A Modified Technique for Inducing Polycystic Ovary Syndrome in Mice
Summary
This protocol introduces a technique for inducing polycystic ovary syndrome (PCOS) models in mice through controlled letrozole release using mini-pumps. Under adequate anesthesia, the mini-pump was implanted subcutaneously, and PCOS was successfully induced in the mice after a certain period of the mini-pump release.
Abstract
Polycystic ovary syndrome (PCOS) is one of the leading causes of infertility in women. Animal models are widely used to study the etiologic mechanisms of PCOS and for related drug development. Letrozole-induced mouse models replicate the metabolic and reproductive phenotypes of patients with PCOS. The traditional method of letrozole treatment in PCOS mice requires daily dosing over a certain period, which can be labor-intensive and cause significant stress to the mice. This study describes a simple and effective method for inducing PCOS in mice by implanting a controlled letrozole-releasing mini-pump. A mini-pump capable of stable, continuous release of a quantitative amount of letrozole was fabricated and implanted subcutaneously in mice under anesthesia. This study demonstrated that the mouse model successfully mimicked PCOS features after letrozole mini-pump implantation. The materials and equipment used in this study are readily available to most laboratories, requiring no special customization. Collectively, this article provides a unique, easy-to-perform method for inducing PCOS in mice.
Introduction
Polycystic ovary syndrome (PCOS) is one of the most common conditions among women of reproductive age1. It affects up to 18% of women globally and is the leading cause of female infertility worldwide2,3. PCOS is characterized by a series of interrelated reproductive abnormalities, including disturbed gonadotropin secretion, chronic anovulation, increased androgen production, and polycystic ovarian morphology4. In addition to gynecological disorders, PCOS also increases the risk of cardiovascular diseases5,6. Despite decades of research, the etiology of PCOS remains unclear7,8.
To gain better insights into the pathogenesis of PCOS and develop novel therapies, the creation of animal models that closely mimic human physiology is of tremendous importance9. Currently reported rodent models of PCOS include those induced by treatments with testosterone, letrozole, and estradiol valerate, among others. Testosterone induces hyperandrogenemia and is more commonly used as a PCOS inducer in rats10. DHEA has been used to induce PCOS in rodents, increasing testosterone levels, LH/FSH (luteinizing hormone/follicle-stimulating hormone) ratios, and causing irregular estrous cycles11. Estradiol valerate (EV) is a long-acting estrogen, and studies using this method show that levels of sex steroid hormones and gonadotropins vary depending on the dose of EV administered12,13,14. Letrozole is a nonsteroidal aromatase inhibitor15. Letrozole treatment induces non-cyclic estrus, increases ovarian weight, body weight, enlarges adipocytes, maximizes follicle development, and elevates testosterone levels in rats16,17. Letrozole-treated mice exhibit increased numbers of sinus follicles and hemorrhagic cysts, as well as elevated concentrations of LH, FSH, estradiol, and progesterone18,19.
Currently, the main methods for letrozole-induced PCOS modeling involve oral administration and subcutaneous injection, both of which require repeated daily dosing20,21,22. These methods are time-consuming and labor-intensive, and the repetitive administration likely causes significant stress to the animals23. Although some studies use letrozole pellets24,25, these products need to be customized and are expensive. This report describes a technique for inducing PCOS in mice using mini-pumps. This method is simple, time-saving, and uses surgical tools and equipment that are readily available in most laboratories.
Protocol
All animal experimental protocols in this study were approved by Animal Ethics Committee of Fudan University. Female C57BL/6J mice, aged 4 weeks, were used here. The details of the animals, reagents, and equipment used in this study are listed in the Table of Materials.
1. Preparation of the mini-pump
- Take the letrozole powder (50 mg) stored in the original packaging bottle, centrifuge the letrozole powder in the bottle at 300 x g for 5 s (at room temperature), then open the cap.
- Slowly add 625 µL of DMSO to the letrozole powder with a pipette and mix well to obtain a stock solution at a concentration of 8 mg/100 µL, totaling 625 µL.
