OP-IVM: Combining In vitro Maturation after Oocyte Retrieval with Gynecological Surgery
Summary
In vitro maturation (IVM) before gynecological operation (OP-IVM) combines IVM following oocyte retrieval with routine gynecological surgery and serves as an extension of conventional IVM applications for fertility preservation.
Abstract
The use of in vitro maturation (IVM) before gynecological operation (OP-IVM) is an extension of conventional IVM that combines IVM following oocyte retrieval with routine gynecological surgery. OP-IVM is suitable for patients undergoing benign gynecological surgery who have the need for fertility preservation (FP) or infertility treatments such as in vitro fertilization and embryo transfer (IVF-ET). In the operating room, patients undergoing benign gynecological surgery are first anesthetized and receive ultrasound-guided immature follicle aspiration (IMFA) treatment. As the subsequent gynecological surgery is performed, the cumulus-oocyte complexes (COCs) are examined, and the immature COCs are transferred into the IVM medium and cultured for 28-32 hours in the IVF laboratory. After assessment, mature oocytes in the MII stage will be selected and cryopreserved in liquid nitrogen for FP or fertilized by intracytoplasmic sperm injection (ICSI) for IVF-ET. By combining IVM with gynecological surgery, immature oocytes that would have been discarded can be saved and used for assisted reproductive technology (ART). The procedure, significance and critical aspects of OP-IVM are described in this article.
Introduction
IVM is an ART in which human immature oocytes are cultured in vitro to maturation for IVF-ET or FP. In IVM, ovulation induction medications are not used, thus reducing pain, financial burden, and complications such as ovarian hyperstimulation syndrome (OHSS)1,2. In addition, IVM is particularly suitable for the FP of cancer patients and the infertility treatment of hormone-sensitive patients who may be unable to or have no time to receive ovulation induction therapy3. Therefore, although the number of oocytes retrieved, clinical pregnancy rate (CPR), and live birth rate (LBR) are lower than those of IVF4,5, IVM has its own unique advantages.
Infertile patients with endometrial lesions, hydrosalpinx, or ovarian cysts usually have gynecological surgery before ART treatment, and their oocytes are usually immature. OP-IVM uses guided transvaginal ultrasound to retrieve the immature oocytes and grow them in vitro until maturation for IVF-ET or FP. OP-IVM combines IVM after oocyte retrieval and gynecological surgery, thereby reducing complications that are common in controlled ovarian hyperstimulation cycles and saving time and money. For fertile patients, OP-IVM could serve as a "fertility insurance" while undergoing routine gynecological surgery.
Furthermore, damages caused by gynecological surgeries, such as electrocautery6,7 and ovarian tumor resection, could be reduced through oocyte retrieval before gynecological surgery. Therefore, compared with routine gynecological surgery, OP-IVM could reduce the number of operations during infertility treatment and prevent the loss of functional oocytes during ovarian surgery.
A previous study has shown that the additional procedure of oocyte retrieval would neither increase surgical complications and adverse pregnancy outcomes, nor prolong the hospital stay8. Some patients have given live birth through OP-IVM8, indicating the feasibility of this method. This paper describes characteristics of patients who may benefit from OP-IVM as well as procedures and critical points of OP-IVM and discusses the evaluation of human oocyte maturity.
Protocol
NOTE: Studies related to the OP-IVM method have been approved by the institutional review board (IRB) of Peking University Third Hospital and the Ethics Committee of Peking University (2014S2004). A summary of OP-IVM is shown in Figure 1. The step-by-step procedure will be introduced in the following section.
1. Introduction of OP-IVM to appropriate patients
- Identify potential patients who may benefit from OP-IVM such as those described in steps 1.1.1-1.1.3.:
- Polycystic ovarian syndrome (PCOS) patients with clomiphene resistance who need laparoscopic ovarian drilling surgery.
- Infertile patients who need benign gynecological surgeries, such as hysteroscopic myomectomy, polypectomy, transcervical resection of septum and laparoscopic tubal surgery, and oophorocystectomy, before ART treatment.
- Patients with cancer or hematological disease who are receiving chemoradiotherapy or radiotherapy.
2. Informed consent
- Provide full information to patients, including why OP-IVM may be beneficial, its procedure, uses of IVM oocytes (IVF-ET or cryopreservation), estimated CPRs and LBRs, and possible complications. Ask patients to give informed consent by signing the consent form.
3. Prepare labels and IVM oocyte medium
- Print identification (ID) labels with the patient's name and dates for culture dishes and tubes.
