Toxicological Assays for Testing Effects of an Epigenetic Drug on Development, Fecundity and Survivorship of Malaria Mosquitoes
Summary
A protocol is developed to examine the effects of an epigenetic drug DZNep on the development, fecundity and survivorship of mosquitoes. Here we describe procedures for the aqueous exposure of DZNep to immature mosquitoes and a blood-based exposure of DZNep to adult mosquitoes in addition to measuring SAH hydrolase inhibition.
Abstract
Insecticidal resistance poses a major problem for malaria control programs. Mosquitoes adapt to a wide range of changes in the environment quickly, making malaria control an omnipresent problem in tropical countries. The emergence of insecticide resistant populations warrants the exploration of novel drug target pathways and compounds for vector mosquito control. Epigenetic drugs are well established in cancer research, however not much is known about their effects on insects. This study provides a simple protocol for examining the toxicological effects of 3-Deazaneplanocin A (DZNep), an experimental epigenetic drug for cancer therapy, on the malaria vector, Anopheles gambiae. A concentration-dependent increase in mortality and decrease in size was observed in immature mosquitoes exposed to DZNep, whereas the compound reduced the fecundity of adult mosquitoes relative to control treatments. In addition, there was a drug-dependent decrease in S-adenosylhomocysteine (SAH) hydrolase activity in mosquitoes following exposure to DZNep relative to control treatments. These protocols provide the researcher with a simple, step-by-step procedure to assess multiple toxicological endpoints for an experimental drug and, in turn, demonstrate a unique multi-prong approach for exploring the toxicological effects of water-soluble epigenetic drugs or compounds of interest against vector mosquitoes and other insects.
Introduction
Malaria is responsible for the highest number of insect-related deaths in the world. An estimated 219 million cases occur annually worldwide, resulting in approximately 660,000 deaths, primarily in Africa 1. Despite concerted efforts, malaria programs face several challenges. While insecticidal treated bednets and indoor residual spraying form key components of the program, resistance to insecticides in local populations impede these efforts 2. The rapid increase in insecticide resistant mosquito populations is largely attributed to the ability of the malaria mosquitoes to adapt quickly to changes in their environment and exploit different niches 3,4,5. To overcome the existing mechanisms of insecticide resistance, the exploration of novel insecticide targets and next generation compounds is warranted. A simple, step-by-step protocol for determining the efficacy of experimental insecticides on the various life stages of malaria mosquitoes would significantly enhance these efforts.
Pharmacological studies of drug effects on cell lines and animal models have established the use of epigenetic drugs as a useful tool for modulating the genetics and physiology of cells and organisms. DNA methylation and histone modification are two epigenetic mechanisms that affect gene expression in multicellular organisms without changing the underlying DNA sequence 6. Post translational modifications such as methylation play a crucial role in maintaining cellular integrity and gene expression, and may affect several fundamental processes 7,8,9. Research in some insect species have highlighted the importance of epigenetics in processes involving oogenesis and stem cell maintenance 10, as well as dosage compensation 11 . However, such aspects in disease vectors are yet to be explored. Using a compound to modulate this system in mosquitoes may provide us with insights into the novel insecticide target pathways. 3-Deazaneplanocin A (DZNep) is a known histone methylation inhibitor, which impact on various types of cancers have been studied 12,13,14,15,16. DZNep is a stable water-soluble epigenetic drug that indirectly inhibits histone lysine N-methyltransferase (EZH2), a component of the polycomb repressive complex 2 (PRC2) in mammalian cells. PRC2 plays an important role in regulating the growth of stem cells in multicellular organisms, and histone methylation is a key aspect of the PRC2 mediated gene silencing. In immunocompromised mice, cells pre-treated with DZNep have been shown to be less tumorigenic 17. This drug is becoming used for studying other diseases, such as non-alcoholic fatty liver disease, in which EZH2 is implicated 18. DZNep is an established S-adenosylhomocysteine (SAH) hydrolase inhibitor 19,20. The inhibition of SAH hydrolase results in an accumulation of SAH and, in turn, leads to the inhibition of methyltransferase activity by limiting available methyl donor groups. SAH is an amino acid derivative utilized by many organisms, including insects, in their metabolic pathways. A recent study has shown that DZNep in low doses may affect diapause and delay development in insects 21.
Here, a robust protocol to investigate the effects of a water-soluble compound on various life stages of mosquitoes is developed. The three parts of this protocol include instructions for examining the effects of a water-soluble compound on immature mosquitoes, adult blood-feeding females, and enzyme activity of adult male and female mosquitoes. First, DZNep is dissolved in water to study immature mosquito development and survival. This is performed at two concentrations to compare any differences arising from 10-fold increase in drug exposure. To explore the effect of drug on adult female mosquitoes, DZNep is added to defibrillated sheep blood and fed the blood artificially to females. Subsequently, the outcome of the drug on fecundity is examined. Finally, an enzyme activity assay is performed using 5,5'-dithiobis-(2-nitrobenzoic acid) (DTNB) as an indicator to determine the effect of DZNep on SAH hydrolase inhibition in adult male and female mosquitoes. While this protocol is developed with a malaria mosquito, Anopheles gambiae, it can be easily adapted to studying the effects of compounds of interest on any species of mosquito or other insects. The techniques detailed in this protocol may not be efficiently applied to a drug with limited or no solubility in water or aqueous media.
