Modulating Shape of Polyester Based Polymersomes using Osmotic Pressure
Summary
Polymersomes are self-assembled polymeric vesicles that are formed in spherical shapes to minimize Gibb’s Free Energy. In the case of drug delivery, more elongated structures are beneficial. This protocol establishes methods to create more rod-like polymersomes, with elongated aspect ratios, using salt to induce osmotic pressure and reduce internal vesicle volumes.
Abstract
Polymersomes are membrane-bound, bilayer vesicles created from amphiphilic block copolymers that can encapsulate both hydrophobic and hydrophilic payloads for drug delivery applications. Despite their promise, polymersomes are limited in application due to their spherical shape, which is not readily taken up by cells, as demonstrated by solid nanoparticle scientists. This article describes a salt-based method for increasing the aspect ratios of spherical poly(ethylene glycol) (PEG)- based polymersomes. This method can elongate polymersomes and ultimately control their final shape by adding sodium chloride in post-formation dialysis. Salt concentration can be varied, as described in this method, based on the hydrophobicity of the block copolymer being used as the base for the polymersome and the target shape. Elongated nanoparticles have the potential to better target the endothelium in larger diameter blood vessels, like veins, where margination is observed. This protocol can expand therapeutic nanoparticle applications by utilizing elongation techniques in tandem with the dual-loading, long-circulating benefits of polymersomes.
Introduction
Shape modulation is a relatively new and efficient way to improve nanoparticle-mediated drug delivery. Not only does the change in morphology increase the surface area of particles, which in turn allows for a greater carrying capacity, but it also has implications across the board to improve stability, circulation time, bioavailability, molecular targeting, and controlled release1. Polymersomes, the nanoparticle of focus in this method, tend to thermodynamically self-assemble into a spherical shape, which has proven to be impractical in cellular uptake and is more easily detected in the immune system as a foreign body. Being able to elongate the structure into a prolate or a rod will allow the drug carrier to evade macrophages by mimicking native cells and more successfully deliver to their desired target2,3,4,5,6,7. The significant benefits of polymersomes, including membrane-bound protection of payloads, stimuli-responsiveness of the membrane, and dual encapsulation of hydrophilic and hydrophobic drugs8,9,10, that make them strong candidates for drug delivery are maintained during shape modulation.
There are many different methods in modulating polymersomes' shapes, and each comes with its respective advantages and disadvantages. However, most of these methods fall into two categories: solvent-driven and salt-driven osmotic pressure change11. Both approaches aim to overcome the bending energy present after polymersomes are formed in a spherical equilibrium shape. By introducing an osmotic pressure gradient, polymersomes can be forced to bend into elongated structures despite strong bending energies11,12.
The solvent-based method explores shape change inspired by the work of Kim and van Hest13. They plasticized polymersomes in an organic solvent and water mixture to trap the organic solvents in the vesicle membrane and drive water out of the vesicle core. Eventually, the particle's internal volume is so low that it elongates. While this method has shown promise, it lacks practicality. This method requires different solvents for each individual polymeric backbone involved in the modulation. Therefore, it is not widely applicable to promote shape change. Conversely, the salt-based method is uniform and utilizes one universal driver that can introduce osmotic pressure to many block copolymer-based polymersomes.
This project utilizes the salt-based method introduced by L'Amoreaux et al14. This protocol involves two rounds of dialysis. One aims at purifying and solidifying poly(ethylene glycol)-b-poly(lactic acid) (PEG-PLA) polymersomes by removing organic solvent that may have gotten trapped in the bilayer during production, and one that promotes the shape change. The second dialysis step introduces a 50 mM NaCl solution that creates an osmotic pressure gradient to drive the shape change. This method is supported by Salva et al., who note that hypertonic stress in a solution will cause the vesicle to shrink15. This method builds on a previously published method14 looking at two different polyester-based polymersomes and various salt gradients from 50-200 mM NaCl. Polyesters are used due to their biocompatibility and biodegradation. The salt gradient has varying effects on the shape depending on the hydrophobicity of the block copolymer backbone. It can be used to create prolates, rods, and stomatocytes. This salt-driven method was chosen because of the ease of replication and experimental versatility.