- Add 0.5 mL of IVM medium supplemented with 0.75 IU/mL of follicle-stimulating hormone (FSH) and 0.75 IU/mL of luteinizing hormone (LH) to each well of a 4-well plate. Cover the medium with oil.
NOTE: Perform all these procedures in laminar flow clean benches. - Prewarm the 4-well plate with IVM oocyte medium at 37 °C in humidified air containing 5% CO2 and 5% O2 at least 6 h before use.
4. Administer anesthesia in an operating room
- Check the name of the patient before administering anesthesia.
- Intravenously anesthetize the patient by anesthesiologist.
5. Perform oocyte retrieval
- Tag the labels with name, date, and ID on the culture dishes and tubes.
- Place the patient in the bladder lithotomy position; constantly disinfect, drape, and scrub the vagina with warm saline.
- Place the ultrasound probe inside the vagina; scan and record the number of follicles in both ovaries. Find the location closest to the ovary as the puncture site, and avoid the intestine, bladder, and large blood vessels.
- Aspirate follicular fluid.
- Wash the needle with pH-stable handling medium before puncturing (see Table of Materials).
- Inject the 19 G, single-lumen aspiration needle into the ovaries under the guidance of ultrasound.
- Puncture larger follicles with clear boundaries closest to the probe. At a low position, quickly inject the rinse solution supplemented with 25 U/mL of heparin. Aspirate follicular fluid with the needle under a pressure of 80-90 mmHg, rotating the needle slightly to aspirate as much follicular fluid as possible. Puncture other follicles from the near to far side on this plane.
NOTE: During aspiration, follicular fluid containing oocytes will flow into a sterile 10 mL test tube under negative pressure. Heparin can reduce the follicular fluid viscosity to facilitate the aspiration process. - Remove the needle from the ovary (keep the needle in the vaginal wall). Adjust the direction of the ultrasound probe, and puncture the remaining follicles on other planes. Try to aspirate all the follicles with a diameter of ~5-9 mm.
NOTE: Adjust the probe's position to keep it closest to the ovaries at all times. Press the vaginal fornix with appropriate force to reduce injury and bleeding. - Pull out the needle after finishing in one ovary, wash the needle with the handling medium, and puncture the other side using the same method.
NOTE: Complete follicle aspiration within 25-30 min.
- Transfer the follicular fluid from the operation room to the IVF laboratory within a few minutes after confirming the patient's name and ID.
NOTE: Transfer aspirated follicular fluid to the IVF laboratory bench as soon as possible to prevent coagulation. - Detect active hemorrhage in the pelvic cavity with B-mode ultrasound after puncturing all the follicles. Insert a speculum, and point the tip at the posterior fornix to detect active bleeding at the puncture site.
NOTE: No active bleeding should be observed at the vaginal puncture site if the ovary position is normal and oocyte retrieval is performed carefully. For light bleeding that continues even after compression, keep a sterile gauze compress in the vagina for 2-4 h. Control excessive bleeding from small arteries using clamps with vascular forceps for 2-4 h. Bleeding in the pelvic cavity or ovary, which seldom happens, should be controlled by electrocoagulation by laparoscopic surgery.
6. Gynecological surgery
- Based on the need and condition of the patient, perform appropriate gynecological surgery after oocyte retrieval.
- Perform laparoscopic ovarian drilling surgery for polycystic ovarian syndrome (PCOS) patients with clomiphene resistance.
- Perform benign gynecological surgery for infertile patients before ART treatment, such as hysteroscopic myomectomy, polypectomy, transcervical resection of septum and laparoscopic tubal surgery, and oophorocystectomy.
- Perform ovariectomy for fertility cryopreservation of patients with cancer or hematological disease who need to receive chemoradiotherapy.
NOTE: These gynecological surgeries are basic and standardized clinical operations. Operation guidelines in various countries and hospitals should be relatively similar.
7. Perform IVM
NOTE: Perform the whole process of IVM on a 37 °C homothermal flat.
- Filter the aspirated follicle fluid with a 70 µm nylon cell strainer. Repeatedly rinse the culture tube and strainer with pre-warmed pH-stable handling medium. Ensure all the immature COCs are completely transferred to the culture dish. Collect the filtered fluid , rinse, and culture in a 100 x 15 mm Petri dish.
- Examine the COCs (Figure 2) under the stereoscope with 40x magnification. Quickly transfer the immatures into a pre-warmed IVM oocyte medium.
