A Modified Sonographic Algorithm for Image Acquisition in Life-Threatening Emergencies in the Critically Ill Newborn
Summary
Here, we present a protocol that can be applied in the neonatal intensive care unit and the delivery room in relation to three scenarios: cardiac arrest, hemodynamic deterioration, or respiratory decompensation. This protocol can be performed with a state-of-the-art ultrasound machine or an affordable handheld device; an image acquisition protocol is carefully detailed.
Abstract
The use of routine point-of-care ultrasound (POCUS) is increasing in neonatal intensive care units (NICUs), with several centers advocating for 24 h equipment availability. In 2018, the sonographic algorithm for life-threatening emergencies (SAFE) protocol was published, which allows the assessment of neonates with sudden decompensation to identify abnormal contractility, tamponade, pneumothorax, and pleural effusion. In the study unit (with a consulting neonatal hemodynamics and POCUS service), the algorithm was adapted by including consolidated core steps to support at-risk newborns, aiding clinicians in managing cardiac arrest, and adding views to verify correct intubation. This paper presents a protocol that can be applied in the NICU and the delivery room (DR) in relation to three scenarios: cardiac arrest, hemodynamic deterioration, or respiratory decompensation.
This protocol can be performed with a state-of-the-art ultrasound machine or an affordable handheld device; the image acquisition protocol is carefully detailed. This method was designed to be learned as a general competence to obtain the timely diagnosis of life-threatening scenarios; the method aims to save time but does not represent a substitute for comprehensive and standardized hemodynamic and radiological analyses by a multidisciplinary team, which might not universally be on call but needs to be involved in the process. From January 2019 to July 2022, in our center, 1,045 hemodynamic consultation/POCUS consults were performed with 25 patients requiring the modified SAFE protocol (2.3%), and a total of 19 procedures were performed. In five cases, trained fellows on call resolved life-threatening situations. Clinical examples are provided that show the importance of including this technique in the care of critical newborns.
Introduction
Ultrasound is a tool that allows an immediate evaluation at the patient's bedside without having to transfer them to another room or floor in the hospital. It can be repeated, it is simple, economical, and precise, and it does not emit ionizing radiation. Ultrasound has been increasingly used by emergency physicians1, anesthesiologists2, and intensivists3 to obtain anatomical and functional images at the patient's bedside. It is a practical tool that is considered by some authors as the fifth pillar of physical examination, as an extension of the human senses4 (inspection, palpation, percussion, auscultation, and insonation)5.
In 2018, the SAFE protocol (for the acronym sonographic algorithm for life threatening emergencies) was published, which allows the assessment of neonates with sudden decompensation (respiratory and/or hemodynamic) to identify alterations in contractility, pericardial effusion with cardiac tamponade (PCE/CT), pneumothorax (PTX), and pleural effusion (PE)6. Our unit is a tertiary-level referral hospital, with most babies needing mechanical ventilation and central catheters; in this context, the SAFE protocol was modified by evaluating the consolidated core steps for a critically ill newborn8, adapting the assistance for cardiac arrest7, taking calcium and glucose, and adding ultrasonographic views to verify intubation. Since 2017, a hemodynamic consultation (HC) and POCUS team has been available in the NICU with dedicated equipment.
Compared to adults, most cases of cardiac arrest in newborns are due to respiratory causes, resulting in pulseless electrical activity (PEA) or asystole. Ultrasound might be a valuable tool adjuvant to traditional resuscitation skills to assess intubation, ventilation, and heart rate (HR)9 and rule out hypovolemia, PCE/CT, and tension PTX. Electrocardiograms have been found to be misleading during neonatal resuscitation, as some newborns may have PEA10,11,12.
The overall goal of this method was to adapt the cited literature to create a sonographic algorithm that can be applied in the NICU and the DR in relation to three scenarios: cardiac arrest, hemodynamic deterioration, or respiratory decompensation. This allows for the expansion of the physical examination by the critical care team to provide a timely diagnosis with correct intubation, including diagnoses of PEA or asystole, abnormal contractility, PCE/CT, PTX, or PE, either using high-end ultrasound equipment (HEUE) or an affordable handheld device (HHD). This algorithm was adapted from the SAFE protocol to be applied both in tertiary level care centers with a NICU-dedicated machine and in the DR and secondary level care centers with reasonably priced portable equipment. This method was designed as a general competence to obtain opportune diagnoses of life-threatening scenarios; the method aims to save time but does not represent a substitute for comprehensive, standardized hemodynamic and radiological analyses performed by a multidisciplinary team, which is essential but not always universally available.
Figure 1 depicts the protocol: a modified sonographic algorithm for life-threatening emergencies in the critically ill newborn. This procedure can be performed with an HEUE or an HHD depending on the healthcare center's resources. In this method, the POCUS team is considered an adjuvant to the attending team; patient management, especially during newborn resuscitation, should be performed according to the latest International Liaison Committee on Resuscitation (ILCOR) recommendations13 and local guidelines, while the sonographer helps as an extra member.
Protocol
This protocol was approved by the institution's human research ethics committee; written consent was obtained for acquiring and publishing anonymized images. Never substitute a traditional maneuver, such as auscultating, for an ultrasound image (they can be done simultaneously or alternately by different operators). The consolidated core steps for a critically Ill newborn are a rapid series of supportive actions that need to be remembered as the POCUS team assesses the patient. Always have a second member of the POCUS team securing the endotracheal tube (ETT). Adapt the scanning to the patient's needs without interfering with resuscitation maneuvers.
1. Ultrasound preparation, specification, and settings14
- Disinfect the transducer and connecting lines to prevent healthcare-associated infections.
NOTE: Always disinfect the equipment before and after use in case of an emergency. - Prepare an HEUE or HHD depending on the situation. See Table 1 for general settings.
- Click on image store after each step on the console or menu on the electronic tablet. Ensure that the imaging obtained is linked to the patient identifier once the emergency is under control.
2. Newborn handling
- Call for help, access the equipment needed for clinical support, and provide warmth (use prewarmed gel).
- Assess airways: Position the infant's head in a neutral position, clear the airway of secretions, and nest the infant whenever possible.
- Oxygen: Administer oxygen as needed to maintain an SpO2 of 90%-95%, or an FiO2 of 100% if the infant is in cardiac arrest.
- Monitor the newborn: Place a pulse oximeter on the infant's right hand, attach cardiopulmonary leads, and use a blood pressure monitor and a cuff of the correct size.
- Obtain the HR, respiratory rate, blood pressure, and axillary temperature8. Obtain point-of-care blood gas analysis (PCBGA) with glucose and calcium.
NOTE: Glucose and calcium disturbances may present as hemodynamic decompensation. The transition from carbohydrate-dependent to fatty acid-dependent metabolism occurs in the first few weeks of life15. In premature infants, contraction depends on the flow of extracellular calcium into the cell as the sarcoplasmic reticulum is physically apart from the L-type channels, the transverse tubules are not present, and the myocytes have a higher surface area to volume ratio16.
3. Verify intubation using the HEUE/HHD in cricothyroid membrane view
- HEUE/HHD
- Select the linear array probe (HEUE 8-18 MHz, HHD 7.5-10 MHz), and press Small Parts on the console or the menu on the electronic tablet.