Protocol
1. Spherical polymersome formation using a solvent injection method
- Dissolution of polyesters in organic solvent
NOTE: Only one polyester should be dissolved in its respective organic solvent at a time to form polymersomes.- Dissolve polyesters PEG-PLA or PEG-b-poly(lactic-co-glycolic acid) (PEG-PLGA) in dimethyl sulfoxide (DMSO) at a concentration of 1.5% weight. Specifically, dissolve 0.015 g of selected polyester in 1 mL of DMSO (15 mg/mL). Full dissolution of the polymer may require periods of up to 15 min of vortexing.
- While the polyester is dissolving in organic solvent, set up the solvent injection apparatus according to Figure 1.
- Place a stir plate directly below the vertical syringe pump. Place a 5 mL glass vial with 1 mL of Type II deionized water and a miniature stir bar on the stir plate.
- Adjust the syringe pump's height to allow for the tip of the needle to be fully immersed in Type II deionized water.
- Set the infusion rate of the syringe pump to 5 µL/min.
NOTE: If a small volume syringe pump is used, the adapter with the syringe can be set up on a ring stand. If a large volume syringe pump is used, the pump can be placed vertically on a lab jack to adjust the height.
- Perform the solvent injection by drawing the organic solvent and polyester solution (step 1.1.1) into a 27 G needle with a ½" needle length.
- Place the needle into the syringe pump and make sure it is entirely secure. Adjust the pusher block to touch the syringe plunger's end.
- Start the stir plate so that the water is spinning at 100 rpm, and then start the syringe pump.
- Once the syringe pump has fully infused the organic solvent and polymer into the stirring water, remove from the stir bar and cap the glass vial.
- Characterize the polymersomes via dynamic light scattering (DLS).
- Take 1 mL of water, now with a small percentage of organic solvent and polymer, and place in a 1 mL cuvette.
- Using the settings from Table 1, perform DLS by placing a 1 mL cuvette into the system and set up the run. Read and collect the polymersome intensity-weighted diameter and polydispersity index (PDI).
NOTE: A plastic cuvette works fine in this case, as the amount of organic solvent is very low. However, a glass cuvette will work as well if any concerns exist.
- Confirm spherical polymersome formation using Transmission Electron Microscopy (TEM) and Scanning Electron Microscopy (SEM).
- Optimize TEM and SEM protocols based on the equipment available. Representative results were obtained at 120 kV in the TEM and 5.0 kV in the SEM.
- If polymersomes are not visible using EM, apply uranyl acetate as a background stain.
NOTE: Details on TEM and SEM imaging for the shape modulation of polyester-based polymersomes can be found in reference14. Information on electron microscopy techniques for soft nanoparticles is detailed in reference16.
2. Dialysis to remove organic solvent
- Wash a 300 kDa dialysis membrane according to protocols provided by the manufacturer.
- Add 1 mL of polymersome solution into the reservoir of the dialysis device.
- Place the dialysis device in a 250 mL beaker with 150 mL of Type II deionized water on a stir plate. Set the stir plate to a speed that allows for gentle movement of the dialysis device and leave to stir overnight.
NOTE: If a vortex is formed during dialysis, the speed needs to be decreased. - After the dialysis is completed, extract the 1 mL polymersome solution from the dialysis device. Characterize the polymersome solution, following step 1.5.
NOTE: Collection of this information is relevant to determine the success of the shape modulation protocol, as one should be able to identify an increase in PDI if the polymersome has been elongated.
3. Dialysis against salt gradients
- Create 150 mL of desired salt buffer, with either 50 mM, 100 mM, or 200 mM concentration of sodium chloride based on the final desired polymersome properties. In general, increased salt concentration leads to increased polymersome elongation.
