Laser Doppler Perfusion Imaging in the Mouse Hindlimb
Summary
Here, we present a protocol that demonstrates the technique and necessary controls for Laser Doppler perfusion imaging to measure blood flow in the mouse hindlimb.
Abstract
Blood flow recovery is a critical outcome measure after experimental hindlimb ischemia or ischemia-reperfusion. Laser Doppler perfusion imaging (LDPI) is a common, noninvasive, repeatable method for assessing blood flow recovery. The technique calculates overall blood flow in the sampled tissue from the Doppler shift in frequency caused when a laser hits moving red blood cells. Measurements are expressed in arbitrary perfusion units, so the contralateral non-intervened upon leg is usually used to help control measurements. Measurement depth is in the range of 0.3-1 mm; for hindlimb ischemia, this means that dermal perfusion is assessed. Dermal perfusion is dependent on several factors—most importantly skin temperature and anesthetic agent, which must be carefully controlled to result in reliable readings. Furthermore, hair and skin pigmentation can alter the ability of the laser to either reach or penetrate to the dermis. This article demonstrates the technique of LDPI in the mouse hindlimb.
Introduction
Skin ulceration with inadequate wound healing is a leading cause of amputations in human patients1. Adequate wound healing requires higher levels of arterial perfusion than are needed to maintain intact skin, which is compromised in patients with peripheral arterial disease2,3,4. Several other rheumatologic conditions and diabetes can also lead to disturbed and inadequate skin microcirculation to heal wounds5,6. Many diabetic patients have concomitant peripheral arterial disease, placing them at especially high risk for amputation. Laser Doppler perfusion imaging (LDPI) is used in clinical situations to evaluate the skin microcirculation, as well as in research situations to evaluate blood flow and blood flow recovery after experimental hindlimb ischemia, ischemia-reperfusion, and microsurgical flaps7.
The LDPI system projects a low power laser beam that is deflected by a scanning mirror to move over a region of interest. This differs from Laser Doppler flowmetry, which provides a perfusion measurement for the small area of tissue in direct contact with the flowmetry probe8. When the laser beam interacts with moving blood in the microvasculature, it undergoes a Doppler frequency shift, which is photodetected by the scanner and converted to arbitrary perfusion units. Because LDPI is a light-based technique, it is limited in terms of depth of penetration to 0.3-1 mm, meaning that for the most part dermal perfusion is assessed7. Dermal flow can be altered by skin temperature and the sympathetic nervous system, which may be affected by various anesthetic agents9. Measurements from the optical laser are also affected by ambient lighting conditions, skin pigmentation, and can be blocked by overlying fur or hair7.
LDPI is the most commonly used research technique to monitor perfusion recovery after ischemia because it is noninvasive, does not require contrast administration, and has quick scan times allowing data collection on multiple animals. This makes it ideal to help determine whether treatments aimed at therapeutic arteriogenesis or angiogenesis are effective in small animal models. Blood flow recovery after hindlimb ischemia as measured by LDPI correlates well with collateral artery development when assessed by other means such as Microfil casting or micro-CT10,11. The goal of this protocol is to demonstrate the assessment of hindlimb perfusion using LDPI.
Protocol
Animal experiments were performed according to a protocol approved by the University of Washington Institutional Animal Care and Use Committee.
1. Scanner preparation
- Adjust the scanner height so that the distance to the scanned subject is approximately 30 cm.
- Turn on the imager and launch the associated software.
- Open the Measurement program. If the software is correctly communicating with the scanner, the infrared laser turn on warning will appear.
- Calibrate the machine with manufacturer provided standards (not shown in the video and will depend on the specific model of machine being used).
- Adjust the scanner settings to be appropriate for the background material and lighting setup in the room.
- Set the gain levels DC FLUX and CONC, per the manufacturer's instructions (not shown in the video).
- Set the Background Threshold by pointing the laser beam at the black background material, and press Auto BK Set.
2. Mouse pre-scanning preparation
- Set up the isoflurane induction chamber with appropriate scavenging of the waste gas.
