Direct and Indirect Culture Methods for Studying Biodegradable Implant Materials In Vitro
Summary
We introduce three methods of direct culture, direct exposure culture, and exposure culture for evaluating the in vitro cytocompatibility of biodegradable implant materials. These in vitro methods mimic different in vivo cell-implant interactions and can be applied to study various biodegradable materials.
Abstract
Over the past several decades, biodegradable materials have been extensively explored for biomedical applications such as orthopedic, dental, and craniomaxillofacial implants. To screen biodegradable materials for biomedical applications, it is necessary to evaluate these materials in terms of in vitro cell responses, cytocompatibility, and cytotoxicity. International Organization for Standardization (ISO) standards have been widely utilized in the evaluation of biomaterials. However, most ISO standards were originally established to assess the cytotoxicity of nondegradable materials, thus providing limited value for screening biodegradable materials.
This article introduces and discusses three different culture methods, namely, direct culture method, direct exposure culture method, and exposure culture method for evaluating the in vitro cytocompatibility of biodegradable implant materials, including biodegradable polymers, ceramics, metals, and their composites, with different cell types. Research has shown that culture methods influence cell responses to biodegradable materials because their dynamic degradation induces spatiotemporal differences at the interface and in the local environment. Specifically, the direct culture method reveals the responses of cells seeded directly on the implants; the direct exposure culture method elucidates the responses of established host cells coming in contact with the implants; and the exposure culture method evaluates the established host cells that are not in direct contact with the implants but are influenced by the changes in the local environment due to implant degradation.
This article provides examples of these three culture methods for studying the in vitro cytocompatibility of biodegradable implant materials and their interactions with bone marrow-derived mesenchymal stem cells (BMSCs). It also describes how to harvest, passage, culture, seed, fix, stain, characterize the cells, and analyze postculture media and materials. The in vitro methods described in this article mimic different scenarios of the in vivo environment, broadening the applicability and relevance of in vitro cytocompatibility testing of different biomaterials for various biomedical applications.
Introduction
For decades, biodegradable materials have been extensively studied and used in biomedical applications such as orthopedic1,2, dental3,4, and craniomaxillofacial5 applications. Unlike permanent implants and materials, biodegradable metals, ceramics, polymers, and their composites gradually degrade in the body over time via different chemical reactions in the physiological environment. For example, biodegradable metals such as magnesium (Mg) alloys1,6,7 and zinc (Zn) alloys8,9 are promising materials for bone fixation devices. Their biodegradability could eliminate the necessity for secondary surgeries to remove the implants after bone healing. Biodegradable ceramics such as calcium phosphate cements (CPCs) have shown exciting potential for the treatment of osteoporotic vertebral compression fractures in percutaneous kyphoplasty10. The CPCs provide mechanical support for the fractured vertebral body and gradually degrade after the fracture has healed.
Biodegradable polymers, such as some polysaccharides and polyesters, have also been widely explored for biomedical applications. For instance, chitosan hydrogel as a biodegradable polysaccharide has exhibited its capabilities for preventing infection and regenerating skin tissue11. Poly-L-lactic acid (PLLA), poly(glycolic acid) (PGA), and poly(lactic-co-glycolic acid) (PLGA) are widely studied polyesters for fabricating 2D or 3D porous scaffolds for tissue engineering applications12,13,14. Moreover, composite materials integrate two or more phases of metals, ceramics, and polymers to provide advanced functions for a wide range of biomedical applications15,16,17. For example, PLGA and calcium phosphate composites can be used to fabricate biodegradable scaffolds for applications such as repairing skull bone defects18. These biodegradable scaffolds and implants could support and promote the growth of cells and tissues and then gradually degrade in the body over time.
As shown in Supplemental Table 1, different biodegradable materials may have varied degradation mechanisms, products, and rates. For example, magnesium alloys, such as Mg-2 wt % Zn-0.5 wt % Ca (ZC21)1, Mg-4 wt% Zn-1 wt% Sr (ZSr41)19, and Mg-9 wt% Al-1 wt% Zinc (AZ91)20, degrade by reacting with water, and their degradation products mainly include Mg2+ ions, OH– ions, H2 gas, and mineral depositions. The degradation rate for biodegradable metals varies depending on their different compositions, geometries, and degradation environments. For example, Cipriano et al.19 reported that ZSr41 wires (Ø1.1 × 15 mm) lost 85% mass while pure Mg wires with the same geometry lost 40% mass after being implanted in the rat tibiae for 47 days. Biodegradable ceramic materials such as hydroxyapatite (HA) and β-tricalcium phosphate (β-TCP) can degrade via solution-driven extracellular liquid dissolution or break down into small particles and then degrade via both extracellular liquid dissolution and cell-mediated resorption processes. The degradation products of these calcium phosphate-based ceramics may include Ca2+ ions, (PO4)3- ions, OH– ions, and mineral depositions21. The degradation rate for calcium phosphate ceramics is significantly affected by their crystal structures. For instance, Van Blitterswijk et al.22 reported that HA with 40 vol.% micropores did not lose any mass while β-TCP with 40 vol.% micropores lost 30 ± 4% mass after being implanted in the tibiae of rabbits for 3 months. Polymers such as PLGA14,23 may degrade due to hydrolysis of the ester linkages in the presence of water, and the degradation products mainly include lactic and glycolic acids. It may take one month for PLGA 50/50 and several months for PLGA 95/5 to achieve complete degradation24.
Cell response and cytocompatibility testing are critical to evaluate and screen these biodegradable implant materials for biomedical applications. However, current standards from the International Organization for Standardization (ISO), such as ISO 10993-5:2009 "Biological evaluation of medical devices-Part 5 Tests for in vitro cytotoxicity", were initially designed to assess the cytotoxicity of nondegradable biomaterials such as Ti alloys and Cr-Co alloys in vitro25. Specifically, ISO 10993-5:2009 only covers the in vitro cytotoxicity tests of the extract, direct contact, and indirect contact tests. In the extract test, the extract is prepared by immersing samples in extraction fluids such as culture media with serum and physiological saline solutions under one of the standard time and temperature conditions. The collected extract or dilution is then added into the cell culture to study cytotoxicity. For the direct contact test, direct contact between sample and cells is achieved by placing the test sample on the established (adhered) cell layer. In the indirect contact test, the culture media containing serum and melted agar is pipetted to cover the established cells. The sample is then placed onto the solidified agar layer with or without a filter.
The ISO standards have shown some limitations when applied to evaluate biodegradable materials in vitro. Unlike nondegradable materials, the degradation behaviors of biodegradable materials are dynamic and may change at a different time or in varied environmental conditions (e.g., temperature, humidity, media composition, and cell type). The extract test only evaluates the cytotoxicity of the degradation products of the material and does not reflect the dynamic process of sample degradation. Both direct and indirect contact tests of the ISO standard only characterize the interactions between the established cells and samples. Moreover, in the indirect contact test, the materials and cells are in different microenvironments that do not reflect the in vivo environment and do not capture the dynamic degradation of biodegradable materials.
The objective of this article is to introduce and discuss the cytocompatibility testing methods for various biodegradable implant materials to address the abovementioned limitations of the methods described in the current ISO standards. The methods presented in this article consider the dynamic degradation behavior of implant materials and the different circumstances of cell-material interactions in vivo. Specifically, this article provides three cytocompatibility testing methods, namely direct culture, direct exposure culture, and exposure culture for various biodegradable materials, including biodegradable polymers, ceramics, metals, and their composites for medical implant applications.
In the direct culture method, cells suspended in the culture media are directly seeded on the samples, thus evaluating the interactions between newly seeded cells and the implants. In the direct exposure culture, the samples are placed directly on the established cell layer to mimic the interactions of implants with established host cells in the body. In the exposure culture, the samples are placed in their respective well inserts and then introduced to the culture wells with established cells, which characterizes the responses of established cells to the changes in the local environment induced by implant degradation when they have no direct contact with implants. The direct culture and direct exposure culture methods evaluate the cells directly or indirectly in contact with the implant materials in the same culture well. The exposure culture characterizes the cells indirectly in contact with the implant materials within a prescribed distance in the same culture well.
This article presents a detailed description of the cytocompatibility testing for different biodegradable materials and their interactions with model cells, that is, bone marrow-derived mesenchymal stem cells (BMSCs). The protocols include the harvesting, culturing, seeding, fixing, staining, and imaging of the cells, along with analyses of postculture materials and media, which apply to a variety of biodegradable implant materials and a wide range of cell types. These methods are useful for screening biodegradable materials for different biomedical applications in terms of cell responses and cytocompatibility in vitro.
