the 5% CO2 incubator (38 °C) for about 5-7 min. After this, FA capsules will become cloudy because of simultaneous gelation of fibrin and alginate.
- Transfer FA capsules into 96 well plates (one capsule per well) containing 100 μL MM per well.
NOTE: To not damage the formed FA capsules, perform this step carefully with the use of surgical forceps. - Every 2 days replace half of the MM (50 μL) with fresh, pre-equilibrated one. Image COCs using an inverted light microscope (at 10x magnification).
NOTE: Culture conditions for COCs FA culture: 38 °C, under a 5% CO2 atmosphere and relative humidity 95%. After 4 days of IVM, the hydrogels appear almost clear due to progressive degradation of the fibrin component. The remaining alginate should be degraded enzymatically – by alginate-lyase, a plant-derived enzyme that specifically degrades alginate and does not affect animal cells.- Remove MM from wells containing the capsules and add 100 μL of 10 IU/mL alginate lyase in DMEM. Leave the culture plate in the incubator for 25-30 min.
- Remove COCs from the dissolved capsules.
NOTE: This can be done with a cut pipette tip. - After several washes in a fresh DMEM, transfer COCs to the inner ring of an IVF dish (5-10 COCs per dish) containing PBS. Now, use them for further morphological or biochemical analysis.
3. Encapsulation in super-hydrophobic fluorinated ethylene propylene (FEP) microbioreactors.
NOTE: Make sure that all IVM procedures are carried out on thermostatically controlled table and COCs are maintained at 38 °C throughout their handling.
- Prepare a 30 mm Petri dish containing 5 g of FEP powder bed-average particle size of 1 μm.
NOTE: It is important to use fresh FEP powder. The re-used FEP tends to aggregate and forms clumps. - Distribute a single droplet of MM (~30 μL in volume) containing 3-5 COCs isolated in step 1.6 onto the bed of the FEP powder. Rotate the plate gently in a circular motion to be sure that these particles completely covered the surface of the liquid drop and liquid marbles formed (LM).
- Pick up formed LM using a pipette with 1,000 μL tip.
NOTE: Cut the pipette tip at the edge, to accommodate it to the diameter of the LM (4-5 mm). The approximate diameter of such prepared tip should be slightly bigger than that of the LM. - Prepare several 60 mm IVF Petri dishes. Add 3-4 mL of sterile water to the outer rings to prepare the humidity chamber. Next, place one LM into the central well.
NOTE: This procedure requires good manual skills and precision – if the marble is placed carelessly or falls even from a low height it will be destroyed. - Incubate marbles for 4 days at 38 °C in 5 % CO2 incubator.
- Change the medium daily following the procedure: apply 30 μL of MM on each LM, which will cause their spreading because direct liquid contact disrupts the hydrophobicity of the coated FEP powders. When the marble content dissolve, transfer COCs released from the bioreactor to a drop of fresh MM in the Petri dish.
NOTE: To precisely transfer released COCs, use a polycarbonate micropipette. - After several washes in MM (3-4 times) to remove FEP particles, transfer them with 30 μL of fresh MM onto the FEP powder bed. Gently rotate the plate in a circular motion to ensure that the powder particles completely covered the surface of the liquid drop and form a new LM.
- Then follow the procedure from steps 3.3-3.4.
NOTE: The transparent coating of FEP powder allows to monitor LM content using light microscope.
- Change the medium daily following the procedure: apply 30 μL of MM on each LM, which will cause their spreading because direct liquid contact disrupts the hydrophobicity of the coated FEP powders. When the marble content dissolve, transfer COCs released from the bioreactor to a drop of fresh MM in the Petri dish.
4. Characterization of COCs after 96-h IVM procedure using two 3D systems
NOTE: To determine the efficiency of the IVM systems used, score COCs morphologically under a light microscope. Additionally, use double fluorescent labeling with calcein AM and ethidium homodimer (EthD-1) dyes for analysis of COCs viability44.
- Morphological examination of oocytes after the IVM by light and fluorescence microscopy.
- Wash COCs (both those derived from culture using FAB and those from LM) with 38 °C 1x PBS.
NOTE: To facilitate handling of COCs and not to damage them during the staining procedure, use a 96 or 48 well plate and perform each staining step by transferring the COCs to subsequent wells. - Incubate them in 100 µL of 1.6 mM calcein AM and 5.5 mM EthD-1 for 30–45 min at 37 °C. Dilute both substances in 1x PBS.