NOTE: Choose an appropriate magnification depending on the operator's habit. - Record the number of cultured immature COCs.
- Inform the patient about the number of cultured COCs. Discuss the collection of semen with the patients and their partners. Perform sperm extraction according to the normal procedure.
- Culture the immature COCs at 37 °C in humidified air containing 5% CO2 and 5% O2 for 28-32 h.
- Assess the oocyte maturity.
- Denude the cumulus cells by repeated pipetting using a glass Pasteur pipette under the stereoscope with 40x magnification. Examine the extrusion of the polar body (PB) to identify the developmental stage of the oocytes. See Figure 2 for representative images of oocytes in COCs, metaphase II (MII), metaphase I (MI), and germinal vesicles (GVs) with clear morphological characteristics.
NOTE: Use a vertical flame (e.g., of a Bunsen burner) to adjust the diameter of the glass Pasteur pipette to the size of an oocyte. - Count MII oocytes and record the number.
- Choose mature oocytes for IVF or vitrification.
NOTE: Collect sperm on the day the oocytes mature for IVF, but not for oocyte vitrification. - Culture the immature oocytes in GV and MI stage in the original IVM culture medium with cumulus cells for another 10-14 h. Repeat steps 7.6.2-7.6.3.
- Denude the cumulus cells by repeated pipetting using a glass Pasteur pipette under the stereoscope with 40x magnification. Examine the extrusion of the polar body (PB) to identify the developmental stage of the oocytes. See Figure 2 for representative images of oocytes in COCs, metaphase II (MII), metaphase I (MI), and germinal vesicles (GVs) with clear morphological characteristics.
8. Perform ICSI or oocyte vitrification
- Follow the standard procedure of the reproductive center8.
9. Culture embryos and perform embryo cryopreservation
- Follow the standard procedure of the reproductive center8.
Representative Results
Until December 2019, OP-IVM was used for fertility preservation of 274 patients. Embryological and reproductive outcomes of 158 patients between 2014 to 2016 were published in a previous paper8. The following example discusses the procedure followed for a PCOS patient receiving OP-IVM in 2016. The patient is a 28-year-old diagnosed with primary infertility, left adnexal cyst, and PCOS. She received laparoscopic cystectomy and OP-IVM on September 28th, 2016; 27 immature COCs were obtained. After a 28 h culture, 7 oocytes in the MII stage were selected and fertilized by ICSI. The number of good-quality 2 pro-nuclei (2PNs) on day 1 and embryos on day 3, and day 5 were 6, 6, and 2, respectively. The blastocysts obtained on day 5 (October 4th, 2016) were frozen. On February 14th, 2017, one blastocyst was thawed. This thawed blastula was alive and transferred into the uterus on February 14th, 2017. Results of a blood human chorionic gonadotropin (hCG) test were positive, and ultrasonography showed intrauterine early pregnancy. The patient delivered a healthy girl by cesarean section at 40 weeks on November 2nd, 2017.
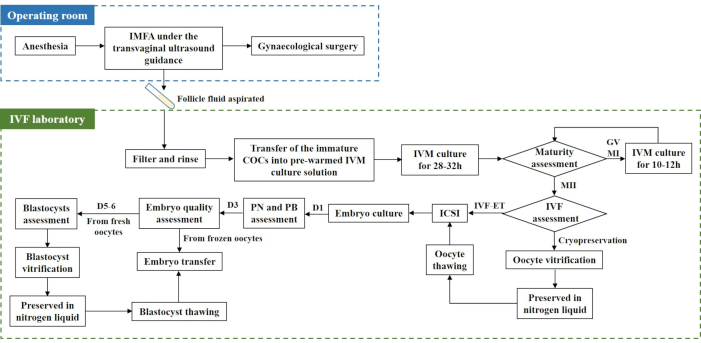
Figure 1: OP-IVM flow chart. Before gynecological surgery, under transvaginal ultrasound guidance, follicle fluid is aspirated through IMFA. The obtained follicle fluid is transferred into the IVF laboratory, filtered, and rinsed. Immature COCs are transferred into a prewarmed IVM culture solution and cultured for 28-32 h. Mature oocytes with PB are used for cryopreservation or IVF-ET. Abbreviations: IMFA = immature follicle aspiration; IVF = in vitro fertilization; IVF-ET = IVF and embryo transfer; COCs = cumulus-oocyte complexes; IVM = in vitro maturation; OP-IVM = IVM before a gynecological operation; GV = germinal vesicle; MI = metaphase I; ICSI = intracytoplasmic sperm injection; PB = polar body; PN = pro-nucleus; D1 = day 1. Please click here to view a larger version of this figure.