- Place the linear transducer, with the notch facing to the right, anteriorly on the neck at the level of the cricothyroid membrane (have a second person take care of the airway). Adjust the scanning depth to 2-4 cm.
- Locate the two thyroid lobes at the level of the cricoid. Identify the outline of the ETT (double rail image, also described as the "comet head and tail")17; observe the ETT in situ, generating a posterior shadow (air-mucosa interface with posterior reverberation and shadowing artifacts). Observe the esophagus on the left of the screen (usually collapsed).
NOTE: If the esophagus is dilated with a posterior shadow, this may correspond to esophageal intubation ("double tract" sign) or a nasal or oral gastric tube (Figure 2). - Verify the depth of the ETT with the weight + 6 formula18.
- Perform a longitudinal lung ultrasound (LUS); check for adequate bilateral pleural sliding, the presence of parenchymal signs (B-Lines, consolidation), and the absence of a lung pulse (explained later in the text).
NOTE: If the patient is being intubated at the time, ultrasound may help in identifying the correct position of the tube after the procedure as previously described, or it can help to observe the tracheal and surrounding tissue movement associated with intubation, the double rail image depicting the ETT in the trachea, and the appearance of the posterior acoustic shadowing in real time. If the patient does not have a nasal or oral gastric tube, and the "double tract" sign is identified, this reflects esophageal intubation.
4. Verifying the ETT depth (HEUE) with the aortic arch suprasternal view
- Select the phased array probe (6-12 MHz).
- Press Neonatal Cardiac mode.
- Adjust the scanning depth to 4-6 cm so the complete aortic arch is seen, and open the complete sector width, as it is needed to identify the ETT and aortic arch in one plane.
- Obtain a suprasternal view with the notch by looking at 1-2 o'clock and moving clockwise on a coronal plane until the view of the ETT and the aortic arch is seen.
- Measure the distance from the ETT tip, and ensure it is 0.5-1 cm from the upper border of the aortic arch (Figure 3).
- Only if the conditions permit, have an experienced sonographer (as additional skills are required) verify the depth by ultrasound. The aortic arch is considered an orientation point to locate the carina. If a deep tube is identified (<1 cm or <0.5 cm in premature infants), alongside the presence of a lung pulse, verify the insertion depth clinically, and then perform gentle movements of 0.2 cm and verify bilateral pleural sliding.
NOTE: This method has been validated in several studies19,20. Video 1 depicts a suspected PTX where a lung pulse was encountered; when verifying the depth, a deep tube was identified and retracted. The lung pulse disappeared, and a PTX was diagnosed. Parenchymal signs appeared after chest tube placement.
- Only if the conditions permit, have an experienced sonographer (as additional skills are required) verify the depth by ultrasound. The aortic arch is considered an orientation point to locate the carina. If a deep tube is identified (<1 cm or <0.5 cm in premature infants), alongside the presence of a lung pulse, verify the insertion depth clinically, and then perform gentle movements of 0.2 cm and verify bilateral pleural sliding.
5. Cardiac arrest assessment based on HEUE with subcostal views, an HHD in parasternal long axis view, and an HEUE/HHD LUS
NOTE: While the attending team is performing neonatal resuscitation according to the ILCOR recommendations, the POCUS team prepares the ultrasound equipment. Intubation may be verified by documenting the endotracheal tube in situ and assessing the depth with the weight + 6 formula. Ultrasound may be used to identify the HR21, qualitatively assess the contractility, and rule out PCE/CT.
- HEUE: Subcostal views are performed as they can be obtained without interfering with chest compressions.
- Select the phased array probe (6-12 MHz). Press Neonatal Cardiac mode, click on the up/down button, use the liver as an acoustic window, and ensure that the right atrium is at the bottom of the screen.
- Adjust the scanning depth to 6 cm and the sector width so that part of the liver and the complete heart are seen. Obtain a subcostal long axis (notch: 5 o'clock), utilizing the liver as an acoustic window to the heart.
- Scan from posterior to anterior recognizing (1) the superior vena cava (SVC), (2) the right and left atriums, (3) the left ventricle and aortic valve, and (4) the crossing right ventricle and pulmonary valve (Figure 4). On B-mode imaging, identify the HR, and qualitatively assess the contractility and the absence of PCE/CT.
- Place the transducer under the xiphoid region with the notch facing 3-5 o'clock, and sweep from side to side to scan the diaphragm and the bottom of the lungs,using the liver as an acoustic window (Figure 5). Assess for PCE/CT and PE.
- Perform LUS searching for parenchymal signs (B-lines, consolidation) during ventilation to rule out PTX (see later in the text).
- HHD: Parasternal long axis view and LUS
- Select the linear array probe (7.5-10 MHz). Press Small Parts on the menu on the electronic tablet.
- Adjust the scanning depth to 4-6 cm. Alternating between chest compressions if needed or after returning to circulation, obtain a parasternal long axis view with the linear handheld probe. Point the notch to the left shoulder, and then rotate clockwise to 3-4 o'clock until the right ventricle is on top of the screen and the descending aorta is at the bottom.
- Identify (1) the right ventricle, (2) the interventricular septum, (3) the aortic valve, (4) the left ventricle, (5) the mitral valve, (6) the left atrium, (7) the pericardium, and (8) the descending aorta (Figure 6). Assess the HR, contractility, and the presence of PCE/CT.
- Perform LUS searching for parenchymal signs (B-lines, consolidation) during ventilation to rule out PTX (see later in the text).
- During cardiac arrest, obtain views twice in relation to neonatal ressucitation22.
- After performing corrective steps to improve the mask ventilation performance, and if still encountering an HR of <100, perform CU to detect the HR and effective cardiac output and ensure a real asystole.
- After advanced cardiopulmonary resuscitation (CPR) with chest compressions and adrenaline dose, perform CU to rule out PCE/CT and hypovolemia, and perform LUS to detect PTX (see later).
NOTE: The descending aorta is a key landmark for distinguishing a left pleural effusion from a pericardial effusion in long axis view. Fluid anterior to the descending aorta (toward the top of the screen) is pericardial effusion, and fluid posterior to the descending aorta is likely pleural effusion23. It may be impossible to obtain a parasternal view in severe cases of pneumomediastinum.
6. Hemodynamic instability (hypoperfusion, hypotension, with or without respiratory deterioration)24
- Hemodynamic instability assessed using HEUE in subxiphoid long axis, four chamber view.
- Select the Phased array probe (6-12 MHz).
- Press Neonatal cardiac mode, click on the up/down button, use the liver as an acoustic window, and ensure that the right atrium is at the bottom of the screen.
- Adjust the scanning depth to 6 cm and the sector width so part of the liver and the complete heart are seen.
- Obtain a subcostal long axis view (notch: 5 o'clock) utilizing the liver as an acoustic window to the heart.
- Scan from posterior to anterior recognizing (1) the superior vena cava (SVC), (2) the right and left atriums, (3) the left ventricle and aortic valve, and (4) the crossing right ventricle and pulmonary valve (Figure 4). On B-mode imaging, identify the HR, and qualitatively assess the contractility and the absence of PCE/CT (Figure 4).
- Press color on the console; adjust the velocity to a scale of 70-80 cm/s. Observe the crossing of the great vessels and adequate outflow without aliasing and acceleration.
