In ovo Expression of MicroRNA in Ventral Chick Midbrain
Summary
Ectopic expression is one technique to elucidate the microRNAs role in brain development. However, targeting specific areas using in ovo electroporation is challenging. Here, we show an efficient way to selectively electroporate ventral and dorsal midbrain regions.
Abstract
Non-coding RNAs are additional players in regulating gene expression. Targeted in ovo electroporation of specific areas provides a unique tool for spatial and temporal control of ectopic microRNA expression. However, ventral brain structures like ventral midbrain are rather difficult to reach for any manipulations. Here, we demonstrate an efficient way to electroporate miRNA into ventral midbrain using thin platinum electrodes. This method offers a reliable way to transfect specific areas of the midbrain and a useful tool for in vivo studies.
Introduction
The recognition of small non-coding RNAs as additional players for gene expression launched a new complexity to genomic programming/gene regulation. Different species of non-coding RNAs have functional importance in neural cells, including small non-coding RNAs1-4. MicroRNAs (miR or miRNA) for example show distinct and changing expression profiles in developing brains5. Targeted in ovo electroporation of chick embryos provides a unique opportunity for temporal and spatial control of gene expression and silencing during development.
This video demonstrates the different steps of performing ectopic expression of miRs in specific areas of the chick midbrain using in ovo electroporation6-10. To ensure a long lasting effect of these small non-coding RNAs in cells, the DNA sequence of miRs were cloned into mono- or bi-cistronic vectors. For in ovo electroporation, miR containing vector is injected into the midbrain neural tube by exposing the embryo after making a small window in the egg shell. To transfect specific areas of the midbrain small plus (anode) and minus (cathode) platinum electrodes are placed at specific positions. For ventral midbrain transfection, the anode is placed underneath the left ventral midbrain and the cathode above the right half of the midbrain before applying a current. The opening in the eggshell is closed with tape and embryos are incubated for as long as required for any analysis. This method was originally described by Muramatsu et al.6 and improved by Momose et al.8 for specific area transfection.
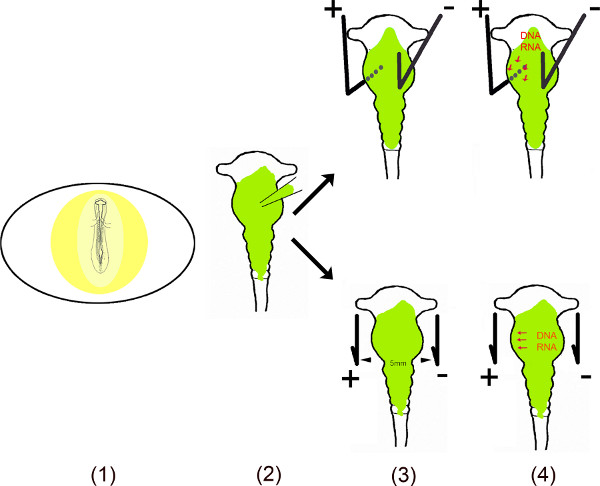
Schematic Overview.
- The embryo in the egg is exposed by cutting a small window into the eggshell.
- The dissolved vector(s) is injected into the midbrain using a micro capillary.
- Two electrodes – placed parallel or under and above the embryo – generate a pulsed electric field.
- The electric field temporally creates pores in the cell membrane, which facilitate entry into the cell by the negatively charged DNA (or RNA) attracted to the anode11,12.
Protocol
1. Requirements for In ovo Electroporation
- Preparing the vector construct: miR sense and antisense primers were designed after the instructions of Ambion, annealed and ligated into ApaI/EcoRI digested pSilencer U6.1 vector. After successful transformation one of the resulting clones was grown and pSilencer plasmid was isolated by alkaline lysis method using a Quiagen midi preparation kit.
- Electrodes: Electrodes can be purchased or easily be built13. We use self-made electrodes of platinum wire (Φ = 0.2, 0.25, or 0.5 mm in diameter) or in some cases an even smaller tungsten wire electrode (Φ = 0.1 mm)8 depending on which area we want to electroporate. To avoid scorching of tissue, we use as a rule the nearer to the tissue the thinner the electrodes. For broad electroporation of midbrain, we use 0.5 mm platinum wires soldered to a brass/stainless steel tubular shaft and fixed to an electrode holder with a distance of 3-5 mm (Figure 1). To electroporate specific areas, we use 0.25 mm platinum wire anodes and 0.2 mm platinum wire or 0.1 mm tungsten for the cathode with an approximate distance of 1 mm or less.