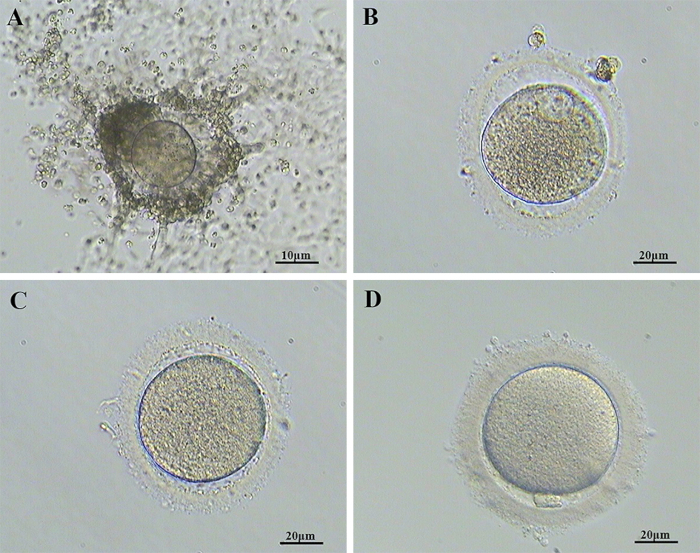
Figure 2: Morphological characteristics of representative images of oocytes. (A) COCs, (B) GV, (C) MI, (D) MII stage. Abbreviations: COCs = cumulus-oocyte complexes; GV = germinal vesicle; MI = metaphase I; MII = metaphase II. Please click here to view a larger version of this figure.
Discussion
The OP-IVM method described in this article extends to conventional IVM applications and combines IVM after oocyte retrieval with routine gynecological surgery. Oocytes that would have been lost in the gynecological surgery can now be used for IVF-ET or FP without additional surgical risks. OP-IVM was first used to retrieve oocytes before ovarian drilling in PCOS patients. Its application soon expanded to infertile patients who need benign gynecological surgery and cancer or hematological disease patients who need chemoradiotherapy. Because of the potential risk of metastasis, OP-IVM is not suitable or recommended for patients with malignant tumors. Based on OP-IVM, a new Chinese mode of egg bank for the FP of infertile patients and cancer patients has been established at this institution.
For OP-IVM, both the urgency of gynecological surgery and the potential of obtaining a higher number of oocytes should be considered. In general, IVM outcome is positively correlated with the number of immature oocytes obtained during oocyte retrieval9. Dominant follicles may inhibit the growth of surrounding smaller follicles and cause atresia10, resulting in a reduced number of retrieved immature oocytes. Therefore, if the gynecological surgery is not urgent, the best window for OP-IVM is when a greater number of small follicles with few dominant follicles are observed by B-mode ultrasonography. In addition, anti-Mullerian hormone (AMH) can be used as a parameter to predict ovarian reserve as the numbers of retrieved oocytes are significantly positively correlated with AMH11,12. In cases where gynecological operation has been prioritized, as many immature oocytes should be retrieved as much as possible. In emergency operations, oocyte retrieval and IVM should be carefully evaluated based on the patient's condition and will.
IMFA under transvaginal ultrasound guidance is a method used to retrieve oocytes that can reduce damage to ovarian function13,14. Laparoscopic ovarian puncture to aspirate immature oocytes is also used in some studies15. However, compared with laparoscopy, IMFA under transvaginal ultrasound takes up less time, is less invasive, is easier to perform16,17, and can achieve more targeted follicle aspiration. Therefore, to minimize the impact on ovarian reserve and increase aspiration accuracy, IMFA under transvaginal ultrasound guidance is recommended as a better method for oocyte retrieval in OP-IVM. However, ICSI is recommended for fertilization. Previous studies have shown that the zona pellucida will harden after being cultured in vitro for long periods, resulting in a reduced rate of successful fertilization18. ICSI has been shown to effectively improve the fertilization rate of IVM oocytes in natural cycles19,20. Although recent studies have shown similar fertilization rates for ICSI and IVF21,22, ICSI holds the advantage of using natural cycles instead of ovarian stimulation. Collected embryos are frozen because of unprepared endometrium, and embryo transfer can be performed later according to the standard procedure at each center.