NOTE: Placing the induction chamber on a warming pad will help prevent mouse temperature loss during anesthesia induction. - Turn on the homeothermic blanket, which is placed in the scanning area underneath a nonreflective surface (in this case a black neoprene fabric). Set the homeothermic blanket to maintain a body temperature of 37 °C.
- Position the temperature probe for the homeothermic blanket and lubricant so they are ready for insertion.
- Place the anesthesia mask and scavenging system in the scanning area.
- Anesthetize the mouse with an isoflurane vaporizer. Set the oxygen rate to 1 L/min of flow and adjust the isoflurane to 4% for anesthesia induction. Turn on the flow to the anesthesia induction chamber, and the mouse breathing rate will slow. Adequate anesthesia is achieved when the mouse loses its righting reflex.
- Transfer the mouse to an anesthetic mask/nose cone with attached waste gas scavenger and adjust the isoflurane to 1.5%.
NOTE: This anesthesia level is generally adequate to keep the mouse lying relatively still during scanning, but is not intended to provide surgical levels of anesthesia, so the depth of anesthesia is not checked. Changing the isoflurane level causes changes in heartbeat, respiration, and dermal perfusion, so a consistent percentage should be used throughout any time course experiment and for all experimental subjects. Alternative anesthetic techniques such as IP injection of ketamine xylazine can also be used, but the same anesthetic technique should be used throughout any time course study as different anesthetics affect dermal perfusion differently. - (Optional depending on scanning area) If the planned region of interest to be scanned is covered by fur, use a small electric trimmer or depilatory cream to remove the hair from the region of interest.
NOTE: The depilatory cream should be completely removed, and the mouse skin dried prior to scanning. - Place the mouse in the appropriate scanning position on the black nonreflective surface covering the homeothermic blanket, confirming that both hindlimbs remain on the heat source throughout equilibration and scanning (Figure 1).
NOTE: It is important to maintain both feet on the homeothermic blanket to prevent regional variation in temperature. - Insert the lubricated rectal temperature probe associated with the homeothermic blanket.
- Equilibrate the mouse temperature to desired scanning temperature (37 °C); approximately 5-10 minutes.
- Select Scanner Setup, which can be accessed from the top menu or from the scanner setup icon. Adjust the scan area by changing the X-Y coordinates to accommodate the region of interest. Scan speed will depend on the scan resolution. Higher resolution will result in longer scan times. For repeat scanning focusing on global perfusion, as opposed to higher resolution focusing on anatomic perfusion, a scan speed of 4 ms/pixel is adequate.
NOTE: Higher resolution and single scan should be considered if the researcher is attempting to directly study the developing collateral circulation (best imaged in the ventral thigh and calf where it is closer to the skin). Repeated scan at lower resolution/speed (e.g., 4 ms/pixel) is adequate when assessing global perfusion to the end organ of the mouse footpad. The software shown in the video automatically loads the previously used template for scanning area, speed, and resolution when restarted, or it can be retrieved from a stored file if different regions of interest are being used for various experiments. - If performing repeat scans, select the Repeat and Line Scan tab. The number of scans can be changed (in this case 3 scans) as well as the repeat interval. The minimum time for the repeat interval would be the estimated scanning time, which is shown in the grayed-out area on the right of the box determined by scan area and scan resolution. Adding a few seconds allows the user to pause and potentially reposition the mouse if needed between scans.
3. Scanning
- Select the Image Scan tab and select the Mark button. The laser will move to outline the scanning area. Adjust the mouse position so that the target to be scanned is within the marked area.
NOTE: For footpad or footpad and calf scanning, prone positioning with the hindlimbs extended provides a more consistent region of interest than supine positioning. The femoral artery and saphenous artery and collaterals are very close to the ventral surface of the thigh and calf, so supine positioning is preferred if using these regions of interest. - Start repeated measurement by selecting the Repeat Scanning icon and press the Play Button to initiate the scan.
