Author Spotlight: Techniques and Challenges in Islet Purification for Xenotransplantation
Summary
This protocol provides a step-by-step guide for the procurement of a porcine pancreas for islet isolation and purification.
Abstract
Pancreatic islet transplantation is an emerging treatment for type I diabetes; however, it is limited by donor matching and availability. Porcine islet xenotransplantation offers a promising alternative to allotransplantation, with the potential for large-scale production of on-demand, functional islets. The yield and viability of isolated islets is highly susceptible to the quality of the donor pancreas and the method of procurement, particularly the duration of warm-ischemia time. To improve organ preservation and subsequent islet yield and viability, we have developed a protocol for surgical perfusion and resection of the porcine pancreas. This protocol employs direct infrarenal aortic cannulation and organ perfusion to both minimize warm-ischemia time and simplify the procedure for operators who do not have extensive surgical expertise. Subsequent arterial perfusion of the pancreas via the aorta flushes stagnant blood from the microvasculature, thereby reducing thrombosis and oxidative damage to the tissue. This manuscript provides a detailed protocol for surgical perfusion and resection of the porcine pancreas, followed by islet isolation and purification.
Introduction
Type 1 diabetes is caused by the autoimmune destruction of pancreatic beta cells1. Consequently, patients are dependent on exogenous insulin, placing them at high-risk glycemic fluctuations with episodes of hyper- and hypoglycemia2. Islet allotransplantation is a potential cure for type I diabetes; however, the limited availability of pancreatic organ donors remains a major barrier to widespread adoption of the procedure2,3. Islet xenotransplantation from porcine donors is a promising alternative as these animals are readily available. Optimization and scaling of porcine pancreatic islet isolation will be critical for progressing islet xenotransplantation.
Although several porcine pancreas procurement techniques have been previously published, many of these procedures describe pancreatectomy after induction of cardiac death or exsanguination4,5,6,7,8,9,10. One major disadvantage of these techniques is the variable warm ischemic time (WIT) occurring between the time of cardiac death and the initiation of intraarterial and/or intraductal infusion of preservation solution. Even 10 min of WIT will negatively impact islet yield and viability5. Minimizing WIT requires prompt perfusion of the pancreas with a preservation solution. During abdominal organ procurement, aortic cannulation followed by intracorporeal perfusion with University of Wisconsin (UW) solution is crucial for removing blood, preventing intravascular thrombosis, protecting against ischemic damage, and minimizing cellular injury11,12.
Previous studies have demonstrated that during porcine pancreas procurement, low-pressure flushing of the celiac trunk and superior mesenteric artery with UW solution improves islet yield and purity4. However, previously published methods for porcine pancreas procurement include open aortic cannulation, which can be technically challenging, particularly for smaller pigs13,14,15. In this manuscript, we present a detailed, step-by-step protocol with accompanying visual aids for the surgical perfusion and procurement of the porcine pancreas, followed by islet isolation and purification. This new technique of aortic cannulation during pancreatic isolation was developed specifically to minimize WIT and allow the pancreatic procurement to be performed in a bloodless field as a means of maximizing islet isolation yield and viability.
Protocol
All procedures involving animals are approved by the Washington University School of Medicine Division of Comparative Medicine. Adult Yorkshire pigs ranging from 35 to 35 kg are ideal for this procedure; however, the protocol can be adapted for pigs of different sizes depending on the experimental context. The entire procedure should be performed in a sterile fashion in an operating room setup.
1. Preoperative preparation and anesthesia
- Sedate the pig with a cocktail of 4 mg/kg tiletamine and zolazepam, 20 mg/kg ketamine, and 2 mg/kg xylazine injected intramuscularly.
- Establish peripheral venous access via the auricular vein.
- Induce general anesthesia with inhaled 1%-3% isoflurane in 100% oxygen and endotracheally intubate the pig.
- Shave and prep the pig with chlorhexidine gluconate from the sternal notch to the bilateral groins.
- Drape the operative field with a clear view of the entire abdomen (Figure 1A).
2. Operative setup and exposure of the retroperitoneum
- Perform a midline laparotomy incision (Figure 1B).
- Use a 10-blade scalpel and electrocautery to make a midline laparotomy from the xiphoid process to the pubis. Take care to curve the incision around the inferior abdominal urogenital opening.
- Perform a right visceral rotation.
- Externalize the right colon and small bowel, wrapping them in a sterile towel.
- Gently retract the colon and bowel to the left to expose the right retroperitoneum while avoiding bowel torsion or malrotation.
- Drain the bladder (optional).
- If the bladder is full and impacting exposure, grip the bladder between two DeBakey forceps and use an 11-blade scalpel to incise the ventral bladder wall.
- Place a Yankauer suction tip into the bladder to drain the urine and decompress the bladder.
- Set up an abdominal retractor set to facilitate optimal exposure of the right retroperitoneum (Figure 1C).