- Solutions: Have the following solutions ready:
- Autoclaved chicken Ringer, pH 7.5 (9.0 g NaCl, 0.42 g KCl, 0.24 g CaCl2, dissolve in 1 L distilled water).
- PBS (8 g NaCl, 0.2 g KCl, 0.24 g KH2PO4, 1.44 g Na2HPO4, dissolve in 1l distilled H2O).
- Fast Green: stock solution 10 mg/ml distilled H2O
- Slow drying ink (less shellac and thus less poisoning for the embryo). Note: A blue dichroic filter attached to a fiber optic lamp as described in Canto-Soler and Adler14 enhances the contrast and can eliminate the need to inject ink.
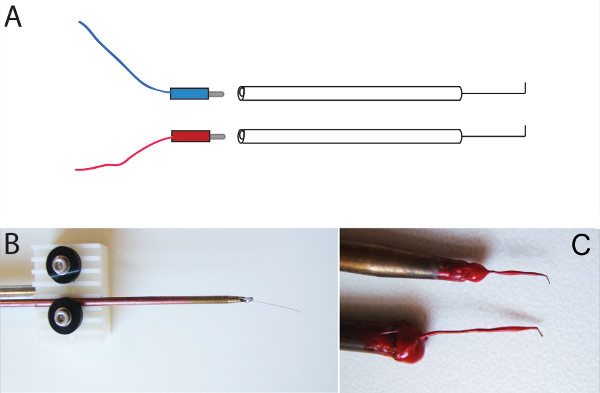
Figure 1. Schematic diagram of electrodes and electrode holder. (A, B, C) Platinum wire was soldered to one side of a stainless steel tubular shaft. A hole was drilled into the other side of the tubular shaft thus that pin plugs can fit in. (A) Red and blue colored wires were connected to the pin plugs. At the opposite end the wire was fitted to banana connectors that work with the electrical sockets of the electroporator. (A, C) The platinum wire was bent in an angel of 90°. A layer of nail varnish isolated the shaft above the bend.
2. Preparations Directly Before In ovo Electroporation
- Prepare the following material:
- Dilute Ink with chicken Ringer 1:6, add Antibiotic-Antimycotic solution (100x from Gibco) 1:100.
- Prepare vector solution: 1-2 μg/μl per specific DNA in H2O supplemented with Fast Green (1:20).
- Sterilize forceps, scissors, tungsten needle and electrodes with 70% ethanol.
- Pull micropipettes for nucleic acid injection from borosilicate glass capillaries (length to outer diameter: 100 mm x 0.9 mm). We use a simple puller PUL-1 (WPI, Berlin) with the settings shown in Table 1.
- Set the parameters of the electroporator as shown in Table 2.
| Parameters | Values |
| Heat | 350 |
| Pull | 100 |
| Velocity | 50 |
| Delay | 200 |
Table 1. Settings for PUL-1
| Parameters | Values |
| Frequency | 50 Hz |
| Delay | 20 – 50 msec* |
| Ancho | 20 msec |
| Voltage | 8-25 V * |
| Pulse | 3 – 5 times |
| *Depending on electrode diameter and distance. Please take that into account when changing one of the parameters. | |
Table 2. Settings for the Pulse Stimulator
- Prepare chick embryos for electroporation:
- Incubate fertilized chicken eggs on their longer side at 37 °C with 65% humidity for 39 hr to acquire embryos between Hamburger & Hamilton (HH) stages15 9-11. The embryo settles at the topmost part of the egg, so you should mark it with a pencil line.
- Disinfect the eggs with 70% ethanol.
- Pierce the egg at its broader side, where the air cavity should be.
- Remove 1-2 ml of egg white. This will detach the embryo from the eggshell.
- Cello-tape the eggshell to avoid shell splinters falling onto the embryo. Use a pair of scissors to cut a small window into the eggshell around the pencil line.