Protocol
This protocol was approved by the Institutional Animal Care and Use Committee (IACUC) at the University of California at Riverside (UCR) for cell and tissue harvesting. A 12-week-old female Sprague-Dawley (SD) rat is shown as an example in the video. Younger female and male rats are preferred.
1. Cell culture preparation
NOTE: The three culture methods described in this article are generally applicable for different cell types that are adherent. Here, BMSCs harvested from rat weanlings will be introduced as an example for cell culture preparation. Depending on their relevance for specific medical applications, different cell types may be utilized, including primary cells harvested from animals or human donors and cell lines from a cell/tissue bank.
- Harvesting BMSCs from rat weanlings
NOTE: The schematic diagram in Figure 1 shows the steps to harvest BMSCs from rat weanlings.- Euthanize the Sprague Dawley (SD) rat by CO2 inhalation.
- Remove the skin and muscle and connective tissues to dissect the femur out of the euthanized rat. Place the femoral bones in a 15 mL conical tube (polypropylene) containing cell culture medium. Place the conical tubes on ice until the time of performing cell extraction.
NOTE: Tibia also can be used to harvest BMSCs. The cell culture medium is Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin (P/S). - Transfer the bones to a Petri dish in the biological safety cabinet. Cut the ends of the bone using a surgical blade and flush the bone marrow into a 50 mL conical tube (polypropylene) by washing the bone marrow cavity with cell culture media using a syringe with a 25½ G needle.
NOTE: Insert a syringe with an 18 G needle into the media with bone marrow. Slowly and gently, take up and dispense the media to break apart the large chunk of marrow until no visible cell/tissue agglomerates are present. - Filter the cell suspension using a 70 µm filter, followed by centrifugation at 126 × g (1,000 rpm) for 5 min to get the cell pellets.
- Aspirate the supernatant media and replenish with 10 mL of fresh media. Gently pipette up and down to resuspend the cells using a 10 mL serological pipette.
- Pipette the suspension directly on the inside bottom of a T-75 flask and add media to bring the volume up to 25 mL. Culture the cells in an incubator in a standard sterile cell culture environment (i.e., 37 °C, humidified atmosphere with 5% CO2 and 95% air).
- After 3-7 days, rinse away the nonadherent cells by aspirating the old medium and replenishing with fresh medium. Continue culturing and feeding the cells with fresh medium until they are ready for cell passage, freezing, or use in an experiment.
- Cell maintenance
- Change the cell culture medium regularly to remove cellular waste products and replenish the nutrients approximately every other day until the cells are 90%-100% confluent. At 90% confluency, passage, freeze, or use the cells in an experiment.
- Passaging cells
NOTE: Passaging, also referred to as subculturing, is a term that applies whenever cells are transferred from one culture to another. Freshly harvested cells are at passage stage 0 (P0). The amounts of trypsin-ethylenediaminetetraacetic acid solution (trypsin-EDTA) and media described in this article are for a T-75 flask.- Check the cells under an optical microscope to confirm that the cells are 90% confluent.
- Aspirate the medium from the cell flask.
- Dispense 10 mL of phosphate-buffered saline (PBS) into the flask using a serological pipette. Gently rock the flask to rinse the cells with PBS; aspirate all the PBS.
NOTE: This step serves as an extra rinse to ensure that no dead cells or cellular waste will be transferred during passaging. - Dispense 3 mL of trypsin-EDTA into the cell flask directly onto the surface of the cells. Gently rock the flask to ensure the entire surface with the cells is covered by the trypsin-EDTA.
- Place the cell flask with trypsin-EDTA into the incubator for 5 min to allow the cells to detach.
- After 5 min in the incubator, check the cells under an optical microscope to confirm that the cells are detached. If some cells have not detached, tap the side of the cell flask gently, and check the flask again.
- Add 9 mL of fresh medium to the cell flask to dilute the trypsin-EDTA. This provides more accessible proteins for the trypsin-EDTA to bind to instead of lysing the cells.
- Pipette out the cells in media and trypsin-EDTA and dispense them into a 15 mL conical tube. Centrifuge the cells at 126 × g (1,000 rpm) for 5 min.
- Without disturbing the cell pellet, aspirate the medium with trypsin-EDTA.
- Add 5-10 mL of fresh media to the centrifuge tube and gently resuspend the cells in the medium using a 10 mL serological pipette.
- Pipette the cells suspended in medium out of the centrifuge tube and split the volume into 2-3 fresh culture flasks. Add enough medium to bring the total medium volume to 25 mL for each flask.
NOTE: The split ratio during subculture may vary depending on the cell types and specific growth characteristics. - Check the cells under an optical microscope and place them back into the incubator.
- Freezing cells
- Check the cells under an optical microscope to confirm that the cells are 90% confluent.
- Repeat steps 1.2.2.2 to 1.2.2.9.
- Add 900 µL of fresh medium to the centrifuge tube with a 100-1000 µL micropipette. Slowly and gently resuspend cells in medium using the same micropipette.
- Transfer the 900 µL cell suspension into a cryovial. Add 100 µL of dimethylsulfoxide (DMSO).
- As soon as possible, place the cryovial into a cylindrical foam container designed to regulate temperature decrease (see the Table of Materials). Place the foam container in the -80 °C freezer.
- Thawing cells
- Thaw the frozen cells in the water bath. Take a sterile 15 mL conical tube filled with 5 mL of fresh medium, place the cells in the conical tube, and centrifuge at 126 × g (1,000 rpm) for 5 min.
- Aspirate the medium containing DMSO.
- Add 5-10 mL of fresh medium to the 15 mL conical tube of cells. Slowly and gently pipette up and down to resuspend the cells.
- Pipette the cells suspended in medium out of the 15 mL conical tube and dispense into a new T-75 flask. When dispensing cells, use a sweeping motion to distribute the cells as evenly as possible on the bottom of the cell flask.
- Add enough fresh medium to bring the total medium volume to 25 mL for each T-75 flask.
- Check the cells under an optical microscope and place the cell flask back into the incubator.
2. Sample preparation and sterilization
- Sample preparation
- Use tissue-culture-treated plates such as 6, 12, 24, 48, or 96-well plates for cell culture experiments described in this article. Select the type of tissue-culture plates and the volume of medium in each well based on different experimental designs such as sample dimension.
- Sterilize or disinfect all the biodegradable samples before the cell culture.
NOTE: Disinfection is acceptable for in vitro studies when the samples are prone to chemical and/or surface changes under some sterilization conditions involving high heat, oxidizer, and/or radicals. Sterilization or disinfection methods for different sample types vary depending on the different properties of the materials, such as polymers, metals, and ceramics. The process for sterilization or disinfection could involve heat, gas, radiation, chemicals, high pressure, or a combination of these.- Biodegradable metals
- In general, use ultraviolet (UV) radiation to disinfect biodegradable metals for in vitro studies.
NOTE: For example, Zhang et al. reported that pure magnesium (Mg) and ZC21 Mg alloy samples were disinfected under UV radiation for 4 h before they were utilized in the cell studies1. For in vivo studies, the samples are generally required to be sterilized. For many biodegradable metals such as magnesium or magnesium alloys, autoclaving using steam should be avoided because these samples could be oxidized or corroded in water6. A quartz dish is recommended for UV disinfection because it provides better UV transparency than most glasses and plastics. - Sterilize Mg alloy samples under dry heat in an oven or autoclave at the temperature range of 100-200 °C.
NOTE: As some metallic alloys could still become oxidized on the surface at high temperatures in the air, high-intensity radiation may be used as an alternative in some cases. However, high-intensity radiation such as alpha or gamma radiation should be avoided when sterilizing thin metallic foils. It may cause atom displacement within the materials, changing the microstructure of materials. - Use ethylene oxide (EtO) gas sterilization as an alternative method for biodegradable metals sensitive to heat and radiation26.
- In general, use ultraviolet (UV) radiation to disinfect biodegradable metals for in vitro studies.
- Biodegradable ceramics
- Generally, disinfect biodegradable ceramics using UV radiation or 70% ethanol solution before in vitro studies.
NOTE: For example, Liu et al. reported that the calcium phosphate samples were disinfected via immersion in 70% ethanol for 1 h and exposure to UV light for 12 h on each side before they were used in in vitro cytocompatibility tests27. - Use autoclaves to sterilize biodegradable ceramics if high temperature and water vapor do not damage their compostions and microstructures.