- Take the polymersome solution that was dialyzed and characterized and re-place into the dialysis device. Place the loaded dialysis device into 150 mL of the desired salt solution and allow to gently stir for 18 h.
NOTE: Shape-modulated polymersomes can be stored and maintain their shape in an isotonic solution for periods of up to 7 days.
4. Shape modulated polymersome characterization
- After the shape modulation, perform polymersome characterization via DLS, TEM, and SEM. If polymersomes are not visible using EM, apply uranyl acetate as a background stain.
- Perform DLS measurements as described in step 1.5, paying particular attention to PDI measurements compared to spherical polymersomes, as a change in PDI suggests effective shape change in polymersomes.
- Ensure the use of appropriate controls for imaging, especially non-shape modulated polymersomes, to ensure the method's success.
Representative Results
Table 2 presents expected results when following the protocol step 1. Note that DMSO is used as a solvent for both PEG-PLA and PEG-PLGA in polymersomes formation. Deviation from this solvent is possible, as other water-miscible solvents will dissolve the copolymers but is expected to change results. It is expected that PDI will be less than 0.2, indicating the formation of monodisperse polymersomes17. Note that increasing hydrophobicity leads to increased deviation in both polymersome diameter and PDI. If upon running the protocol, polymersome diameters vary dramatically from those reported in this table; the most typical culprit is a low concentration of nanoparticles, shown by a low count rate for that sample.
Figure 2 demonstrates non-shape modulated polymersomes' appearance before the addition of salt gradients. Presented here are representative results from PEG-PLA based polymersomes following protocol step 1. Regardless of the block copolymer used, the TEM should indicate an overall spherical structure, with a thicker exterior line, indicative of a membrane. Without NaCl, PEG-PLA polymersomes present as spherical structures in SEM with a brush-like exterior layer of PEG observed through the rough surface presentation.
Figure 3 shows expected changes in polymersomes post-dialysis (step 2). Regardless of the concentration of organic solvent used in polymersome formation, one-hour dialysis in water to remove the solvent will lead to the same overall average diameter, with solvent removal decreasing the polymersome diameter. When larger initial concentrations of organic solvent are used, larger diameter decreases are expected.
Figure 4 provides representative DLS results post-shape change (step 4). Figure 4A shows that when making PEG-PLA polymersomes, modest changes in PDI are expected, which could indicate a change in shape but require imaging to confirm what specific shapes are forming with the polymersomes. Dialyzing PEG-PLA polymersomes against 50 mM NaCl can lead to the formation of prolates with aspect ratios around 2, although this is not a consistent result, demonstrated by large deviation in PDI14. Larger concentrations of salt can lead to the formation of more stomatocyte-like shapes, which is consistent with the current literature18. When dialyzing PEG-PLGA polymersomes, which are slightly more hydrophobic than PEG-PLA polymersomes, against salt, the increase in PDI is more consistent with elongation, with all explored salt gradients leading to an increase in PDI. Having a change in PDI (ΔPDI) above one is encouraging towards the formation of elongated polymersomes. Again, imaging should be used to confirm which shapes are being created. Figure 4B shows that similar results should be observed when using a 50 mM salt gradient to cause a shape change, regardless of polyester hydrophobicity, while 100 mM and 200 mM NaCl salt gradients display the direct trend that ΔPDI increases with increasing polyester hydrophobicity (i.e., PEG-PLGA should have a higher ΔPDI after salt dialysis than PEG-PLA).