- Confirm the scanning distance in the pop-up window and click OK to start scanning.
- Monitor the mouse during scanning for mouse movement; if the mouse moves sufficiently that the hindpaws are no longer in the scanning region in the middle of a scan, restart the scan. Small variations in mouse hindpaw position can be accommodated for in the analysis software.
- Monitor the mouse temperature during the scanning process as it may fluctuate even with the use of the homeothermic blanket. If there is too much variation in the mouse temperature, this may result in significant variation between scans. Generally, a temperature range of 36.8-37.2 °C will result in acceptable data.
- Save the captured scan under the Save as window with a file name that includes mouse identifier and timepoint for easier data analysis. Enter mouse and timepoint details if desired in the subject details window.
- Turn off the isoflurane and remove the rectal temperature probe.
- Disinfect the rectal temperature probe with 70% ethanol so it is ready for use in the next mouse.
- Allow the mouse to recover from anesthesia to the point where it displays a righting reflex by flipping from the supine position to the prone position prior to returning it to the cage.
NOTE: Recovery can be carried out either on a warming blanket for isoflurane since recovery is very quick or in a warmed recovery cage for ketamine/xylazine.
4. Capturing LDPI data (Figure 3)
- Open the imaging review software program.
- Go to the file menu, open, and locate the saved file.
- Select the ROI icon from the toolbar.
- Select the Add Polygon button.
- Trace the region of interest (ROI) for the control hindlimb using the mouse. Polygon tracing does not have to be exact as the gray background will not be included in the calculated averages.
- Repeat steps 4.3-4.5 for the surgical hindlimb.
- Choose the Statistics icon to open the Image ROIs Statistics Results (PU) window.
- Export the results for Polygon 1 (control hindlimb) and Polygon 2 (surgical hindlimb) to a data collection worksheet via copy/paste.
5. Analysis
- Capture the data as Surgical/Control ratio for each scan.
- Use the averaged surgical/control for all three scans for the data point for that particular mouse at that timepoint. Because of biologic variability in the response to hindlimb ischemia, in general 8-10 mice are required per timepoint to achieve reproducible results with ~10% standard error.
NOTE: Before allowing the mouse to recover from anesthesia, it is worthwhile to perform a quick analysis of the repeated scans to check if the data is too variable (e.g., more than 100-150 perfusion units different between scans 1-3). High variation between repeated scans suggests the mouse was not completely equilibrated during the scan (Figure 2), and a repeat scan can be performed without losing a datapoint, which would occur if the images are not analyzed until a later date. Changing the color palette to optimize the dynamic range of displayed flux values may be necessary to better display scan variation (Figure 2).
Representative Results
Successful LDPI should result in consistent repeated measures scans, with no more than 100-150 perfusion unit variation (corresponding to about 10% of the usual mean perfusion for the mouse footpad) between the three scans (Figure 2). As demonstrated in Figure 2, repeat scans help determine that the mouse has been appropriately equilibrated so that the ischemic/control ratio best reflects the underlying blood flow as opposed to variation in dermal perfusion caused by temperature variation. Using single scans for datapoints will increase the variability leading to the need for more experimental mice. When used for hindlimb ischemia, the surgical hindlimb should have decreased global perfusion when compared to the control hindlimb. Results are expressed as a ratio of surgical hindlimb perfusion/control hindlimb perfusion. As mice initially vasodilate and develop their intrinsic collateral network over time, blood flow recovery by LDPI should be seen over a postoperative time course (Figure 4). The degree of recovery is dependent on the mouse strain and severity of the hindlimb ischemia model.