NOTE: Some ceramics might be affected by autoclaving. For example, phase change and surface roughening were found when yttria-stabilized zirconia ceramics were autoclaved at 121 °C for 15 min. Additionally, CPCs cannot be sterilized via autoclaving with steam because the samples will react with water. - Use alternative sterilization methods such as Cobalt-60 radiation for the abovementioned yttria-stabilized zirconia ceramics and CPC samples28.
- Generally, disinfect biodegradable ceramics using UV radiation or 70% ethanol solution before in vitro studies.
- Biodegradable polymers
- In general, disinfect biodegradable polymers using UV radiation or 70% ethanol before using them in cell studies in vitro.
NOTE: Some polymers may undergo chemical changes under UV radiation. For sterilization, gamma-ray treatment such as Cobalt-60 radiation may be used. For example, starch powders were sterilized under Cobalt-60 radiation before being utilized in in vitro cell studies29. - Autoclave polymer materials that can withstand high temperature and moisture.
NOTE: For instance, polymers such as polypropylene can be autoclaved as they can tolerate autoclaving temperatures (i.e., 121-134 °C). Some polymers such as polycaprolactone (PCL) cannot be autoclaved because of their relatively low melting points (i.e., around 60 °C)30. - Use EtO gas to sterilize polymeric materials sensitive to heat or radiation sterilization.
- In general, disinfect biodegradable polymers using UV radiation or 70% ethanol before using them in cell studies in vitro.
- Biodegradable metals
3. Cell culture methods
- Direct culture methods
NOTE: The schematic diagram in Figure 2A shows the steps of the direct culture method. In this article, BMSCs were cultured on a Mg-derived plate placed inside the wells of a 12-well tissue-culture-treated plate as an example to illustrate the culture method.- Follow the steps described in steps 1.2.2.1-1.2.2.10 to obtain the cell suspension.
- Use a 90% confluent flask to determine the cell concentration in the cell suspension using a hemocytometer. Dilute the cell suspension using fresh medium to a prescribed cell concentration needed for the cell study in vitro.
NOTE: The seeding density of cells is determined by the experimental design. For example, cell densities of 2,000-40,000 cells/cm2 have been used in different cell studies with biodegradable materials. - Place the samples (Mg plate) in the center of the 12-well treated tissue-culture plates. Sequentially, rinse the culture plates with 2 mL of PBS and 2 mL of DMEM to calibrate the osmotic pressure under sterile conditions. Add 3 mL of the diluted cell suspension into each well onto the samples of interest.
- Culture the cells in an incubator under standard cell culture conditions for 24 h.
NOTE: The culture time may be longer or shorter than 24 h depending on the experimental design.
- Direct exposure cultures
NOTE: The schematic diagram in Figure 2B shows the steps of direct exposure culture.- As described in steps 3.1.1 and 3.1.2, prepare the cell suspension with the required concentrations of cells based on the experimental design for different cell types and intended applications.
- Rinse the culture plates with 2 mL of PBS and 2 mL of DMEM sequentially to calibrate the osmotic pressure under sterile conditions. Add 3 mL of the diluted cell suspension into each well. Culture the cells in the humidified incubator under standard cell culture conditions for 24 h or until the cells reach 50-80% confluence.
NOTE: The cell confluence level may vary for different cell types and experimental design. - After 24 h, rinse the cells in the well plate with PBS using a pipette to remove floating dead cells.
- Place the disinfected or sterilized samples directly on the adhered cells. Add 3 mL of fresh medium into each well.
- Culture the cells under standard cell culture conditions for another 24 h.
NOTE: The culture time may be longer or shorter than 24 h depending on the experimental design.
- Exposure cultures
NOTE: The schematic diagram in Figure 2C shows the steps of the exposure culture method.- Initial steps for the cell preparation are the same as the exposure culture. As described in steps 3.1.1 and 3.1.2, prepare the cell suspension with the desired cells. Similar to steps 3.2.1 and 3.2.2, seed the cells in the well plate at the desired density and culture them in an incubator under standard cell culture conditions for 24 h.
- After 24 h, rinse the adherent cells with PBS to remove floating dead cells, followed by the addition of 3 mL of fresh medium into each well.
- Afterwards, place the samples in the well inserts with a membrane pore size of 0.4 µm and place the well inserts with the samples into each well with the cells.
- Culture the cells under standard cell culture conditions for another 24 h.
NOTE: The culture time may be longer or shorter than 24 h depending on the experimental design.
4. Postculture characterization of cells
NOTE: For direct culture and direct exposure culture, fix, stain, image, and analyze the cells adherent on both well plates and samples. For exposure culture, analyze the cells adhered to the well-plates.
- Cell fixation
- Collect the postculture medium from each well into a corresponding 15 mL conical tube for further analysis. Collect all the samples after culture for further analysis.
- Rinse the cells adherent on both samples and well plates 3 times using PBS.
- Add 1 mL of 4% paraformaldehyde (PFA, 10% neutral buffered formalin) into each well plate. Put the lid back on the well plate and allow the PFA to react for 20 min.
- After 20 min, aspirate the PFA and dispense it into a waste bottle. Rinse the well plate 3 times using PBS to remove the PFA, and transfer the waste to the waste bottle.
- Cell staining
- Prepare the working stocks of the staining agents following the manufacturers' instructions.
NOTE: For example, Alexa Fluor 488 Phalloidin is used to stain F-actin, and 4′, 6-diamidino-2-phenylindole (DAPI) is used to stain cell nuclei. Staining time may be reduced if the samples degrade rapidly in the staining solutions. - Add 200-400 µL of diluted Alexa Fluor 488 Phalloidin staining agent to each well to cover the cells on the well plate and the sample. Wrap the well plate in aluminum foil to prevent light exposure, and allow the Alexa Fluor 488 Phalloidin to react for 20 min at room temperature.
- Collect the Alexa Fluor 488 Phalloidin staining agent and dispense it into the corresponding waste bottle. Rinse the well plate 3 times using PBS to remove the excess Alexa Fluor 488 Phalloidin, and dispense the used PBS into the corresponding waste bottle.
- Add 200-400 µL of diluted DAPI to each well to cover the cells in the well and on the sample. Wrap the well plate in aluminum foil and allow the DAPI to react for 5 min at room temperature.
- Collect the DAPI and dispense it into the corresponding waste bottle. Rinse the well plate 3 times using PBS to remove the DAPI, and dispense the used PBS into the corresponding waste bottle.
- Prepare the working stocks of the staining agents following the manufacturers' instructions.
- Cell imaging
- After staining, image the cells using a fluorescence microscope. Whenever possible, take phase-contrast images of cells in addition to fluorescence images. Image the cells on the biodegradable samples as soon as possible or immediately after staining to avoid or reduce possible changes caused by the continuous degradation of samples. Store the cells in the well plates in a buffer solution at 2-8 °C after fixation, and image the cells as soon as possible after staining to avoid the loss of fluorescence signals.
- For direct culture and direct exposure culture, image and evaluate two types of cells: (1) the cells on the samples (in direct contact with the samples) and (2) the cells adherent on the well plate surrounding the samples (indirect contact with the samples), as shown in Figure 3A.
- For exposure culture, as shown in Figure 3B, use an image guide when taking the fluorescence images of cells to determine if the cell response would be different in response to the dynamic degradation gradient of the samples. Image and analyze the cells located in the area within the inner ring (3.5 mm away from the center) and the outer ring (7 mm away from the center) separately.
NOTE: The image guide is used to define the distance between the cells and samples. - For each sample and well in the culture plates, randomly take at least five images from each area of interest where the cells are either in direct contact or indirect contact with the samples at a predefined distance.
- Image analysis
- For all the cell images obtained from step 4.3, quantify the cell morphology by measuring the cell spreading area and aspect ratio using software such as ImageJ for image analysis.
- Count the number of cells in each image area. Calculate the cell adhesion density under direct and indirect contact conditions as the number of cells per unit area.
5. Postculture analyses of media and samples
- Measure the pH of postculture medium.
NOTE: Some samples change the pH value of the medium during their degradation. For example, biodegradable metals such as magnesium alloys usually increase the pH value of the medium due to their degradation31. In contrast, biodegradable polymers such as PLGA often decrease the pH value of the medium when they degrade32. Measuring the pH value of postculture medium can indicate the degradation of these samples in vitro.- Before cell fixation, collect the postculture medium as in step 4.1.1.
- Measure the pH values of the postculture medium in each well immediately after collection, using a precalibrated pH meter.
NOTE: The pH of the media may drift over time because of environmental conditions such as CO2 level and temperature, which should be taken into consideration.