Figure 5 provides some examples of polymersome shapes expected when running the protocol. Presented are representative TEM images of shape-modulated PEG-PLA polymersomes after dialysis in 0, 50, 100, or 200 mM NaCl. Recall that self-assembled polymersomes are less controllable than solid particles. Therefore, it is expected to see deviations in sizes and shapes in each sample, which are not observed when modulating the shape of more solid nanoparticles5,7,19,20,21. Knowledge from both Figure 4 and Figure 5 demonstrates this for PEG-PLA polymersomes dialyzed with 50 mM of NaCl; this sample presents with stomatocytes and elongated rods. As salt is increased to 100 mM, an increased number of rods formation with a decreased number of stomatocytes is observed. Finally, with dialysis against 200 mM NaCl, PEG-PLA polymersomes more consistently form prolates with modest aspect ratios (between 2 and 3). Running this protocol will lead to a distribution of nanoparticle shapes, as is the nature of self-assembled nanomedicine.
| Setting | Value |
| Material Refractive Index | Set to your material; 1.450 for polymer |
| Dispersant | [NaCl] used; complex solvent set up in DLS software |
| Temperature | 25 °C |
| Equilibration Time | 120 s |
| Measurement angle | 173 ° Back Scatter |
| Measurement duration | Automatic |
| Data Processing | General purpose |
Table 1: Parameters to use when measuring the size and polydispersity index for polymersomes via dynamic light scattering before and after shape modulation.
| Polyester Block Co-Polymer | Diameter | PDI | ||||
| d, nm | – | |||||
| PEG-b-PLA | 202.5 | ± | 12.0 | 0.06 | ± | 0.06 |
| PEG-b-PLGA | 139.6 | ± | 25.9 | 0.16 | ± | 0.06 |
| PEG-b-PCL | 320.9 | ± | 98.8 | 0.14 | ± | 0.06 |
Table 2: Average polymersome diameter and polydispersity index after solvent injection. This data is typical for PEG-PLA and PEG-PLGA polymersomes after solvent injection, following step 1.5. in the protocol.

Figure 1: Solvent injection apparatus. Created with BioRender. Please click here to view a larger version of this figure.

Figure 2: TEM and SEM images of spherical PEG-PLA polymersomes pre-salt dialysis. For TEM, Spherical particles were dried from a 0 mM NaCl suspension and stained with uranyl acetate. TEM Images were taken at 120 kV/60,000x direct magnification. SEM images were taken at 5.0 kV. Images are adapted from previously published results14. Please click here to view a larger version of this figure.
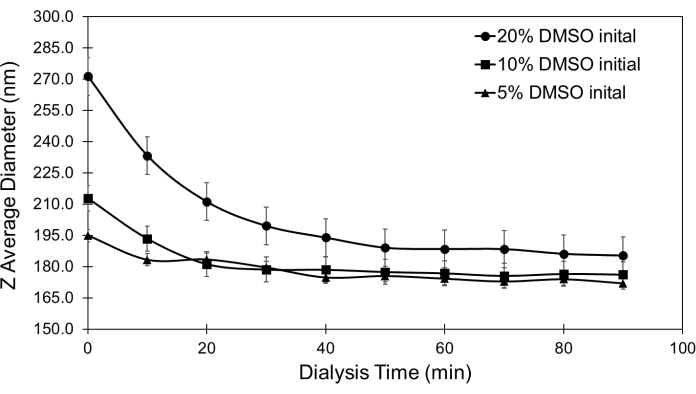
Figure 3: Spherical PEG-PLA based polymersome Z-Average diameter during dialysis. Dialysis removes organic solvent, which solidifies polymersome membranes and decreases polymersome diameter, as demonstrated for PEG-PLA based polymersomes. This figure is adapted from a pre-print22 and published in Nanotechnology14. Please click here to view a larger version of this figure.