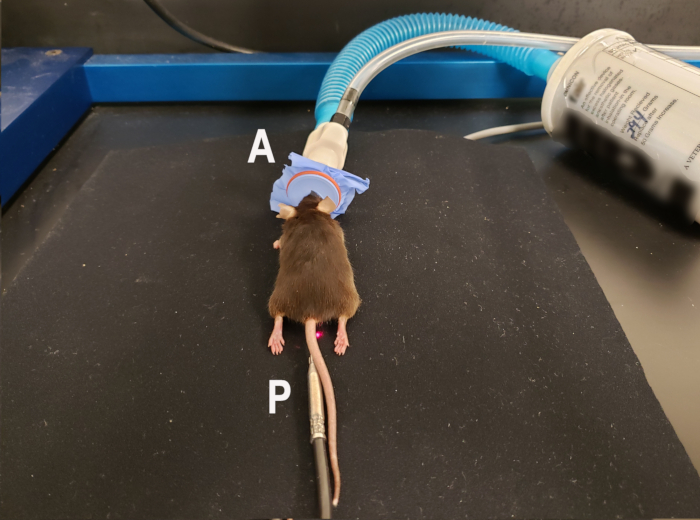
Figure 1. Mouse positioning for laser Doppler perfusion imaging of the ventral footpads. Anesthetized mouse using isoflurane nose cone (A) is placed in the prone position with hindlimbs extended to allow scanning of the ventral footpads. Rectal temperature probe (P) for homeothermic blanket is in place to maintain consistent body temperature during the scan. The homeothermic blanket pad is underneath the nonreflective neoprene material used to provide background for the scan. Laser indicating the middle of the scanning region is visible next to the mouse's tail. Please click here to view a larger version of this figure.
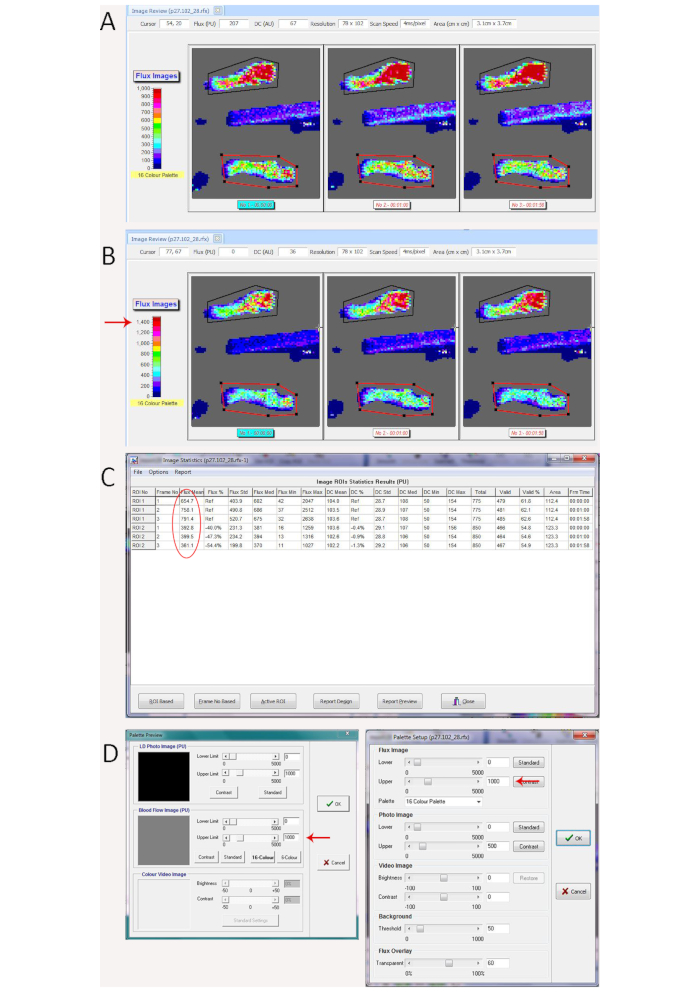
Figure 2. Demonstration of scan variation from mouse temperature change better seen with color palette adjustment. (A) Laser Doppler repeated scans with significant variation caused by mouse core temperature variation during the repeated scans, which is visible based on the perfusion units translated to color on the repeated flux scans. (B) Changing the dynamic range on the color palette (shown on the left of the scan window) from 0-1000 in A to 0-1500 (red arrow) in B makes the variation more obvious. (C) Statistics showing mean perfusion values for the region of interest (circled in red) for the control hindlimb (Polygon 1 in black on the rfx image in A and B) ranges between 655 for the 1st scan to 791 on the 3rd scan and mean perfusion for the region of interest for the ischemic hindlimb (Polygon 2 in red on the rfx image in A and B) showed less variation (361 to 400), leading to significant differences in the ischemic/control ratio between the repeated scans (0.60, 0.53, and 0.46). (D) Window to change the dynamic range of the color palette in the measurement software (left panel) and image review software (right panel). Red arrows show where to increase or decrease the upper range. Please click here to view a larger version of this figure.