NOTE: If using a different package size of letrozole powder, please calculate the required volume of DMSO in advance to configure the stock solution. - Count the number of required mice according to the experimental design.
- Draw up the total amount of letrozole stock solution required with a pipette, depending on the number of required mice, and inject it into the centrifuge tube slowly.
NOTE: 4-week-old mice for an 8-week exposure were selected to induce the PCOS mice model. The total dose of letrozole is 8 mg for each mouse, which requires two consecutive pumps to be implanted (one pump releases for 4 weeks), i.e., 4 mg/4 weeks per pump. As mentioned in step 1.2, the concentration of the letrozole stock solution is 8 mg/100 µL. To prepare the pumps, 50 µL of stock solution is required for each pump as calculated below: a total of 4 mg letrozole divided by 8 mg/100 µL. - Draw up an equal amount of PEG300, add it to the letrozole solution, and mix well (it can be vortexed appropriately).
- Prepare the control vehicle solution for the control group (50 µL of DMSO + 50 µL of PEG300 for each pump without letrozole).
- Prepare an adequate number of sterile centrifuge tubes according to the number of pumps required; one 15 mL centrifuge tube needs to be prepared for each pump.
- Draw saline with a syringe and add 5 mL of saline to each centrifuge tube. Then, cap the centrifuge tubes and place them on a tube rack for further use.
- Wear a new pair of sterile gloves, take out the mini-pumps in the desired quantity, and place them on a clean, sterile countertop or container.
- Gently pinch the cap that matches the pump body with the thumb and forefinger, and hold the pump body with the other hand.
- Slowly poke the filling port of the pump several times with the thin tube attached to the cap (Figure 1B).
NOTE: If not unclogged, fluid may not flow freely in the tube after filling. - Take the needle that matches the pump and a 1 mL sterile syringe. Attach the needle to the syringe and make it tight (Figure 1C).
- Carefully and slowly draw up the final solution of letrozole configured above with the newly assembled syringe (Figure 1D).
NOTE: Aspiration of the letrozole solution needs to be done slowly and evenly. Rapid aspiration may generate air bubbles, which may affect the subsequent drug solution injection. - Inject the letrozole solution into the mini-pump.
- Carefully pinch a mini-pump with one hand so that it stands upright, and with the other hand, hold the aforementioned 1 mL syringe and slowly insert it into the very bottom of the pump (Figure 1E).
NOTE: If the needle is not inserted into the bottom of the pump, the liquid in the pump can easily bubble during injection, which will affect the pump's subsequent release of the drug. - Then slowly inject the configured solution; the injection can be noticed when the opening of the pump slightly bubbles.
- Inject more slowly when the injection volume approaches 90 µL. Once the pump is fully filled (about 100 µL), withdraw the syringe.
- Carefully insert the cap into the pump body and make it tight; at this point, a small amount of solution will spill out. Carefully wipe it away.
- Carefully pinch a mini-pump with one hand so that it stands upright, and with the other hand, hold the aforementioned 1 mL syringe and slowly insert it into the very bottom of the pump (Figure 1E).
- Take the pump loaded with the drug, with the outlet facing upwards (the end with the cap), take the previously prepared centrifuge tube, and fully immerse the pump in sterile saline (Figure 1F). Then, cap the centrifuge tube and place it on the tube rack.
NOTE: Take care to avoid contaminating the pump throughout the process. - After all the mini-pumps are made, place all the centrifuge tubes in a 37 °C thermostat for 48 h.
2. Preparation for operation and anesthesia
- Prepare and sterilize all required instruments on the day of surgery, including forceps, hemostats, surgical scissors, needle holders, and 4-0 absorbable surgical sutures.
- Before using the isoflurane anesthesia machine (Figure 2B), carefully check whether the pipeline connection is correct and ensure the pipeline is intact and does not leak.
- Unscrew the filling sealing cap at the anesthesia machine's evaporator, then slowly add the anesthetic.
- Keep the anesthetic level between the upper and lower limits labeled on the anesthesia machine.
- Convert the three-way valve switch to ensure that the airflow from the anesthesia machine evaporator is connected to the anesthesia induction box.