- Inject a bit of ink underneath the area opaca to visualize the embryo.
- Make a cut into the vitelline membrane with a sharpened tungsten needle to achieve a better accessibility of the embryo for nucleic acid injection and electroporation.
3. Electroporation and Incubation
- Add a few drops of warm Ringer’s solution onto the embryos. This will lower the embryos, prevent overheating, sticking of the electrodes to the tissue and provide a medium for the current between the two electrodes.
- Inject the vector solution into the lumen of the neural tube at midbrain level. If your DNA expression vector contains no reporter gene (e.g. EGFP), add a slightly lower concentrated reporter gene vector (1 μg/μl miR expressing vector to 0.9 μg/μl reporter gene vector) to the DNA solution. This will ensure that you recognize transfected cells later8. We use the so-called pCAX vector, a kind gift from C. Krull 13.
- For a broad transfection of the midbrain, place the electrodes left and right of the embryo at the height of the midbrain. The distance between the electrodes should be around 3-5 mm. The other settings are 15V, 20 msec pulses and 50 msec delay for HH 10. Changing the position of the two electrodes slightly along the dorso-ventral (DV) axis of the midbrain assures a more a broad transfection of cells the DV axis transfection.
- For ventral transfection, add some Ringer to raise the head from the yolk membrane and place the anode beneath ventral midbrain. If the head does not rise push the electrode under the membrane that separates yolk and embryo to avoid scorching of the neural tube. The cathode should be placed above the midbrain dorso-laterally opposite to the anode. Do not touch the neural tube. We transfect the left ventral half of the midbrain to avoid any difficulties with heart development. The distance between the electrodes is around 1-0.5 mm, the rest of the settings are 10V, 20 msec width and 50 msec delay.
- Apply another drop of Saline on top of the embryos to cool it down and remove air bubbles.
- Seal the eggs with the tape and incubate for at least 4 hr; the minimum time required for vector expression.
4. Fixation of Chicken Embryos
- Remove the embryos from their eggs.
- Clean embryos under a stereomicroscope and assess the extent of transfection with a fluorescent stereo microscope.
- Fix the embryos over night in 4% PFA at 4°C. Proceed with sectioning and/or staining (in situ hybridization, immunohistochemistry) to analyse changes in the transfected area.
Representative Results
The extent of the midbrain area transfected with miR is visible through the expression of GFP from a reporter vector injected along with the miR expressing vector (Figure 2). Using platinum electrodes with a diameter of 0.5 mm and placed parallel to the AP axis of midbrain leads normally to the transfection of a broad area along the DV- and AP- axis, including hindbrain, midbrain and sometimes diencephalon (Figures 2A and B). The size of the electrodes results in a wide electric field applied to the brain region and thus a wide transfected area.
It is difficult to accurately target an area like for example the ventral midbrain. Reducing the distance between the 0.5 mm electrodes to achieve a smaller electric field is often toxic for the embryo and leads to artifacts. We used electrodes with smaller diameters (0.2 mm, 0.25 mm platinum or 0.1 mm sharpened tungsten wire), which offer more localized transfection like that of the ventral brain, which is difficult to reach (Figures 2C-I). Smaller electrodes reduce the electric field applied and enable to insert the electrodes much closer to the tissue. Figure 2F shows an example, where the entire ventral midbrain had been transfected. In Figures 2G and 2H the transfected ventral areas are smaller, but part of the hindbrain (2F) or diencephalon (2H) are also transfected. In Figure 2G, the anode was placed under the mid- and hindbrain to transfect both ventral areas. Figure 2H shows an example of an anode not well isolated with nail varnish along the shaft. Thus, not only the ventral area around the MHB expresses miR/GFP but also the dorsal anterior mes- and di-encephalon, where the shaft of the anode had been placed. The specific ventral miR/GFP distribution in Figures 2C-I is a result from the local electric field we apply with the smaller electrodes. Taken together, with smaller electrodes and electric fields we can express DNA/RNA in more restricted areas like ventral midbrain.