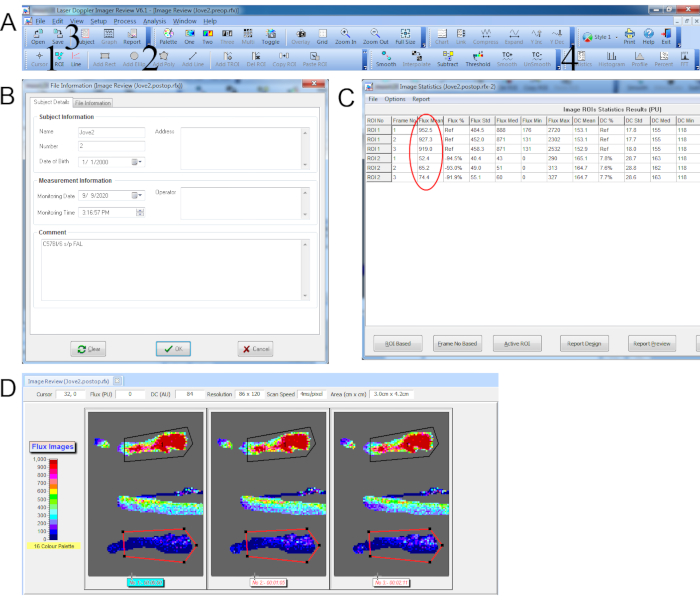
Figure 3. Data capture for laser Doppler perfusion imaging with repeated scans. (A) Top toolbar with 1. Add ROI icon. 2. Add Polygon icon. 3. Subject details icon (accesses window pop-up in B). 4. Statistics icon (accesses window pop-up in C). (B) Subject details window. (C) Statistics window showing the mean perfusion values (circled in red) for each ROI. (D) Repeated scan with polygon ROI traced around control hindpaw (black) and ischemic hindpaw (red). Please click here to view a larger version of this figure.
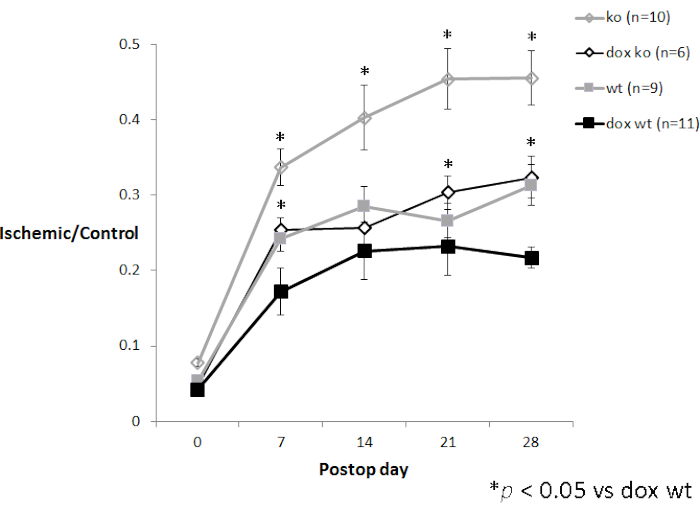
Figure 4. Time course experiment with LDPI data. P27 knockout mice (3-5 month old female CDKN1b-/- mice on a C57Bl/6 background) after femoral artery ligation with (n=6) and without (n=10) oral doxycycline treatment compared to age-matched female wildtype C57Bl/6 mice with (n=11) and without (n=9) oral doxycycline treatment (unpublished data from the author). Error bars represent standard error of the mean (SEM). Please click here to view a larger version of this figure.
