Electrospinning Growth Factor Releasing Microspheres into Fibrous Scaffolds
Summary
This protocol combines electrospinning and microspheres to develop tissue engineered scaffolds to direct neurons. Nerve growth factor was encapsulated within PLGA microspheres and electrospun into Hyaluronic Acid (HA) fibrous scaffolds. The protein bioactivity was tested by seeding the scaffolds with primary chick Dorsal Root Ganglia and culturing for 4-6 days.
Abstract
This procedure describes a method to fabricate a multifaceted substrate to direct nerve cell growth. This system incorporates mechanical, topographical, adhesive and chemical signals. Mechanical properties are controlled by the type of material used to fabricate the electrospun fibers. In this protocol we use 30% methacrylated Hyaluronic Acid (HA), which has a tensile modulus of ~500 Pa, to produce a soft fibrous scaffold. Electrospinning on to a rotating mandrel produces aligned fibers to create a topographical cue. Adhesion is achieved by coating the scaffold with fibronectin. The primary challenge addressed herein is providing a chemical signal throughout the depth of the scaffold for extended periods. This procedure describes fabricating poly(lactic-co-glycolic acid) (PLGA) microspheres that contain Nerve Growth Factor (NGF) and directly impregnating the scaffold with these microspheres during the electrospinning process. Due to the harsh production environment, including high sheer forces and electrical charges, protein viability is measured after production. The system provides protein release for over 60 days and has been shown to promote primary nerve cell growth.
Introduction
One of the ongoing challenges in neural tissue engineering is creating a nerve conduit (NC) that mimics the extra cellular matrix, where nerves grow naturally. Research has shown that cells respond to several factors in their environment including mechanical, topographical, adhesive and chemical signals1-3. One of the primary challenges in this field is determining the appropriate combination of signals and fabricating a system that can maintain cues for an extended period to support cell growth4. Peripheral neurons are known to prefer a soft substrate, be directed by aligned fibers, and respond to nerve growth factor (NGF)5-7. NCs that can provide chemical cues for weeks have been shown to provide improved functional recovery closer to that of allografts, the current gold standard for nerve repair8,9.
Various materials and production methods can be used to produce mechanical and topographical cues10-13. Mechanical cues are inherent to the material chosen, making selection of the appropriate material for the application critical1,13. Production methods to control topographical cues include phase separation, self-assembly and electrospinning1,13. For microscale applications, microfluidics, photopatterning, etching, salt leaches, or gas foams can also be used14-17. Electrospinning has emerged as the most popular way to engineer fibrous substrates for tissue culture due to its flexibility and ease of production13,18-23. Electrospun nanofibers are fabricated by applying a high voltage to a polymer solution causing it to repel itself and stretch across a short gap to discharge24. An aligned scaffold can be created by collecting the fibers on a grounded rotating mandrel and nonaligned scaffolds are collected on a stationary plate25. Adhesion signaling can be achieved by coating the fibrous scaffold with fibronectin or conjugating an adhesion peptide, such as RGD, to the HA before electrospinning26.
Chemical signals, such as growth factors, are the most difficult to maintain over extended periods because they need a source for controlled release. Many systems have been attempted to add controlled release to electrospun fibrous networks with varying levels of success. These methods include blend electrospinning, emulsion electrospinning, core shell electrospinning and protein conjugation27. Additionally, electrospinning is traditionally done in a volatile solvent, which can affect the viability of the protein28, therefore maintaining bioactivity of the protein must be considered.
This approach specifically addresses combining mechanical, topographical, chemical and adhesive signals to create a tunable scaffold for peripheral nerve growth. Scaffold mechanics is precisely controlled by synthesizing methacrylated Hyaluronic Acid (HA). The methacrylation sites are used to attach photo reactive crosslinkers. The crosslinked material is no longer water soluble and is exclusively broken down by enzymes29. The amount of crosslinking changes the degradation rate, mechanics and other physical properties of the material. Using HA with 30% methacrylation, which has a tensile modulus of ~500 Pa, creates a soft substrate that is close to the native mechanics of neural tissue and is typically preferred by neurons26,29. Electrospinning on a rotating mandrel is used to create aligned fibers for a topographical cue. Using electrospinning along with microspheres provides chemical signals within the scaffold over extended periods. To support neurite growth microspheres containing NGF are used to create the chemical signal. Unlike most electrospun materials HA is soluble in water so the NGF does not encounter harsh solvents during production. To add an adhesive signal, the scaffold is coated with fibronectin. The completed system contains all four types of signals described above: soft (mechanical) aligned (topographical) fibers with NGF releasing microspheres (chemical) coated with fibronectin (adhesive). Production and testing of this system is described in this protocol.