NOTE: Because this is a two-color fluorescence assay, perform this step in the dark. - After incubation, wash the COC twice in PBS and then immerse it in an antifade mounting medium designed to prevent rapid photobleaching of fluorescent proteins and fluorescent dyes (Table of Materials).
NOTE: Protect the edges of cover glass against drying with any nail polish. - Visualize stained samples under a confocal laser scanning microscope.
NOTE: To excite the fluorescence of both probes (i.e., calcein and ethidium homodimer-1, EthD-1) tune an argon laser to 488nm. The emitted fluorescence is separated by a triple dichroic filter 488/561/633 and then measured at 505−570 nm for the green one (calcein), and at 630−750 nm for the red one (EthD-1). - Classify COCs into four categories, using modified classification that is applied to ovarian follicles in culture based on confocal fluorescence imaging depending on the percentage of dead granulosa cells35. V1: COCs with estimated 100% viable CCs; V2: COCs with 10% of dead CCs; V3: COCs with 10–50% of dead CCs; V4: COCs with 50% of dead CCs.
NOTE: Technically, it is difficult to assess the viability of the oocyte with the live-dead assay; therefore, it should be estimated e.g., by analyzing mitochondrial ultrastructure or activity.
- Wash COCs (both those derived from culture using FAB and those from LM) with 38 °C 1x PBS.
- Examination of oocytes ultrastructure after the IVM by transmission electron microscopy.
- Fix COCs in 100 µL 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.2, room temperature) for 2 h.
- Immerse COCs in 100 µL 0.1 M sodium cacodylate buffer (overnight at 4 °C) and rinse in the same buffer.
- Postfix COCs with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer (room temperature) for 1 h.
- After staining in uranyl acetate (1 %), dehydrate COCs in an increasing series of ethanol dilutions (50%, 60%, 70%, 90% and 100%), infiltrate them overnight at room temperature, followed by two short changes of resin and finally, embed them in LR White.
- Next, to orientate and evaluate samples, stain semi-thin sections (1 µm) with methylene blue/azure II.
- Finally, examine ultrathin, doubly stained sections at the ultrastructural level with the TEM.
Representative Results
In COCs using both IVM systems, the granulosa cells adhered tightly to each other, and most of the recovered COCs had intact layers of cumulus cells (Figure 1A,B). Additionally, substantial proportion of cumulus cells were retained.
The results obtained from the COCs viability analysis confirmed that both systems applied for the encapsulation of porcine oocytes in 3D in vitro conditions ensured optimal growth conditions (Figure 2). In both groups, only high-viability oocytes were observed, V1 = 13 %, V2 = 87 % for FAB and 10 % V1, and 90 % V2 for LM, respectively (Table 1).
Mitochondria, observed by transmission electron microscopy (TEM), were evenly distributed in oocytes, their shape after IVM were shell-like45. Only a few of them were elongated, moreover, their clustering was sporadically observed (Figure 1C,D). Endoplasmic reticulum was observed, either associated with mitochondria or free in the oocyte cytoplasm. Lipid droplets appeared as small dark round structures and Golgi apparatus were emerged with dilated cisternae (Figure 1C,D).
| V1 | V2 | V3 | V4 | ||||
| % | number | % | number | % | number | % | |
| FAB | 13% | 7 | 87% | 48 | – | – | |
| LM | 10% | 9 | 90% | 80 | – | – | |
| COCs total number: 144 | 16a | 128b | 0 | 0 | |||
Table 1. Results on the viability of COCs after encapsulation using fibrin-alginate beads (FAB) and microbioreactors FEP, Liquid Marbles (LM). The results were given as average. Values with superscripts (a, b) differed statistically significantly (p <0.05).