Discussion
Consistent technique is critical for obtaining reliable results with LDPI. The same anesthetic, temperature settings, mouse position, and region of interest should be used throughout the entire time course. Different anesthetic agents will result in higher or lower perfusion values9. Isoflurane anesthesia is convenient because of its rapid onset and emergence as well as overall safety. A consistent percentage of isoflurane should be used as depth of anesthesia with this vasodilatory agent may alter skin perfusion. If the region of interest includes fur, then the same method of hair removal should be used each time, as chemically depilated mice will have higher perfusion values than mice whose fur was removed with electric clippers7. Mouse temperature has a large effect on perfusion imaging, with mice at 36 °C having significantly lower perfusion values than mice at 38 °C7,12. The ischemic hindlimb may also react differently to regional temperature variation than the control hindlimb (Figure 2). In this protocol, a homeothermic blanket is used to maintain mouse temperature during scanning, which provides more consistent temperature control during the scanning process than pre-equilibrating the mouse on a 37 °C warming plate for five minutes and then scanning on a non-warmed surface as shown by Niiyama et al.13
If only the footpads are chosen as the region of interest, then prone positioning is preferred because of reproducibility in the scanned region of interest. The advantage of this approach is that it studies the area furthest from the heart, and the most clinically relevant area corresponding to where ischemic foot ulcers are common. The footpads are hairless, so clipping or depilation is not necessary, simplifying preparation and time for measurement. Also, non-white mice may have patches of pigmentation in the skin of the calf or thigh, which can interfere with LDPI measurement. If the chosen region of interest includes the calf and thigh, then supine positioning is preferred because the femoral and saphenous artery run along the ventral surface of the hindlimb and can be imaged using LDPI7. From the supine position, consistent imaging of the foot is difficult to capture, as the side and top surfaces may be variably imaged.
Collateralization and blood flow recovery after hindlimb ischemia is dependent on a number of different factors including hindlimb ischemia model, mouse strain, gender, and age. Certain strains of mice such as C57Bl/6 have robust baseline collaterals, with a less dramatic drop in perfusion after induction of acute hindlimb ischemia, whereas others such as BALB/c have poor collaterals14,15. Female mice have worse recovery than male mice. Older mice also have worse blood flow recovery than younger mice16. Therefore, mice need to be strain, age, and gender matched for reliable conclusions to be drawn regarding blood flow recovery using LDPI data. Even with rigorous matching and using inbred strains of mice, there is a certain amount of biologic variability to the mouse response to hindlimb ischemia, so adequate mouse numbers (usually 8-10 mice per timepoint) are required for valid data. Furthermore, normalization of LDPI does not necessarily mean restoration of normal levels of arterial flow as the measurement is done in anesthetized mice who do not have any skeletal muscle demand. Finally, because of the limitations in depth of penetration, detailed anatomic studies of collateral pathways that may run through the deeper musculature of the thigh and calf are not possible with LDPI.
Several other methods have been used to assess blood flow recovery including perfusion-based imaging of the skin such as laser speckle imaging17,18,19 or deeper structures such as contrast-enhanced ultrasound of skeletal muscle20, MRI21, and (13)N-ammonia PET22. Also used are anatomic-based imaging of collateral vessels such as micro-CT10, OCT23, and contrast-enhanced ultrasound with intravital microscopy24. Because of fast scan time, relative ease of data capture and analysis, and avoidance of the need for intravenous contrast, LDPI is the predominant method used by most groups in the literature. Weaknesses include that the technique measures blood flow velocities and provides data in arbitrary perfusion units rather than measuring absolute tissue perfusion, the scanning depth is relatively shallow, and it provides relatively poor anatomic detail.