3. Retroperitoneal dissection
- Begin retroperitoneal dissection (Figure 2A).
- Using electrocautery, incise the peritoneum to the right of the midline, from the level of the right kidney to the aortic bifurcation.
- Sharply expose the infrarenal aorta, inferior vena cava (IVC), and right renal artery and vein
- Obtain circumferential control of the distal infrarenal aorta with a large vessel loop.
- Ligate the IVC (Figure 2B).
- Obtain circumferential control of the mid-infrarenal IVC.
- Ligate the IVC using a 2-0 silk tie.
- Ligate the renal vessels (Figure 2C).
- Obtain circumferential control of the right and left renal arteries and veins.
- Ligate these structures using 2-0 silk ties.
- If there are multiple renal arteries, similarly isolate and ligate each of these vessels.
4. Intrathoracic dissection
- Incise the right hemidiaphragm (Figure 3A).
- Gently retract the liver inferiorly to expose the right hemidiaphragm. Apply two Allis clamps to the membranous portion of the right hemidiaphragm and incise the diaphragm in between the two clamps using electrocautery.
- Extend the diaphragm myotomy to approximately 7 cm horizontally.
- Obtain circumferential control of the intrathoracic IVC (Figure 3B).
- Place a 2-0 silk tie around the intrathoracic IVC. Snap the ends of the suture with a small hemostat without occluding the IVC.
- Extend the diaphragmotomy to the left hemidiaphragm (Figure 3C).
- Use two Allis clamps to retract edge of the diaphragmotomy inferiorly and extend to the left hemidiaphragm.
NOTE: Avoid entering the pericardium during diaphragmotomy.
- Use two Allis clamps to retract edge of the diaphragmotomy inferiorly and extend to the left hemidiaphragm.
- Expose the descending thoracic aorta in the left posteromedial chest region.
5. Infrarenal aortic cannulation, visceral isolation, and perfusion
- Systemically anticoagulate the pig.
- Administer systemic heparin (100 U/kg) via peripheral venous access. Wait 3 min before proceeding with aortic cannulation.
- Access the infrarenal aorta using an 18 G micropuncture needle (Figure 4A).
- Pass a j-wire through the needle into the aorta and remove the needle over the wire.
- Place a 10Fr sheath over the wire into the aorta and remove the wire (Figure 4B).
- Connect IV tubing to the 10Fr sheath and in a sterile fashion, infuse UW solution through the sheath. Infuse UW solution at a rate of 40-60 drops/min.
NOTE: Flow will not begin until the thoracic aorta is crossclamped. Ensure the intravenous tubing is not on tension to avoid displacement of the intraaortic sheath. - Perform visceral isolation and perfusion in rapid succession.
- Clamp the infrarenal aorta using an aortic clamp.
- Clamp the descending thoracic aorta using an aortic clamp.
- Tie off the intrathoracic IVC with the previously placed 2-0 tie.
- Administer 100 mg/kg KCl for euthanasia via peripheral venous access.
NOTE: Cardiac death commences with the administration of KCl. - Make a venotomy in the infrarenal IVC, superior to the 2-0 tie, and insert pool-tipped suction through the venotomy (Figure 4C).
- Perfuse the viscera with 2-3 L of cold UW solution.
- Proceed with dissection for total pancreatectomy during the perfusion.
NOTE: The viscera, and pancreas in particular, should become pale as the UW solution is perfused into the solid organ region (Figure 5A).
6. Total pancreatectomy
- Release the small bowel from retraction as the UW solution is perfusing.
- Begin dissection of duodenal, connecting, and splenic lobes of the pancreas during the visceral perfusion. Retract the stomach superiorly to expose the pancreas. With a combination of blunt dissection and electrocautery, dissect the pancreas from its omental attachments.
NOTE: The body of the pancreas effectively wraps around the portal vein. Take care to avoid the portal vein running posteriorly to the duodenal lobe and then anteriorly to the bridge and connecting lobes. - Isolate the pancreatic duct (Figure 5B). Locate the pancreatic duct where it enters the duodenum. Ligate the duct using two 3-0 silk ties and transect the duct in between the ties.
- Complete pancreatectomy (Figure 5C).
- Dissect the remainder of the duodenal lobe from the C-loop of the duodenum until it is freely mobile.
- Separate the pancreas from its remaining attachments. Carefully ligate and divide the small vessels from the superior mesenteric artery to the pancreas.
- Separate the pancreas from the portal vein. Dissect the body of the pancreas and the connecting lobe from the portal vein. Ligate any venous branches from the portal vein to the pancreas, including the splenic vein.
- Transect the bridge of the pancreas between the connecting lobe and splenic lobe to allow free mobilization from the portal vein.
NOTE: Take care not to disrupt the pancreatic duodenal lobe from the splenic lobe.