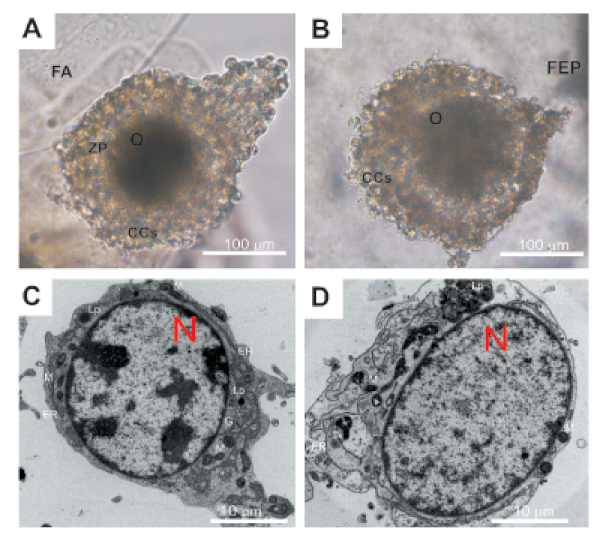
Figure 1: Representative images showing the morphology and ultrastructure of COCs after a 96 h of encapsulation. (A) morphology of COCs inside FAB – light microscopy; (B) morphology of COCs inside LM – light microscopy; C: oocyte ultrastructure (TEM) after in vitro culture inside FAB; D: oocyte ultrastructure (TEM) after in vitro culture inside LM. O: oocyte; CCs: cumulus cells; ZP: zona pellucida; N: nucleus; M: mitochondria; G: Golgi apparatus; ER: endoplasmic reticulum; Lp: lipid droplet; FA: alginate and fibrin filaments; FEP: powder of copolymer of hexafluoropropylene and tetrafluoroethylene. Please click here to view a larger version of this figure.
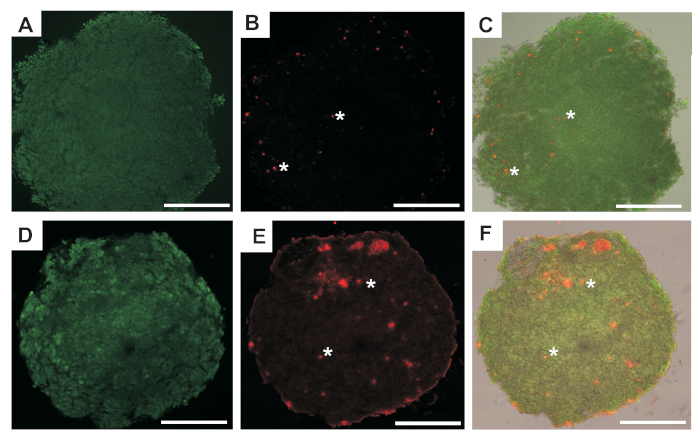
Figure 2: Representative fluorescent images of COCs after 96 h of encapsulation. (A,B) The images obtained for the same COCs after in vitro culture using FAB with two filters to visualize green fluorescence or red fluorescence for live or dead cells respectively and (C) merge of both. (D,E) The images obtained for the same COCs after in vitro culture using LM with two filters to visualize green fluorescence or red fluorescence for live or dead cells, respectively and (F) merge of them. Scale bars = 50 µm. White asterisks indicate cells undergoing apoptosis. Please click here to view a larger version of this figure.
Discussion
The ability to maintain in vitro growth not only of the oocyte but also of cumulus cells surrounding it and simultaneously supporting its maturation is exceedingly essential for the successful assisted reproductive technologies and for furthering the understanding of somatic cell/oocyte interactions especially in species undergoing prolonged follicular growth such as human beings or pigs. Continuously improved IVM techniques are becoming useful tool to preserve reproductive options in cases of polycystic ovarian syndrome (PCOS), premature ovarian failure, or definitive infertility (oncotherapy). In addition, since certain ovarian dysfunctions might be caused by dysregulated follicular growth, understanding the molecular and cellular mechanisms that control proper development of the oocyte may provide important insight into the pathophysiology and rational treatment of these conditions. In vitro culture systems that would support the maintenance of COCs 3D architecture during IVM, require a step of encapsulation in a matrix or inside a bioreactor. This work describes a protocol that can be applied for the culture of porcine oocytes. Both 3D in vitro culture systems presented here to induce growth and development of porcine COCs are user-friendly and allow for the effective control of both oocyte and cumulus cells survival.
The first in vitro culture method of COCs using their encapsulation in fibrin-alginate hydrogel beads (FAB) presented here allows for a 3D oocyte growth in an actively cell-responsive matrix environment. Previously published protocols based on alginate hydrogels were usually used for the investigation of the development of ovarian follicles isolated from numerous animal species with very promising results32. Interestingly, regardless of the final composition of the alginate hydrogel32 or the encapsulation method itself46, these studies showed that alginate bead-based 3D culture systems were able to support follicular growth and their survival.