LDPI is most commonly used to assess recovery after various hindlimb ischemia models7. It has also been used in ischemia-reperfusion research both in the hindlimb25 as well as in deeper splanchnic organs or the spinal cord26,27,28. Assessment of deep structures however requires surgical exposure of the structure to be scanned, making repeated measurements more difficult because of scarring. A further application is assessment of flap reperfusion after microsurgery29.
In conclusion, LDPI is an effective, easily performed, and repeatable method for measuring hindlimb dermal perfusion as a reflection of overall arterial perfusion. Consistent technique is required when using LDPI to obtain reliable data.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was carried out with the use of facilities and resources at the VA Puget Sound Health Care Center. The work is that of the author and does not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government. Dr Tang is currently funded via the VA (Merit 5 I01 BX004975-02).
Materials
| Black nonreflective material | Fabric store, black neoprene recommended by company | ||
| F/air cannister | A.M. Bickford Inc | 80120 | |
| Homeothermic blanket with rigid metal probe | Harvard Apparatus | Also comes with flexible probe, but this is less durable | |
| Isoflurane Anesthesia machine | Drager | Multiple manufacturers | |
| Isoflurane induction chamber | VetEquip | 941444 | 2 L chamber |
| Moor laser Doppler perfusion imager | Moor Instruments | MoorLDI2-IR | Higher resolution imager (MoorLDI2-HIR) |
| Mouse Anesthesia nose cone | Multiple manufacturers | ||
| Nair | Nair | ||
| Oxygen tank | Multiple manufacturers | ||
| Surgilube | Multiple distributors |
References
- Varma, P., Stineman, M. G., Dillingham, T. R. Epidemiology of limb loss. Physical Medicine and Rehabilitation Clinics of North America. 25 (1), 1-8 (2014).
- Farber, A. Chronic Limb-Threatening Ischemia. New England Journal of Medicine. 379 (2), 171-180 (2018).
- Abularrage, C. J., et al. Evaluation of the microcirculation in vascular disease. Journal of Vascular Surgery. 42 (3), 574-581 (2005).
- Houben, A., Martens, R. J. H., Stehouwer, C. D. A. Assessing Microvascular Function in Humans from a Chronic Disease Perspective. Journal of the American Society of Nephrology. 28 (12), 3461-3472 (2017).
- Mahe, G., Humeau-Heurtier, A., Durand, S., Leftheriotis, G., Abraham, P. Assessment of skin microvascular function and dysfunction with laser speckle contrast imaging. Circulation: Cardiovascular Imaging. 5 (1), 155-163 (2012).
- Murray, A. K., Herrick, A. L., King, T. A. Laser Doppler imaging: a developing technique for application in the rheumatic diseases. Rheumatology (Oxford). 43 (10), 1210-1218 (2004).
- Greco, A., et al. Repeatability, reproducibility and standardisation of a laser Doppler imaging technique for the evaluation of normal mouse hindlimb perfusion. Sensors (Basel). 13 (1), 500-515 (2012).
- Sonmez, T. T., et al. A novel laser-Doppler flowmetry assisted murine model of acute hindlimb ischemia-reperfusion for free flap research. PLoS One. 8 (6), 66498 (2013).
- Gargiulo, S., et al. Effects of some anesthetic agents on skin microcirculation evaluated by laser Doppler perfusion imaging in mice. BMC Veterinary Research. 9, 255 (2013).
- Ankri-Eliahoo, G., Weitz, K., Cox, T. C., Tang, G. L. p27(kip1) Knockout enhances collateralization in response to hindlimb ischemia. Journal of Vascular Surgery. 63 (5), 1351-1359 (2016).
- McEnaney, R. M., Shukla, A., Madigan, M. C., Sachdev, U., Tzeng, E. P2Y2 nucleotide receptor mediates arteriogenesis in a murine model of hind limb ischemia. Journal of Vascular Surgery. 63 (1), 216-225 (2016).