- Transect the bridge of the pancreas between the connecting lobe and splenic lobe to allow free mobilization from the portal vein.
- Immerse the entire pancreas in cold sterile UW solution, placing the container immediately on ice. Transport the pancreas to a sterile hood for islet isolation. Proceed with pancreatic islet isolation (Figure 6).
7. Pancreatic duct cannulation
- Place the pancreas on a sterile tray on ice inside the biosafety cabinet.
- Dissect excess fibrous tissue and blood away from the pancreas.
- If necessary, cut the duct transversely to better visualize the lumen. Locate and advance the cannula into the duct with an appropriately sized catheter (16 G).
- Secure the catheter in place with a 2.0 silk tie but avoid occluding the lumen.
- Prepare three decontamination solutions and submerge the cannulated pancreas in each decontamination solution for 60 s.
8. Perfusion and distention of the pancreas
- Prepare the enzyme digestion solution (Figure 6).
- Mix 100 mg of commercially available collagenase 1 and 2 solutions (see Table of Materials for further details) with the desired amount of 1x HBSS and 10,000 U Heparin solution. If the isolated pancreatic tissue is between 100 g and 200 g, mix the enzyme with 200 mL total of 1x HBSS and Heparin solution.
- Infuse the pancreas with the enzyme solution.
- With controlled moderate force, infuse enzyme solution in 60 mL boluses into the pancreatic duct. Continue this process until the entirety of the enzyme solution is infused into the pancreas.
NOTE: Effective infusion should cause the pancreas tissue to distend. - If some enzyme solution leaks out of the pancreas tissue into the sterile tray, re-inject this remaining solution into the pancreas in step 8.3 or place it in the Ricordi chamber in step 9.2.
- With controlled moderate force, infuse enzyme solution in 60 mL boluses into the pancreatic duct. Continue this process until the entirety of the enzyme solution is infused into the pancreas.
- Directly inject any remaining enzyme solution into any poorly distended pancreatic parenchyma segments using a 22 G needle.
9. Digestion of the pancreas
- Place circuit reservoir containing 500 mL of 1x HBSS in a water bath set to 47 °C to preheat the reservoir solution.
NOTE: The enzyme solution functions optimally at 36-38 °C and this temperature needs to be reached within 5 min of starting the peristaltic pump. The temperature of the contents of the Ricordi, including the enzyme-infused pancreas, is initially 4 °C. By pre-heating the circuit reservoir to 47 °C, the contents of the reservoir and the Ricordi chamber will mix, bringing the entire circuit temperature up to 36-38 °C within 5 min of starting the peristaltic pump. - Remove and discard the catheter from the pancreatic duct and cut the pancreas into ~1-3 cm pieces.
- Prepare the digestion circuit. The peristaltic pump will circulate solution from the digestion circuit reservoir, through the heating coil, and into the Ricordi chamber (Figure 6).
- Place the pancreas pieces with nine 1.5 cm silicon nitride marbles inside the Ricordi chamber and fill to the filter level with the remaining enzyme solution.
- To seal the Ricordi chamber, place the mesh filter and rubber gasket, followed by the lid onto the Ricordi chamber. Secure the lid of the Ricordi chamber by tightening the screws and ensure that the chamber is well sealed before proceeding.
- Place the heating coil in a 47 °C hot water bath.
- Place the inflow and outflow cannulas into the digestion circuit reservoir containing 500 mL of 1x HBSS.
- Initiate the pump at a flow rate of 450 mL/min until the entire chamber is filled with the digestion solution. Decrease the flow rate to 100 mL/min as the fluid circulates within the circuit.
- Manually shake the digestion chamber to agitate the tissue with the silicon nitride marbles.
- Starting with 8 min of digestion, take a 1 mL sample from the circuit reservoir for Dithizone (DTZ) staining to monitor digestion progress.
- To prepare DTZ staining, dissolve 10 mg of DTZ in 2 mL of Dimethyl Sulfoxide (DMSO).
- Add 10 µL of the prepared DTZ stain to 1 mL of the digestion sample. Inspect the sample under 4x magnification. Islets will appear dark red to black with DTZ staining.
NOTE: Digestion is complete when most or all the observed islets are 'free' and intact, with the acinar tissue dispersed into smaller clusters.
- When digestion is judged complete, transfer the coil to the ice bath, remove the inflow cannula from the reservoir, and increase the pump speed to 210-250 mL/min. Let the digested tissue drain from the Ricordi chamber into the circuit reservoir.
NOTE: It is typical to see very little tissue remaining in the Ricordi chamber after digestion. Typically, <10% of undigested pancreatic tissue will remain, although this may vary due to specimen-specific factors. - Weigh the remaining tissue to determine the efficiency of the digestion. Transfer the digested tissue from the circuit reservoir into 250 mL conical tubes that are maintained cool on ice.