Since our microRNA expression vector contains no reporter gene, we mix in a GFP expressing vector in a ratio of 1 μg/μl to 0.9 μg/μl, respectively. The almost equal ratio ensures that the microRNA and GFP transfected area almost completely overlap (see Momose et al.8 and own experience). To be able to judge the extent of the transfected area becomes important when only a small area containing specific cell types is transfected or the transfected cells are used for qRT-PCR. A much less concentration of the reporter gene might lead to cells expressing GFP under the detection level. The strength of expression of any transfected vector depends not only on the concentration of the used vector but also on the promoter. Our vectors are U6-promoter driven (expressing miRNA) and ß-actin promoter driven (expressing fluorescent proteins) and are both ubiquitously and constitutively active in the chick embryo.
Most of our embryos are electroporated at the left side except for Figure 2C (mirror image). For time-lapse experiments, electroporation of the right side of the embryo is recommended since most embryos turn their head towards the right and lie on their left side.
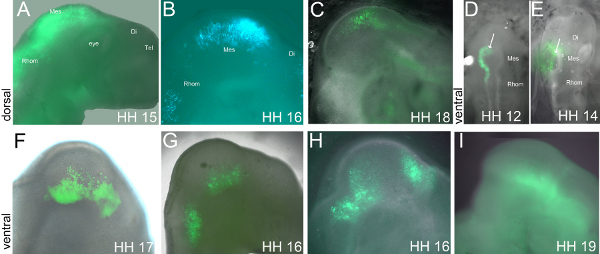
Figure 2. Representative results. Embryos were electroporated between HH10 and HH11 and fixed at the stages indicated in the pictures. (A-C) show electroporations of dorsal midbrain, and (D-I) of ventral midbrain. The arrows in (D,E) indicate ventrally located, GFP positive cells. For more details see text. Mes – mesencephalon/midbrain, Di – diencephalon, Tel – telencephalon, Rhom – rhombencephalon/hindbrain.
Discussion
This video demonstrates an effective method to transfect plasmid into the neuroepithelial cells of specific areas of the chick midbrain. Rectangular electric pulses of low voltage can introduce DNA into cells of the chick neural tube in ovo6,16. However, the accuracy of DNA targeting is often hindered by the wide electric field, which rises through the relatively large electrodes (Φ = 0.5 mm). We tried to tackle that problem by using electrodes smaller in diameter following the guidelines of Momose et al.8. For ventral midbrain transfection, we place the cathode under the membrane of the area pellucida or if the age of the embryo allows it directly below the ventral midbrain. The last scenario needs a bit of practice since midbrain/neural tube tissue should not be touched and damaged by the anode. Adding Ringer can often help to lift the head of the embryo slightly off the membrane covering the area pellucida.
We hold both electrodes with our hands. However, that requires calm and steady hands and a bit of practice. Since the position of the cathode is above the embryo, this electrode can be fixed to a micromanipulator. That leaves only one electrode to be put into position manually. Electrodes too close to the embryo/neural tube will literally scorch the embryo and lead to unwelcome anatomical malformation. Hence, the strength of the voltage should be carefully adjusted to each series of experiments and care should be taken when placing the electrodes. These precautions will prevent any morphological, cellular or molecular malformation caused by the procedure itself.
Although to consider are gene concentrations in the cell. Different gene doses might have different effects on cells and tissue. There could be a threshold effect involved (see Agoston et al.17, for example) or different, graded effects. We normally test different concentrations of the miR expressing (0.5-2 μg/μl) vector and analyse changes in morphology or protein/gene-expression. Effects can be tested in vivo using immunohistochemistry, sensor vectors18 or RNA in situ hybridization.
This technique also offers to test strength and location of any activity of a specific miR in vivo using for example a dual fluorescence reporter/sensor vector as described in De Pietri Tonelli et al.18. They use a vector that contains a GFP-reporter and a RFP miR sensor. The RFP is followed by a complimentary miR sequence. While GFP expression identifies the successfully transfected cells, red fluorescence (RFP) indicates the absence of the investigated miR, and disappearance of red fluorescence betrays the presence of the miR. This technique can also be used to test the specificity of miR target in vivo, by using 3’UTR of the target sequence in the downstream of RFP. Taken together this technique is highly appropriate for developmental studies on gene expression and function in different tissues.
Divulgaciones
The authors have nothing to disclose.