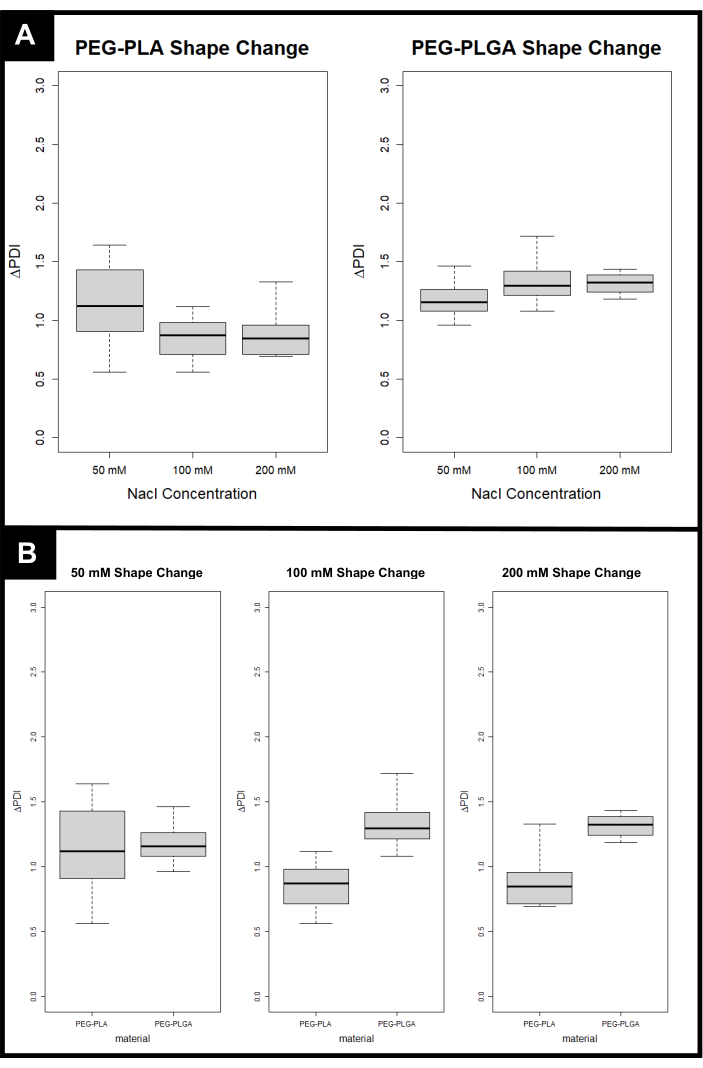
Figure 4: Expected PDI change (ΔPDI) for each polyester-based polymersomes after the addition of salt gradients. (A) ΔPDI versus Concentration of NaCl for each Polyester-Based Polymersome. (B) ΔPDI versus Polyester for each Concentration of NaCl. Please click here to view a larger version of this figure.
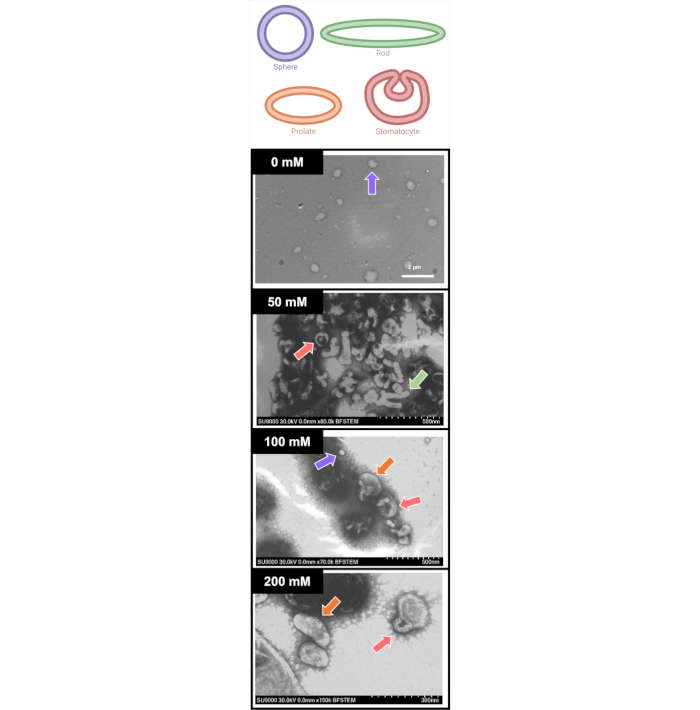
Figure 5: Sample TEM images of shape modulated PEG-PLA polymersomes after being dialyzed against 0, 50, 100, and 200 mM NaCl. In order to aid in the visualization of shapes formed through the salt dialysis, a key is provided at the top of the figure, denoting potential shapes of spheres (purple), rods (green), prolates (orange), and stomatocytes (red). As is usual for self-assembled systems, a variety of shapes and sizes are formed. Before the salt dialysis, spheres are consistently observed. The use of 50 mM and 100 mM sodium chloride gradients leads to a wide variety of shapes, including stomatocytes (50 and 100 mM), rods (50 mM), spheres (100 mM) and prolates (100 mM) denoted by arrows colored according to the key provided. Finally, the use of a 200 mM sodium chloride gradient leads to the formation of mainly prolate shapes, with some stomatocytes. Please click here to view a larger version of this figure.