10. Collection of pancreatic tissue
- Centrifuge the digested pancreatic tissue in 250 mL conical tubes at 250 × g for 1 min at 4 °C without brake.
- Remove the tubes from the centrifuge with caution and aspirate the supernatant using a serological pipette while avoiding disruption of the pellet.
- Resuspend the pellets in each conical tube gently and combine them into one 250 mL conical tube. If there is ≤50 mL of tissue, resuspend the pellet to a total volume of 100 mL in Islet Collection Solution. If there is >50 mL of tissue, resuspend the pellet to a total volume of 200 mL in Islet Collection Solution. In the case that over 100 mL of tissue is collected, run two Cell Processor purification cycles.
- Resuspend the tissue in RPMI medium to make a total volume of 100 mL (assuming 50 mL of tissue was collected) and load in the Cell Processor.
11. Islet purification using the continuous gradient method
- Prime the Cell Processor per manufacturer's instructions.
- Load 125 mL of High Density gradient solution into the continuous gradient former, then load the High Density gradient solution into the Cell Processor using a pump set at 200 mL/min.
- Start the Cell Processor centrifuge function at 1,000 RPM.
- Release the air from the system and re-prime with High-density Gradient Solution.
- Add 125 mL of High-density gradient solution and 130 mL of Low-density Solution to the gradient former.
- Load the 100 mL of digested tissue suspension.
- Increase the Cell Processor speed to 2,000 RPM for 3 min.
- Prepare 16 50 mL conical tubes for purified islet fraction collection. Add 25 mL of RPMI medium to each tube and arrange all tubes in ascending order.
- Collect the purified islet fractions in the prepared collection conical tubes.
12. Islet assessment and staining
- Collect 100-200 µL aliquots from each of the purified islet fractions and place them in a 24-well tissue culture plate for DTZ staining to assess islet content and from each purification fraction. Inspect each fraction using light microscopy with 4x magnification. The islets will appear dark red to black with DTZ staining.
- Once the islets are identified, select the fractions with the highest content of islets. Centrifuge the purified fraction at 250 × g for 1 min at 4 °C without brake.
- Carefully aspirate the supernatant and resuspend the islet pellet in islet growth media. Transfer the purified islets to 1-4 T-125 culture flasks.
- Following purification, evaluate the islets for quantity, viability, and function.
- Use an islet cell counter to evaluate the islet number and size distribution (Figure 7A). An entire pancreas will typically yield 1-2 million islet equivalents.
- For viability staining, mix 2 µL of calcein AM and 2 µL of ethidium homodimer-1 (CaAM/EthD-1) with 1,000 µL of PBS (Figure 7B). Allow the islets to be stained for 30 min, then evaluate them with under 4x or 10x magnification. Viable islets will stain green while nonviable islets will stain red.
13. Cell culture of pancreatic Islets
- During the initial 48 h, culture the islets in recovery medium at 37 °C to provide sufficient nutrients (~1,000 islets/mL of recovery medium).
- From days 2-21, culture the islets in standard growth medium at 37 °C.
Representative Results
The operative setup and midline laparotomy is shown in Figure 1. The laparotomy incision should be curved to avoid the urogenital opening (Figure 1B). When setting up the retractor, attach the post for the Omni or Bookwalter retractor to the left, inferior corner of the table. Ideal retraction includes two retractor blades for the right abdominal wall, two for the left abdominal wall, and 1-2 for the right colon and small bowel wrapped in a sterile towel (Figure 1C). Body wall, splanchnic, or malleable retractor blades can be used, depending on the availability of an assistant and operator preference. If an Omni or Bookwalter retractor is not available, 1-2 self-retaining Balfour retractors can be used to retract the abdominal wall on either side. An assistant can then apply retraction to the right colon and small bowel wrapped in a sterile towel.
The retroperitoneal dissection carried out in Figure 2 can be performed with a mix of blunt and sharp dissection. While it is important to obtain circumferential control of the infrarenal IVC and aorta, this dissection does not need to be continued down to the bifurcation. The intrathoracic portion of the procedure is the most technically challenging (Figure 3), as injury to the intrathoracic or retrohepatic IVC can cause significant bleeding and obscure the operative field. When identifying the descending thoracic aorta, bluntly separate the aorta from the esophagus using your thumb and index finger. To avoid the avulsion of small intercostal arteries and subsequent bleeding, circumferential control is not essential as long as the descending thoracic aorta is accessible with a clearly defined space to accommodate an aortic clamp.