- Click on 2D, and obtain a four-chamber view with the notch of the transducer directed toward the left axilla at the 2-3 o'clock position as viewed from the apex. Identify (1) the right atrium, (2) the tricuspid valve, (3) the right ventricle, (4) the interventricular septum, (5) the left atrium, (6) the mitral valve, and (7) the left ventricle (Figure 7). Subjectively evaluate the contractility by examining the change in ventricular cavity size during systole.
- Click on the M mode button. To evaluate the contractility, using the track ball, place the cursor on the tricuspid and mitral annulus to calculate the tricuspid and mitral annular systolic excursion (TAPSE/MAPSE), and compare it to the nomograms according to gestational age25,26.
- Assess the cardiac filling and fluid status. Differentiate a normal filled heart versus an underfilled one by evaluating the end diastolic area, where obliteration of the cavity (empty "kissing" ventricles) suggests hypovolemia, whereas an overloaded heart often appears dilated with poor contractility.
- Determine further management with a hemodynamic/pediatric cardiology consult27. Rule out PCE/CT by looking for a large pericardial effusion (circumferential) with altered contractility, which is indicative of PCE/CT.
- HHD with parasternal long axis view
- Select the linear array probe (7.5-10 MHz). Press Small Parts on the menu on the electronic tablet.
- Adjust the scanning depth to 4-6 cm. Obtain a parasternal long axis view with the linear handheld probe. Point the notch to the left shoulder, and then rotate clockwise to 3-4 o'clock until the right ventricle is on top of the screen and the descending aorta is at the bottom.
- Identify (1) the right ventricle, (2) the interventricular septum, (3) the aortic valve, (4) the left ventricle, (5) the mitral valve, (6) the left atrium, (7) the pericardium, and (8) the descending aorta (Figure 6). Subjectively evaluate the contractility by examining the change in the ventricular cavity size during systole.
- Assess the cardiac filling and fluid status. Differentiate a normal filled heart versus an underfilled one by evaluating the end diastolic area, where obliteration of the cavity (empty "kissing" ventricles) suggest hypovolemia, whereas an overloaded heart appears dilated and often has poor contractility.
- Determine further management with a hemodynamic/pediatric cardiology consult. Rule out PCE/CT, as indicated by fluid anterior to the descending aorta.
NOTE: See the representative results for notes on assessing the cardiac function. Figure 8 shows images of systolic right atrial collapse and diastolic right ventricular collapse during PCE/CT28.
7. Exclusive respiratory symptoms (normal blood pressure and perfusion)
- Using HEUE/HHD for LUS, longitudinal and transversal scans. Lung ultrasound semiology has been described by Liu and collaborators (Table 2)29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45
- Select the linear array probe (HEUE 8-18 MHz, HHD 7.5-10 MHz). Press Small Parts on the console or on the menu on the electronic tablet. Turn off harmonics.
- Adjust the scanning depth to 4-6 cm. Divide the thorax into six regions using the anterior and posterior axillary lines, as well as the parasternal lines. Identify the following: a) the anterior region from the parasternal line to the anterior axillary line, and then use the intermammary line to divide into the upper and lower anterior regions; b) the lateral region from the anterior to posterior axillary line.
- Perform a longitudinal scan with the notch facing up (perpendicular to the ribs) and with medial to lateral slide in both the anterior and posterior regions. Obtain clips of 6-10 s. Rotate the transducer 90° (notch to the right) to scan from top to bottom through the intercostal spaces.
- Assess pleural sliding to search for a PTX. Identify the to-and-fro motion of the pleural line, which synchronizes with the respiratory movement. The presence of parenchymal signs (B-lines, consolidation) rules out PTX. Perform M-mode to search for the "Bar code" sign (Figure 9).
- Rotate the transducer 90°, and place the transducer between the second and third intercostal spaces to obtain the anterior superior transverse plane with the notch pointing to the right. The sternum and mediastinal structures (the thymus, SVC, aorta, and pulmonary artery and branches) are observed in a healthy newborn (Figure 10).
- On longitudinal lateral scans, identify the presence of a PE, which is characterized by the accumulation of fluid in the pleural cavity (Figure 11).
NOTE: On some HHDs, the harmonics function allows the user to increase the frequency from 7.5 MHz to 10 MHz so that it can be maintained in preterm infants. Ultrasound allows the detection of pleural fluid in amounts as small as 3-5 mL, which cannot be identified by radiographs. Be mindful of the ultrasound depth, as modern machines allow great amplification, and the amount of fluid might be overestimated.
8. Drainage (HEUE/HHD)
NOTE: In all cases, use sterile technique.
- Perform emergency procedures if there is significant hemodynamic instability, impending deterioration, or cardiac arrest.
- Use an 18-20 G needle or an angiocatheter connected to a 20 mL syringe and a three-way stopcock. Keep the newborn comfortable, and ensure adequate pain control if possible. Swab the area with chlorhexidine.
- PCE/CT46
- Place a high-frequency linear transducer horizontally in the subcostal area with the marker pointing caudally.
NOTE: The optimal place for echocardiography-guided pericardiocentesis is the largest, shallowest fluid pocket with no intervening vital structures. - Palpate the xiphoidal process, and insert the needle (visualized piercing the pericardial sac) just below it at an angle of 30° to the skin, with the needle tip pointing toward the left shoulder. Once a flashback is obtained, stop advancing the needle, and continue to aspirate the maximal amount of fluid using the syringe.
- Place a high-frequency linear transducer horizontally in the subcostal area with the marker pointing caudally.
- PTX33
- Identify a suitable puncture point away from the sliding portion if a lung point is present, ensuring that only an A-line pattern with no pleural sliding exists ("Barcode sign" in M-mode). Adopt a supine, prone, or side position, allowing the air on the affected side to rise.
- Insert the needle into the intercostal space at the superior margin of the lower rib to avoid damage to the neurovascular bundle. Evacuate the pleural air by needle aspiration, and consider the placement of a chest tube based on the situation.
- PE41
- Identify a suitable puncture point; choose the deepest pool of fluid. Adopt a supine or lateral position, with the upper part of the body slightly elevated, allowing fluid to accumulate due to gravity at the lowest point of the pleural space.
- Insert the needle into the intercostal space at the superior margin of the lower rib to avoid damage to the neurovascular bundle. Evacuate the pleural fluid by needle aspiration, and consider the placement of a chest tube based on the situation.
Representative Results
The inspection of cardiac function by "eyeballing" can be applied to qualitatively assess the global cardiac systolic function. Any suspicion of impaired cardiac function should lead to an urgent HC with pediatric cardiology for the assessment of congenital heart disease (CHD). Treatment must be started according to the pathophysiology, and the treatment should be integrated and modified according to a comprehensive anatomical and functional echocardiography study27. If ductal-dependent CDH is suspected, prostaglandins must be started, and a pediatric cardiology consult must be scheduled. In the study center, pediatric cardiology and neonatal hemodynamics consultation services are available.
From January 2019 to July 2022, a total of 1,045 HC/POCUS studies were carried out in our hospital, of which 25 corresponded to the protocol (2.3%). The type of decompensation was classified as respiratory in 14 newborns, hemodynamic in 8 newborns, and cardiac arrest-related (one PEA and one tamponade) in 3 newborns. The ultrasound protocol diagnoses were PTX (12), PE (4), PCE/CT (3), altered contractility (2), cardiac arrest-related (2), mobilization of the endotracheal tube (1), and hypoglycemia (1).