The process begins with the production of the microspheres with a Water-in-Oil-in-Water Double Emulsion. The emulsion is stabilized with a surfactant, Polyvinyl Alcohol (PVA). The inner water phase contains the protein. As it is added to the oil phase, containing the PLGA shell material dissolved in Dichloromethane (DCM), the surfactant creates a barrier between the phases protecting the protein from the DCM. This emulsion is than dispersed in another water phase containing PVA to create the outer surface of the microspheres. The stable emulsion is stirred to allow the DCM to evaporate. After rinsing and lyophilizing you are left with the dry microspheres containing the protein.
After the microspheres are completed they are ready to be electrospun into scaffolds. First you prepare the electrospinning solution. The viscosity of the solution is critical to proper fiber formation. Solutions of pure HA do not meet this requirement; PEO is added as a carrier polymer to allow for electrospinning. The microspheres are added to the solution and electrospun resulting in a fibrous scaffold with microspheres distributed throughout.
Once the production is complete, the protein should be tested to verify its viability. To do this, a primary cell that responds to NGF can be used. This protocol uses Dorsal Root Ganglia (DRG) from 8-10 day old chicken embryos. The cell bundles are seeded onto scaffolds containing microspheres filled with NGF or ones that are empty. If the NGF is still viable you should see enhanced neurite growth on the NGF containing scaffolds. If the NGF is no longer viable it will not promote neurites to extend and should appear similar to the control.
The exact procedure described herein is focused on neural support, however, with simple modifications to the material, electrospinning method, and proteins the system can be optimized for various cell and tissue types.
Protocol
1. Water/Oil/Water Double Emulsion Microsphere Production
- First prepare 2% and 0.5% w/v solutions of polyvinyl alcohol (PVA) in deionized water. Stir the solutions at 50 °C until clear, this may take several hours. Prepare a solution of 2% v/v Isopropyl Alcohol in deionized water.
- Prepare an aqueous solution of desired hydrophilic protein. The table below provides example formulations.

Table 1: Example Protein Solutions. The following protein solutions have been successfully encapsulated and electrospun using this protocol. Other hydrophilic protein solutions can be used as needed.
- Place 40 ml of the 0.5% PVA solution into a 50 ml centrifuge tube and set aside.
- In a 15 ml centrifuge tube dissolve 300 mg of 65:35 poly(lactic-co-glycolic acid) (PLGA) in 3 ml of Dichloromethane. A vortex mixer can be used to accelerate PLGA dissolution.
- Combine 200 µl of protein solution and 4 µl of 2% PVA solution. Pour the protein mixture into the PLGA solution (step 1.4). The solutions will remain mostly separate.
- Place the tube into a beaker of ice water. Using a wand sonicator at ~10 Watts (RMS), agitate the solution for a few seconds (5-10) until a uniform creamy white emulsion is created.
- Pour the emulsion into the 50 ml tube containing 0.5% PVA (step 1.3). Mix the solution at high speed on a vortex mixer for ~20 sec. The solution will develop a cloudy appearance.
- Transfer the emulsion to a 200 ml beaker and place on a stir plate at 350 rpm for 2 min. Add 50 ml of 2% isopropyl alcohol to the beaker on the stir plate. Allow the mixture to continue stirring for a minimum of 1 hr to allow the DCM to evaporate and the PLGA to harden.
- Transfer the microsphere solution into centrifuge tubes.
- Centrifuge at 425 x g for 3 min. The microspheres will collect at the bottom of the tube and appear white. Carefully remove the supernatant from the tube, above the microspheres, and store in a 500 ml bottle.
- Rinse the microspheres with deionized water by filling the tube three quarters full and shaking it to redistribute the microspheres in the liquid.
- Repeat steps 1.10 & 1.11 four times.
- Following the final rinse, remove the supernatant again and place in the 500 ml bottle with the other samples. Freeze the microspheres collected in the centrifuge tube, then lyophilize for at least 24 hr.
- Visualize the microspheres under a light microscope or with a Scanning Electron Microscope. Microspheres no larger than 60 µm are for effective electrospinning. If microspheres are too large, longer sonicating or vortexing times may be required in step 1.6 or 1.7.
