Alliages nanocristallins et stabilité de la taille des nano-grains
English
Share
Overview
Source: Sina Shahbazmohamadi et Peiman Shahbeigi-Roodposhti-Roodposhti, School of Engineering, University of Connecticut, Storrs, CT
Les alliages de moins de 100 nm sont connus sous le nom d’alliages de nanocristalline. En raison de leurs propriétés physiques et mécaniques améliorées, il y a une demande toujours croissante de les employer dans diverses industries telles que les semi-conducteurs, les biocapteurs et l’aérospatiale.
Pour améliorer le traitement et l’application des alliages de nanocristalline, il est nécessaire de développer près de 100% de matériaux en vrac denses qui nécessite un effet synergique de température et de pression élevées. En augmentant la température et la pression appliquées, les petits grains commencent à croître et à perdre leurs propriétés distinguées. Ainsi, il est technologiquement important de parvenir à un compromis entre la liaison interpartiique avec une porosité minimale et la perte de la taille du grain à l’échelle nanométrique lors de la consolidation à des températures élevées.
Dans cette étude, nous visons à éliminer l’oxygène de la solution solide pour améliorer la stabilité de la taille des nanograins à des températures élevées. L’alliage Nano-cristallin Fe-14Cr-4Hf sera synthétisé dans un environnement protégé afin d’éviter la formation de particules d’oxyde.
Principles
Procedure
Results
Fig. 3 shows the XRD data for ball-milled OF-Fe14Cr4Hf annealed for one hour at 900°C. There is sharpening of the peaks along with slight peak shifts. It is due to relaxation of lattice strain as the annealing temperature rises. When the annealing temperature rises, several small peaks are revealed between the four major BCC peaks. These would indicate the formation of secondary phases.
Fig. 4a-c shows TEM images and diffraction pattern for OF-Fe14Cr4Hf annealed for 1 hour at 900°C. Nanoscale particles in a size range up to about 20nm are present.
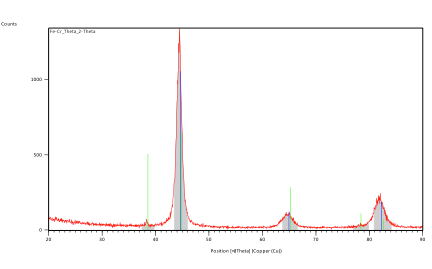
Figure 3: XRD patterns for OF-Fe14Cr4Hf annealed for one hour at 900°C.

Figure 4: TEM images and diffraction pattern for OF-Fe14Cr4Hf annealed at 900°C for 60 min.
Applications and Summary
The experiment demonstrates how the nano-grain size stability of the nominally oxygen free nanocrystalline materials may improves compare to the alloys with significant amount of oxygen. In this study the OF powders synthesized in a protected atmosphere to minimize the interaction between oxygen and solid solution leads to increase the segregation of alloying elements to the grain boundaries and improve the thermodynamic grain size stability. TEM microscope introduced itself as a cost-effective, time-saving and powerful tools to characterize the grain boundaries and nanoparticles.
Fatigue strength and creep resistance are the key properties required for aircraft components that may have a direct influence on aircraft life time. To increase the life of aircraft it is critically important to employ materials with elevated fatigue/creep strength/resistance, achievable mainly due to a reduction in the grain sizes. High-temperature stable nanomaterials, with grain size in the order of less than 10^-7 m, may provide fatigue life three times more than conventional materials. Furthermore, this new generation of nanocrystalline materials is stronger and able to operate at relatively high temperatures leading to a significant increase in aircrafts speed and fuel efficiency.
The high temperature stable nanocrystalline materials are perfect candidates for space crafts as well. Various parts of the space crafts (e.g. rocket engines, thrusters and vectoring nozzles) are working at higher temperatures compared to aircrafts.
Satellites, with dual applications of civilian and defense, are also a reasonable target for high temperature stable nanomaterials. Thruster rockets using in the satellite to change their orbits, need nanomaterials that could tolerate elevated temperatures. On board ignitors, developed from conventional materials, may wear out quickly and lose their efficiency, whereas proposed nanomaterials last longer.