The protocol and interventions were performed by an expert neonatologist with advanced ultrasound training in 8 patients, by neonatology fellows supervised by an expert in 12 patients, and by fellows exclusively in 5 patients (including the resolution of three tension PTX cases and two tamponade drains). Most (96%) of the patients survived the event, and 68% survived to discharge. Overall, 19 procedures were performed (five chest tubes, three chest tube corrections, four pneumothorax needle drainages, four pleural effusion needle drainages, and three tamponade needle drainages), an endotracheal tube adjustment was performed, and one glucose bolus was administered. The chest X-ray (CXR) corresponding to each event was found in the electronic system at a median (interquartile range) of 58 (27-97) min. Table 3 details the institution's experience with this protocol.
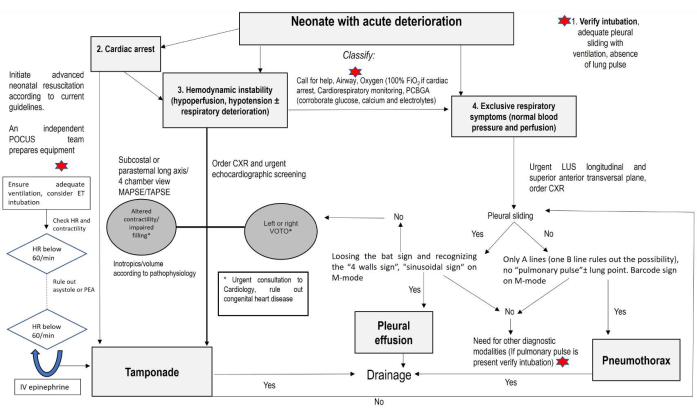
Figure 1: Algorithm: A modified sonographic algorithm for life-threatening emergencies in the critically ill newborn. Start by assessing the airway if the newborn is intubated, perform the consolidated core steps to ensure the newborn is monitored, and obtain the PCBGA. If the infant is in cardiac arrest, assistance (image acquisition) can be provided in two steps: a) performing corrective steps to detect the HR and effective cardiac output and ensure a real asystole; b) performing advanced CPR to rule out PCE/CT and hypovolemia and performing LUS to detect PTX. If hemodynamic instability (hypoperfusion, hypotension, with or without respiratory deterioration) is present, assess contractility, assess the left or right VOTO, and rule out PCE/CT. If negative or exclusive respiratory symptoms (normal blood pressure and perfusion) are present, rule out PTX and PE. Abbreviations: PCBGA = point of care blood gas analysis; POCUS = point of care ultrasound; ET = endotracheal; HR = heart rate; PEA = pulseless electrical activity; MAPSE = mitral annular systolic excursion; TAPSE = tricuspid annular systolic excursion; CXR = chest X-ray; VOTO = ventricular outflow tract obstruction; PCE/CT = pericardial effusion/cardiac tamponade; PTX = pneumothorax; PE = pleural effusion. Please click here to view a larger version of this figure.
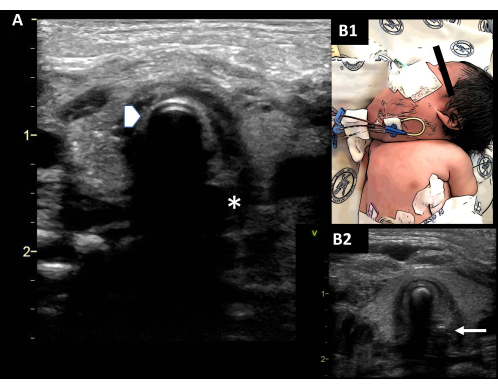
Figure 2: Verifying intubation. (A) Observe the outline of the ETT (double rail image, arrowhead), which generates a posterior shadow. The esophagus on the left of the screen is collapsed (asterisk). (B1) Difficult airway in a newborn with lymphangioma. (B2) The ETT is observed in situ; a small orogastric tube is observed (arrow). Abbreviation: ETT = endotracheal tube. Please click here to view a larger version of this figure.
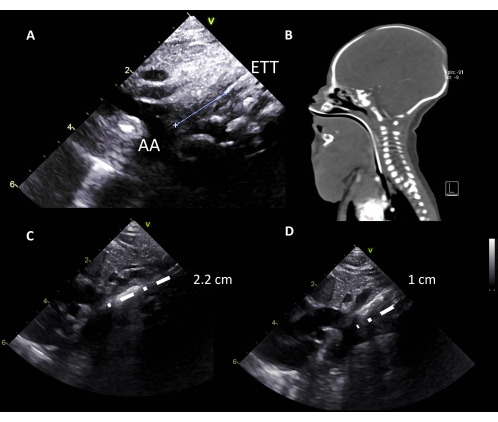
Figure 3: ETT depth. (A) The aortic arch is considered an orientation point to locate the carina, and the ETT is located at 1 cm from the AA. (B) Difficult airway in a newborn with lymphangioma; a high ETT is detected. (C) A high ETT (2.2 cm from the AA) is seen on the ultrasound and corrected. (D) Correctly placed ETT (1 cm from the AA). Abbreviations: AA = aortic arch; ETT = endotracheal tube. Please click here to view a larger version of this figure.
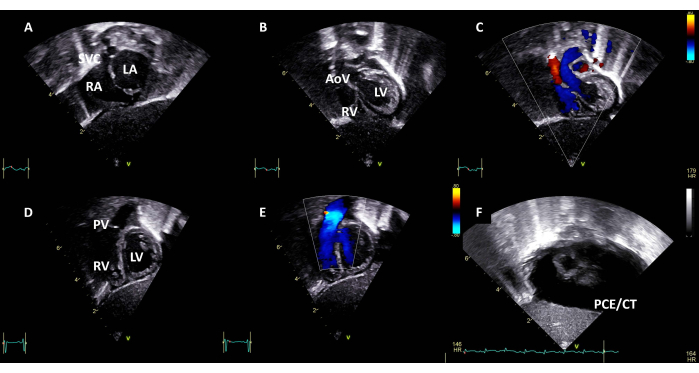
Figure 4: Subcostal long axis view. Sweeping from posterior to anterior, identify (A) the superior vena cava, the right and left atriums; (B) the right and left ventricles and the aortic valve; (C) color Doppler, indicating left ventricular outflow tract without obstruction; (D) and the crossing right ventricle and pulmonary valve. (E) Color doppler, indicating right ventricular outflow tract without obstruction. (F) Subcostal view with PCE/CT. Abbreviations: SVC = superior vena cava; RA = right atrium; LA = left atrium; RV = right ventricle; LV = left ventricle; AoV = aortic valve; PV = pulmonary valve; PCE/CT = pericardial effusion with cardiac tamponade. Please click here to view a larger version of this figure.
