Isolation of Small Noncoding RNAs from Human Serum
Summary
This protocol describes a method for extracting small RNAs from human serum. We have used this method to isolate microRNAs from cancer serum for use in DNA arrays and also singleplex quantitative PCR. The protocol utilizes phenol and guanidinium thiocyanate reagents with modifications to yield high quality RNA.
Abstract
The analysis of RNA and its expression is a common feature in many laboratories. Of significance is the emergence of small RNAs like microRNAs, which are found in mammalian cells. These small RNAs are potent gene regulators controlling vital pathways such as growth, development and death and much interest has been directed at their expression in bodily fluids. This is due to their dysregulation in human diseases such as cancer and their potential application as serum biomarkers. However, the analysis of miRNA expression in serum may be problematic. In most cases the amount of serum is limiting and serum contains low amounts of total RNA, of which small RNAs only constitute 0.4-0.5%1. Thus the isolation of sufficient amounts of quality RNA from serum is a major challenge to researchers today. In this technical paper, we demonstrate a method which uses only 400 µl of human serum to obtain sufficient RNA for either DNA arrays or qPCR analysis. The advantages of this method are its simplicity and ability to yield high quality RNA. It requires no specialized columns for purification of small RNAs and utilizes general reagents and hardware found in common laboratories. Our method utilizes a Phase Lock Gel to eliminate phenol contamination while at the same time yielding high quality RNA. We also introduce an additional step to further remove all contaminants during the isolation step. This protocol is very effective in isolating yields of total RNA of up to 100 ng/µl from serum but can also be adapted for other biological tissues.
Introduction
In recent years, there has been a growing push to discover novel biomarkers for the early detection of human diseases. Much attention has been focused on using small RNAs such as microRNAs2 (miRNAs or miRs) as potential markers. These small RNAs are found in body fluids such as serum and studies have shown they are resilient to degradation and are stable over a range of varying environmental conditions3. Given these features, serum or circulating miRNAs are the ideal biomarker4,5. Currently there are two main approaches for the isolation of small RNA from biological fluids. The first approach uses column-based technology to bind and elute the small RNAs6, while the second approach uses the long-standing protocol with phenol and guanidinium thiocyanate reagents7. We have developed a simple, effective, column-free protocol to isolate small RNAs from human serum. The isolated RNA is immediately usable in downstream applications, including DNA oligonucleotide arrays and RNA sequencing.
This protocol was developed because we were confronted with several issues when using phenol-based methods to isolate RNA from serum. The traditional Chomczynski approach is frequently used in most laboratories with a range of reagents available from most commercial vendors. However considering their widespread use, stringent guidelines have not been developed to consistently produce high quality RNA from bodily fluids, in particular blood or serum.
Common problems associated with isolating RNA from serum include low RNA yields and contamination with reagents used during the isolation, particularly phenol. Our approach eliminates these phenol contaminants to provide high quality RNA for downstream analysis such as quantitative PCR (qPCR) and RNA sequencing. We have further tested this RNA on miRNA arrays.
Protocol
Note: Human serum samples from healthy patients or patients with cancer were obtained with informed consent under approved human ethical protocols from the Royal Prince Alfred Hospital Sydney (Protocol number X10-0016 and HREC/10/RPAH/24) and the University of Technology, Sydney. Serum samples were collected from patients before surgery from various Sydney hospitals and placed in storage at -80 °C.
1. Small RNA Isolation from Serum
Total RNA was prepared from human serum using a modified version of the Tri-Reagent RT LS protocol.
- Thaw frozen serum sample on ice and then transfer 400 µl of the freshly thawed serum into a labeled microcentrifuge tube.
- Dilute the serum with 100 µl of RNase free H2O and add proteinase K at a concentration of 1 mg/ml.
- Incubate at 37 °C for 20 min to allow protein digestion by proteinase K.
- To ensure complete solubilization, add 1.5 times its volume of Tri-Reagent RT LS and 100 µl of 4-bromoanisole.
- Briefly invert the homogenate, perform repetitive pipetting for 5 sec and transfer into a labeled 2 ml Heavy Phase Lock tube.
- Spin the homogenate at 12,000 × g for 20 min at 4 °C.
- Carefully decant at least 1 ml of the resulting aqueous solution into a fresh DNA Lobind tube. The organic and interphase should be trapped underneath the white gel of the Phase Lock tube.
- Add 5.0 µl of glycogen (5 mg/ml) and 500 µl of 100% isopropanol to the aqueous solution, mix by inversion, and incubate O/N at -20 °C.
- Following O/N incubation, centrifuge the sample for 20 min at 12,000 × g in a 4 °C centrifuge.