The purpose of ART is to treat infertility and deliver healthy babies. Previous research about the reproductive outcomes of OP-IVM showed that the numbers of retrieved oocytes and oocyte maturation rates are comparable with those observed for conventional IVM cycles using FSH and hCG. However, OP-IVM has a lower CPR and a lower LBR than conventional IVM8,23,24,25. This may be explained by constrained oocyte maturation due to the poor development potential of immature oocytes in natural cycles. However, hormone stimulation is not an option for some patients who are unable to or who do not have the time to receive the treatment. In addition, vitrification may damage the oocytes' developmental potential, resulting in low CPRs and LBRs26,27.
Moreover, gonadotropins may enlarge the ovaries and increase the risk of bleeding during oocyte retrieval, adversely affecting the exposure of operative fields in laparoscopy. The LBR of the OP-IVM method is relatively low. More studies are needed to examine the best operative timing and modify the IVM culture system to improve OP-IVM outcomes. Overall, OP-IVM combines IVM with gynecological surgery so that immature oocytes that would have been damaged or discarded can be saved and used for ART. OP-IVM has the advantages of avoiding complications caused by ovarian stimulating medications and reducing the number of operations for infertility treatment. Therefore, OP-IVM should be considered as a potentially valuable treatment method for certain patients and studied more in depth to better understand its effects and outcomes.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the China National Key R&D Program (no. 2017YFC1002000, 2018YFC1004001, 2019YFA0801400), the National Science Foundation of China (no. 81571386, 81730038), the CAMS Innovation Fund for Medical Sciences (2019-I2M-5-001), and the Special Research Project of Chinese Capital Health Development (2018-2-4095).
Materials
| 19 G single-lumen aspiration needles | Cook, Australia | K-OPS-7035-REH-ET | |
| 4-well plate | Corning | ||
| 70 μm nylon cell strainer | Falcon, USA | 352350 | |
| CO2 Incubator | Thermo | ||
| Culture oil | Vitrolife, Sweden | 10029,OVOIL | Step 3.2. |
| FSH & LH | Ferring Reproductive Health, Germany | MENOPUR® | |
| Glass Pasteur pipette | Hilgenberg GmbH, Germany | 3154102-26 | |
| G-MOPS medium | pH-stable handling medium for washing the needle before puncturing | ||
| IVM medium | Origio, Denmark | ART-1600-B | |
| Laminar Flow Clean Benches | ESCO | ||
| Petri dish | Thermo Fisher Scientific, Denmark | 263991 | |
| pH stable handing media designed to support the handling and manipulation of oocytes and embryos outside the incubator | Vitrolife, Sweden | 10130, G-MOPS PLUS | Step 7.1. |
| Rinse solution | Cook, Australia | K-SIFB-100 | |
| Stereoscope | Nikon |
References
- Ho, V. N. A., Braam, S. C., Pham, T. D., Mol, B. W., Vuong, L. N. The effectiveness and safety of in vitro maturation of oocytes versus in vitro fertilization in women with a high antral follicle count. Human Reproduction. 34 (6), 1055-1064 (2019).
- Yang, Z. Y., Chian, R. C. Development of in vitro maturation techniques for clinical applications. Fertility and Sterility. 108 (4), 577-584 (2017).
- Hatirnaz, S., et al. Oocyte in vitro maturation: A sytematic review. Turkish Journal of Obstetrics and Gynecology. 15 (2), 112-125 (2018).
- Gremeau, A. S., et al. In vitro maturation or in vitro fertilization for women with polycystic ovaries? A case-control study of 194 treatment cycles. Fertility and Sterility. 98 (2), 355-360 (2012).
- Ho, V. N. A., Braam, S. C., Pham, T. D., Mol, B. W., Vuong, L. N. The effectiveness and safety of in vitro maturation of oocytes versus in vitro fertilization in women with a high antral follicle count. Human Reproduction. 34 (6), 1055-1064 (2019).
- Sahin, C., et al. Which should be the preferred technique during laparoscopic ovarian cystectomy. Reproductive Sciences. 24 (3), 393-399 (2017).
- Xiao, J., et al. Impact of hemostatic methods on ovarian reserve and fertility in laparoscopic ovarian cystectomy. Experimental and Therapeutic. 17 (4), 2689-2693 (2019).
- Song, X. -. L., et al. Enhancing the scope of in vitro maturation for fertility preservation: transvaginal retrieval of immature oocytes during endoscopic gynaecological procedures. Human Reproduction. 35 (4), 837-846 (2020).