- Open the air pump and the evaporator, then adjust the concentration of isoflurane for anesthesia induction (generally 3%-4%).
- Wait for about 1 min until the anesthetic fills the induction box, then put the mouse into the induction box and wait until complete anesthesia (this process takes about 2-3 min).
- Check that the mice are adequately anesthetized by gently shaking the box. If the mice are turned over and show no signs of returning to the recumbent position, anesthesia is induced.
3. Implantation of the letrozole mini-pump
- Put on a surgical mask and sterile gloves.
- Remove the pre-anesthetized mice from the induction box and apply the depilatory cream to the back of the mice, then wait for 1 min. Gently wipe off the depilatory cream and hair with wet gauze.
- At the same time, change the three-way valve switch to ensure that the airflow from the anesthesia machine evaporator is connected to the anesthesia mask and adjust the maintenance concentration (generally 2%).
- After dehairing, hold the mouse with the dominant hand and place its head/nose into the anesthesia mask.
- Check the depth of anesthesia by testing the pedal withdrawal reflex.
NOTE: All procedures involving anesthesia must be performed using gas filters fitted with activated charcoal tablets, and the procedure should be performed in a fume hood. - Secure the mouse in the prone position on the operating plate.
- Apply povidone-iodine swabs three times to the exposed skin (Figure 2C).
- At the midline of the back of the neck, favoring the skin on the hairless side, pick up the skin with forceps and make an incision of approximately 0.5 cm laterally with a scalpel or surgical scissors (Figure 2D).
- Then open the epidermis underneath the incision and access it with a hemostat or needle holder to free the skin, making room for a mini-pump (roughly 2 cm away from the incision) (Figure 2E).
- Take the pump out of the saline in the centrifuge tube with sterile forceps, with the open end of the pump facing downward, and be ready to insert the pump subcutaneously in the mice (Figure 2F).
- Clamp up one side of the skin incision with forceps and push the pump into the subcutaneous space. There should be no resistance when entering the pump. Once inside, use the fingers to slightly and gently squeeze the pump inward as far as possible (Figure 1G).
- Suture the incision site carefully using 4-0 surgical sutures (Figure 2H).
NOTE: Take care not to damage the subcutaneous pump body when suturing. - After suturing, disinfect the skin at the surgical site twice with iodophor swabs.
4. Animal recovery
- After surgery, gently transfer the mice from the mattress used during anesthesia to a dry, clean mattress. Ensure that the mice are in a warm and quiet environment.
- Closely monitor the conditions of the mice, such as the depth and frequency of their breaths.
- Observe the behavior of the mice as their mobility begins to return. Finally, ensure that they can stand and walk without any obvious abnormal behaviors such as dizziness or uncoordinated movements.
- After the mice have recovered, gently transfer them to pre-prepared cages. Place sterile jelly and food at the bottom of the cage for the mice to feed on.
NOTE: The stretching of the surgical site may prevent the mice from easily drinking and eating during the early postoperative period, so place jelly and food on the bottom of the cage. - After 8 weeks of letrozole treatment (i.e., two consecutive mini-pump implantations), test the mice models.
Representative Results
The experimental protocol and some critical steps are shown in Figure 1 and Figure 2. Serum testosterone levels are displayed in Figure 3A. Letrozole mini-pump-treated mice (hereafter referred to as LTZ mice) exhibited significantly elevated serum testosterone levels compared to female control mice. Meanwhile, histological analysis of ovaries showed that LTZ mice showed polycystic ovaries with a significant reduction in corpora lutea number (Figure 3B,C), indicating anovulation. Moreover, LTZ mice exhibited significant estrous acyclicity compared to female control mice (Figure 3D). The evaluation of body weight revealed that LTZ mice were heavier than control mice (Figure 3E). Glucose tolerance test results and AUC calculations demonstrated that LTZ mice developed glucose intolerance compared to control mice (Figure 3F,G). Serum lipid assay indicated that LTZ mice exhibited a change in serum lipid profile compared to control mice (Figure 3H).