- Discard the clear supernatant and perform a "flash" spin for 2 min at 16,000 × g in a 4 °C centrifuge.
- Carefully remove the clear solution surrounding the pellet using a pipette.
- Wash the pellet with 1 ml of 70% ethanol and centrifuge at 10,000 × g for 10 min. Decant the wash solution and repeat wash step.
2. RNA Resuspension into Solution
- Resuspend the pellet in 10 µl of RNase free H2O, ensuring the pellet is completely dissolved. To ensure that the RNA is completely solubilized the sample can be heated to 55 °C for 5 min. During this time, mix the sample with repeated pipetting. For a higher total RNA yield, pool two RNA preparations from the same patient.
- Quantitate the resuspended RNA using a UV-Vis spectrophotometer and assess the RNA quality using a 2100 Bioanalyzer. Store the pooled RNA samples at -80 °C for use in downstream applications.
Representative Results
Figure 1 represents a typical UV/Vis spectrum of RNA isolated from serum. From this profile we noted protein contamination at 280 nm with phenol and organic contaminants both at 270 nm and 230 nm, respectively. Residue guanidinium thiocyanate was also noted at 260 nm. To reduce contaminants, a series of optimization steps were made to the standard Tri-Reagent RT-LS procedure. We added 5 mg/ml of glycogen to increase both the total RNA yield and reduce contamination (black line, Figure 1A).
Further we observed that after the removal of isopropanol there was roughly 100-300 µl of wash buffer surrounding the RNA pellet. An additional centrifuge spin was added to the protocol and the residue wash solution was carefully removed. This resulted in a profile shift from 270 nm to 280 nm (red trace, Figure 1B). We deduced that this "flash" spin had reduced the phenol contamination at 270 nm and concluded that the 280 nm peak most likely represented protein contamination.
To further increase RNA yield and reduce protein carry through we added a Phase Lock Gel step. This step allowed for the complete transfer of the aqueous phase without organic contaminants (Figure 1B). As serum contains a large abundance of protein, we evaluated if diluting the original serum volume would make any difference (Figure 1C). In a total extract volume of 500 µl, we mixed 400 and 250 µl of serum with dH2O. The larger volume yield nearly doubled the amount of total RNA when compare to using 250 µl of serum. There was also a reduction in contaminants when decreasing the starting volume (Note: As the only variable in this optimization step was the volume of serum, the 230 nm peak is directly associated with serum volumes and not an artifact from the Tri-Reagent isolation).
The small RNA content of these preparations was then assessed using the Bioanalyzer in combination with the Small RNA Kit. From the Bioanalyzer trace, there is a distinct peak at approximately 20 nts which represents the microRNA population (Figure 2A). Using this protocol, this microRNA component represented 93% of the total small RNA population. This is a high level of purity and the total RNA can now be used for varying molecular assays such as arrays and qPCR reactions. A summary of the workflow is presented in Figure 2B.
We then measured microRNA expression in four different biological samples using either an oligonucleotide array or qPCR. Figure 3A represents a microRNA heat map of these four samples. As in our previous study, hierarchical clustering was used to analyze the expression data and group samples based on their microRNA expression profile8. Quantitative PCR (qPCR) was also used to assess microRNA levels in these preparations. Using the same four samples, we performed singleplex TaqMan qPCR reactions to detect miR-21-5p, miR-486-5p, miR-15b-5p, miR-16-5p and let-7a. As shown in the amplification plots (Figures 3B and 3C) all miRNAs were successfully detected.
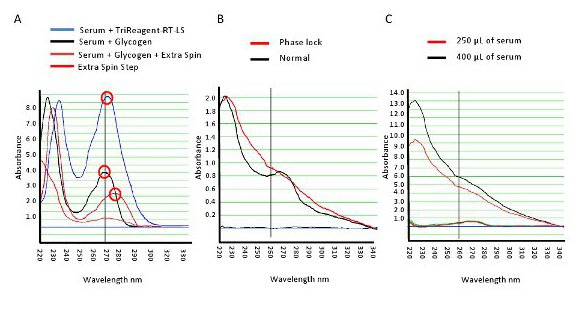
Figure 1. Spectrophotometric profile of RNA from human serum. A. A typical UV profile of RNA isolated from human serum (blue line). Various optimization steps were tested to reduce contaminates and increase total RNA yield. B. Compares the use of a Phase Lock Gel to a normal isolation. C. Profile of two measurements with different starting volumes of serum. Increasing the volume had marked impact on RNA yields. Please click here to view a larger version of this figure.