Alginate hydrogels were gentle on the follicles, not affecting their further survival or development in vitro. Similarly, the in vitro culture protocol of COCs described for the first time here, is suitable to generate good quality pig oocytes, as it was documented at both morphological and ultrastructural levels. The presence of fibrinogen in an alginate solution and their simultaneous gelling, results in the formation of an interpenetrating network which creates and further stabilizes the three-dimensional environment. Due to the specific structural support inside such a matrix, cell-to-cell contacts and paracrine communication between oocytes and cumulus cells are maintained similarly to these present in vivo.
The two most critical steps in the maturation protocol described in this document are the transfer of FA capsules from incubation chambers into 96-well plates containing MM (step 2.10.) and the removal of COCs from the dissolved capsules after enzymatic degradation of the remaining alginate, using alginate lyase (step 2.11.). Both steps require significant manual skill and precision to avoid, in the first case the destruction of capsules, in the second – damage to COCs.
The main strength of this method, in addition to establishing optimal conditions for the oocyte maturation of a large domestic animal, is that during analysis using TEM, light microscopy or confocal microscopy, no capsule removal is necessary. Alginate is not a barrier when imaging. Leaving COCs in capsules facilitates manipulation during the staining procedure.
The second COC in vitro culture presented here is the one with the use of LM, which generally are nonstick droplets covered by micro- scaled particles and obtained by simply rolling small volumes of a liquid in a very hydrophobic powder47. Previous studies with the use of LM for culture, among others fibroblasts48, red blood cells49, organoid of a tumor38, pancreatic cells50 or sheep oocytes36 have been performed using polytetrafluoroethylene (PTFE) as a hydrophobic polymer for the preparation of the droplets. PTFE is widely used in the clinic (e.g., cardiovascular grafts).
In this work, we present, for the first time, a reliable method for microbioreactors made of FEP particles for in vitro culture of porcine COCs. The use of FEP in the presented protocol, which is very similar in composition and properties to the PTFE, makes preparation of LM much easier. This powder is softer than PTFE and what is especially important, it is transparent. This in turn simplifies the work during imaging of structures cultured with its use. The ease of forming LM using FEP is a definite advantage of this method.
The most critical step in the maturation protocol described in this document is picking up and transferring formed LM into 60 mm IVF Petri dish using a pipette tip (steps 3.3. and 3.4.). These steps require significant manual skill and precision to avoid destruction of LM.
Possibly, the main limitation of the 3D oocyte maturation induction protocols presented here is the relatively great difficulty in obtaining proper research material collected from animals not treated with antibiotics, anabolic steroids (i.e., nadrolone, boldenone) or melengestrol acetate which promote their rapid growth. These steroids, despite their use being banned in Europe, are still widely utilized in the industrial livestock breeding during the last two months of the fattening period, which causes, among others, disturbed sexual maturation.
In conclusion, both 3D systems presented here maintain the optimal gaseous in vitro culture environment. They also maintain COCs 3D organization by preventing their flattening and consequent disruption of gap junctions, thus preserving the functional relationship between the oocyte, and surrounding follicular cells, what is crucial for proper oocyte maturation. Thus, both models are a valuable tool for basic research in reproductive biology and may also have clinical relevance, leading to improved infertility treatment. Additionally, they can also be applied for developing novel biotechnology methods, as well as for livestock improvement.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors are very grateful to: dr Waclaw Tworzydlo (Department of Developmental Biology and Invertebrate Morphology, Institute of Zoology and Biomedical Research, Jagiellonian University) for technical facilities in TEM; to Ms. Beata Snakowska (Department of Endocrinology, Institute of Zoology and Biomedical Research, Jagiellonian University) for technical assistance; to the Department of Cell Biology and Imaging, Institute of Zoology and Biomedical Research, Jagiellonian University, JEOL JEM 2100HT (JEOL, Tokyo, Japan). This work was supported by grant 2018/29/N/NZ9/00983 from National Science Centre Poland.