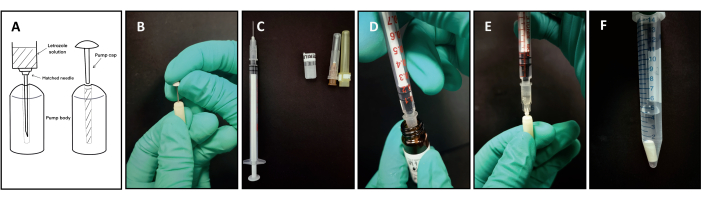
Figure 1: Materials and critical steps for preparing the mini-pump. (A) Schematic illustration showing the preparation of letrozole mini-pumps. (B) The mini-pump was unblocked by a thin tube attached to the cap. (C) A needle matching the pump was attached to the needle end of a 1 mL syringe. (D) Letrozole solution was slowly aspirated without bubbling. (E) Letrozole solution was injected slowly from the very bottom of the pump. (F) The filled pumps were capped and stored in saline with the cap end pointing upwards. Please click here to view a larger version of this figure.

Figure 2: Materials and critical steps of implanting the pumps in the mice for PCOS induction. (A) Schematic illustration showing implantation of letrozole mini-pumps. (B) Anesthesia equipment: air pump, anesthesia machine, induction box, and gas filters. (C) Anesthetized mice were fixed and sterilized. (D) A 0.5 cm cut near the dorsal midline. (E) The skin was freed to make space for mini-pump insertion. (F) A mini-pump was clipped out of the saline with clean and sterile forceps. (G) The mini-pump was implanted under the previously freed skin. (H) The skin was closed with 4-0 sutures. (I) The implanted mouse was allowed to recover. Please click here to view a larger version of this figure.
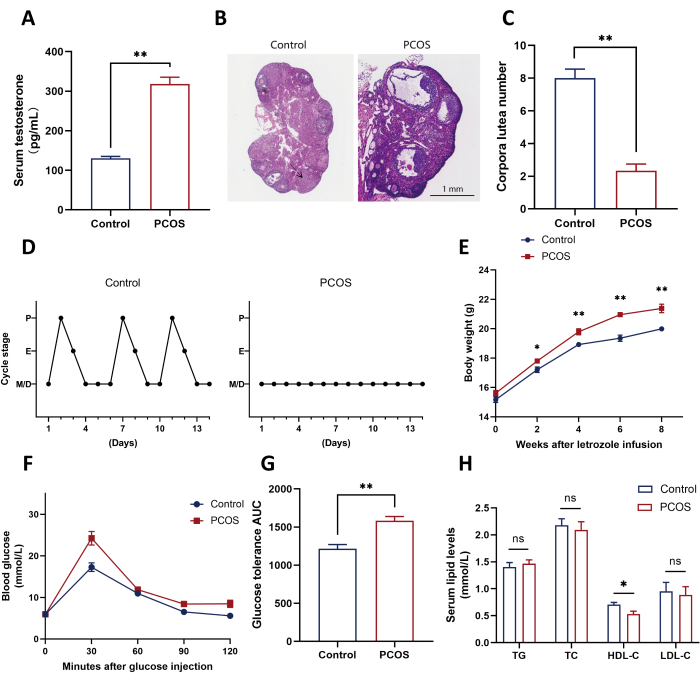
Figure 3: Characterization of reproductive and metabolic phenotypes in the letrozole mini-pump implanted mouse models. (A) Plasma testosterone levels in the LTZ-treated mice were significantly higher than those in the control mice (n = 9 animals per group). (B) Histological analysis of the ovaries.The arrow indicates corpora lutea. Scale bar: 1 mm. (C) Number of corpora lutea in the indicated groups (n = 9 animals per group). (D) LTZ mice showed acyclicity compared with control female mice. (E) The body weight in the indicated groups (n = 10 animals per group). (F,G) Glucose tolerance test results in indicated groups (n = 8 animals per group). (H) Serum lipid levels in indicated groups (n = 8 animals per group). Data are expressed as mean ± SEM. AUC indicates area under the curve; E, estrus stage; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; P, proestrus stage; M/D, metestrus/diestrus stage; TC, total cholesterol; and TG, triglycerides. Please click here to view a larger version of this figure.