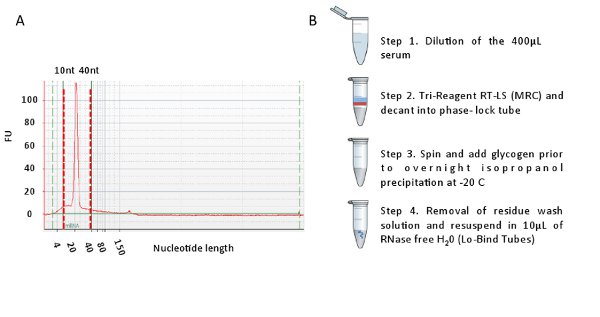
Figure 2. A. Representative Bioanalyzer trace showing a single spike at approximately 21 nucleotides (x-axis). This spike represents the miRNA fraction in the small RNA population from serum. B. Summary of the workflow to isolate total RNA from serum. Please click here to view a larger version of this figure.
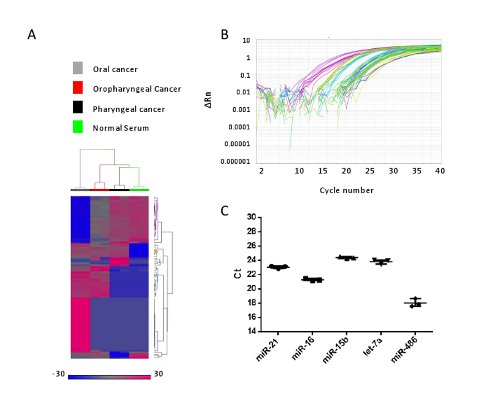
Figure 3. The microRNAs isolated using this method were utilized for array profiling and qPCR analysis. A. Three head and neck cancers and one normal sera were isolated for total RNA (including microRNAs) and subjected to array profiling using the 8 X 60K miRNA chip. This was repeated in technical replicates. After raw data Quality Control (QC) processing, expression of these microRNAs was analyzed using Hierarchical Clustering (HCL) and presented as a heat map. Red indicates up regulation and blue indicates down regulation of the microRNAs. B. Representative qPCR amplification curves for the detection of miR-21, miR-486, miR-15, miR-16 and let-7a in four human sera. C. All five miRNAs were detected and raw Ct values were then plotted for the normal serum. This pattern of detection was similar for the other three samples (Data not shown). Please click here to view a larger version of this figure.
Discussion
The population of microRNAs constitutes approximately 0.4-0.5% of the total RNA found in serum. Further there is also a high protein content found in human serum. To improve RNA and reduce both protein and phenol contaminates we have modified the traditional Chomczynski approach9 with the addition of several steps.
Total RNA was isolated from serum using the standard Tri-Reagent RT-LS (Molecular Research Centre) however when performed in our laboratory, this method yielded RNA in low quantities with various contaminants.
Figure 1 shows a UV spectrophotometric RNA profile with evidence of protein contamination at 280 nm, phenol contamination at 270 nm, and other organic contaminants at 230 nm. The reagent, guanidinium thiocyanate, was also detected at 260 nm.
To address the problems of low serum RNA yields and reduce contamination, a series of optimization steps were added to the standard Tri-Reagent RT-LS procedure. Firstly, we found that diluting the serum 1 in 5 reduced protein contamination. This step has been recommended by other studies10 and has been shown to reduce protein contamination. Furthermore, we used 1 mg/ml concentration of proteinase K. We had tested a range of concentrations from 100, 500, and 1,000 μg. There was no difference between the concentrations thus we utilized higher concentrations and shorter incubation periods.
The second modification was to introduce the use of the 2.0 ml Phase Lock tubes. The heavy Phase Lock Gel traps the majority of contaminants such as phenol and proteins in the organic phase. It provides a density barrier for the maximum transfer of the aqueous phase without the risk of contamination. The application of the Phase Lock Gel as an aqueous/organic phase barrier material can decrease the processing time and further improve DNA recovery by as much as 30%, while at the same time eliminating the user from exposure to harmful volatile organics11. The third modification resulted in a considerable improvement in RNA quality. The introduction of a "flash" centrifugation spin at 12,000 × g was important in removing any residual contamination from the RNA pellet prior to the ethanol washes.
When comparing our approach to other published methodologies6,12,13, we have introduced the use of a Phase Lock Gel to maximize recovery of small noncoding RNAs from the aqueous phase. One of the limitations of our protocol and others is that replicate isolations may be needed to obtain enough RNA for expression analysis. From our experiences, we prefer the Trizol based methods over the column-based kits and numerous studies have compared the merits of both systems6,14-16. Of notably interest was a recent study which concluded that miRNAs with low GC content, such as miR-141, miR-29b, miR-21, miR-106b, miR-15a, and miR-34a, are selectively lost during sample preparation using the Trizol method17. This problem was solved by the addition of magnesium during sample isolation.