Cannulation of the infrarenal aorta is performed using the Seldinger technique (Figure 4). This can be done with the infrarenal aorta clamped proximally and distally (Figure 4A), or directly without clamping. Be careful not to insert the needle through the back wall of the aorta. A larger sheath can also be used if the aorta is of sufficient size. It is important to purge air from the IV extension tubing and connect the IV tubing from the UW solution to the 10Fr sheath in a sterile fashion. Ensure the IV channel is open, although flow will not initiate until the aorta is clamped (Figure 4B). When making the venotomy in the infrarenal IVC, it is important for the primary surgeon to securely grip the IVC using DeBakey forceps, so as not to lose the site of the venotomy and have a large blood volume pour into the operative field. A venotomy away from the pancreas, with the prompt placement of pool suction, maintains a bloodless operative field with clear tissue planes for the pancreatectomy (Figure 4C). Initial drainage will be dark venous blood, but this will lighten as the pig is exsanguinated and UW solution is perfused through the viscera. Be sure to use a large suction canister for this portion of the procedure.
The viscera, and in particular, the pancreas, will become increasingly pale as the UW solution is perfused (Figure 5A). The pancreatic duct can be identified by following the course of the duodenal lobe into the duodenum. The duct should be divided between two silk ties to avoid contamination with enteric contents (Figure 5B). While the pancreatic parenchyma should appear pale and bloodless, the remaining connective tissue surrounding the pancreas should be cleaned prior to islet isolation (Figure 5C).
The harvested pancreas can then undergo a multi-step islet isolation and purification process (Figure 6). Figure 7A shows the representative islet size distribution after the islet isolation and purification process. Figure 7B shows purified islets using brightfield microscopy, and after live/dead staining, with green and red staining representing viable and non-viable islet cells, respectively. Islet yield and purity can be evaluated using an islet cell counter. Representative yields are provided from three independent porcine pancreatic isolations are summarized in Figure 7C.
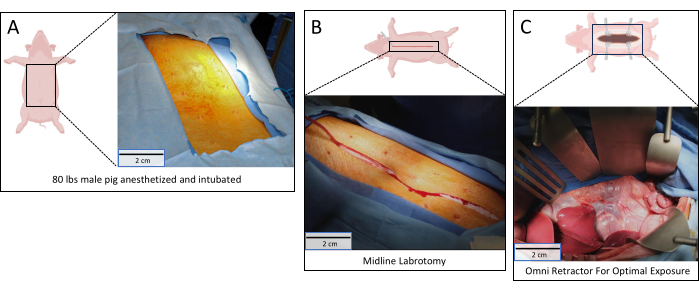
Figure 1: Operative setup. (A) Pig positioned in supine position with the abdomen sterilely prepped; (B) Midline laparotomy made from xiphoid to pubis; (C) Retroperitoneum exposed with Omni retractor in place. Scale bars = 2 cm. Please click here to view a larger version of this figure.
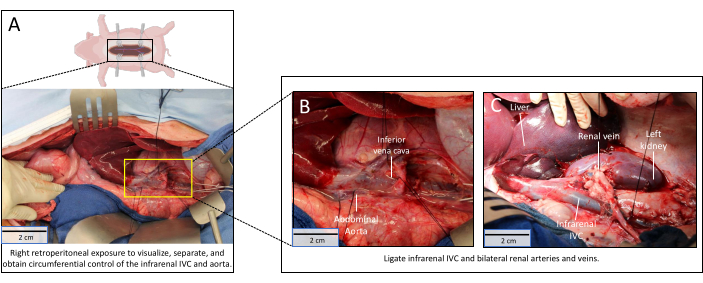
Figure 2: Retroperitoneal dissection. (A) Retroperitoneum dissection complete with circumferential control of the infrarenal IVC and renal veins. The yellow rectangle is magnified in Figure 2B; (B) Magnified image of infrarenal IVC and renal veins; (C) Renal veins tied off. Scale bars = 2 cm. Abbreviation: IVC = inferior vena cava. Please click here to view a larger version of this figure.
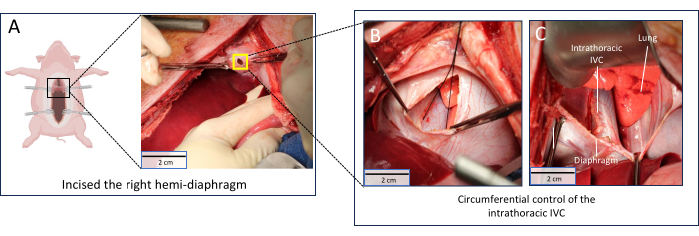
Figure 3: Obtaining supraceliac control. (A) Right hemidiaphragm incised. The yellow square is magnified in Figure 3B; (B) Extension of right hemidiaphragm incision to expose intrathoracic IVC and descending thoracic aorta; (C) Circumferential control of intrathoracic IVC obtained. Scale bars = 2 cm. Abbreviation: IVC = inferior vena cava. Please click here to view a larger version of this figure.