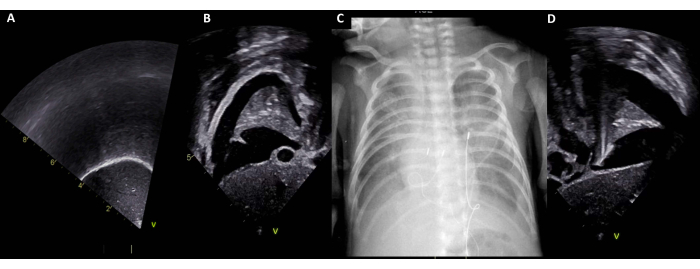
Figure 5: Transdiaphragmatic window. (A) Normal right transdiaphragmatic window. (B) Right PE. (C) Corresponding CXR with bilateral PE. (D) Left PE. Abbreviations: PE = pleural effusion; CXR = chest X-ray. Please click here to view a larger version of this figure.
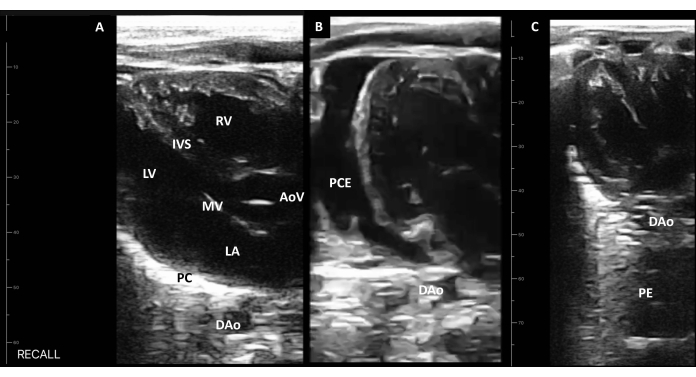
Figure 6: Handheld device long axis view. (A) Identify the right ventricle, the interventricular septum, the aortic valve, the left ventricle, the mitral valve, the left atrium, the pericardium, and the descending aorta. (B) The PCE identified as fluid anterior to the DAo. (C) The PE posterior to the DAo. Abbreviations: LA = left atrium; RV = right ventricle; LV = left ventricle; AoV = aortic valve; IVS = interventricular septum; MV = mitral valve; PC = pericardium; DAo = descending aorta; PCE = pericardial effusion; PE = pleural effusion. Please click here to view a larger version of this figure.
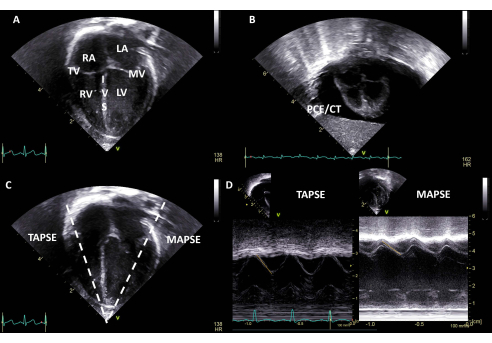
Figure 7: Four chamber view. (A) Identify the right atrium, the tricuspid valve, the right ventricle, the interventricular septum, the left atrium, the mitral valve, and the left ventricle. (B) Four chamber view with PCE/CT. (C) An M-mode image can be obtained on the tricuspid and mitral annulus to calculate the TAPSE/MAPSE. (D) TAPSE and MAPSE are depicted; the measurement in millimeters (mm) can be compared to gestational age nomograms. Abbreviations: SVC = superior vena cava; RA = right atrium; LA = left atrium; RV = right ventricle; LV = left ventricle; PCE/CT = pericardial effusion with cardiac tamponade; TV = tricuspid valve; MV = mitral valve; IVS = interventricular septum; TAPSE = tricuspid annular systolic excursion; MAPSE = mitral annular systolic excursion. Please click here to view a larger version of this figure.
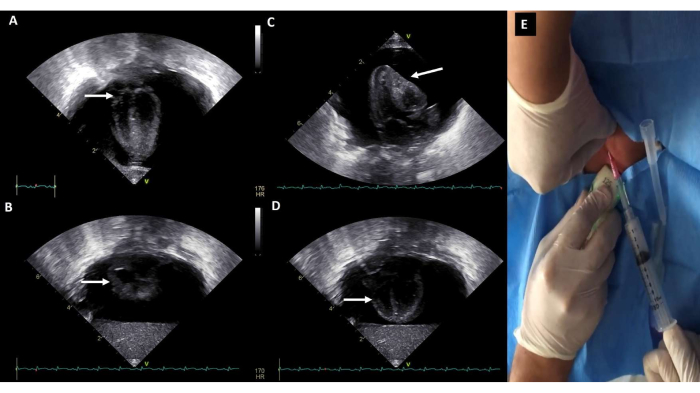
Figure 8: Pericardial effusion with cardiac tamponade. Large circumferential pericardial effusion. (A,B) A systolic right atrial collapse and (C,D) diastolic right ventricular collapse are observed qualitatively. (E) Pericardiocentesis. Please click here to view a larger version of this figure.
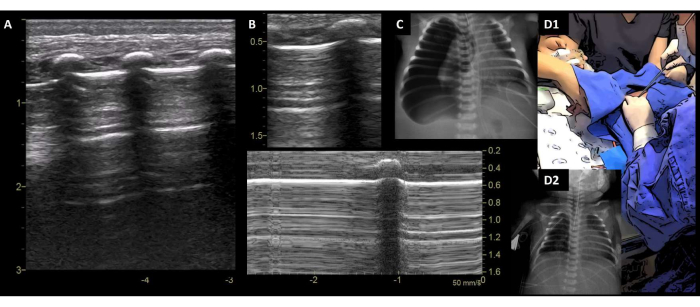
Figure 9: Pneumothorax. (A) PTX is diagnosed with absent pleural sliding, only A-lines, and no "lung pulse". (B) M-mode image shows the "Bar code sign". (C) Corresponding X-rays. (D1) Chest tube insertion. (D2) PTX resolved on a control CXR. Abbreviations: PTX = pneumothorax; CXR = chest X-ray. Please click here to view a larger version of this figure.
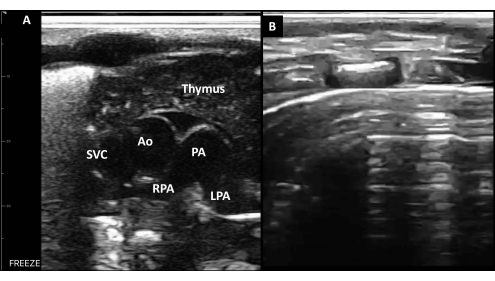
Figure 10: Anterior-superior transverse plane. (A) In a healthy newborn, the sternum and mediastinal structures, including the thymus, the superior vena cava, the aorta, and the pulmonary artery with its right and left branch, can be observed. (B) A-lines in the anterior transverse plane without sliding is a sensitive sign of anterior PTX. Abbreviations: SVC = superior vena cava; Ao = aorta; PA = pulmonary artery; RPA = right PA branch; LPA = left PA branch. Please click here to view a larger version of this figure.