Discussion
Self-assembled systems are notoriously uncontrollable. Their final properties, including size, shape, and structure, are driven by the chosen amphiphile's hydrophobic properties and the solvent environment selected. Amphiphilic block copolymers tend towards spherical shapes, which minimizes Gibb's free energy and leads to the thermodynamic equilibrium23, thus forming polymersomes. Because of their equilibrium nature, polymersomes are significantly more challenging to elongate or alter in shape and, therefore, less studied than solid nanoparticle counterparts. The solvent-driven method has been extensively studied in tandem with PEG-b-polystyrene (PS) based polymersomes12,13,24,25,26,27. However, due to the need to re-optimize protocols for a solvent-based approach for each block copolymer used, this method has not been widely adopted to change the shapes of polymersomes made from alternative materials. Other methods used to modulate polymersome shape are highly specific to the polymersome chemistry; some introduce perylene groups24,28 or other crosslinkers29,30,31 to initiate the desired collapse and elongation. The specificity of the approach to the polymeric material limits its widespread applicability. This method uses salt as the driver for creating an osmotic pressure gradient between the internal, vesicle-encapsulated water and the external saltwater. This can be more universally applied than the alternative solvent-driven method, as various polymers require various solvents to ensure success in the shape modulation. Salt-based shape modulation of polymersomes has worked with PEG-b-PS18, poly(dimethyl siloxane)-g-poly(ethylene oxide)15, and now, with a recent publication, polyester-based polymersomes14. The chemistry-independent success of this method is promising towards the development of more targeted drug delivery systems. Notably, the applicability of this approach to the use of polyesters could have widespread biologic benefits, as these materials are biodegradable.
The first dialysis step is crucial. The solvent injection method of polymersome formation utilizes water-miscible solvents that can become trapped in the hydrophobic membrane of polymersomes during formation, making their membranes more permeable to things like salt. The initial dialysis of formed polymersomes in water ensures that the solvent has been removed from the polymer backbone, demonstrated by a decrease in polymersome diameter (Figure 3). This makes the polymersome less permeable to salt molecules introduced in the second dialysis step, which forms the pressure gradient across the polymersome membrane.
When creating shape-modulated polymersomes, it is expected that each sample will possess polymersomes that are of various shapes and sizes. The benefits of membrane-bound drug delivery, including but not limited to payload protection, extended circulation time, and increased cellular uptake, outweigh the drawbacks of polydispersity. This fact necessitates that images are taken in tandem with DLS data each time polymersomes are prepared. It has been noted that an increase in PDI denotes a shift from spherical to non-spherical particles32, but a direct correlation between ΔPDI and specific shape has not yet been identified. This is especially important with lower salt concentrations, where there is a large batch to batch variability in ΔPDI (Figure 4).