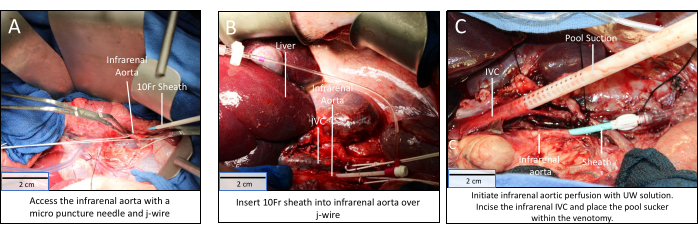
Figure 4: Infrarenal aortic cannulation, visceral isolation, and perfusion. (A) Proximal and distal control of the infrarenal aorta obtained; (B) 10Fr sheath placed in the infrarenal aorta by Seldinger technique; (C) Visceral perfusion via the infrarenal aortic sheath and drainage via venotomy and pool sucker in infrarenal IVC. Scale bars = 2 cm. Abbreviation: IVC = inferior vena cava. Please click here to view a larger version of this figure.
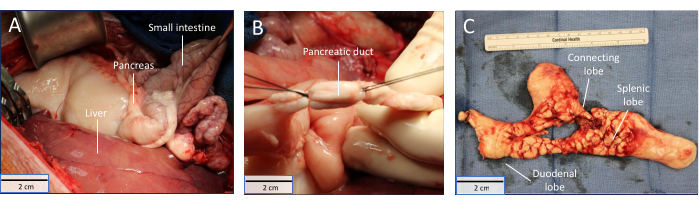
Figure 5: Total pancreatectomy. (A) Viscera becoming paler during perfusion; (B) pancreatic duct isolated; (C) Explanted porcine pancreas. Scale bars = 2 cm. Please click here to view a larger version of this figure.
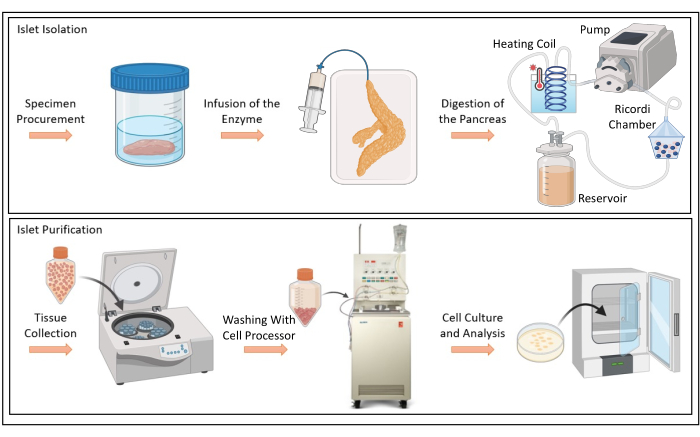
Figure 6: Islet isolation and purification. Schematic depiction of infusion of enzyme solution, digestion of porcine pancreas, tissue collection, cell washing with Cell Processor, and cell culture and analysis. Please click here to view a larger version of this figure.
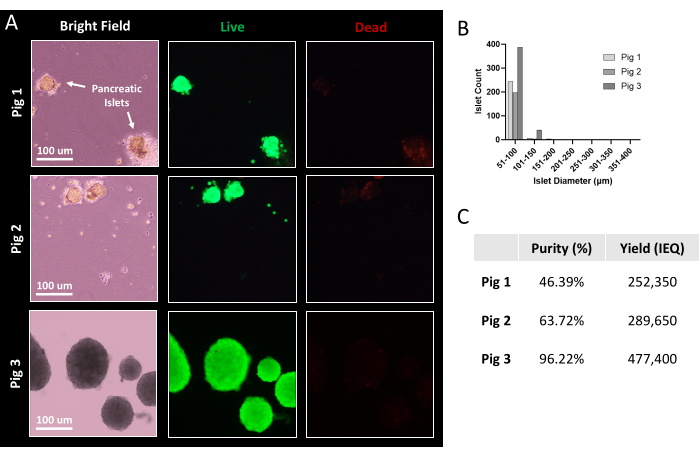
Figure 7: Representative islet viability and yield after purification. (A) Porcine islets after magnified bright-field imaging and live/dead staining. Green demonstrates live cells; red demonstrates dead cells. Scale bars = 100µm (B) Histogram distribution of porcine islet size following purification. (C) Table indicating islet purity and yield from three porcine pancreatic islet isolations. Abbreviation: IEQ = islet equivalent, the standard unit for islet quantification. Please click here to view a larger version of this figure.
Discussion
Pancreatic islet xenotransplantation using porcine donors is a promising strategy for the treatment of Type I diabetes. Islet isolation is challenging, and final islet viability and yield are highly susceptible to hypotension and tissue ischemia encountered during organ procurement16,17. To optimize pancreas procurement and preservation, this protocol presents a new method for aortic cannulation, visceral isolation, and perfusion during the procurement of the porcine pancreas prior to islet isolation.