Discussion
This report demonstrates a simple protocol for inducing PCOS in mice using easily accessible materials. The mouse PCOS model is essential for exploring PCOS mechanisms and screening drugs26. Of the available methods for inducing PCOS models in mice, letrozole induction is one of the most commonly used. The use of letrozole can develop and maintain a hyperandrogenic condition by inhibiting the conversion of testosterone to estradiol or by increasing testosterone synthesis27. The letrozole-induced mouse model shows metabolic and reproductive similarities to patients with PCOS28. However, conventional techniques of letrozole treatment in the mouse PCOS model require daily oral feeding or injection of letrozole drug into mice for 21 days or more29,30. Long-term continuous drug allocation with timed dosing of feeding or injection is time-consuming and labor-intensive, and repeated administration of drugs can be notoriously stressful for mice, introducing non-experimental factors affecting the model results23,31. Although some rat and mouse PCOS studies use implantable letrozole pellets28,32, the products used need to be customized and may not be available in a timely manner. The current study proposes a technique of implanting letrozole mini-pumps to induce PCOS in mice. The method is simple, time-saving, and uses surgical tools and equipment readily available in most laboratories.
Critical steps and troubleshooting
It is essential to note a few key steps to achieve optimal results when practicing this method. Firstly, unclog the pump using the thin tube attached to the matching cap before use. Previous experience indicates that a clogged pump can disrupt drug loading and release. Additionally, each step in configuring the letrozole solution must be executed with care to ensure the final solution is precisely at the desired concentration. Throughout the process of assembling the pump, attention must be paid to prevent contamination. Furthermore, careful adjustments are necessary before and during anesthesia to ensure adequate anesthesia for the experimental mice and to minimize the adverse effects of anesthesia on the operated mice.
Advantages and limitations
The method described in this study offers several advantages: (1) it saves time and allows for continuous letrozole release post-pump implantation; (2) it avoids the stress of repeated drug administration on experimental animals and minimizes the influence of non-experimental factors on modeling results; (3) the necessary materials, surgical tools, and equipment are commonly available in most laboratories; (4) the method effectively induces PCOS phenotypes in mice. However, this method also has limitations: (1) the anesthesia and surgical procedure required for mini-pump implantation may cause irritation to the mice, and the presence of the mini-pump under the skin could disturb their activity; (2) the efficacy of this modeling method may be impacted by contamination or damage to the pump body during the process. Based on the authors' experience, meticulous execution of the protocol steps can lead to a high success rate in inducing the PCOS mouse model.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The study was supported by National Key Research and Development Program of China (grants 2021YFC2700701), the National Natural Science Foundation of China (grants 82088102, 82071731, 82171613, 8227034, 81601238), Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (grant 2019-I2M-5-064), the Science and Technology Commission of Shanghai Municipality (grants 21Y11907600), Shanghai Municipal Commission of Health and Family Planning (grant 20215Y0216), Collaborative Innovation Program of Shanghai Municipal Health Commission (grant 2020CXJQ01), Clinical Research Plan of Shanghai Hospital Development Center (grant SHDC2020CR1008A), Shanghai Clinical Research Center for Gynecological Diseases (grant 22MC1940200), Shanghai Urogenital System Diseases Research Center (grant 2022ZZ01012), Shanghai Frontiers Science Research Base of Reproduction and Development, The Science and Technology Commission of Quzhou Municipality (grant 2022K54), Open Fund Project of Key Laboratory of Reproductive Genetics, Ministry of Education, Zhejiang University (grant KY2022035), and Open Fund Project of Guangdong Academy of Medical Sciences (grant YKY-KF202202).