- Analyze the medium compositions for the degradation behavior of biodegradable samples. For some samples that cause a color change of the medium, measure the optical density (O.D.) value of the postculture medium using a spectrophotometer or a microplate reader to determine the degradation behavior. Alternatively, use ultraviolet-visible spectroscopy (UV-VIS) and Fourier transform infrared spectroscopy-attenuated total reflectance (FTIR-ATR) to analyze the degradation products in the postculture medium.
NOTE: Measuring the degradation products in the post-culture medium is valuable for understanding the degradation behavior of the samples. One of the most common approaches is to measure the ions of interest in the postculture medium using an inductively coupled plasma-optical emission spectrometer (ICP-OES). The ICP-OES can be used to measure the compositions of postculture medium of metals and ceramics but may not be suitable for polymers. Polymers usually consist of carbon, hydrogen, and oxygen, and accurate quantification of these elements is difficult for ICP-OES.- Following step 5.1.2 for pH measurement, collect and dilute the medium using a desirable dilution factor for optimal measurements of ion concentrations.
- Measure the concentrations of the ions of interest in the postculture medium using an ICP-OES.
- Postculture analysis of samples
NOTE: After in vitro cell study, the biodegradable samples may change in dimension, mass, surface morphology, microstructure, and composition. Postculture analysis of samples helps understand the degradation mechanism of samples.- After cell culture, take the photographs of the samples to show possible changes in sample dimension, color, morphology, and other visible characteristics.
- Dry or dehydrate the postculture samples and measure the sample mass, dimension, and volume to quantify any changes in mass, dimension, and volume.
- Use a scanning electron microscope (SEM) to characterize the microstructure and morphology of the samples. Use energy dispersive X-Ray spectroscopy (EDX) and X-ray diffraction (XRD) to characterize the composition and phase of the degradation products on the samples. Use FTIR-ATR to detect the chemical bonding on the sample surfaces.
Representative Results
Figure 4 shows the representative fluorescence images of BMSCs under direct and indirect contact conditions using different culture methods. Figure 4A,B show the BMSCs under direct and indirect contact conditions after the same 24 h direct culture with ZC21 magnesium alloys1. The ZC21 alloys consist of 97.5 wt% Magnesium, 2 wt% Zinc, and 0.5 wt% calcium. The cells that have no direct contact with the ZC21 alloy samples spread better than those that have direct contact with the samples. As shown in Figure 4C,D, the cells under direct and indirect contact conditions all exhibit normal morphology after a 24 h direct exposure with hyaluronic acid (HyA) hydrogels crosslinked by Fe3+ ions. However, the number of cells under the indirect contact condition is lower than that under the direct contact condition33. Another study reported the effects of degradation of ZSr41 alloys (ф = 1.1 mm) on BMSCs after a 24 h exposure culture19. The ZSr41 alloys consist of 95 wt% magnesium, 4 wt% zinc, and 1 wt% strontium. Figure 4E shows the representative fluorescence images of BMSCs adherent to the culture well at a location 3.5 mm away from the well center, after a 24 h exposure culture with the biodegradable pins19.
Figure 5 shows the example data for quantified cell adhesion density. As shown in Figure 5A, in the 24 h direct exposure (24h_DE) culture, BMSCs in direct contact with the ZC21 have significantly greater cell adhesion density than any other group. In the 24 h direct culture (24h_D), BMSCs in direct contact with the ZC21 show significantly higher cell adhesion density than the Mg group, significantly lower than the Glass reference group, but no statistical difference compared with the Ti alloy (T64) control. As shown in Figure 5B, in the indirect contact condition of direct exposure culture, BMSC adhesion density is significantly higher for the ZC21 group than the Mg group. However, it shows no significant difference compared with the T64 and cells-only control groups. In the indirect contact condition of direct culture, BMSC adhesion density is significantly higher for the ZC21 group than for the Mg group but shows no significant difference compared with the T64 and cells-only control groups1.
Figure 6A shows the pH value of postculture medium after the direct exposure culture and direct culture. For the direct exposure culture, the pH values of medium range from 8.3 to 8.4 for all samples. In the direct culture, the pH values of medium range from 7.9 to 8 across the groups. Figure 9B shows the Mg2+ ion concentration in the postculture medium. In both the direct exposure culture and the direct culture, the Mg2+ ion concentrations in the ZC21 and Mg groups are significantly higher than in any other control groups1. Figure 7 shows the XRD patterns for ZSr41 and pure Mg after a 3-day exposure culture. In Figure 7A, the crystalline phases of Mg, Ca(OH)2, ZnO, MgO∙H2O, Ca(HPO4)(H2O)2, and Ca5(PO4)3(OH) (i.e., hydroxyapatite or HA), Mg17Sr2 are found on the surface of ZSr41. In Figure 7B, the crystalline phases of Mg, Ca(OH)2, Mg3(PO4)2, Mg7(PO4)2(OH)8, Ca2P2O7∙5H2O are on the surface of pure Mg19. Figure 8A shows the overlay of SEM images and EDX maps of surface elemental composition for MgO-coated Mg and the control of Mg substrates and Glass after 24 h of direct culture with BMSCs. Figure 8B shows the quantitative surface elemental composition of the sample surfaces, indicating different depositions formed during cell culture34.
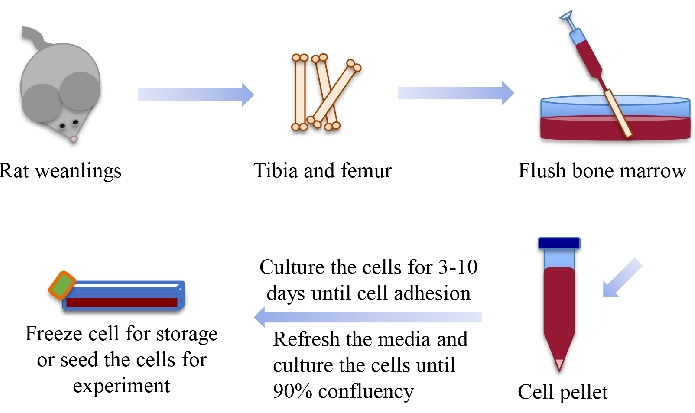
Figure 1: Schematic diagram showing the steps to harvest BMSCs from rat weanlings. This figure is modified from 35. Abbreviation: BMSCs = bone marrow-derived mesenchymal stem cells. Please click here to view a larger version of this figure.
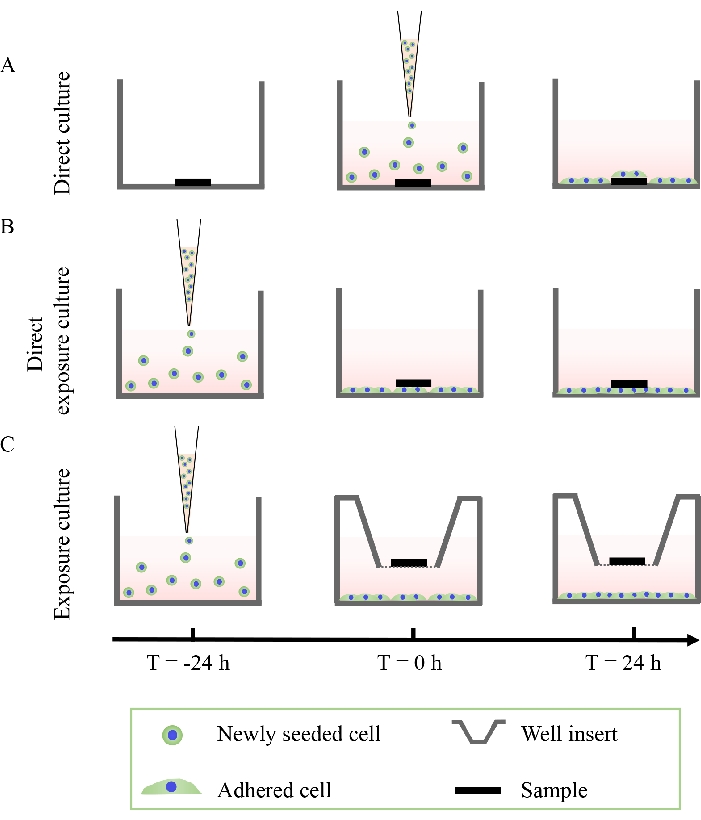
Figure 2: Schematic diagram showing the three cell culture methods. (A) Direct culture, (B) direct exposure culture, and (C) exposure culture. This figure is modified from 36. Please click here to view a larger version of this figure.