- Store the dried microspheres in a -20 °C freezer.
- Optional: Use a protein assay, per manufacturer instructions, to test the amount of protein in the 500 ml bottle from step 1.1030. This is used to calculate the percent of protein encapsulated in the microspheres, by subtracting the amount in the solution from what was used in the production process.
Note: To visualize the protein location in the microsphere add Rhodamine 2 µg/ml to the PLGA solution31 and encapsulate a FITC conjugated protein. Figure 1 shows an example.
2. Electrospinning with Microspheres
- Prior to preparing electrospinning solution, create a 0.5% w/v solution of photoinitiator, in deionized water by dissolving at 37 °C. This process can take several hours.
- Create a 2% w/v methacrylated hyaluronic acid (MeHA) (see Burdick et al. for synthesis)29, 3% w/v 900 kD poly(ethylene oxide) (PEO) and 0.05% w/v photo initiator solution in deionized water.
- Calculate the correct amount of MeHA and PEO for the desired volume. For example, 10 ml of electrospinning solution requires 200 mg of MeHA and 300 mg of PEO.
- Dissolve the PEO in deionized water at 90% of the final volume desired (9 ml for this example). This may take several hours, a heated stir plate or water bath at 37 °C can be used to accelerate the process.
- Next add the MeHA and use a vortex mixer to stir the solution until clear. This will only take a few minutes.
- Finally add the 0.5% photo initiator solution to fill the remaining 10% volume (1 ml for this example).
- Add microspheres at the desired concentration up to 400 mg/ml. Mix the solution on a vortex mixer until the microspheres are evenly distributed in the solution.
- Transfer the solution to a syringe and attach a 6 inch 18 gauge blunt tip needle.
- Place the syringe in a syringe pump and set it to dispense at 1.2 ml/hr.
- Tape a layer of aluminum foil on the collection plate or mandrel. This allows for easy clean up and storage of the finished scaffold. A rotating mandrel is used to create aligned fibers. A flat plate or stationary mandrel will result in randomly arranged fibers.
- Connect the ground wire from a high voltage power source to the collection apparatus. Connect the positive lead to the needle.
- Adjust the syringe pump and collection surface so that there is 15 cm between the needle tip and collection surface.
- Start the polymer pumping, when the solution is visible at the end of the syringe, turn on the voltage source and set the voltage to 24 kV. CAUTION: Once the voltage is turned on do not touch any metal part of the system. Charge may also jump short distances from electrified parts to skin.
- Run the solution until desired scaffold thickness is achieved. When complete turn off voltage source and syringe pump.
- Remove foil with scaffold attached. Completed scaffolds containing protein are stored in a -20 °C freezer.
3. Protein Bioactivity Testing
- Prepare cell culture media. Add 10% v/v Fetal Bovine Serum, 1% v/v L-Glutamine, and 1% v/v Penicillin-Streptomycin to Dulbecco’s Modified Eagle’s Medium.
- Select glass coverslips that fit completely into a well plate.
- Use 3-(Trimethoxysilyl)propyl methacrylate to treat the coverslips as described by the manufacturer. Methacrylation enhances scaffold adherence to the coverslips.
- Attach methacrylated coverslips to the collection area of the electrospinner with removable double sided tape before electrospinning. Spinning onto the coverslips eases handling and viewing.
- Electrospin to desired thickness as described above.
- After electrospinning carefully remove coverslips from mandrel. Place the scaffold coated coverslips into a clear nitrogen chamber and ensure that all oxygen is purged.
- Place the chamber and scaffold under a 10 mW/cm2 365 nm light for 15 min. After crosslinking place into appropriately sized well plate. Ensure that the scaffold side is facing up.
- Place scaffolds under a germicidal lamp for 30 min to sterilize. If desired fibronectin or other protein is used as a coating to enhance cell adhesion. Follow manufacturer’s instructions to coat scaffolds.
- Harvest Dorsal Root Ganglia (DRG) as previously described by Hollenbeck32. One DRG will be needed for each scaffold covered coverslip tested.
- Place 100-200 µl of media on each scaffold in the well plate. Carefully place one DRG on each scaffold in the media droplet. For thick scaffold more media may be needed; DRG need to be fully submerged and not floating.
- Incubate the scaffold and DRG at 37 °C for 4 hr to allow the cell to adhere to the scaffold.
- Fill the media to the appropriate level for the well and place back into the incubator. Continue incubating for 4-6 days.