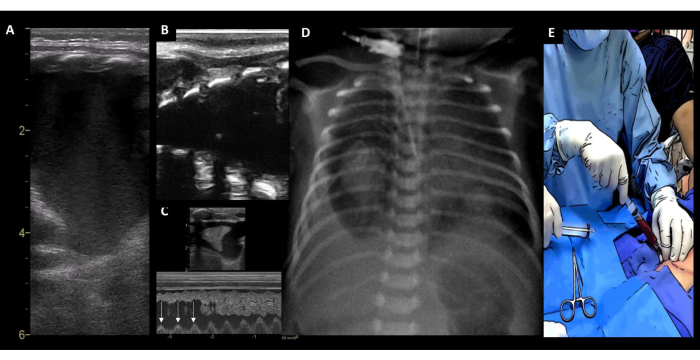
Figure 11: Pleural effusion. (A) PE identified by the absence of the bat sign and the "four walls sign" (high-end ultrasound equipment). (B) Same PE identified with a hand-held device. (C) M-mode image showing the "sinusoidal sign" (with each respiratory cycle, the lung surface line moves toward the pleural line, arrow). (D) Corresponding CXR. (E) Drainage of the hemothorax. Abbreviations: PE = pleural effusion; CXR = chest X-ray. Please click here to view a larger version of this figure.
Video 1: Lung pulse, deep ETT, and pneumothorax. A preterm newborn with respiratory decompensation and a suspected PTX, but a lung pulse was encountered; in verifying the ETT depth, a deep tube was recognized and retracted. The lung pulse disappeared, and a PTX was diagnosed. Parenchymal signs appeared after chest tube placement. The corresponding X-rays are shown. Please click here to download this Video.
Table 1: Ultrasound settings. Please click here to download this Table.
Table 2: Lung ultrasound semiology29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45. Abbreviations: PTX = pneumothorax; SVC = superior vena cava; PE = pleural effusion; ETT = endotracheal tube. Please click here to download this Table.
Table 3: Center experience. Abbreviations: DT = deterioration type; GA = gestational age; PDL = postnatal day of life; SF = supervised fellow; A = attending neonatologist; NF = neonatology fellow; SE = survived event; SD = survived discharge; Y = yes; N = no; RDS = respiratory distress syndrome; PDA = patent ductus arteriosus; VSD = ventricular septal defect; PO = post operated; ROP = retinopathy of prematurity; IVH = intraventricular hemorrhage; ETT = endotracheal tube; NEC = necrotizing enterocolitis. Please click here to download this Table.
Discussion
Compared to children and adults, most cases of acute deterioration/cardiac arrest are due to respiratory causes in newborns. The original SAFE protocol was modified in our unit, a tertiary referral care neonatal center, due to this unit expecting several ventilated patients with indwelling catheters. The protocol has been adapted to different scenarios and equipment for use in low- and medium-income countries. As an institution with a neonatal hemodynamics and POCUS program, and after giving LUS workshops in different states of the Republic, we noted the need to integrate ultrasound to enhance neonatal care.
The critical steps in the protocol include the categorization of the patient into three starting scenarios (cardiac arrest, hemodynamic deterioration, or respiratory decompensation) and the addition of some steps where ultrasound might help the critical care/resuscitation team.
One of the steps included is verifying intubation, which can be performed at several points of the algorithm according to the patient's needs. Transtracheal ultrasound has a sensitivity of 98.7% (95% confidence interval [CI]: 97.8%-99.2%) and a specificity of 97.1% (95% CI: 92.4%-99.0%)47. Once the ETT is detected in situ, the depth can be checked with the Tochen formula18. Additionally, correct intubation is confirmed by documenting adequate pleural sliding on both sides, as well as the presence of parenchymal signs (B-lines, consolidation) and the absence of a lung pulse. Ultrasound can be used to verify the depth of the ETT only if a skilled sonographer is present, the condition of the patient allows it, and deterioration is considered dependent on the airway (e.g., the presence of a lung pulse). In a study with neonates weighing 1,282 g ± 866 g, considering a tube "deep" (<1 cm) compared to CXR showed a sensitivity of 86% and a specificity of 96%48. In this work, the tube was demonstrated in situ in all the cases with an intubated patient. Only in one case was a displaced ETT the cause of respiratory decompensation.
We consider the POCUS team as a valuable adjuvant to the attending team performing neonatal resuscitation. As mentioned earlier, the POCUS team might help by detecting the HR and effective cardiac output and ensuring a real asystole or PEA in the first step10,11,12,21,22. After advanced CPR, the POCUS team may help rule out PCE/CT and hypovolemia (empty right and left ventricles) and perform LUS to detect PTX21,22. In one of our cases, the POCUS team was called to a preterm infant who was being ventilated. The cardiac monitor indicated an HR of 80 bpm, but the ultrasound image detected asystole (PEA). Immediate chest compressions were started as the attending team was ventilating only because the monitor indicated an HR ˃60 bpm.
Ultrasound provides useful, additional information to the conventional treatment of a crashing infant. Modern PCBGA provides the levels of glucose, calcium, and electrolytes, so reversible causes can be immediately addressed considering the 7Hs, including hypovolemia (POCUS), hypoxia (PCBGA), hydrogenation/acidosis (PCBGA), hypothermia (clinical), hypoglycemia (PCBGA), hypo/hyperkalemia (PCBGA), hypocalcemia (PCBGA), and the 2Ts, including tamponade and tension pneumothorax. In one of our cases, in a newborn classified with hemodynamic decompensation (pale, hypotensive, lethargic), the etiology was hypoglycemia detected with PCBGA.
PCE/CT is infrequent but linked to high mortality. PCE/CT is closely related to the presence of a central line and the tip position (as the pericardial fluid found is normally consistent with the infusate) and commonly affects very-low-birthweight (VLBW) infants49. Survival improves when PCE/CT is detected early and treated promptly50,51. In units caring for VLBW infants and surgical patients, a dedicated ultrasound machine is recommended for immediate access. When a significant PCE causing CT is found, normally a blind procedure can be performed safely. Nevertheless, the fact that the same probe used for diagnosing helps in guiding the procedure improves patient safety and decreases the complication rate to a minimum52. In our series, three PCE/CT cases were diagnosed, with two survivors (draining with parenteral nutrition in one case and normal saline with antibiotics in the other) and one death (hemopericardium). A large PE causing hemodynamic instability or cardiac arrest is infrequent, but in case it presents, ultrasound diagnostic performance for fluid is high, and drainage can be performed safely. In some scenarios of neonatal resuscitation, such as hydrops, ultrasound guidance is essential.
The subjective evaluation of cardiac contractility, ventricular filling, and outflow assessment can guide the neonatologist to begin with a pathophysiology-suitable treatment and to perform an appropriate pediatric cardiology and hemodynamic consultation. It is of great value to identify an underfilled heart and differentiate it from volume overload and altered contractility, as the treatment is different24. In our unit, we advocate for the practice of advanced neonatal hemodynamics with highly trained members of the team; however, all our neonatology fellows must acquire basic POCUS skills as they are the primary care providers. In this series, one newborn was observed to have altered contractility and ventricular dilatation, which led to a prompt diagnosis of an aortic coarctation.
The LUS diagnostic accuracy for PTX is very high and can even reach 100% in terms of sensitivity, specificity, and positive and negative predictive values. As its superiority is staggering compared to CXR and transillumination with respect to time, there is enough evidence to consider LUS as the first-line diagnostic test53. Either with HEUE or an HHD, procedures can be safely performed while avoiding sliding portions where aerated lung is present. Using this algorithm, 12 PTX cases were successfully diagnosed and treated.