- Magnusson, &. #. 1. 9. 7. ;., Källen, K., Thurin-Kjellberg, A., Bergh, C. The number of oocytes retrieved during IVF: a balance between efficacy and safety. Human Reproduction. 33 (1), 58-64 (2018).
- Larose, H., et al., Wellik, D. M., et al. Gametogenesis: A journey from inception to conception. Current Topics in Developmental Biology. 132, 257-310 (2019).
- Broer, S. L., Broekmans, F. J. M., Laven, J. S. E., Fauser, B. C. J. M. Anti-Müllerian hormone: ovarian reserve testing and its potential clinical implications. Human Reproduction Update. 20 (5), 688-701 (2014).
- Arce, J. C., La Marca, A., Mirner Klein, B., Nyboe Andersen, A., Fleming, R. Antimüllerian hormone in gonadotropin releasing-hormone antagonist cycles: prediction of ovarian response and cumulative treatment outcome in good-prognosis patients. Fertility and Sterility. 99 (6), 1644-1653 (2013).
- Katayama, K. P., et al. Ultrasound-guided transvaginal needle aspiration of follicles for in vitro fertilization. Obstetrics and Gynecology. 72 (2), 271-274 (1988).
- Dellenbach, P., et al. Transvaginal sonographically controlled follicle puncture for oocyte retrieval. Fertility and Sterility. 44 (5), 656-662 (1985).
- Hirata, T., et al. Concomitant ovarian drilling and oocyte retrieval by laparoendoscopic single-site surgery led to live birth using in vitro maturation of oocyte and transfer of frozen-thawed blastocyst in woman with polycystic ovary syndrome. Journal of Obstetrics and Gynaecology Research. 40 (5), 1431-1435 (2014).
- Deutinger, J., et al. Follicular aspiration for in vitro fertilization: sonographically guided transvaginal versus laparoscopic approach. European Journal of Obstetrics, Gynecology, and Reproductive Biology. 26 (2), 127-133 (1987).
- Tanbo, T., Henriksen, T., Magnus, O., Abyholm, T. Oocyte retrieval in an IVF program. A comparison of laparoscopic and transvaginal ultrasound-guided follicular puncture. Acta Obstetricia et Gynecologica Scandinavica. 67 (3), 243-246 (1988).
- Nagy, Z. P., et al. Pregnancy and birth after intracytoplasmic sperm injection of in vitro matured germinal-vesicle stage oocytes: case report. Fertility and Sterility. 65 (5), 1047-1050 (1996).
- Söderström-Anttila, V., Mäkinen, S., Tuuri, T., Suikkari, A. -. M. Favourable pregnancy results with insemination of in vitro matured oocytes from unstimulated patients. Human Reproduction. 20 (6), 1534-1540 (2005).
- Hwang, J. L., Lin, Y. H., Tsai, Y. L. In vitro maturation and fertilization of immature oocytes: a comparative study of fertilization techniques. Journal of Assisted Reproduction and Genetics. 17 (1), 39-43 (2000).
- Walls, M., Junk, S., Ryan, J. P., Hart, R. IVF versus ICSI for the fertilization of in-vitro matured human oocytes. Reproductive Biomedicine Online. 25 (6), 603-607 (2012).
- Park, J. H., Jee, B. C., Kim, S. H. Comparison of normal and abnormal fertilization of in vitro-matured human oocyte according to insemination method. Journal of Obstetrics and Gynaecology Research. 42 (4), 417-421 (2016).
- Ho, V. N. A., Pham, T. D., Le, A. H., Ho, T. M., Vuong, L. N. Live birth rate after human chorionic gonadotropin priming in vitro maturation in women with polycystic ovary syndrome. Journal of Ovarian Research. 11 (1), 70 (2018).
- Zheng, X., et al. Effect of hCG priming on embryonic development of immature oocytes collected from unstimulated women with polycystic ovarian syndrome. Reproductive Biology and Endocrinology. 10, 40 (2012).
- Tannus, S., et al. Predictive factors for live birth after in vitro maturation of oocytes in women with polycystic ovary syndrome. Archives of Gynecology and Obstetrics. 297 (1), 199-204 (2018).
- Cohen, Y., et al. Decreased pregnancy and live birth rates after vitrification of in vitro matured oocytes. Journal of Assisted Reproduction and Genetics. 35 (9), 1683-1689 (2018).
- Yazdanpanah, F., Khalili, M. A., Eftekhar, M., Karimi, H. The effect of vitrification on maturation and viability capacities of immature human oocytes. Archives of Gynecology and Obstetrics. 288 (2), 439-444 (2013).