Materials
| C57BL/6J Mice | Shanghai Model Organisms Center | N/A | Age: 4 weeks |
| Centrifugation tube | Biological Hope | 1850-K | 15ML |
| Depilatory cream | ZIKER BIOTECHNOLOGY | ZK-L2701 | Depilation agent for laboratory animals |
| Dimethyl sulfoxide | Biosharp | BS087 | Used ofr dissolution |
| Forceps | RWD | F12028 | Surgical instrument |
| Hemostats | Biosharp | BS-HF-S-125 | Surgical instrument |
| Isoflurane | RWD | 20071302 | Used for anesthesia |
| Letrozole powder | Sigma | L6545-50MG | Primary acting drugs |
| Needle and the 4-0 absorbable suture | JINGHUAN | CR413 | Surgical instrument |
| Needle holder | ShangHaiJZ | J32010 | Surgical instrument |
| Nitrile Gloves | Biosharp | BC040-L | Used for aseptic operation |
| Osmotic Pumps | ALZET | 1004 | Letrozole storage and sustained release |
| PEG(Poly(ethylene glycol)) | Solarbio | P8250 | Used ofr dissolution |
| Physiological Saline Solution | Biosharp | BL158A | Mini-pump storage |
| Pipette | Eppendorf | 3123000268-A | 100 μL-1000 μL |
| Pipette | TopPette | 7010101008 | 10 μL-100 μL |
| Povidone-iodine swabs | SingleLady | GB26368-2010 | Skin disinfection |
| Scissors | Biosharp | BS-SOR-S-100P | Surgical instrument |
| Small Animal Anesthesia Machine | RWD | R500IP | Used for anesthesia |
| Sterile gauze | ZHENDE | BA69087 | Used for wiping liquids |
| Syringe | Bofeng Biotech | BD300841 | 1 mL |
References
- Dapas, M., Dunaif, A. Deconstructing a syndrome: Genomic insights into PCOS causal mechanisms and classification. Endocr Rev. 43 (6), 927-965 (2022).
- Teede, H., et al. Anti-müllerian hormone in PCOS: A review informing international guidelines. Trends Endocrinol Metab. 30 (7), 467-478 (2019).
- Joham, A. E., et al. Polycystic ovary syndrome. Lancet Diabetes Endocrinol. 10 (9), 668-680 (2022).
- Pan, J. X., et al. Aberrant expression and DNA methylation of lipid metabolism genes in PCOS: A new insight into its pathogenesis. Clin Epigenetics. 10, 6 (2018).
- Gao, L., et al. Calcitriol attenuates cardiac remodeling and dysfunction in a murine model of polycystic ovary syndrome. Endocrine. 52 (2), 363-373 (2016).
- Gao, L., et al. Polycystic ovary syndrome fuels cardiovascular inflammation and aggravates ischemic cardiac injury. Circulation. 148 (24), 1958-1973 (2023).
- Rosenfield, R. L., Ehrmann, D. A. The pathogenesis of polycystic ovary syndrome (PCOS): The hypothesis of PCOS as functional ovarian hyperandrogenism revisited. Endocr Rev. 37 (5), 467-520 (2016).
- Wang, F., et al. Alternative splicing of the androgen receptor in polycystic ovary syndrome. Proc Natl Acad Sci U S A. 112 (15), 4743-4748 (2015).
- Noroozzadeh, M., Behboudi-Gandevani, S., Zadeh-Vakili, A., Ramezani Tehrani, F. Hormone-induced rat model of polycystic ovary syndrome: A systematic review. Life Sci. 191, 259-272 (2017).
- Tyndall, V., et al. Effect of androgen treatment during foetal and/or neonatal life on ovarian function in prepubertal and adult rats. Reproduction. 143 (1), 21-33 (2012).
- Jang, M., et al. Oriental medicine Kyung-ok-ko prevents and alleviates dehydroepiandrosterone-induced polycystic ovarian syndrome in rats. PLoS One. 9 (2), e87623 (2014).
- Brawer, J. R., Munoz, M., Farookhi, R. Development of the polycystic ovarian condition (PCO) in the estradiol valerate-treated rat. Biol Reprod. 35 (3), 647-655 (1986).
- Mirabolghasemi, G., Kamyab, Z. Changes of the uterine tissue in rats with polycystic ovary syndrome induced by estradiol valerate. Int J Fertil Steril. 11 (1), 47-55 (2017).
- Li, X., et al. Amitriptyline plays important roles in modifying the ovarian morphology and improving its functions in rats with estradiol valerate-induced polycystic ovary. Arch Pharm Res. 42 (4), 344-358 (2019).