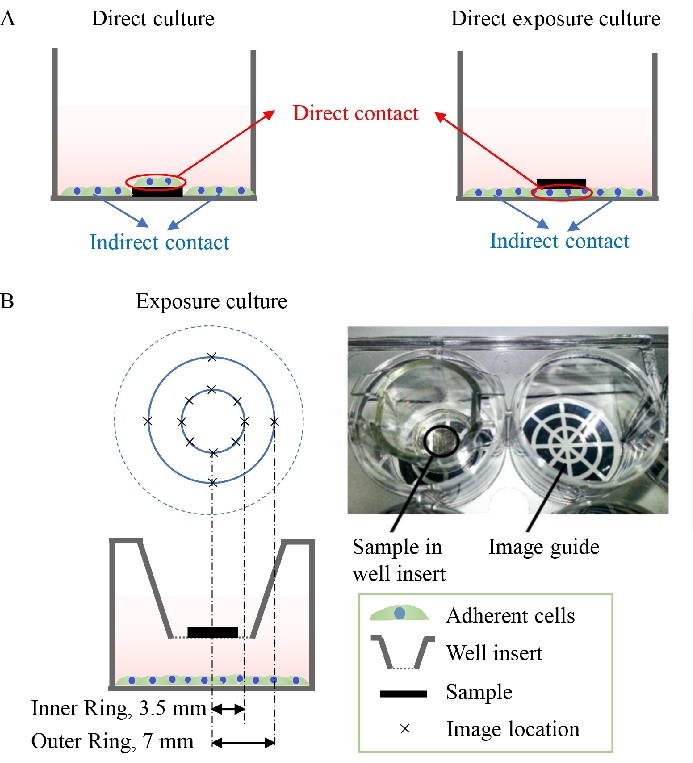
Figure 3: Schematic diagrams showing direct culture and direct exposure culture. (A) Cells under direct contact and indirect contact conditions in direct culture and direct exposure culture. (B) Utilization of an imaging guide to take pictures of the cells adhered to the well plate at different distances away from the center of the samples in exposure culture. Figure 3B is modified from 37. Please click here to view a larger version of this figure.
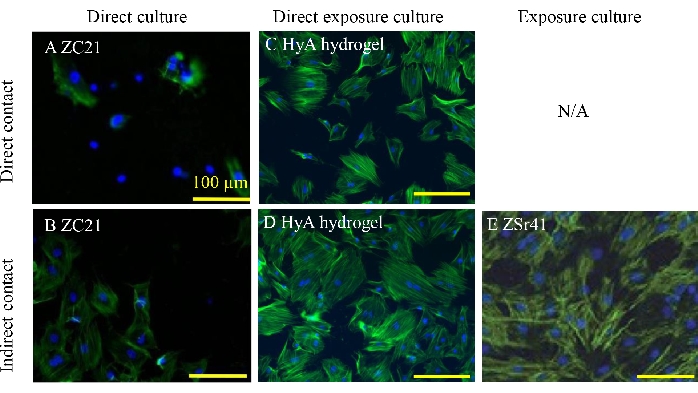
Figure 4: Representative fluorescence images of BMSCs. (A, B) Direct and indirect contact conditions after a 24 h direct culture with ZC21 alloys. (C, D) Direct exposure culture with HyA hydrogels. (E) On the culture plate after a 24 h exposure culture with ZSr41 alloys. Scale bars = 100 µm. A and B are reproduced from 1; C and D are reproduced from 33; and E is reproduced from 19. Abbreviations: BMSCs = bone marrow-derived mesenchymal stem cells; HyA = hyaluronic acid. Please click here to view a larger version of this figure.
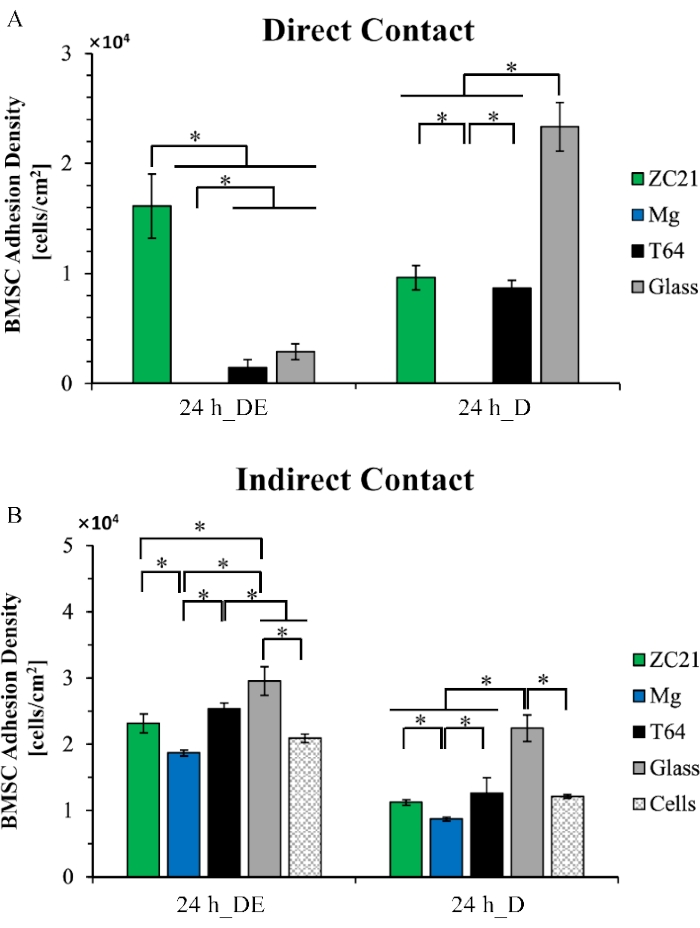
Figure 5: Quantitative results for cell adhesion density of BMSCs. (A) Direct contact and (B) indirect contact conditions after the 24 h direct exposure culture (24 h_DE) and direct culture (24 h_D). This figure is reproduced from 1. Abbreviations: BMSCs = bone marrow-derived mesenchymal stem cells; DE = direct exposure culture; D = direct culture. Please click here to view a larger version of this figure.
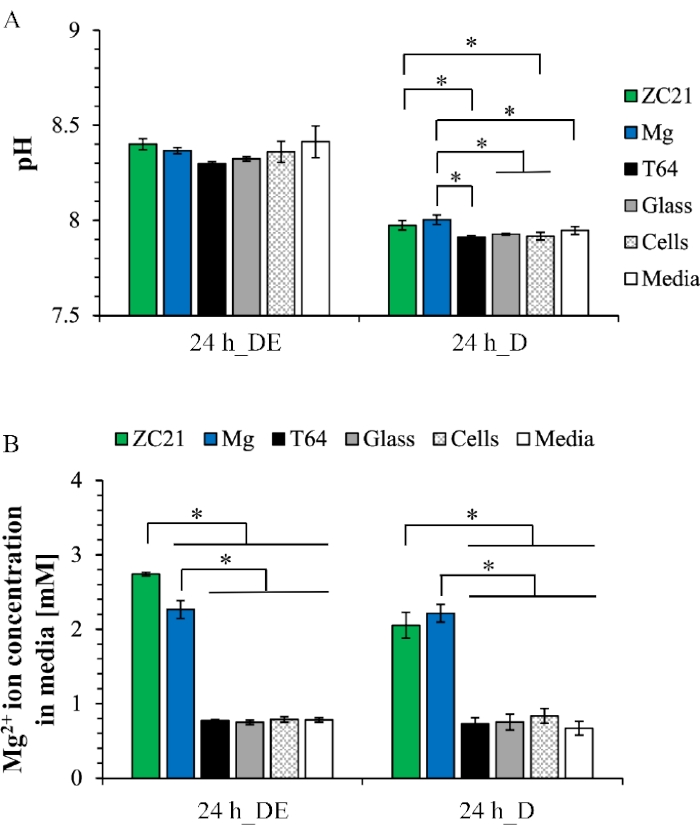
Figure 6: Representative results for postculture analyses of medium after the 24 h direct exposure culture and direct culture. (A) pH values and (B) Mg2+ ion concentrations. This figure is reproduced from 1. Abbreviations: DE = direct exposure culture; D = direct culture. Please click here to view a larger version of this figure.