- After the incubation period carefully remove the media from each well and gently wash once with PBS. Fix cells for 30 min using 4% w/v paraformaldehyde.
- Stain cells using an antibody stain for neurofilament. This will allow visualizing of neurite outgrowth for quantification. DAPI can also be used to view nuclei of the cells. An example staining protocol was described by Sundararaghavan and colleagues14.
- Visualize the cells using a fluorescent microscope.
- Place well plate on the stage of the microscope.
- Locate the cell mass using the filter and excitation settings for DAPI.
- Once the cell is switch the filter to FITC to visualize the extended neurites. Using the stitch function on the microscope collect and combine as many images as necessary to see the entire structure. Repeat for DAPI, FITC and bright field.
Representative Results
Microspheres 50±14 µm in diameter with an over 85% protein encapsulation have been consistently produced and electrospun into scaffolds. Size was determined by imaging samples of microspheres from three separate production batches. The images where captured on an optical microscope and lengths where measured using commercial lab software. Figure 1 shows a histogram of the size distribution. Encapsulation rate was also tested from three separate microsphere batches, by quantifying the protein that escaped during the production process.
Figure 2 shows representative microspheres with Rhodamine (2 µg/ml) in the PLGA shell and bovine serum albumin (BSA)-FITC encapsulated within. The images were taken using a fluorescent microscope. The shell can be clearly seen (red) with the protein concentrated on the interior (green).
To evaluate encapsulation, the microspheres were filled with BSA and tested for protein release with a Bradford Protein Assay. PLGA breaks down by hydrolysis of ester bonds, creating larger gaps for protein escape. The microspheres continue releasing for over 60 days (Figure 3). The release begins with an initial burst then continues as microspheres break down.
After electrospinning the microspheres can be seen throughout the 3D fibrous structure. Figure 4 shows an SEM image of the complete scaffold where the spheres can be seen in multiple layers (indicated by arrows). Distribution of microspheres in the scaffolds was measured to be 15.3 ± 3.6 spheres/mm2. The scaffold holds the microspheres in place preventing migration away from the target site. As the microspheres break down they release NGF into the scaffold to encourage neurite growth33,34. This creates a continual delivery of protein throughout the scaffold to support the growth of cells and encourage cells to infiltrate into the scaffold.
Finally, Dorsal Root Ganglia (DRG) were used to test the viability of NGF in the microspheres, within the scaffold. Figure 5 shows a DRG seeded on a scaffold containing microspheres loaded with NGF next to one containing microspheres with no protein in them. There are longer neurite extensions visible extending from the DRG on the NGF scaffold, indicating that the NGF remained viable and is stimulating growth.
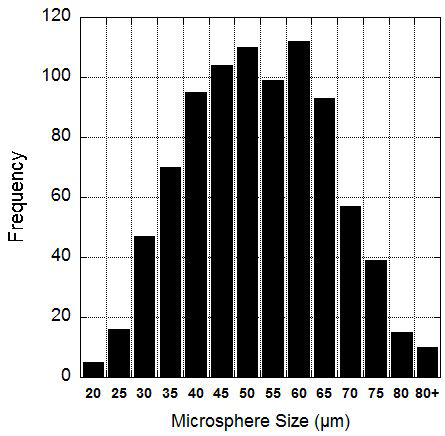
Figure 1. Microsphere size distribution. Microspheres produced by this protocol are 50±14 µm in diameter. The graph shows a histogram of the microsphere diameter in 5 µm bins.
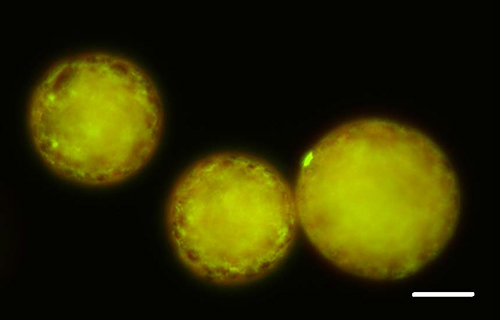
Figure 2. Fluorescent Image of Microspheres. 2 µg/ml Rhodamine was included in the PLGA solution to visualize the microsphere shell (red) and BSA-FITC was encapsulated to visualize the protein (green). Protein can clearly be seen within the sphere. Scale bar = 20 µm. Please click here to view a larger version of this figure.