Porcine islet isolation success is dependent on optimal procurement and preservation of the donor pancreas. The removal of all blood from the pancreas tissue is crucial for optimizing islet isolation as residual blood in the tissue leads to complement activation and thrombosis of pancreatic microvasculature, which can deteriorate islet yield and viability4,18. Previous techniques also include cannulation of the infrarenal aorta with intra-arterial flushing13,14,15,19. However, a major advantage of the technique presented here is the use of Seldinger technique for aortic cannulation to maintain a bloodless operative field. This not only minimizes hemodynamic instability before aortic cross clamping but also keeps the dissection planes pristine for the pancreatectomy, enabling faster organ recovery and minimizing WIT. This technique is also adaptable based on the size of the pig, as a smaller-caliber sheath could be used for mini-pigs with smaller-diameter aortas.
An additional strength of this technique is the significant reduction of WIT. Prior reports suffer from extended periods of WIT given pancreatectomy following the induction of cardiac arrest or exsanguination. Even when combined with ex vivo intraarterial and intraductal flushing, these procurement techniques are hindered by WIT, to which the porcine pancreas is particularly sensitive due to species-specific abundance of proteolytic enzymes and high fat content20. Prolonged WIT can also alter pancreatic tissue distention, likely due to an increase in resistance in the pancreatic ducts. This can significantly reduce the amount of enzyme reaching the distal pancreatic ducts, the overall efficiency of the digestion, and the yield of the islet isolation5.
Some limitations and pitfalls may be encountered during the procurement, but the majority can be avoided with careful planning and clearly defining personnel roles at the start of the procedure. Although the operative procurement can be carried out by two operators, a third assistant, who is not sterile, is needed to prepare the UW solution and container for the pancreas. When performing the intrathoracic portion of the procedure, it is important for the liver to be handled with gentle retraction. Injury and fracture of the liver can lead to persistent bleeding throughout the procedure. If this occurs, the liver can be packed with multiple lap pads to limit bleeding and limit blood obscuring the pancreatic procurement site. Of note, the identification and isolation of the pancreatic duct are critical for this procedure.
For the purification to go smoothly, two well-trained laboratory personnel should be available to perform the experiment. We recommend that one person maintain sterility during the procedure, while the other can be nonsterile. This will help with the handoff of critical materials during the procedure while maintaining sterility during pancreatic isolation.
During the perfusion procedure, the lumbar arteries that emerge from the infrarenal aorta are typically not identified and individually ligated. This may lead to a negligible volume of UW solution to disperse into non-viscera. However, we believe that minimizing the aortic dissection time and the risk of vascular injury during lumbar vessel manipulation outweigh the slight inefficiency of the UW perfusion not reaching the pancreatic tissue.
Once the pancreatic duct is identified, a catheter must be cannulated through the duct to proceed with the infusion of the pancreas. If the pancreatic duct is damaged, the parenchyma will not distend during subsequent perfusion of digestive solution during islet isolation. In the case of poor distention, islet isolation should be aborted. If the distention is favorable, the remaining enzyme can be injected into the parenchyma. In summary, this protocol provides a method for visceral isolation and perfusion during procurement of the porcine pancreas for islet isolation and purification. The use of the Seldinger technique for aortic cannulation minimizes hemodynamic changes and blood loss during procurement. This method can be used in combination with islet isolation to provide porcine islets for future studies and, ultimately, xenotransplantation.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This manuscript was supported by a clinical innovation grant from the Mid-America Transplant Foundation.
Materials
| 1 M NaOH, 100 mL | Millipore Sigma | 1310-73-2 | |
| 10x HBSS, 500 mL (15 to 30 °C) | Thermo Fisher | 14065056 | |
| 1x D-PBS, 500 mL | Thermo Fisher | 14200075 | |
| 1x HBSS, 1 L | Thermo Fisher | 14025076 | |
| 250 mL Conical Tubes | Millipore Sigma | CLS430236-6EA | |
| Amphotericin B [50 mg] | Millipore Sigma | PHR1662 | |
| Antiseptic Povidone Iodine 10% | Millipore Sigma | 25655-41-8 | |
| BioRad Gradient Former | BioRad | 395 | |
| Calcium Chloride dihydrate | Thermo Fisher | 10035-04-8 | |
| COBE Cell Processor | Ebay | 317690 | |
| Digestion circuit reservoir, Gibco bottle, 1 L | Thermo Fisher | 10341001 | |
| Dimethylsulfoxide (DMSO) | Thermo Fisher | 85190 | |
| Dithizone | Thermo Fisher | 60-10-6 | |
| Gradient Former Connection Kit | BioRad | 1652008 | |
| Heparin [1000 U/mL] | Emergency Medical Products | 0409-2720-01 | |
| HEPES Buffer (1 M), 100 mL | Thermo Fisher | 15630080 | |
| Human Serum Albumin 25%, 100 mL | Celprogen | HSA2001-25-1 | |
| Liberase (100 mg) | Millipore Sigma | 5401020001 | |
| LIVE/DEAD Viability/Cytotoxicity Kit | Thermo Fisher | L3224 | |
| Lympholyte 1.1 Media 500 mL | Cedarlane Labs | CL5020 | |
| Masterflex LS 16 Tubing | Masterfle | 96419-16 | |
| Masterflex Peristaltic Pump | Masterflex | 07522-30 | |
| PenStrep (2 mg/mL) | Celprogen | PS-30-002 | |
| Pulmozyme (1 mg/mL, 2.5 mL/vial) | Dornase Alfa | 8931278 | |
| Recovery Medium, PIM(R) | Prodo Laboratories, Inc. | PIM-R001GMP | |
| RPMI 1640 | Millipore Sigma | R8758-1L | |
| Standard Culture Medium, PIM(S) | Prodo Laboratories, Inc. | PIM-S001GMP | |
| University of Wisconsin (UW) Solution, 3 L | Global Transplant Solutions | 1000-0046-06 | |
| Waterbath | Sigma Alderich | CLS6783-1EA |
References
- Gillespie, K. M. Type 1 diabetes: pathogenesis and prevention. CMAJ. 175 (2), 165-170 (2006).