There is mostly moderate evidence regarding the use of cardiac, lung, vascular, cerebral, and abdominal POCUS54. POCUS protocols need to be individualized according to different centers' needs in close collaboration with cardiology and radiology to ensure quality care. It is fundamental to include POCUS skills on the curriculum for neonatology fellows as many complications occur on call. Immediate equipment availability is essential to ensure a successful program.
This protocol warrants further external validation to prove its generalizability. This modified protocol has limitations as it is focused on cardiopulmonary deterioration in the NICU and relies on prompt expert consultation (HC, pediatric cardiology). Recently, a protocol has been published on hemodynamic precision in the neonatal intensive care unit using targeted neonatal echocardiography (TnECHO)55. This expert consultative model in which a neonatologist performs an HC (a comprehensive and standardized echocardiographic assessment with a recommendation based on advanced hemodynamics knowledge) needs advanced training. The objective of this protocol is to present it as a general competence to ensure that the neonatologist on call (in a unit with an ultrasound in the NICU) has the ability to diagnose and treat life threatening emergencies. Additionally, the recently published sonographic assessment of life-threatening emergencies-revised (SAFE-R)56 has added the recognition of acute critical aortic occlusion, acute abdominal complications, and severe intraventricular hemorrhage.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank Dr. Nadya Yousef, Dr. Daniele De Luca, Dr. Francesco Raimondi, Dr. Javier Rodriguez Fanjul, Dr. Almudena Alonso-Ojembarrena, Dr. Shazia Bhombal, Dr. Patrick McNamara, Dr. Amish Jain, Dr. Ashraf Kharrat, the Neonatal Hemodynamics Research Center, Dr. Yasser Elsayed, Dr. Muzafar Gani, and the POCUSNEO group for their support and feedback.
Materials
| Conductivity gel | Ultra/Phonic, Pharmaceutical innovations, New Jersey, United States | 36-1001-25 | |
| Handheld linear probe, 10.0 MHz | Konted, Beijing, China | C10L | handheld device |
| Hockey stick probe 8–18 MHz, L8-18I-SC Probe | GE Medical Systems, Milwaukee, WI, United States | H40452LZ | high-end ultrasound equipment |
| iPad Air 2 | Apple Inc | MGWM2CL/A | electronic tablet |
| Phased array probe 6-12 MHz, 12S-D Phased Array Probe | GE Medical Systems, Milwaukee, WI, United States | H45021RT | high-end ultrasound equipment |
| Vivid E90 v203 Console Package | GE Medical Systems, Milwaukee, WI, United States | H8018EB | Vivid E90 w/OLED monitor v203 Console |
References
- Kameda, T., Kimura, A. Basic point-of-care ultrasound framework based on the airway, breathing, and circulation approach for the initial management of shock and dyspnea. Acute Medicine & Surgery. 7 (1), 481 (2020).
- Adler, A. C., Matisoff, A. J., DiNardo, J. A., Miller-Hance, W. C. Point-of-care ultrasound in pediatric anesthesia: Perioperative considerations. Current Opinion in Anaesthesiology. 33 (3), 343-353 (2020).
- Sen, S., Acash, G., Sarwar, A., Lei, Y., Dargin, J. M. Utility and diagnostic accuracy of bedside lung ultrasonography during medical emergency team (MET) activations for respiratory deterioration. Journal of Critical Care. 40, 58-62 (2017).
- Soldati, G., Smargiassi, A., Mariani, A. A., Inchingolo, R. Novel aspects in diagnostic approach to respiratory patients: Is it the time for a new semiotics. Multidisciplinary Respiratory Medicine. 12 (1), 15 (2017).
- Narula, J., Chandrashekhar, Y., Braunwald, E. Time to add a fifth pillar to bedside physical examination: Inspection, palpation, percussion, auscultation, and insonation. JAMA Cardiology. 3 (4), 346-350 (2018).
- Raimondi, F., Yousef, N., Migliaro, F., Capasso, L., de Luca, D. Point-of-care lung ultrasound in neonatology: Classification into descriptive and functional applications. Pediatric Research. 90 (3), 524-531 (2021).
- Kharrat, A., Jain, A. Guidelines for the management of acute unexpected cardiorespiratory deterioration in neonates with central venous lines in situ. Acta Paediatrica. 107 (11), 2024-2025 (2018).
- Boulton, J. E., Coughlin, K., O’Flaherty, D., Solimano, A. . ACoRN: Acute care of at-risk newborns: A resource and learning tool for health care professionals. , (2021).
- Johnson, P. A., Schmölzer, G. M. Heart rate assessment during neonatal resuscitation. Healthcare. 8 (1), 43 (2020).
- Luong, D., et al. Cardiac arrest with pulseless electrical activity rhythm in newborn infants: A case series. Archives of Disease in Childhood. Fetal and Neonatal Edition. 104 (6), F572-F574 (2019).
- Levitov, A., et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-Part II: Cardiac ultrasonography. Critical Care Medicine. 44 (6), 1206-1227 (2016).
- Hodgson, K. A., Kamlin, C. O. F., Rogerson, S., Thio, M. ECG monitoring in the delivery room is not reliable for all patients. Archives of Disease in Childhood. Fetal and Neonatal Edition. 103 (1), F87-F88 (2018).
- Wyckoff, M. H., et al. Neonatal life support 2020 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Resuscitation. 142, S185-S221 (2020).
- Liu, J., et al. Specification and guideline for technical aspects and scanning parameter settings of neonatal lung ultrasound examination. The Journal of Maternal-Fetal & Neonatal Medicine. 35 (5), 1003-1016 (2022).
- Schmidt, M. R., et al. Glucose-insulin infusion improves cardiac function during fetal tachycardia. Journal of the American College of Cardiology. 43 (3), 445-452 (2004).
- Wiegerinck, R. F., et al. Force frequency relationship of the human ventricle increases during early postnatal development. Pediatric Research. 65 (4), 414-419 (2009).
- Galicinao, J., Bush, A. J., Godambe, S. A. Use of bedside ultrasonography for endotracheal tube placement in pediatric patients: A feasibility study. Pediatrics. 120 (6), 1297-1303 (2007).
- Tochen, M. L. Orotracheal intubation in the newborn infant: A method for determining depth of tube insertion. The Journal of Pediatrics. 95 (6), 1050-1051 (1979).
- Zaytseva, A., Kurepa, D., Ahn, S., Weinberger, B. Determination of optimal endotracheal tube tip depth from the gum in neonates by X-ray and ultrasound. The journal of maternal-fetal & neonatal medicine. 33 (12), 2075-2080 (2020).
- Sandig, J., Bührer, C., Czernik, C. Evaluation of the endotracheal tube by ultrasound in neonates. Zeitschrift fur Geburtshilfe und Neonatologie. 226 (3), 160-166 (2022).
- Bobillo-Perez, S., et al. Delivery room ultrasound study to assess heart rate in newborns: DELIROUS study. European Journal of Pediatrics. 180 (3), 783-790 (2021).
- Rodriguez-Fanjul, J., Perez-Baena, L., Perez, A. Cardiopulmonary resuscitation in newborn infants with ultrasound in the delivery room. The Journal of Maternal-Fetal & Neonatal Medicine. 34 (14), 2399-2402 (2021).
- Lewandowski, B. J., Jaffer, N. M., Winsberg, F. Relationship between the pericardial and pleural spaces in cross-sectional imaging. Journal of Clinical Ultrasound. 9 (6), 271-274 (1981).