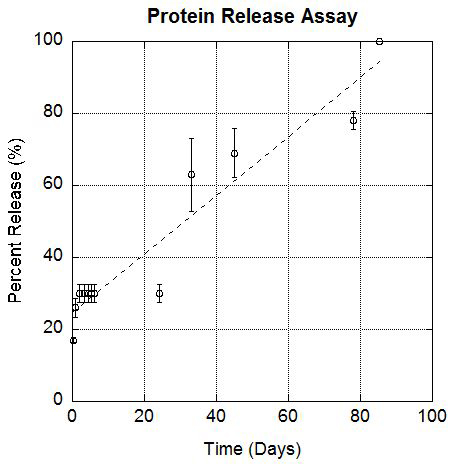
Figure 3. Protein Release Curve. BSA release from the microspheres over 60 days was measured using a Bradford Protein Assay. The initial burst is likely due to BSA that was attached to the outer surface. As the PLGA breaks down, protein is released over time.
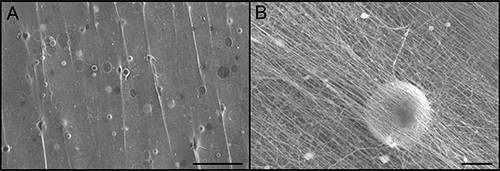
Figure 4. Scaffold containing Microspheres. The electrospinning process allows microspheres to be incorporated throughout the depth of the scaffold. (A) Arrows indicating the location of microspheres. Scale bar = 500 µm. (B) High magnification image of one microsphere with the nanofibers from the scaffold above it. Scale bar = 20 µm. Please click here to view a larger version of this figure.
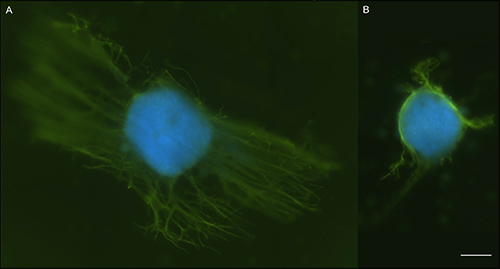
Figure 5. NGF Bioactivity Test. Dorsal Root Ganglia primary cells were harvested from 8 day old chick eggs and seeded on scaffolds with NGF filled microspheres (A) or empty microspheres (B). DRGs were stained with a neurofilament antibody, FITC secondary antibody and DAPI to visualize the cell nuclei. Images show increased neurite outgrowth in the presence of NGF loaded microspheres as indicated by FITC stained neurites (green). This shows released NGF is viable and can promote growth. Images were taken with a confocal microscope. Scale bar = 200 µm. Please click here to view a larger version of this figure.
Discussion
Many studies have shown that nerve cells can be directed by topographical cues (fiber alignment) and chemical cues (growth factors)1,2,10,11,35. Electrospinning is a facile method to create aligned fibers. Growth factors encourage nerve growth but in order to include them into nerve conduits (NC), a method for sustained release is required. To create a more robust system with both cues, these two signals should be combined. Several methods have been previously studied to provide extended release of protein within the electrospun fibrous scaffold for nerve regeneration but none to date have been capable of sustained release for over 60 days. Additionally, patterning of growth factor release has not been possible. Herein we describe a system to combine electrospinning with PLGA microsphere delivery to provide both signals.
Traditional methods to fabricate PLGA microspheres, such as the protocol outlined by de Boer et al., produced much larger microspheres33 (300±100 µm) that could not withstand the electrospinning process. Several attempts to incorporate them into the scaffolds were unsuccessful. This led to the use of a sonicator, in place of the vortex mixer, to better disperse the first emulsion and create smaller spheres. Once the microspheres were small enough, they easily passed through the needle and electrospun along with the rest of the solution. The primary electrospinning solution in this protocol is dissolved in water, rather than a harsh solvent, to prevent damage to the NGF and provide a hospitable substrate for cell growth28.
Many methods have been tested for providing topographical and chemical signals to nerve injury sites. Some of the early attempts at providing NGF to injuries involved putting solutions of free NGF or microspheres containing NGF into the center of a solid silicon nerve conduit (NC)36. These supported nerve growth; however they did not provide additional cues or allow the ability to pattern the location of the growth factors in the NC. In a more recent example Yan et. al. used poly[(lactic acid)-co-[(glycolic acid)-alt-(L-lysine)]] (PLGL) modified by grafting Gly-Arg-Gly-Asp-Gly (RGD peptide) and NGF to fabricate PLGL-RGD-NGF nerve conduits37. Similarly, these provided the chemical signal that promoted nerve growth, but did not provide other cues. Another study used NGF encapsulated in microspheres with electrospun scaffolds above and below them38. This system does provide both the topographical cues of a fibrous scaffold in combination with NGF as a chemical cue. However, the release only lasted a week, which is not long enough to support peripheral nerve repair, and the location of the microspheres is not well controlled.