Acknowledgements
We acknowledge K. Mikic, who contributed to the initial phase of this movie and M. Nicolescu for the miR picture. C. Huber was supported by a fellowship of the IZKF of the Universtitätsklinikum Tübingen, A. Alwin Prem Anand by the Fortüne programme of the Universtitätsklinikum Tübingen.
Materials
| Name | Company | Model | |
| Borosillicate glass capillaries | Hartenstein | Model: 0.9 mm | |
| Microcapillary puller | WPI, Berlin | Model: Pul1-E | |
| Electroporator | Intracel | Model: TSSIC | |
| Stereomicroscope – fluorescence | LEICA | Model: MZFLIII | |
| Stereomicroscope | Zeiss | Model: Stemi | |
| Camera and software | Zeiss | Model: Axiocam MRc/ Axiovision Re. 4.8 |
Referencias
- Kutter, C., Svoboda, P. miRNA, siRNA, piRNA: Knowns of the unknown. RNA Biol. 5, 181-188 (2008).
- Szell, M., Bata-Csorgo, Z., Kemeny, L. The enigmatic world of mRNA-like ncRNAs: their role in human evolution and in human diseases. Semin. Cancer Biol. 18, 141-148 (2008).
- Li, X., Jin, P. Roles of small regulatory RNAs in determining neuronal identity. Nature reviews. Neuroscience. 11, 329-338 (2010).
- Riedmann, L. T., Schwentner, R. miRNA, siRNA, piRNA and argonautes: news in small matters. RNA Biol. 7, 133-139 (2010).
- Coolen, M., Bally-Cuif, L. MicroRNAs in brain development and physiology. Curr. Opin. Neurobiol. 19, 461-470 (2009).
- Muramatsu, T., Mizutani, Y., Ohmori, Y., Okumura, J. Comparison of three nonviral transfection methods for foreign gene expression in early chicken embryos in ovo. Biochem. Biophys. Res. Commun. 230, 376-380 (1997).
- Itasaki, N., Bel-Vialar, S., Krumlauf, R. Shocking’ developments in chick embryology: electroporation and in ovo gene expression. Nat. Cell Biol. 1, 203-207 (1999).
- Momose, T., et al. Efficient targeting of gene expression in chick embryos by microelectroporation. Dev. Growth Differ. 41, 335-344 (1999).
- Nakamura, H., Watanabe, Y. Misexpression of genes in brain vesicles by in ovo electroporation. Dev. Growth Differ. 42, 199-201 (2000).
- Voiculescu, O., Papanayotou, C., Stern, C. D. Spatially and temporally controlled electroporation of early chick embryos. Nat. Protoc. 3, 419-426 (2008).
- Neumann, E., Schaefer-Ridder, M., Wang, Y., Gene Hofschneider, P. H. transfer into mouse lyoma cells by electroporation in high electric fields. The EMBO journal. 1, 841-845 (1982).
- Potter, H., Weir, L., Leder, P. Enhancer-dependent expression of human kappa immunoglobulin genes introduced into mouse pre-B lymphocytes by electroporation. Proceedings of the National Academy of Sciences of the United States of America. 81, 7161-7165 (1984).
- Krull, C. E. A primer on using in ovo electroporation to analyze gene function. Dev. Dyn. 229, 433-439 (2004).
- Canto-Soler, M. V., Adler, R. Optic cup and lens development requires Pax6 expression in the early optic vesicle during a narrow time window. Developmental biology. , 294-2119 (2006).
- Hamburger, V., Hamilton, H. L. A series of normal stages in the development of the chick embryo. 1951. Dev Dyn. 195, 231-272 (1992).
- Muramatsu, T., Nakamura, A., Park, H. M. In vivo electroporation: a powerful and convenient means of nonviral gene transfer to tissues of living animals (Review). Int J Mol Med. 1, 55-62 (1998).
- Agoston, Z., Li, N., Haslinger, A., Wizenmann, A., Schulte, D. Genetic and physical interaction of Meis2, Pax3 and Pax7 during dorsal midbrain development. BMC Dev Biol. 12, 10 (2012).
- De Pietri Tonelli, D., et al. Single-cell detection of microRNAs in developing vertebrate embryos after acute administration of a dual-fluorescence reporter/sensor plasmid. Biotechniques. 41, 727-732 (2006).