Materials
| General | |||
| Antibiotic Antimycotic (100x) 100ml | Thermo Fisher | 15240062 | 2.5% final concentration for Handling Medium. 1% in PBS (step 1.2) |
| DMEM/F12 (500ml) | Sigma-Aldrich | D8062 | Handling and Maturation Medium |
| DPBS (w/o Ca, Mg), 1x, 500ml | Thermo Fisher | 14190144 | |
| FCS (100 ml) | Thermo Fisher | 16140063 | 10% final concentration for both Handling Medium and Maturation Medium. (steps: 1.5. 2.6.) |
| PBS (1x, pH 7.4) 500ml | Thermo Fisher | 10010023 | |
| TBS Stock Solution (10x, pH 7.4) 500 ml | Cayman Chemicals | 600232 | 1x final concentration. Other brand can be use |
| Maturation Medium | |||
| hCG (1 VIAL of 10 000 U) | Sigma-Aldrich | CG10 | |
| PMSG | BioVendor | RP1782721000 | |
| Fibrin-alginate beads | |||
| Alginate Lyase | Sigma-Aldrich | A1603 | (Step 2.11.1) |
| Thrombin | Sigma-Aldrich | T9326-150UN | (Step 2.1) |
| Calcium Chloride | Sigma-Aldrich | C5670 | (Step 2.1) |
| Fibrinogen (250mg) | Sigma-Aldrich | F3879 | (Step 2.2) |
| Sodium Alginate | Sigma-Aldrich | W201502 | (Step 2.3) use for alginate solution |
| Liquid Marble | |||
| FEP | Dyneon GmbH 3M AdMD | A-66670 | |
| Morphological examination | |||
| LIVE/DEAD Viability/Cytotoxicity Kit, for mammalian cells | Thermo Fisher | L3224 | (Step 4.1.) Emitted fluorescence: 494 nm for calcein, 528 nm for EthD-1; measure: 517 nm for calcein, 617 nm – EthD-1 |
| VECTASHIELD Antifade Mounting Medium | Vector Laboratories | H-1000 | mounting medium |
| Ultrastructure examination | |||
| Glutaraldehyde solution | Sigma-Aldrich | G5882 | 2.5% final concentraion (Step 4.2.1.) |
| LR White resin | Sigma-Aldrich | L9774 | (Step 4.2.4.) |
| Methylene blue | Sigma-Aldrich | M9140 | (Step 4.2.5.) |
| Osmium Tetroxide | Sigma-Aldrich | O5500 | (Step 4.2.3.) |
| Sodium cacodylate trihydrate | Sigma-Aldrich | C0250 | Use for preparing 0.1M sodium cacodylate buffer (pH 7.2) |
| Uranyl Acetate | POCH | 868540111 | (Step 4.2.4.) |
| Specific instruments, tools | |||
| 30 mm Pteri dish | TPP | 93040 | |
| 60 mm IVF Petri dish | Falcon | 353653 | |
| Ez-Grid Premium Cell Handling Pipettor | RI Life Sciences | 8-72-288 | |
| Ez-Tip | RI Life Sciences | 8-72-4155/20 | |
| Heating Table | SEMIC | Other brands can be used | |
| Incubator | Panasonic | MCO-170AIC-PE | Other brands can be used |
| Sterile petri dish (10 cm) | NEST Biotechnology | 704002 | |
| Sterile syringe filters with 0.2 µm | GOOGLAB SCIENTIFIC | GB-30-022PES | |
| Thermos | Quechua | 5602589 | Other brands can be used |
References
- Jeruss, J. S., Woodruff, T. K. Preservation of fertility in patients with cancer. The New England Journal of Medicine. 360 (9), 902-911 (2009).
- Paramio, M. T., Izquierdo, D. Current status of in vitro embryo production in sheep and goats. Reproduction in Domestic Animals. 49 (4), 37-48 (2014).
- Gilchrist, R. B., Thompson, J. G. Oocyte maturation: emerging concepts and technologies to improve developmental potential in vitro. Theriogenology. 67 (1), 6-15 (2007).
- Nogueira, D., Sadeu, J. C., Montagut, J. In vitro oocyte maturation: current status. Seminars in Reproductive Medicine. 30 (3), 199-213 (2012).
- Farsi, M. M., Kamali, N., Pourghasem, M. Embryological aspects of oocyte in vitro maturation. International Journal of Molecular and Cellular Medicine. 2 (3), 99-109 (2013).
- You, J., et al. Treatment with the proteasome inhibitor MG132 during the end of oocyte maturation improves oocyte competence for development after fertilization in cattle. PLOS One. 7 (11), 48613 (2012).
- Donnay, I., et al. Effect of prematuration, meiosis activating sterol and enriched maturation medium on the nuclear maturation and competence to development of calf oocytes. Theriogenology. 62 (6), 1093-1107 (2004).