Other researchers have focused on incorporating the chemical signals into the fibers. One method creates and emulsion of the hydrophilic protein with a hydrophobic polymer. This method was able to provide topographical and chemical cues, however 60% of the protein was released in the first 12 days39. Another example used coaxial electrospinning to encapsulate NGF within poly(lactic acid-caprolactone) (P(LLA-CL)) fibers40,41. This method provided results similar to the autograph in a rat sciatic nerve model. However, special equipment is need for coaxial electrospinning and it is unclear at what rate the protein was released, and if this could be sustained. The combination of topographical cues in the form of aligned electrospun fibers and chemical cues through microspheres that release for over 2 months as described here, provides multiple synergistic cues and is highly tunable to provide a customized environment.
There are several issues that need to be considered when electrospinning; environmental factors can affect electrospinning of any solution. An unexpected change in humidity, for instance, can have a significant effect on the electrospinning results and cause inconsistent fibers. Having a separate room that can be environmentally controlled is very helpful. A temperature of 70 °F with 50% humidity was used for these experiments. Another potential problem with an electrospinning system is short circuits; they often create obvious sparks between the syringe and metal pieces on the pump. To prevent this, place plastic or another insulator between them; food storage bag weight plastic works well. Similarly, stray electrical charges in the environment can cause material to accumulate in unexpected locations. To avoid this you can create an enclosure with acrylic sheets or other insulating material. More information on adjusting electrospinning conditions can be found in the review by Sill24.
The next steps for this procedure will include encapsulating other growth factors to create more dynamic scaffolds containing multiple growth factors. For example, NGF has been shown to work synergistically with Glial Cell Derived Growth Factor to support sensory and motor neurons3. Using PLGA microspheres, we can control the release rate by altering the L:G ratio, which would allow us to deliver various growth factors at different rate. A gradient of the microspheres can also be generated to build a more complex support system that directs cell behavior. Additionally, adhesion factors will be conjugated directly to the scaffold material through a Michael’s addition reaction26. Finally, the system will be tested with an in vivo rat sciatic nerve model.
Offenlegungen
The authors have nothing to disclose.
Acknowledgements
This work was partially funded through the Richard Barber Foundation and a Thomas Rumble Fellowship (TJW).
Materials
| DAPI | Invitrogen | D1306 | |
| Irgacure 2959 | BASF | 24650-42-8 | Protect from light |
| PEO 900 kDa | Sigma-Aldrich | 189456 | |
| Methacryloxethyl thiocarbamoyl rhodamine B | Polysciences, Inc. | 23591-100 | Prepare stock solution in DMSO |
| Syringe Pump | KD Scientific | KDS100 | |
| Power Source | Gamma High Voltage | ES30P-5W | |
| Motor | Triem Electric Motors, Inc | 0132022-15 | Must attach to a custom built mandrel |
| Tachometer | Network Tool Warehouse | ESI-330 | Use to monitor mandrel speed |
| Omnicure UV Spot Cure System with collimating adapter | EXFO | S1000 | |
| Needles | Fisher Scientific | 14-825-16H | |
| Coverslips | Fisher Scientific | 12-545-81 | |
| Polyvinyl Alcohol | Sigma-Aldrich | P8136-250G | |
| Isoporopyl Alcohol | Sigma-Aldrich | I9030-500mL | |
| Bovine Serum Albumin (BSA) | Fisher Scientific | BP9703-100 | |
| BSA-FITC | Sigma-Aldrich | 080M7400 | |
| β-Nerve Growth Factor (NGF) | R&D Systems | 1156-NG | |
| 65:35 Poly-Lactic-Glycolic-Acid (PLGA) | Sigma-Aldrich | 1001554270 | |
| Dichloromethane | Sigma-Aldrich | 34856-2L | |
| Coomassie (Bradford) Protein Assay | Thermo Scientific | 1856209 | |
| 3-(Trimethoxysilyl)propyl methacrylate | Sigma-Aldrich | 1001558456 | |
| Fibronectin | Sigma-Aldrich | F2006 | |
| DMEM | Lonza | 12-604F | |
| FBS | Atlanta Biologicals | S11150 | |
| PBS | Hyclone | SH30256.01 | |
| Glutamine | Fisher Scientific | G7513 | |
| Pen-Strep | Sigma-Aldrich | P4333 | |
| Paraformaldehyde | Alfa Aesar | A11313 |
Referenzen
- Wrobel, M. R., Sundararaghavan, H. G. Directed migration in neural tissue engineering. Tissue Eng Part B Rev. , (2013).