- Shapiro, A. M., Pokrywczynska, M., Ricordi, C. Clinical pancreatic islet transplantation. Nat Rev Endocrinol. 13 (5), 268-277 (2017).
- Shapiro, A. M., et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med. 343 (4), 230-238 (2000).
- Anazawa, T., et al. Improved method of porcine pancreas procurement with arterial flush and ductal injection enhances islet isolation outcome. Transplant Proc. 42 (6), 2032-2035 (2010).
- Dufrane, D., et al. Parameters favouring successful adult pig islet isolations for xenotransplantation in pig-to-primate models. Xenotransplantation. 13 (3), 204-214 (2006).
- Hilling, D. E., Rijkelijkhuizen, J. K., Tons, H. A., Terpstra, O. T., Bouwman, E. Porcine islet isolation outcome is not affected by the amount and distribution of collagen in the pancreas. Xenotransplantation. 17 (3), 250-255 (2010).
- Lamb, M., et al. In vitro maturation of viable islets from partially digested young pig pancreas. Cell Transplant. 23 (3), 263-272 (2014).
- Otsuka, R., et al. Pancreas preservation with a neutrophil elastase inhibitor, alvelestat, contributes to improvement of porcine islet isolation and transplantation. J Clin Med. 11 (15), 4290 (2022).
- Vanderschelden, R., Sathialingam, M., Alexander, M., Lakey, J. R. T. Cost and scalability analysis of porcine islet isolation for islet transplantation: comparison of juvenile, neonatal and adult pigs. Cell Transplant. 28 (7), 967-972 (2019).
- Weegman, B. P., et al. Temperature profiles of different cooling methods in porcine pancreas procurement. Xenotransplantation. 21 (6), 574-581 (2014).
- Hart, N. A., et al. Determination of an adequate perfusion pressure for continuous dual vessel hypothermic machine perfusion of the rat liver. Transpl Int. 20 (4), 343-352 (2007).
- Tullius, S. G., et al. Accumulation of crystal deposits in abdominal organs following perfusion with defrosted University of Wisconsin solutions. Am J Transplant. 2 (7), 627-630 (2002).
- Ching, C. D., et al. A reliable method for isolation of viable porcine islet cells. Arch Surg. 136 (3), 276-279 (2001).
- Lu, Y., et al. Adult pig islet isolation. J Vis Exp. (176), (2021).
- Taylor, M. J., Baicu, S., Greene, E., Vazquez, A., Brassil, J. Islet isolation from juvenile porcine pancreas after 24-h hypothermic machine perfusion preservation. Cell Transplant. 19 (5), 613-628 (2010).
- Iwanaga, Y., Sutherland, D. E., Harmon, J. V., Papas, K. K. Pancreas preservation for pancreas and islet transplantation. Curr Opin Organ Transplant. 13 (4), 445-451 (2008).
- Pratschke, J., et al. Brain death and its influence on donor organ quality and outcome after transplantation. Transplantation. 67 (3), 343-348 (1999).
- vander Windt, D. J., et al. Early islet damage after direct exposure of pig islets to blood: has humoral immunity been underestimated. Cell Transplant. 21 (8), 1791-1802 (2012).
- Wright, M. J., Cavanagh, T. J., Fetterhoff, T. J., Wile, K. J. Effect of blood content on porcine pancreatic dissociation and islet yield. Transplant Proc. 26 (6), 3442 (1994).
- Toso, C., et al. Isolation of adult porcine islets of Langerhans. Cell Transplant. 9 (3), 297-305 (2000).

.