- Mamounas, E. P., et al. Use of letrozole after aromatase inhibitor-based therapy in postmenopausal breast cancer (NRG oncology/NSABP B-42): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 20 (1), 88-99 (2019).
- Deveci, H. S., et al. Histological evaluation of rat larynx in experimental polycystic ovary syndrome model. Eur Arch Otorhinolaryngol. 269 (8), 1945-1950 (2012).
- Du, D. F., Li, X. L., Fang, F., Du, M. R. Expression of anti-müllerian hormone in letrozole rat model of polycystic ovary syndrome. Gynecol Endocrinol. 30 (12), 885-889 (2014).
- Kelley, S. T., Skarra, D. V., Rivera, A. J., Thackray, V. G. The gut microbiome is altered in a letrozole-induced mouse model of polycystic ovary syndrome. PLoS One. 11 (1), e0146509 (2016).
- Arroyo, P., Ho, B. S., Sau, L., Kelley, S. T., Thackray, V. G. Letrozole treatment of pubertal female mice results in activational effects on reproduction, metabolism and the gut microbiome. PLoS One. 14 (9), e0223274 (2019).
- Pyun, B. J., et al. Tetragonia tetragonioides (pall.) Kuntze regulates androgen production in a letrozole-induced polycystic ovary syndrome model. Molecules. 23 (5), 1173 (2018).
- Patel, R., Shah, G. Insulin sensitizers modulate GNRH receptor expression in PCOS rats. Arch Med Res. 49 (3), 154-163 (2018).
- Chaudhari, N., Dawalbhakta, M., Nampoothiri, L. GNRH dysregulation in polycystic ovarian syndrome (PCOS) is a manifestation of an altered neurotransmitter profile. Reprod Biol Endocrinol. 16 (1), 37 (2018).
- Turner, P. V., Brabb, T., Pekow, C., Vasbinder, M. A. Administration of substances to laboratory animals: Routes of administration and factors to consider. J Am Assoc Lab Anim Sci. 50 (5), 600-613 (2011).
- Lee, Y. H., et al. Welsh onion root (Allium fistulosum) restores ovarian functions from letrozole induced-polycystic ovary syndrome. Nutrients. 10 (10), 1430 (2018).
- Yang, H., et al. Traditional medicine (mahuang-tang) improves ovarian dysfunction and the regulation of steroidogenic genes in letrozole-induced PCOS rats. J Ethnopharmacol. 248, 112300 (2020).
- Corrie, L., et al. Recent updates on animal models for understanding the etiopathogenesis of polycystic ovarian syndrome. Life Sci. 280, 119753 (2021).
- Corbin, C. J., Trant, J. M., Walters, K. W., Conley, A. J. Changes in testosterone metabolism associated with the evolution of placental and gonadal isozymes of porcine aromatase cytochrome p450. Endocrinology. 140 (11), 5202-5210 (1999).
- Ryan, G. E., Malik, S., Mellon, P. L. Antiandrogen treatment ameliorates reproductive and metabolic phenotypes in the letrozole-induced mouse model of PCOS. Endocrinology. 159 (4), 1734-1747 (2018).
- Bhattarai, P., Rijal, S., Bhattarai, J. P., Cho, D. H., Han, S. K. Suppression of neurotransmission on gonadotropin-releasing hormone neurons in letrozole-induced polycystic ovary syndrome: A mouse model. Front Endocrinol (Lausanne). 13, 1059255 (2022).
- Annie, L., Gurusubramanian, G., Roy, V. K. Inhibition of visfatin by fk866 mitigates pathogenesis of cystic ovary in letrozole-induced hyperandrogenised mice. Life Sci. 276, 119409 (2021).
- Mcdonald, L. T., et al. Early blood profile of C57BL/6 mice exposed to chronic unpredictable stress. Front Psychiatry. 10, 230 (2019).
- Torres, P. J., et al. Exposure to a healthy gut microbiome protects against reproductive and metabolic dysregulation in a PCOS mouse model. Endocrinology. 160 (5), 1193-1204 (2019).

.