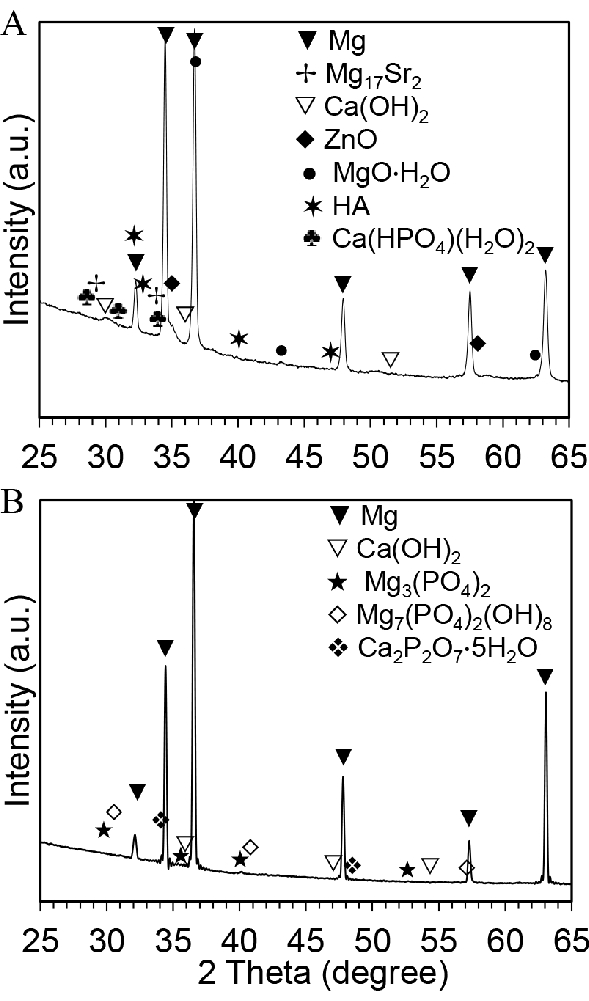
Figure 7: Representative postculture results for phase analyses of biodegradable metallic samples after 3 days of culture with BMSCs. (A) X-ray diffraction spectrum for ZSr41. (B) XRD spectrum for pure Mg. This figure is reproduced from 19. Abbreviations: BMSCs = bone marrow-derived mesenchymal stem cells; XRD = X-ray diffraction. Please click here to view a larger version of this figure.

Figure 8: Representative postculture results for surface analyses of samples after 24 h of direct culture with BMSCs, including surface microstructure, morphology, and composition. (A) Overlay of SEM images and EDX maps of surface elemental composition for MgO-coated Mg., non-coated Mg control, and Glass reference. (B) Surface elemental composition (at. %) quantified from EDX analyses. Scale bars = 200 µm. Reproduced from 34. Abbreviations: BMSCs = bone marrow-derived mesenchymal stem cells; SEM = scanning electron microscopy; EDX = energy dispersive X-Ray spectroscopy. Please click here to view a larger version of this figure.
Supplemental Table 1: Degradation mechanisms, products, and rates for different types of materials, and the results collected for the postculture sample and medium analysis. Please click here to download this Table.
Discussion
Different cell culture methods can be used to evaluate the in vitro cytocompatibility of biomaterials of interest for various aspects of applications in vivo. This article demonstrates three in vitro culture methods, i.e., direct culture, direct exposure culture, and exposure culture, to mimic different in vivo scenarios where biodegradable implant materials are used inside the human body. The direct culture method is mainly used to evaluate the behavior of newly seeded cells directly adherent to and surrounding the implant materials. The direct exposure culture method mimics the in vivo scenario where the implant materials come into direct contact with the established cells and tissues. The exposure culture method can be used to show how the degradation products from the implant materials and the changes in the local microenvironment can affect the established cells and tissues that are not directly in contact with the implant materials.
In the direct culture, the newly seeded cells under both direct and indirect contact conditions are evaluated. In the direct exposure culture, established cells can be evaluated under both direct and indirect contact conditions. In the exposure culture, only established cells under indirect contact conditions can be evaluated. The newly seeded cells under direct contact conditions in the direct culture are affected by material properties and material-induced changes in medium such as the changes in ion concentration and pH value.
The abovementioned material properties may include surface morphology, hydrophilicity, surface free energy, stiffness, and composition. The newly seeded cells under indirect contact conditions in the direct culture method and all the established cells in the direct exposure culture and exposure culture methods are mainly affected by material-induced changes in the medium. The three different methods described in this article are closer to the practical scenario of the in vivo environment than conventional methods such as the medium extract method. The medium extract method only evaluates the cytotoxicity of the degradation products of the material and does not reflect the dynamic process of sample degradation. In the culture methods described in this article, as the cells are cultured with the implant materials, the dynamic change of the biodegradable materials and medium environment can affect the cells in situ.
Although no in vitro studies can completely replace in vivo studies, in vitro studies are complementary and can provide valuable data in a low-cost and efficient manner. In vivo studies usually include all multiple variables in a model, whereas in vitro cell culture can study the effects of a single factor on cell-material interactions. The methods introduced in this article can mimic different scenarios of the relevant in vivo studies. We can create the linkages between different variables to provide supplements for in vivo studies. An in vivo model usually only includes the same tissue in an animal type. However, in vitro studies can include different cell types in one culture, which can study the combined effects of different variables on cell-material interactions. Moreover, it is relatively difficult to study the effects of dynamic environmental changes on cell-material interactions in in vivo models. The methods described in this article can investigate the effects of dynamic changes such as ion concentrations in the medium on cell behavior38.
The methods presented in this article are applicable for understanding in vitro cytocompatibility of all types of materials, including polymers, metals, ceramics, composites, and nanoparticles, and determining their interactions with different cells, bacteria, or fungi based on the intended applications. For example, Xu et al. evaluated the in vitro cytocompatibility of HyA-based hydrogels with BMSCs via the direct exposure culture method33. Cell adhesion densities and cell morphologies were analyzed under direct and indirect contact conditions. The cytotoxicity of HyA-based hydrogel composites might be related to the concentrations of Fe3+ and H+ ions released from the crosslinked HyA hydrogels during the cell culture experiment. Tian et al. cultured human urothelial cells (HUCs) with four different Mg alloys for 24 h and 48 h using the exposure culture method and their insoluble degradation products of MgO and Mg(OH)2 for 24 h using direct exposure culture to investigate cytocompatibility and degradation behaviors of Mg alloys containing zinc (Zn) and strontium (Sr) for potential ureteral stent application39. In this study, ZSr41_B containing 4 wt% Zn and 0.5 wt% Sr was found to have better cytocompatibility with HUCs among all the other Mg-4Zn-xSr alloys in both 24 h and 48 h exposure cultures. The results also showed that no visible adherent cells were found on the well plate when the concentrations of magnesium oxide (MgO) and magnesium hydroxide (Mg(OH)2) exceeded 1.0 mg/mL after 24 h of direct exposure culture. Therefore, Tian et al. concluded that reducing the degradation rates of Mg alloys is necessary to control the possible side effects toward future clinical translation. Wetteland et al. created a polymer-based nanocomposite by dispersing hydroxyapatite (nHA) and nMgO nanoparticles in a biodegradable PLGA polymer40. This nanocomposite was studied by culturing BMSCs with different samples using the direct culture method. The results demonstrated that improved dispersion of nanoparticles in the polymer could improve BMSC adhesion on nHA/PLGA but decrease the cell viability on nMgO/PLGA. Based on the results of in vitro cell studies, Wetteland et al. reported valuable insight for engineering optimal ceramic/polymer nanocomposites for different biomedical applications.
Cell morphologies and cell numbers can be observed and quantified in fluorescence images using software for quantitative image analysis such as ImageJ. We can investigate the effects of different materials on cell adhesion and morphology by quantifying the cell adhesion densities, cell aspect ratios, and cell spreading areas for different sample groups. The morphology of cells from the blank control group, where only the cells are cultured in medium, could serve as a standard of reference without any influence from sample materials. We can determine whether the sample materials would affect cell adhesion and morphology in vitro by comparing the cell adhesion densities and cell morphologies of the sample groups with those of the blank control. Cell spreading area reveals the preference of cell adhesion to the sample surface, showing how the cells interact with the sample materials. In this article, we reduced the reaction time for DAPI staining to be less than the vendor-recommended optimal time because biodegradable samples, such as pure magnesium, degrade rapidly in aqueous solutions. The morphology of cells adhered to the biodegradable materials may change if the staining process takes too long and the water exposure time is too long for the samples. Moreover, for the cells adherent to biodegradable materials, cell images should be taken promptly to reduce any possible changes in cell adhesion and morphology because of sample degradation.