- Ledda, S., Bogliolo, L., Leoni, G., Calvia, P., Naitana, S. Influence of vasoactive intestinal peptide (VIP), atrial natriuretic peptide (ANP) and insulin-like growth factor-I (IGF-I) on in vitro maturation of prepubertal and adult sheep oocytes. Zygote. 4 (4), 343-348 (1996).
- Uhm, S. J., et al. Epidermal growth factor can be used in lieu of follicle-stimulating hormone for nuclear maturation of porcine oocytes in vitro. Theriogenology. 73 (8), 1024-1036 (2010).
- Fair, T., Hyttel, P., Greve, T. Bovine oocyte diameter in relation to maturational competence and transcriptional activity. Molecular Reproduction and Development. 42 (4), 437-442 (1995).
- Sirard, M. A. Follicle environment and quality of in vitro matured oocytes. Journal of Assisted Reproduction and Genetics. 28 (6), 483-488 (2011).
- Banwell, K. M., Thompson, J. G. In vitro maturation of Mammalian oocytes: outcomes and consequences. Seminars in Reproductive Medicine. 26 (2), 162-174 (2008).
- de Smedt, V., Crozet, N., Gall, L. Morphological and functional changes accompanying the acquisition of the meiotic competence in ovarian goat oocyte. Journal of Experimental Zoology. 269 (2), 128-139 (1994).
- Luca, X., et al. Relationship between antral follicular size, oocyte diameters and nuclear maturation of immature oocyte in pigs. Theriogenology. 58 (5), 870-885 (2002).
- Abeydeera, L. R. In vitro production of embryo in swine. Theriogenology. 57 (1), 256-273 (2002).
- Carabatsos, M. J., Sellitto, C., Goodenough, D. A., Albertini, D. F. Oocyte-granulosa cell heterologous gap junctions are required for the coordination of nuclear and cytoplasmic meiotic competence. Developmental Biology. 226 (2), 167-179 (2000).
- Hashimoto, S., et al. Effects of cumulus cell density during in vitro maturation of the developmental competence of bovine oocytes. Theriogenology. 49 (8), 1451-1463 (1998).
- Zuccotti, M., Merico, V., Cecconi, S., Redi, C. A., Garagna, S. What does it take to make a developmentally competent mammalian egg. Human Reproduction Update. 17 (4), 525-540 (2011).
- Albertini, D. F., Barrett, S. L. Oocyte-somatic cell communication. Reproduction. Supplement. 61, 49-54 (2003).
- Cavilla, J. L., Kennedy, C. R., Byskov, A. G., Hartshorne, G. M. Human immature oocytes grow during culture for IVM. Human Reproduction. 23 (1), 37-45 (2008).
- Hirao, Y., et al. In vitro growth and development of bovine oocyte-granulosa cell complexes on the flat substratum: effects of high polyvinylpyrrolidone concentration in culture medium. Biology of Reproduction. 70 (1), 83-91 (2004).
- Xu, M., et al. In vitro oocyte maturation and preantral follicle culture from the luteal-phase baboon ovary produce mature oocytes. Biology of Reproduction. 84 (4), 680-697 (2011).
- Ka, H., Sawai, K., Wang, W. H., Im, K. S., Niwa, K. Amino acids in maturation medium and presence of cumulus cells at fertilization promote male pronuclear formation in porcine oocytes matured and penetrated in vitro. Biology of Reproduction. 57, 478-483 (1997).
- Shimada, M., Zeng, W. X., Terada, T. Inhibition of PI 3-kinase or MEK leads to suppression of p34cdc2 kinase activity and meiotic progression beyond the MI stage in porcine oocytes surrounded with cumulus cells. Biology of Reproduction. 65, 442-448 (2001).
- Coy, P., Romar, R. In vitro production of pig embryos: a point of view. Reproduction Fertility and Development. 14, 275-286 (2002).
- Reinsberg, J., Ackermann, D., Vander Ven, H. Pitfalls in assessment of progesterone production by granulosa cells cultured in contact with silicone rubber or paraffin oil. Archives Gynecology Obstetrics. 270, 174-178 (2004).
- Shimada, M., Kawano, N., Terada, T. Delay of nuclear maturation and reduction in developmental competence of pig oocytes after mineral oil overlay of in vitro maturation media. Reproduction. 124, 557-564 (2002).
- Cekleniak, N. A., et al. Novel system for in vitro maturation of human oocytes. Fertility and Sterility. 75, 1185-1193 (2001).
- Cukierman, E., Pankov, R., Yamada, K. M. Cell interactions with three-dimensional matrices. Current Opinion in Cell Biology. 14, 633-639 (2002).