- Padgett, M. E., McCord, T. J., McClung, J. M., Kontos, C. D. Methods for Acute and Subacute Murine Hindlimb Ischemia. Journal of Visualized Experiments. (112), e54166 (2016).
- Niiyama, H., Huang, N. F., Rollins, M. D., Cooke, J. P. Murine model of hindlimb ischemia. Journal of Visualized Experiments. (23), e1035 (2009).
- Chalothorn, D., Faber, J. E. Strain-dependent variation in collateral circulatory function in mouse hindlimb. Physiological Genomics. 42 (3), 469-479 (2010).
- Helisch, A., et al. Impact of mouse strain differences in innate hindlimb collateral vasculature. Arteriosclerosis, Thrombosis, and Vascular Biology. 26 (3), 520-526 (2006).
- Faber, J. E., et al. Aging causes collateral rarefaction and increased severity of ischemic injury in multiple tissues. Arteriosclerosis, Thrombosis, and Vascular Biology. 31 (8), 1748-1756 (2011).
- Forrester, K. R., Stewart, C., Tulip, J., Leonard, C., Bray, R. C. Comparison of laser speckle and laser Doppler perfusion imaging: measurement in human skin and rabbit articular tissue. Medical & Biological Engineering & Computing. 40 (6), 687-697 (2002).
- Briers, J. D. Laser Doppler, speckle and related techniques for blood perfusion mapping and imaging. Physiological Measurement. 22 (4), 35-66 (2001).
- Heeman, W., Steenbergen, W., van Dam, G., Boerma, E. C. Clinical applications of laser speckle contrast imaging: a review. Journal of Biomedical Optics. 24 (8), 1-11 (2019).
- Nguyen, T., Davidson, B. P. Contrast Enhanced Ultrasound Perfusion Imaging in Skeletal Muscle. Journal of Cardiovascular Imaging. 27 (3), 163-177 (2019).
- Zaccagnini, G., et al. Magnetic Resonance Imaging Allows the Evaluation of Tissue Damage and Regeneration in a Mouse Model of Critical Limb Ischemia. PLoS One. 10 (11), 0142111 (2015).
- Penuelas, I., et al. PET as a measurement of hindlimb perfusion in a mouse model of peripheral artery occlusive disease. Journal of Nuclear Medicine. 48 (13), 1216-1223 (2007).
- Jia, Y., Qin, J., Zhi, Z., Wang, R. K. Ultrahigh sensitive optical microangiography reveals depth-resolved microcirculation and its longitudinal response to prolonged ischemic event within skeletal muscles in mice. Journal of Biomedical Optics. 16 (8), 086004 (2011).
- Turaihi, A. H., et al. Combined Intravital Microscopy and Contrast-enhanced Ultrasonography of the Mouse Hindlimb to Study Insulin-induced Vasodilation and Muscle Perfusion. Journal of Visualized Experiments. (121), e54912 (2017).
- Liu, C., et al. Enhanced autophagy alleviates injury during hindlimb ischemia/reperfusion in mice. Experimental and Therapeutic Medicine. 18 (3), 1669-1676 (2019).
- Liu, D. L., Svanberg, K., Wang, I., Andersson-Engels, S., Svanberg, S. Laser Doppler perfusion imaging: new technique for determination of perfusion and reperfusion of splanchnic organs and tumor tissue. Lasers in Surgery and Medicine. 20 (4), 473-479 (1997).
- Jing, Y., Bai, F., Chen, H., Dong, H. Using Laser Doppler Imaging and Monitoring to Analyze Spinal Cord Microcirculation in Rat. Journal of Visualized Experiments. (135), e56243 (2018).
- Zhang, D., Li, S., Wang, S., Ma, H. An evaluation of the effect of a gastric ischemia-reperfusion model with laser Doppler blood perfusion imaging. Lasers in Medical Science. 21 (4), 224-228 (2006).
- Fitzal, F., et al. Circulatory changes after prolonged ischemia in the epigastric flap. Journal of Reconstructive Microsurgery. 17 (7), 535-543 (2001).