In summary, our protocol uses the traditional methods to isolate small RNAs from serum. These serum miRNAs can be derived from micro-particles, exosomes or dying cells. This protocol would isolate all these miRNAs to produce high quality RNA which can be used for various downstream molecular applications. This extraction method is fairly straightforward, relies on common hardware found in most laboratories, and is simple enough for most novices interested in measuring miRNAs expression in human serum.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Samantha Khoury and Pamela Ajuyah are supported by the Australian Postgraduate Award. We would also like to acknowledge the Translational Cancer Research Network, Lowy Cancer Research Centre, University of New South Wales and the Northern Translational Cancer Research Unit for their additional support of Samantha Khoury.
Materials
| Name of the Material/Equipment | Company | Catalog Number | Comments/ Description (optional) |
| Tri-Reagent RT LS | Molecular Research Centre, USA | TR 118 | – |
| RNase free H20 | GIBCO Invitrogen | 10977-023 | – |
| Proteinase K | Finnzymes, Finland | EO0491 | – |
| Heavy Phaselock Tube | 5PRIME | 2302830 | 2ml capacity |
| DNA Lobind tube | Eppendorf | 0030 108.078 | 1.5ml capacity |
| Glycogen | Invitrogen, USA | 10814-010 | 5mg/ml |
| RNA grade Isopropanol | Sigma Aldrich, USA | I9516 | 100% |
| Refrigerated Centrifuge | John Morris | – | – |
| Nanodrop UV-Vis spectrophotometer | Thermo Fisher Scientific, USA | – | – |
| RNA grade Ethanol | Sigma Aldrich, USA | E7023 | 70% |
| Agilent 2100 Bioanalyser | Agilent, USA | – | – |
References
- Babu, S. Monitoring extraction efficiency of small RNAs with the Agilent 2100 Bioanalyzer and the Small RNA Kit. Agilent Application Note. , (2010).
- Tran, N., Hutvagner, G. Biogenesis and the regulation of the maturation of miRNAs. Essays Biochem. 54, 17-28 (2013).
- Chim, S. S., et al. Detection and characterization of placental microRNAs in maternal plasma. Clin. Chem. 54, 482-490 (2008).
- Lodes, M. J., et al. Detection of cancer with serum miRNAs on an oligonucleotide microarray. PLoS One. 4, (2009).
- Chen, X., et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 18, 997-1006 (2008).
- Burgos, K. L., et al. Identification of extracellular miRNA in human cerebrospinal fluid by next-generation sequencing. RNA. 19, 712-722 (2013).
- Chomczynski, P., Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162, 156-159 (1987).
- Zhang, X., et al. Alterations in miRNA processing and expression in pleomorphic adenomas of the salivary gland. Int. J. Cancer. 124, 2855-2863 (2009).
- Chomczynski, P., Sacchi, N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat. Protoc. 1, 581-585 (2006).
- Kirschner, M. B., et al. Haemolysis during sample preparation alters microRNA content of plasma. PLoS One. 6, (2011).
- Murphy, N. R., Hellwig, R. J. Improved nucleic acid organic extraction through use of a unique gel barrier material. Biotechniques. 21, 934-936 (1996).
- Taylor, C. J., Satoor, S. N., Ranjan, A. K., Pereira e Cotta, M. V., Joglekar, M. V. A protocol for measurement of noncoding RNA in human serum. Experimental Diabetes Research. 2012, (2012).
- Ichikawa, M., Akiyama, H. A Combination of Extraction Reagent and DNA Microarray That Allows for the Detection of Global MiRNA Profiles from Serum/Plasma. Methods Mol. Biol. 1024, 247-253 (2013).
- Fromm, B., Harris, P. D., Bachmann, L. MicroRNA preparations from individual monogenean Gyrodactylus salaris-a comparison of six commercially available totalRNA extraction kits. BMC Res. Notes. 4, 217 (2011).
- Li, Y., Kowdley, K. V. Method for microRNA isolation from clinical serum samples. Anal. Biochem. 431, 69-75 (2012).
- McAlexander, M. A., Phillips, M. J., Witwer, K. W. Comparison of Methods for miRNA Extraction from Plasma and Quantitative Recovery of RNA from Cerebrospinal Fluid. Frontiers in Genetics. 4, 83 (2013).
- Kim, Y. K., Yeo, J., Kim, B., Ha, M., Kim, V. N. Short structured RNAs with low GC content are selectively lost during extraction from a small number of cells. Mol. Cell. 46, 893-895 (2012).