- Desai, N., et al. Three-dimensional in vitro follicle growth: Overview of culture models, biomaterials, design parameters and future directions. Reproductive Biology and Endocrinology. 8 (119), (2010).
- Combelles, C. M., Fissore, R. A., Albertini, D. F., Racowsky, C. In vitro maturation of human oocytes and cumulus cells using a co-culture three-dimensional collagen gel system. Human Reproduction. 20, 1349-1358 (2005).
- Dorati, R., et al. Formulation and stability evaluation of 3D alginate beads potentially useful for cumulus-oocyte complexes culture. Journal of Microencapsulation. 33, 137-145 (2016).
- Morselli, M. G., Canziani, S., Vigo, D., Luvoni, G. C. A three-dimensional alginate system for in vitro culture of cumulus-denuded feline oocytes. Reproduction in Domestic Animals. 52 (1), 83-88 (2017).
- Pangas, S. A., Saudye, H., Shea, L. D., Woodruff, T. K. Novel approach for the three-dimensional culture of granulosa cell-oocyte complexes. Tissue Engineering. 9 (5), 1013-1021 (2003).
- Shen, P., et al. A new three-dimensional glass scaffold increases the in vitro maturation efficiency of buffalo (Bubalus bubalis) oocyte via remodeling the extracellular matrix and cell connection of cumulus cells. Reproduction in Domestic Animals. 55 (2), 170-180 (2020).
- Kniazeva, E., et al. Primordial follicle transplantation within designer biomaterial grafts produce live births in a mouse infertility model. Scientific Reports. 5, 17709 (2015).
- Tian, J., Fu, N., Chen, X. D., Shen, W. Respirable liquid marble for the cultivation of microorganisms. Colloids and Surfaces B Biointerfaces. 106, 187-190 (2013).
- Arbatan, T., Al-Abboodi, A., Sarvi, F., Chan, P. P., Shen, W. Tumor inside a pearl drop. Advanced Healthcare Materials. 1 (4), 467-469 (2012).
- Sarvi, F., et al. Cardiogenesis of embryonic stem cells with liquid marble micro-bioreactor. Advanced Healthcare Materials. 4 (1), 77-86 (2015).
- Ledda, S., et al. A novel technique for in vitro maturation of sheep oocytes in a liquid marble microbioreactor. Journal of Assisted Reproduction and Genetics. 33 (4), 513-518 (2016).
- Ooi, C. H., Nguyen, N. T. Manipulation of liquid marbles. Microfluid Nanofluid. 19, 483-495 (2015).
- Lin, P., Rui, R. Effects of follicular size and FSH on granulosa cell apoptosis and atresia in porcine antral follicles. Molecular Reproduction Development. 77 (8), 670-678 (2010).
- Eppig, J. J., O’Brien, M. J. Development in vitro of mouse oocytes from primordial follicles. Biology of Reproduction. 54 (1), 197-207 (1996).
- Dolmans, M. M., et al. Evaluation of Liberase, a purified enzyme blend, for the isolation of human primordial and primary ovarian follicles. Human Reproduction. 21 (2), 413-420 (2006).
- Reader, K. L., Stanton, J. A., Juenge, J. L. The role of oocyte organelles in determining developmental competence. Biology. 6, 35-57 (2017).
- Shikanov, A., Xu, M., Woodruff, T. K., Shea, L. D. A method for ovarian follicle encapsulation and culture in a proteolytically degradable 3-dimensional system. Journal of Visualized Experiments. (49), e2695 (2011).
- Aussillous, P., Quere, D. Liquid Marbles. Nature. 411, 924-927 (2001).
- Serrano, M. C., Nardecchia, S., Gutiérrez, M. C., Ferrer, M. L., del Monte, F. Mammalian cell cryopreservation by using liquid marbles. ACS Applied Materials & Interfaces. 7, 3854-3860 (2015).
- Arbatan, T., Li, L., Tian, J., Shen, W. Liquid marbles as micro-bioreactors for rapid blood typing. Advance Healthcare Materials. 1 (1), 80-83 (2012).
- Brevini, T. A. L., Manzoni, E. F. M., Ledda, S., Gandolfi, F., Turksen, K. Use of a Super-hydrophobic Microbioreactor to Generate and Boost Pancreatic Mini Organoids. Organoids. Methods in Molecular Biology. , 291-299 (2017).