This method has only been applied to modulating the shape of the two polyester block copolymers that form polymersomes presented in this paper (PEG-PLA and PEG-PLGA). Other types of PEG-based polymersome systems have been altered for the shape via other salt gradients or solvent gradients, which are reviewed elsewhere33, though there has not been much study into the shape modulation of non-PEG based polymersome systems. Polymersomes are the equilibrium state of PEG-PLA and PEG-PLGA in this paper due to their molecular weights, which provide hydrophilic fractions that lead to membrane bound vesicles10. Thus, following the method directly with an alternative polymer may yield different results. In general, this method can be used to control the shapes of notoriously uncontrollable, self-assembled polymersomes formed from polyester block copolymers.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This project was funded in part by the National Institutes of Health Project number 5P20GM103499-19 through the Student Initiated Research Project Program. This work was also partially supported by Clemson's Creative Inquiry Program. We also acknowledge Nicholas L'Amoreaux and Aon Ali who initially worked on creating this protocol, publishing their first paper cited here14.
Materials
| 15*45 vials screw thread w/cap attached | Fisherbrand | 9609104000 | |
| Dimethyl Sulfoxide | Fisher Chemical | D128-1 | |
| Dimethyl Sulfoxide | BDH | BDH1115-1LP | |
| Isoremp stirrers, hotplates, and stirring hotplates | Fisher scientific | CIC00008110V19 | |
| LEGATO 130 SYRINGE PUMP | kd Scientific | 788130 | |
| PEG(1000)-b-PLA(5000), Diblock Polymer | Polysciences Inc | 24381-1 | note the molecular weights when replicating |
| Poly(ethylene glycol) (2000) Methyl ether-block-poly(lactide-co-glycolide) (4500) | Sigma aldrich | 764825-1G | note the molecular weights when replicating |
| Single-Use Syringe/BD PrecisionGlide Needle combination, sterile, BD medical | BD medical | BD305620 | Tuberculin |
| Sodium Chloride | BDH | BDH9286 | |
| Zetasizer Nano ZS | Malvern |
References
- Varma, L. T., et al. Recent advances in self-assembled nanoparticles for drug delivery. Current Drug Delivery. 17 (4), 279-291 (2020).
- Salatin, S., Maleki Dizaj, S., Yari Khosroushahi, A. Effect of the surface modification, size, and shape on cellular uptake of nanoparticles. Cell Biology International. 39 (8), 881-890 (2015).
- Baio, J. E., et al. Reversible activation of pH-sensitive cell penetrating peptides attached to gold surfaces. Chemical Communications. 51 (2), 273-275 (2015).
- Zhou, Y., et al. Mesoporous silica nanoparticles for drug and gene delivery. Acta Pharmaceutica Sinica B. 8 (2), 165-177 (2018).
- Champion, J. A., Mitragotri, S. Role of target geometry in phagocytosis. Proceedings of the National Academy of Sciences of the United States of America. 103 (13), 4930-4934 (2006).
- Banerjee, A., Qi, J., Gogoi, R., Wong, J., Mitragotri, S. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. Journal of Controlled Release. 238, 176-185 (2016).
- Kolhar, P., et al. Using shape effects to target antibody-coated nanoparticles to lung and brain endothelium. Proceedings of the National Academy of Sciences. 110 (26), 10753-10758 (2013).
- Meng, F., Zhong, Z., Feijen, J. Stimuli-responsive polymersomes for programmed drug delivery. Biomacromolecules. 10 (2), 197-209 (2009).
- Iqbal, S., Blenner, M., Alexander-Bryant, A., Larsen, J. Polymersomes for therapeutic delivery of protein and nucleic acid macromolecules: from design to therapeutic applications. Biomacromolecules. 21 (4), 1327-1350 (2020).
- Discher, D. E., Ahmed, F. Polymersomes. Annual review of biomedical engineering. 8, 323-341 (2006).
- Seifert, U., Berndl, K., Lipowsky, R. Shape transformations of vesicles: Phase diagram for spontaneous- Curvature and bilayer-coupling models. Physical Review A. 44 (2), 1182-1202 (1991).
- Rikken, R. S. M., et al. Shaping polymersomes into predictable morphologies via out-of-equilibrium self-assembly. Nature Communications. 7, 1-7 (2016).