Transcript
Nanocrystalline alloys are in high demand in industries such as semiconductor, biosensors and aerospace due to their enhanced physical and mechanical properties. Alloys with grain size less than 100 nanometers are known as nanocrystalline alloys.
To produce produce industrial parts with these alloys, nanocrystalline powders are processed using elevated temperature and pressure combined to develop close to 100 percent dense bulk materials. Nanograins however start to grow at high temperatures causing the material to loose it’s enhanced properties. To combat this issue high density interparticle bonding with minimum porosity must be obtained at high temperature while minimizing the loss of nanoscale grain size.
This video reveals a new approach to improve the nanograin size stability of Fe14Cr4Hf alloy at elevated temperatures.
Nano materials tend to be unstable causing grain size to increase at elevated temperatures. This results in the material losing it’s superior mechanical properties. The instability of nano materials is the result of two factors that cause the material to go far beyond an equilibrium condition. Both grain size and mechanical processing lead to these altered thermodynamic properties. The smaller grains in nano materials have more grain boundary per volume than larger grains and thus a higher gibbs free energy.
Mechanical alloying techniques used to produce these materials also increase the energy available to drive grain growth. The thermodynamic instability caused by these factors drives the movement of grain boundaries especially at elevated temperatures causing grains to grow. To be useful nano materials must be developed that are stable at high temperature. One way of stabilizing grain size is to introduce alloying elements and eliminate oxygen from the solid solution. When oxygen is present, alloying elements form oxides within the grains preventing all of the alloying elements from reaching the grain boundaries. By eliminating oxygen, elements are free to segregate to grain boundaries stabilizing the size of the nanograins.
Studies have shown that if a nonequilibrium stabilizer solute such as hafnium is introduced to a nanocrystalline iron ten chromium alloy it segregates to the grain boundaries at elevated temperatures. This decreases the gibbs free energy of the grain boundaries resulting in a metastable equilibrium state and thus more stable nanocrystalline materials. It has been found that the elimination of oxygen further enhances this stabilization.
To compare nanograin size stability at different temperatures, samples are heat treated over a range of temperatures. Grain size is then analyzed using transmission electron microscopy images and x-ray diffraction. The Scherrer equation is used to calculate grain size based on x-ray diffraction results. Using this equation the size of nanograins is related to the broadening of a peak in the diffraction pattern.
Now that you understand the principles behind the stabilization of nanocrystalline materials, let’s see how this method is applied in the laboratory.
Use high purity low oxygen content bulk materials iron, chromium, and hafnium enclosed in a glove box to minimize oxygen contamination. Load 6.4 and 7.9 mm 440c stainless steel milling balls and powder in to a stainless steel vial creating a ball to powder weight ratio of ten to one. The sealed vial needs to be kept under protective atmosphere in the glove box.
Transfer the vial to the high energy specs ball milling machine. Carry out ball milling for 20 hours. Return the vial to the glove box and transfer the milled powder to a small glass vial. Seal the glass vial for annealing. Anneal the ball milled Fe14Cr4Hf for 60 minutes at temperatures between 500 and 1200 degrees celsius at steps of 100 degrees celsius. Run XRD analysis of multiple samples from each annealing temperature as well as samples of the milled material. Employ a five millimeter dye and punch with hydraulic press to press the powder for microscopic analysis.
Now that you appreciate the importance of nanocrystals maintaining their grain size at high temperature let’s take a look at some applications where they can be utilized. The life of aircraft can be increased utilizing nanocrystalline materials. Improved fatigue life, strength, and higher operating temperatures lead to a significant increase in aircraft speed and fuel efficiency.
These materials are also perfect candidates for spacecraft components that must work at higher temperatures. For example, onboard igniters on satellites developed from conventional materials may wear out quickly with no possibility of repair. Whereas nano materials will last longer prolonging the life of the mission.
You’ve just watched Jove’s introduction to nano crystal stability. You should now understand the need for maintaining grain size at elevated temperature, ways in which it is accomplished and how grain size is measured.
Thanks for watching.