- Schmidt, C. E., Leach, J. B. Neural tissue engineering: strategies for repair and regeneration. Annual Review of Biomedical Engineering. 5, 293-347 (2003).
- Madduri, S., di Summa, P., Papaloizos, M., Kalbermatten, D., Gander, B. Effect of controlled co-delivery of synergistic neurotrophic factors on early nerve regeneration in rats. Biomaterials. 31, 8402-8409 (2010).
- Madduri, S., Gander, B. Growth factor delivery systems and repair strategies for damaged peripheral nerves. J Control Release. 161, 274-282 (2012).
- Madigan, N. N., McMahon, S., O’Brien, T., Yaszemski, M. J., Windebank, A. J. Current tissue engineering and novel therapeutic approaches to axonal regeneration following spinal cord injury using polymer scaffolds. Respir Physiol Neurobiol. 169, 183-199 (2009).
- Sundararaghavan, H. G., Monteiro, G. A., Firestein, B. L., Shreiber, D. I. Neurite growth in 3D collagen gels with gradients of mechanical properties. Biotechnol. Bioeng. 102, 632-643 (2009).
- Hudson, T. W., Evans, G. R., Schmidt, C. E. Engineering strategies for peripheral nerve repair. Clin Plast Surg. 26, 617-628 (1999).
- Kokai, L. E., Bourbeau, D., Weber, D., McAtee, J., Marra, K. G. Sustained growth factor delivery promotes axonal regeneration in long gap peripheral nerve repair. Tissue Eng Part A. 17, 1263-1275 (2011).
- Bronzino, J. D., Peterson, D. R. . The Biomedical Engineering Handbook, Third Edition – 3 Volume Set: Tissue Engineering and Artificial Organs. , (2006).
- Bell, J. H. A., Haycock, J. W. Next generation nerve guides: materials, fabrication, growth factors, and cell delivery. Tissue Eng Part B Rev. 18, 116-128 (2012).
- Ruiter, G. C. W., Malessy, M. J. A., Yaszemski, M. J., Windebank, A. J., Spinner, R. J. Designing ideal conduits for peripheral nerve repair. Neurosurgical focus. 26, (2009).
- Olakowska, E., Woszczycka-Korczyńska, I., Jędrzejowska-Szypułka, H., Lewin-Kowalik, J. Application of nanotubes and nanofibres in nerve repair. A review. Folia Neuropathol. 48, 231-237 (2010).
- Gunn, J., Zhang, M. Polyblend nanofibers for biomedical applications: perspectives and challenges. Trends Biotechnol. 28, 189-197 (2010).
- Sundararaghavan, H. G., Masand, S. N., Shreiber, D. I. Microfluidic generation of haptotactic gradients through 3D collagen gels for enhanced neurite growth. Journal of Neurotrauma. 28, 2377-2387 (2011).
- Sundararaghavan, H. G., Metter, R. B., Burdick, J. A. Electrospun fibrous scaffolds with multiscale and photopatterned porosity. Macromol Biosci. 10, 265-270 (2010).
- Edalat, F., Sheu, I., Manoucheri, S., Khademhosseini, A. Material strategies for creating artificial cell-instructive niches. Current Opinion in Biotechnology. 23, 820-825 (2012).
- Annabi, N., et al. Synthesis of highly porous crosslinked elastin hydrogels and their interaction with fibroblasts in vitro. Biomaterials. 30, 4550-4557 (2009).
- Castaño, O., Eltohamy, M., Kim, H. -. W. Electrospinning technology in tissue regeneration. Methods Mol. Biol. 811, 127-140 (2012).
- Chew, S. Y., Wen, J., Yim, E. K. F., Leong, K. W. Sustained release of proteins from electrospun biodegradable fibers. Biomacromolecules. 6, 2017-2024 (2005).
- Han, D., Gouma, P. -. I. Electrospun bioscaffolds that mimic the topology of extracellular matrix. Nanomedicine. 2, 37-41 (2006).