- Kim, K. T., et al. Polymersome stomatocytes: Controlled shape transformation in polymer vesicles. Journal of the American Chemical Society. 132 (36), 12522-12524 (2010).
- L’Amoreaux, N., Ali, A., Iqbal, S., Larsen, J. Persistent prolate polymersomes for enhanced co-delivery of hydrophilic and hydrophobic drugs. Nanotechnology. 31 (17), 175103 (2020).
- Salva, R., et al. Polymersome shape transformation at the nanoscale. ACS Nano. 7 (10), 9298-9311 (2013).
- Skoczen, S. L., Stern, S. T. Characterization of Nanoparticles Intended for Drug Delivery. Methods in Molecular Biology. 1682, (2018).
- Bhattacharjee, S. DLS and zeta potential – What they are and what they are not. Journal of Controlled Release. 235, 337-351 (2016).
- Men, Y., Li, W., Lebleu, C., Sun, J., Wilson, D. A. Tailoring polymersome shape using the Hofmeister effect. Biomacromolecules. 21 (1), 89-94 (2020).
- Decuzzi, P., et al. Size and shape effects in the biodistribution of intravascularly injected particles. Journal of Controlled Release. 141 (3), 320-327 (2010).
- Liu, Y., Tan, J., Thomas, A., Ou-Yang, D., Muzykantov, V. R. The shape of things to come: Importance of design in nanotechnology for drug delivery. Therapeutic Delivery. 3 (2), 181-194 (2012).
- Tao, L., et al. Shape-specific polymeric nanomedicine: Emerging opportunities and challenges. Experimental Biology and Medicine. 236 (1), 20-29 (2011).
- L’Amoreaux, N., Ali, A., Iqbal, S., Larsen, J. Persistent prolate polymersomes for enhanced co-delivery of hydrophilic and hydrophobic drugs. BioRxiv. , 796201 (2019).
- Chandler, D. Interfaces and the driving force of hydrophobic assembly. Nature. 437 (7059), 640-647 (2005).
- Wong, C. K., Mason, A. F., Stenzel, M. H., Thordarson, P. Formation of non-spherical polymersomes driven by hydrophobic directional aromatic perylene interactions. Nature Communications. 8 (1), (2017).
- Abdelmohsen, L. K. E. A., et al. Shape characterization of polymersome morphologies via light scattering techniques. Polymer. 107, 445-449 (2016).
- Men, Y., et al. Nonequilibrium Reshaping of polymersomes via polymer addition. ACS Nano. 13 (11), 12767-12773 (2019).
- Meeuwissen, S. A., Kim, K. T., Chen, Y., Pochan, D. J., van Hest, J. C. M. Controlled shape transformation of polymersome stomatocytes. Angewandte Chemie. 123 (31), 7208-7211 (2011).
- Wong, C. K., et al. Faceted polymersomes: A sphere-to-polyhedron shape transformation. Chemical Science. 10 (9), 2725-2731 (2019).
- Chidanguro, T., Ghimire, E., Simon, Y. C. Shape-transformation of polymersomes from glassy and crosslinkable ABA triblock copolymers. Journal of Materials Chemistry B. 8 (38), 8914-8924 (2020).
- Van Oers, M. C. M., Rutjes, F. P. J. T., Van Hest, J. C. M. Tubular polymersomes: A cross-linker-induced shape transformation. Journal of the American Chemical Society. 135 (44), 16308-16311 (2013).
- Che, H., et al. Pathway dependent shape-transformation of azide-decorated polymersomes. Chemical Communications. 56 (14), 2127-2130 (2020).
- Haryadi, B. M., et al. Nonspherical nanoparticle shape stability is affected by complex manufacturing aspects: Its implications for drug delivery and targeting. Advanced Healthcare Materials. 8 (18), 11-13 (2019).
- Katterman, C., Pierce, C., Larsen, J. Combining nanoparticle shape modulation and polymersome technology in drug delivery. ACS Applied Bio Materials. , (2021).