Besides collecting results of cells, postculture medium and sample analyses are important because they will provide valuable data for analyzing the degradation mechanism, products, and rates of the implant materials. For example, biodegradable polymers such as PLGA may generate acidic degradation byproducts such as monomeric or oligomeric hydroxyl-carboxylic acids during the cell culture32, which may influence cell growth and proliferation. In contrast, biodegradable metals, such as magnesium and its alloys, produce hydroxide ions and hydrogen gas during their degradation31, which can significantly increase the local pH, and severe alkalinity may have adverse effects on local cell functions. Various biodegradable ceramics may also increase the pH of the medium41. In general, cells require a specific pH range in culture medium to function properly, and it is known that increased or decreased pH values in body fluids are harmful to life42. Measuring the pH of postculture medium is valuable for understanding any potential harm that biodegradable sample materials may cause in cell culture. Therefore, it is necessary to measure the pH value of the postculture medium to understand the potential mechanisms of how these biodegradable materials affect cells.
It is important to measure the crucial ion concentrations in the postculture medium for biodegradable materials. For example, Cortez Alcaraz et al. measured the Mg2+ and Ca2+ ion concentrations of the postculture medium when they studied magnesium oxide nanoparticle-coated magnesium samples using direct culture with BMSCs34. The concentrations of magnesium ions indicate the degradation properties of different samples in vitro during cell culture, and the concentrations of calcium ions can provide information about calcium deposition during cell proliferation. Xu et al. measured Fe3+ ion concentrations of the postculture medium when they studied HyA hydrogels using direct exposure culture with BMSCs. They utilized Fe3+ ions to adjust the crosslinking densities of HyA33. Fe3+ ions may decrease the pH value of the culture medium, and high concentrations of Fe3+ ions might be toxic to the cells. Therefore, it is important to measure the concentrations of the ions of interest to improve the cytocompatibility of biodegradable materials and their associated degradation products.
We may collect different data to analyze the cell-material interactions for different materials. For example, as shown in Supplemental Table 1, Mg alloys degrade by reacting with water, and the degradation products may include Mg2+ and OH– ions, H2 gas, and some other insoluble degradation products such as Mg(OH)2. XRD, SEM, and EDX, which could be used to determine the mineral deposition formed on the material. We may study the effects of concentration of Mg2+ ions and pH values in medium on the cell behaviors. Moreover, we may use these results to study the gas evolution during metal degradation. In vitro studies have reported the critical tolerance level of H2 gas to be <0.01 mL/cm2/day, and this has been widely used to screen magnesium alloys for temporary implant applications. Essentially, the amount of gas evolution is dependent on the degradation rate of the magnesium alloys. In another example, PLGA degrades due to the hydrolysis of its ester linkages in the presence of water. The degradation products of lactic acid and glycolic acid, as well as the pH values in the medium, could be studied to analyze cell-material interactions. The methods described in this article include the measurement of released ions and the pH values in the cell culture media and the mass change of the materials, which can be used to estimate the degradation rate of the materials.
Different materials usually behave differently in vitro and in vivo, and the methods for cytocompatibility studies should be selected based on the application environment and material type. For orthopedic applications, it is desirable to evaluate the interactions between the relevant bone cells and implants when they are in direct contact with each other. The direct culture method could be utilized to investigate the interactions between the newly seeded cells and the implant. In cardiovascular applications, as the established cells will directly or indirectly come into contact with the implanted stent materials, direct exposure culture and exposure culture methods may be used to evaluate the cytocompatibility of biodegradable metals for cardiovascular applications. We believe that the in vitro methods described in this article are feasible to provide initial evidence for the cytocompatibility of biodegradable implant materials. The culture methods need to be modified for different materials with varied degradation mechanisms, products, and rates. For instance, the culturing time for different materials can be modified based on varied degradation rates of different material types. Different results may be collected based on different degradation mechanisms and products of the materials.
In summary, it is important to analyze the cells, medium, and sample materials qualitatively and quantitatively, before and after in vitro cell culture, to understand the effects of biodegradable implant materials and their degradation products on cytocompatibility. The three culture methods presented in this article can be used for studying a wide range of biodegradable materials, including biodegradable polymers, ceramics, and metals for medical implant and tissue engineering applications. These in vitro cell studies are valuable for screening biodegradable materials, optimizing the design of implantable devices and scaffolds at the early stage of product development, and reducing potential toxicity to cells.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors appreciate the financial support from the U.S. National Science Foundation (NSF CBET award 1512764 and NSF PIRE 1545852), the National Institutes of Health (NIH NIDCR 1R03DE028631), the University of California (UC) Regents Faculty Development Fellowship, and Committee on Research Seed Grant (Huinan Liu), and UC-Riverside Dissertation Research Grant (Jiajia Lin). The authors appreciate the Central Facility for Advanced Microscopy and Microanalysis (CFAMM) at the UC-Riverside for the use of SEM/EDS and Dr. Perry Cheung for the use of XRD instruments. The authors also appreciate Thanh Vy Nguyen and Queenie Xu for partial editing. The authors also would like to thank Cindy Lee for recording the narration for the video. Any opinions, findings, and conclusions, or recommendations expressed in this article are those of the authors and do not necessarily reflect the views of the National Science Foundation or the National Institutes of Health.
Materials
| 10 mL serological pipette | VWR | 490019-704 | |
| 12-well tissue-culture-treated plates | Thermo Fisher Scientific | 353043 | |
| 15 mL conical tube (Polypropylene) | VWR | 89039-666 | |
| 18 G needle | BD | 305196 | |
| 25½ G needle | BD | 305122 | |
| 4′,6-diamidino-2- phenylindole dilactate (DAPI) | Invitrogen | D3571 | |
| 50 mL conical tube (Polypropylene) | VWR | 89039-658 | |
| 70 μm nylon strainer | Fisher Scientific | 50-105-0135 | |
| Alexa Flour 488-phalloidin | Life technologies | A12379 | |
| Biological safety cabinet | LABCONCO | Class II, Type A2 | |
| Centrifuge | Eppendorf | Rotor F-35-6-30, Centrifuge5430 | |
| Clear Fused Quartz Round Dish | AdValue Technology | FQ-4085 | |
| CO2 incubator | SANYO | MCO-19AIC | |
| CoolCell Freezer Container | Corning | 432000 | foam container designed to regulate temperature decrease |
| Cryovial | Thermo Fisher Scientific | 5000-1020 | |
| Dimethyl Sulfoxide (DMSO) | Sigma-Aldrich | 472301 | |
| Dulbecco’s modified Eagle’s medium (DMEM) | Sigma-Aldrich | D5648 | |
| EDX analysis software | Oxford Instruments | AztecSynergy | |
| Energy dispersive X-ray spectroscopy (EDX) | FEI | 50mm2 X-Max50 SDD | |
| Fetal bovine serum (FBS) | Thermo Fisher Scientific Inc. | SH30910 | |
| Fluorescence microscope | Nikon | Eclipse Ti | |
| Formaldehyde | VWR | 100496-496 | |
| Hemacytometer | Hausser Scientific | 3520 | |
| ImageJ software | National Institutes of Health and the Laboratory for Optical and Computational Instrumentation (LOCI, University of Wisconsin) | ||
| Inductively coupled plasma optical emission spectrometry (ICP-OES) |
PerkinElmer | Optima 8000 | |
| Optical microscope | VWR | VistaVision | |
| Penicillin/streptomycin (P/S) | Thermo Fisher Scientific, Inc., | 15070063 | |
| pH meter | VWR | model SB70P | |
| Phosphate Buffered Saline (PBS) | VWR | 97062-730 | |
| Scanning electronic microscope (SEM) | FEI | Nova NanoSEM 450 | |
| surgical blade | VWR | 76353-728 | |
| Tissue Culture Flasks | VWR | T-75, MSPP-90076 | |
| Transwell inserts | Corning | 3460 | |
| Trypsin-ethylenediaminetetraacetic acid solution (Trypsin-EDTA) | Sigma-Aldrich | T4049 | |
| X-ray diffraction instrument (XRD) | PANalytical | Empyrean Series 2 |
References
- Zhang, C., et al. Antimicrobial bioresorbable Mg-Zn-Ca alloy for bone repair in a comparison study with Mg-Zn-Sr Alloy and pure Mg. ACS Biomaterials Science & Engineering. 6 (1), 517-538 (2019).
- Xu, C., et al. A green biocompatible fabrication of highly porous functional ceramics with high strength and controllable pore structures. Journal of Materials Science & Technology. 32 (8), 729-732 (2016).
- Asgari, M., et al. Biodegradable metallic wires in dental and orthopedic applications: a review. Metals. 8 (4), 212 (2018).
- Shi, Y., Liu, J., Yu, L., Zhong, L. Z., Jiang, H. B. β-TCP scaffold coated with PCL as biodegradable materials for dental applications. Ceramics International. 44 (13), 15086-15091 (2018).