- Prabhakaran, M. P., et al. Electrospun biocomposite nanofibrous scaffolds for neural tissue engineering. Tissue Eng Part A. 14, 1787-1797 (2008).
- Xie, J., MacEwan, M. R., Schwartz, A. G., Xia, Y. Electrospun nanofibers for neural tissue engineering. Nanoscale. 2, 35-44 (2010).
- Yao, L., O’Brien, N., Windebank, A., Pandit, A. Orienting neurite growth in electrospun fibrous neural conduits. J. Biomed. Mater. Res. Part B Appl. Biomater. 90, 483-491 (2009).
- Sill, T. J., von Recum, H. A. Electrospinning: applications in drug delivery and tissue engineering. Biomaterials. 29, 1989-2006 (2008).
- Ifkovits, J. L., Sundararaghavan, H. G., Burdick, J. A. Electrospinning fibrous polymer scaffolds for tissue engineering and cell culture. Journal of Visualized Experiments: JoVE. , (2009).
- Sundararaghavan, H. G., Burdick, J. A. Gradients with depth in electrospun fibrous scaffolds for directed cell behavior. Biomacromolecules. 12, 2344-2350 (2011).
- Meinel, A. J., Germershaus, O., Luhmann, T., Merkle, H. P., Meinel, L. Electrospun matrices for localized drug delivery: current technologies and selected biomedical applications. Eur J Pharm Biopharm. 81, 1-13 (2012).
- Ji, W., et al. Bioactive electrospun scaffolds delivering growth factors and genes for tissue engineering applications. Pharm. Res. 28, 1259-1272 (2011).
- Burdick, J. A., Chung, C., Jia, X., Randolph, M. A., Langer, R. Controlled degradation and mechanical behavior of photopolymerized hyaluronic acid networks. Biomacromolecules. 6, 386-391 (2005).
- Péan, J. M., et al. Optimization of HSA and NGF encapsulation yields in PLGA microparticles. International Journal of Pharmaceutics. 166, 105-115 (1998).
- Cartiera, M. S., Johnson, K. M., Rajendran, V., Caplan, M. J., Saltzman, W. M. The Uptake and Intracellular Fate of PLGA Nanoparticles in Epithelial Cells. Biomaterials. 30, 2790-2798 (2009).
- Hollenbeck, P. J., Bamburg, J. R. . Neurons: Methods and Applications for the Cell Biologist. , (2003).
- Boer, R., et al. Rat sciatic nerve repair with a poly-lactic-co-glycolic acid scaffold and nerve growth factor releasing microspheres. Microsurgery. 31, 293-302 (2011).
- Pujic, Z., Goodhill, G. J. A dual compartment diffusion chamber for studying axonal chemotaxis in 3D collagen. Journal of Neuroscience Methods. 215, 53-59 (2013).
- Madduri, S., di Summa, P., Papaloïzos, M., Kalbermatten, D., Gander, B. Effect of controlled co-delivery of synergistic neurotrophic factors on early nerve regeneration in rats. Biomaterials. 31, 8402-8409 (2010).
- Xu, X., et al. Polyphosphoester microspheres for sustained release of biologically active nerve growth factor. Biomaterials. 23, 3765-3772 (2002).
- Yan, Q., Yin, Y., Li, B. Use new PLGL-RGD-NGF nerve conduits for promoting peripheral nerve regeneration. Biomed Eng Online. 11, (2012).
- Gungor-Ozkerim, P. S., Balkan, T., Kose, G. T., Sarac, A. S., Kok, F. N. Incorporation of growth factor loaded microspheres into polymeric electrospun nanofibers for tissue engineering applications. J Biomed Mater Res A. , (2013).
- Li, X., et al. Encapsulation of proteins in poly(L-lactide-co-caprolactone) fibers by emulsion electrospinning. Colloids Surf B Biointerfaces. 75, 418-424 (2010).
- Wang, C. -. Y., et al. The effect of aligned core-shell nanofibres delivering NGF on the promotion of sciatic nerve regeneration. J Biomater Sci Polym Ed. 23, 167-184 (2012).
- Liu, J. -. J., Wang, C. -. Y., Wang, J. -. G., Ruan, H. -. J., Fan, C. -. Y. Peripheral nerve regeneration using composite poly(lactic acid-caprolactone)/nerve growth factor conduits prepared by coaxial electrospinning. J Biomed Mater Res A. 96, 13-20 (2011).