- Singh, Y., Bhombal, S., Katheria, A., Tissot, C., Fraga, M. V. The evolution of cardiac point of care ultrasound for the neonatologist. European Journal of Pediatrics. 180 (12), 3565-3575 (2021).
- Koestenberger, M., et al. Systolic right ventricular function in preterm and term neonates: Reference values of the tricuspid annular plane systolic excursion (TAPSE) in 258 patients and calculation of Z-score values. Neonatology. 100 (1), 85-92 (2011).
- Koestenberger, M., et al. Longitudinal systolic left ventricular function in preterm and term neonates: Reference values of the mitral annular plane systolic excursion (MAPSE) and calculation of z-scores. Pediatric Cardiology. 36 (1), 20-26 (2015).
- Giesinger, R. E., McNamara, P. J. Hemodynamic instability in the critically ill neonate: An approach to cardiovascular support based on disease pathophysiology. Seminars in Perinatology. 40 (3), 174-188 (2016).
- Alerhand, S., Adrian, R. J., Long, B., Avila, J. Pericardial tamponade: A comprehensive emergency medicine and echocardiography review. The American Journal of Emergency Medicine. 58, 159-174 (2022).
- Liu, J., et al. Protocol and guidelines for point-of-care lung ultrasound in diagnosing neonatal pulmonary diseases based on international expert consensus. Journal of Visualized Experiments. (145), e58990 (2019).
- Almudena, A. O., Alfonso María, L. S., Estefanía, R. G., Blanca, G. H. M., Simón Pedro, L. L. Pleural line thickness reference values for preterm and term newborns. Pediatric Pulmonology. 55 (9), 2296-2301 (2020).
- Rodríguez-Fanjul, J., Balcells Esponera, C., Moreno Hernando, J., Sarquella-Brugada, G. La ecografía pulmonar como herramienta para guiar la surfactación en neonatos prematuros. Anales de Pediatría. 84 (5), 249-253 (2016).
- Lichtenstein, D. A., Lascols, N., Prin, S., Mezière, G. The "lung pulse": An early ultrasound sign of complete atelectasis. Intensive Care Medicine. 29 (12), 2187-2192 (2003).
- Liu, J., et al. International expert consensus and recommendations for neonatal pneumothorax ultrasound diagnosis and ultrasound-guided thoracentesis procedure. Journal of Visualized Experiments. (157), e60836 (2020).
- Cattarossi, L., Copetti, R., Brusa, G., Pintaldi, S. Lung ultrasound diagnostic accuracy in neonatal pneumothorax. Canadian Respiratory Journal. 2016, 6515069 (2016).
- Alrajab, S., Youssef, A. M., Akkus, N. I., Caldito, G. Pleural ultrasonography versus chest radiography for the diagnosis of pneumothorax: Review of the literature and meta-analysis. Critical Care. 17 (5), R208 (2013).
- Raimondi, F., et al. Lung ultrasound for diagnosing pneumothorax in the critically ill neonate. The Journal of Pediatrics. 175, 74-78 (2016).
- Liu, J., et al. Lung ultrasonography to diagnose pneumothorax of the newborn. The American Journal of Emergency Medicine. 35 (9), 1298-1302 (2017).
- Lichtenstein, D., Mezière, G., Biderman, P., Gepner, A. The "lung point": An ultrasound sign specific to pneumothorax. Intensive Care Medicine. 26 (10), 1434-1440 (2000).
- Montero-Gato, J., et al. Ultrasound of pneumothorax in neonates: Diagnostic value of the anterior transverse plane and of mirrored ribs. Pediatric Pulmonology. 57 (4), 1008-1014 (2022).
- Kurepa, D., Zaghloul, N., Watkins, L., Liu, J. Neonatal lung ultrasound exam guidelines. Journal of Perinatology. 38 (1), 11-22 (2018).
- Soffiati, M., Bonaldi, A., Biban, P. La gestione del drenaggio pleurico [Management of pleural drainage]. Minerva Pediatrica. 62 (3), 165-167 (2010).
- Lichtenstein, D. A. Ultrasound examination of the lungs in the intensive care unit. Pediatric Critical Care Medicine. 10 (6), 693-698 (2009).
- Cantinotti, M., et al. Overview of lung ultrasound in pediatric cardiology. Diagnostics. 12 (3), 763 (2022).
- Liu, J., Ren, X. L., Li, J. J. POC-LUS guiding pleural puncture drainage to treat neonatal pulmonary atelectasis caused by congenital massive effusion. The Journal of Maternal-Fetal & Neonatal Medicine. 33 (1), 174-176 (2020).
- Lichtenstein, D. A. BLUE-protocol and FALLS-protocol: Two applications of lung ultrasound in the critically ill. Chest. 147 (6), 1659-1670 (2015).
- Osman, A., Ahmad, A. H., Shamsudin, N. S., Baherin, M. F., Fong, C. P. A novel in-plane technique ultrasound-guided pericardiocentesis via subcostal approach. The Ultrasound Journal. 14 (1), 20 (2022).
- Gottlieb, M., Holladay, D., Peksa, G. D. Ultrasonography for the confirmation of endotracheal tube intubation: A systematic review and meta-analysis. Annals of Emergency Medicine. 72 (6), 627-636 (2018).
- Chowdhry, R., Dangman, B., Pinheiro, J. M. B. The concordance of ultrasound technique versus X-ray to confirm endotracheal tube position in neonates. Journal of Perinatology. 35 (7), 481-484 (2015).
- Hou, A., Fu, J. Pericardial effusion/cardiac tamponade induced by peripherally inserted central catheters in very low birth weight infants: A case report and literature review. Frontiers in Pediatrics. 8, 235 (2020).
- Nowlen, T. T., Rosenthal, G. L., Johnson, G. L., Tom, D. J., Vargo, T. A. Pericardial effusion and tamponade in infants with central catheters. Pediatrics. 110, 137-142 (2002).
- Kayashima, K. Factors affecting survival in pediatric cardiac tamponade caused by central venous catheters. Journal of Anesthesia. 29 (6), 944-952 (2015).
- Pérez-Casares, A., Cesar, S., Brunet-Garcia, L., Sanchez-de-Toledo, J. Echocardiographic evaluation of pericardial effusion and cardiac tamponade. Frontiers in Pediatrics. 5, 79 (2017).
- Musolino, A. M., et al. Ten years of pediatric lung ultrasound: A narrative review. Frontiers in Physiology. 12, 721951 (2022).
- Singh, Y., et al. International evidence-based guidelines on point of care ultrasound (POCUS) for critically ill neonates and children issued by the POCUS Working Group of the European Society of Paediatric and Neonatal Intensive Care (ESPNIC). Critical Care. 24 (1), 65 (2020).
- Makoni, M., Chatmethakul, T., Giesinger, R., McNamara, P. J. Hemodynamic precision in the neonatal intensive care unit using targeted neonatal echocardiography. Journal of Visualized Experiments. (191), e64257 (2023).
- Yousef, N., Singh, Y., de Luca, D. Playing it SAFE in the NICU SAFE-R: A targeted diagnostic ultrasound protocol for the suddenly decompensating infant in the NICU. European Journal of Pediatrics. 181 (1), 393-398 (2022).