- Wu, C. -. C., et al. A self-reinforcing biodegradable implant made of poly (ɛ-caprolactone)/calcium phosphate ceramic composite for craniomaxillofacial fracture fixation. Journal of Cranio-Maxillofacial Surgery. 44 (9), 1333-1341 (2016).
- Jiang, W., et al. In vitro evaluation of MgSr and MgCaSr alloys via direct culture with bone marrow derived mesenchymal stem cells. Acta Biomaterialia. 72, 407-423 (2018).
- Zhang, C., et al. Magnesium-based biodegradable microelectrodes for neural recording. Materials Science and Engineering: C. 110, 110614 (2020).
- Jia, B., et al. In vitro and in vivo studies of Zn-Mn biodegradable metals designed for orthopedic applications. Acta Biomaterialia. 108, 358-372 (2020).
- Yang, H., et al. Alloying design of biodegradable zinc as promising bone implants for load-bearing applications. Nature Communications. 11 (1), 1-16 (2020).
- Liu, H., et al. Injectable, biomechanically robust, biodegradable and osseointegrative bone cement for percutaneous kyphoplasty and vertebroplasty. International Orthopaedics. 42 (1), 125-132 (2018).
- Anjum, S., Arora, A., Alam, M., Gupta, B. Development of antimicrobial and scar preventive chitosan hydrogel wound dressings. International Journal of Pharmaceutics. 508 (1-2), 92-101 (2016).
- Barroca, N., et al. Electrically polarized PLLA nanofibers as neural tissue engineering scaffolds with improved neuritogenesis. Colloids and Surfaces B: Biointerfaces. 167, 93-103 (2018).
- Liu, Y., et al. Polydopamine-modified poly (l-lactic acid) nanofiber scaffolds immobilized with an osteogenic growth peptide for bone tissue regeneration. RSC Advances. 9 (21), 11722-11736 (2019).
- Liu, H., Webster, T. J. Enhanced biological and mechanical properties of well-dispersed nanophase ceramics in polymer composites: from 2D to 3D printed structures. Materials Science and Engineering: C. 31 (2), 77-89 (2011).
- Xu, C., et al. Bioinspired mechano-sensitive macroporous ceramic sponge for logical drug and cell delivery. Advanced Science. 4 (6), 1600410 (2017).
- Xu, C., Bai, Y., Yang, H., Yang, L. Mechanically modulated, ultra-high precision logic delivery of molecules by bio-inspired macroporous ceramic sponge. MRS Advances. 2 (19-20), 1125-1130 (2017).
- Zhang, N., Xu, C., Azer, A., Liu, H. Dispersibility and characterization of polyvinyl alcohol-coated magnetic nanoparticles in poly (glycerol sebacate) for biomedical applications. Journal of Nanoparticle Research. 21 (12), 1-11 (2019).
- Kim, S. S., et al. A poly (lactide-co-glycolide)/hydroxyapatite composite scaffold with enhanced osteoconductivity. Journal of Biomedical Materials Research Part A. 80 (1), 206-215 (2007).
- Cipriano, A. F., et al. Degradation of bioresorbable Mg-4Zn-1Sr intramedullary pins and associated biological responses in vitro and in vivo. ACS Applied Materials & Interfaces. 9 (51), 44332-44355 (2017).
- Surmeneva, M. A., et al. Bone marrow derived mesenchymal stem cell response to the RF magnetron sputter deposited hydroxyapatite coating on AZ91 magnesium alloy. Materials Chemistry and Physics. 221, 89-98 (2019).
- Sheikh, Z., et al. Mechanisms of in vivo degradation and resorption of calcium phosphate based biomaterials. Materials. 8 (11), 7913-7925 (2015).
- Klein, C., Driessen, A., De Groot, K., Van den Hooff, A. Biodegradation behavior of various calcium phosphate materials in bone tissue. Journal of Biomedical Materials Research. 17 (5), 769-784 (1983).
- Lanao, R. P. F., Leeuwenburgh, S. C., Wolke, J. G., Jansen, J. A. Bone response to fast-degrading, injectable calcium phosphate cements containing PLGA microparticles. Biomaterials. 32 (34), 8839-8847 (2011).
- Vey, E., et al. Degradation kinetics of poly (lactic-co-glycolic) acid block copolymer cast films in phosphate buffer solution as revealed by infrared and Raman spectroscopies. Polymer Degradation and Stability. 96 (10), 1882-1889 (2011).
- Standard, I. Biological evaluation of medical devices-Part 5: Tests for in vitro cytotoxicity. Geneve, Switzerland: International Organization for Standardization. , (2009).
- Liu, X., Zhou, W., Wu, Y., Cheng, Y., Zheng, Y. Effect of sterilization process on surface characteristics and biocompatibility of pure Mg and MgCa alloys. Materials Science and Engineering: C. 33 (7), 4144-4154 (2013).
- Liu, H., Yazici, H., Ergun, C., Webster, T. J., Bermek, H. An in vitro evaluation of the Ca/P ratio for the cytocompatibility of nano-to-micron particulate calcium phosphates for bone regeneration. Acta Biomaterialia. 4 (5), 1472-1479 (2008).
- Liu, H., et al. Enhancing effects of radiopaque agent BaSO4 on mechanical and biocompatibility properties of injectable calcium phosphate composite cement. Materials Science and Engineering: C. 116, 110904 (2020).
- Xu, C., et al. A versatile three-dimensional foam fabrication strategy for soft and hard tissue engineering. Biomedical Materials. 13 (2), 025018 (2018).
- Speranza, V., Sorrentino, A., De Santis, F., Pantani, R. Characterization of the polycaprolactone melt crystallization: complementary optical microscopy, DSC, and AFM studies. The Scientific World Journal. , 720157 (2014).
- Cipriano, A. F., et al. Anodization of magnesium for biomedical applications-Processing, characterization, degradation and cytocompatibility. Acta Biomaterialia. 62, 397-417 (2017).
- Li, H., Chang, J. pH-compensation effect of bioactive inorganic fillers on the degradation of PLGA. Composites science and technology. 65 (14), 2226-2232 (2005).
- Xu, C., Hung, C., Cao, Y., Liu, H. H. Tunable crosslinking, reversible phase transition, and 3D printing of hyaluronic acid hydrogels via dynamic coordination of innate carboxyl groups and metallic ions. ACS Applied Bio Materials. 4 (3), 2408-2428 (2021).
- Cortez Alcaraz, M. C., et al. Electrophoretic deposition of magnesium oxide nanoparticles on magnesium: processing parameters, microstructures, degradation, and cytocompatibility. ACS Applied Bio Materials. 2 (12), 5634-5652 (2019).
- Rutherford, D., et al. Synthesis, characterization, and cytocompatibility of yttria stabilized zirconia nanopowders for creating a window to the brain. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 108 (3), 925-938 (2020).
- Tian, Q., Deo, M., Rivera-Castaneda, L., Liu, H. Cytocompatibility of magnesium alloys with human urothelial cells: a comparison of three culture methodologies. ACS Biomaterials Science & Engineering. 2 (9), 1559-1571 (2016).
- Nguyen, T., Cipriano, A., Guan, R. G., Zhao, Z. Y., Liu, H. In vitro interactions of blood, platelet, and fibroblast with biodegradable magnesium-zinc-strontium alloys. Journal of Biomedical Materials Research Part A. 103 (9), 2974-2986 (2015).
- Jiang, W., Lin, J., Chen, A. H., Pan, J., Liu, H. A portable device for studying the effects of fluid flow on degradation properties of biomaterials inside cell incubators. Regenerative Biomaterials. 6 (1), 39-48 (2019).
- Tian, Q., et al. Responses of human urothelial cells to magnesium-zinc-strontium alloys and associated insoluble degradation products for urological stent applications. Materials Science and Engineering: C. 96, 248-262 (2019).
- Wetteland, C. L., Liu, H. Optical and biological properties of polymer-based nanocomposites with improved dispersion of ceramic nanoparticles. Journal of Biomedical Materials Research Part A. 106 (10), 2692-2707 (2018).
- Wetteland, C. L., Nguyen, N. -. Y. T., Liu, H. Concentration-dependent behaviors of bone marrow derived mesenchymal stem cells and infectious bacteria toward magnesium oxide nanoparticles. Acta Biomaterialia. 35, 341-356 (2016).
- Aoi, W., Marunaka, Y. The importance of regulation of body fluid pH in the development and progression of metabolic diseases. Advances in Medicine and Biology. 77, 177-189 (2014).
- Wang, H. . Hydroxyapatite degradation and biocompatibility. , (2004).