Protocol
NOTE: The three parts of the protocol describe the aqueous exposure of the DZNep drug to larval mosquitoes, a blood-based exposure of DZNep to adult females to study its effect of fecundity, and SAH hydrolase inhibition by DZNep measured using a simple colorimetric technique. A schematic representation of these assays is shown in Figure 1.
1. Immature Mosquito Development and Survivorship Assays
NOTE: This section explains the use of a water-soluble drug that affects mosquito larvae development and survival.
- Hatch A. gambiae eggs at 28 °C in distilled water (dH2O) and rear them to 2nd instar larvae.
- Take a six-well DNAse, RNAse free cell culture plate and pour 10 ml dH2O to each well.
- Select mosquito larvae at 2nd instar and add 15 larvae per well. Before adding 2nd instar larvae to plate, put them on a paper towel for 2-3 sec to remove excess water.
- Label wells with color-coded tapes to randomize experiment. Label 2 wells per test concentration on each plate (Figure 2).
- Prepare a 1 mM DZNep hydrochloride (MW = 298.73) stock solution by dissolving 0.3 mg DZNep in 1 ml of dH2O. Add stock solution of DZNep.HCl to labeled wells to achieve a final concentration of 0.5 µM and 5.0 µM.
NOTE: The stock solution can be stored at 4 °C for short term (1 day to a month) or -20 °C for long term (longer than one month).
NOTE: Addition of the stock solution for aforementioned concentrations will not change the volume of water in the wells significantly. However in case of much higher concentrations, it is advisable to dissolve the compound in the larval medium to avoid any unnecessary dilution of the compound. - Add equal amount of flake fish food to each well. Cover the plates and incubate them at 28 °C in an incubator.
- Record the mortality for the DZNep-treated and untreated larval mosquitoes after a 24-hr exposure period. Remove and discard any dead larvae. Repeat this recording every day until all larvae die or pupate and emerge as adults. Record the size of larvae every second day, i.e., day 0, 2, 4, 6, 8, until they pupate.
- Analyze the larval development and mortality data using an adequate statistical analysis software.
NOTE: A bar graph can be used to represent the mortality data in Microsoft Excel. Multivariate Analysis of Variance (MANOVA) performed with a software such as JMP or SPSS will reveal if different doses of the drug result in significantly different survival of mosquito larvae.
2. Fecundity Assay by Administering Drug Through Artificial Feeder to Female Mosquitoes
NOTE: This section details the addition of the drug to blood and feeding it to adult female mosquitoes using an artificial feeder system.
- Set up adult mosquito cages with equal number of male and female mosquitoes for control and test cage. Provide 10% sugar solution soaked in cotton balls to both the cages till the females are ready for feeding (3-5 days is optimum for Anopheles gambiae).
- 2 hr prior to the blood feeding, remove the cotton balls to ensure females are hungry.
- Assemble two artificial blood feeders that consist of an inverted wide bottom glass funnel (diameter = 50 mm, length = 70 mm) and a surrounding plastic sealed with industrial glue. Attach plastic tubes (diameter = 7 mm) to two feeders’ outlets (diameter= 10 mm) via connectors for inflow and outflow of water. Label the two feeders as “Control” and “5 μM DZNep.”
- Using a commercially available heating element, set up the program for feeding. In “Menu” go to “Settings” and select “Setpoints”; preloaded programs will appear. Select “SP1” and set the temperature for 37 °C.
NOTE: The heating element ensures that the blood administered is maintained at a constant temperature. Additional program settings may be used for different mosquito or insect species. - Fill a bucket with water and immerse the heating element into the water.
- Cut a square piece of Parafilm (50 mm x 50 mm) and stretch it to make a thin membranous film. Cover the bottom of the artificial feeders with the Parafilm. Cut another piece of Parafilm (50 mm x 10 mm), stretch it and seal the edges of the feeders.
- Assemble the system, connecting the feeders and heating element by the tubes. Once completed, switch on the system and select “SP1”. Press “Enter” to start the system.
NOTE: The water will get heated to 37 °C and maintained at that temperature. - Monitor the temperature of water using a thermometer in the water bucket.
NOTE: The connectors and tubes help maintain a constant circulation of water through the system so that the temperature of the blood remains at 37 °C. - Fill a flat bottom tray with 10% bleach solution that will be used to decontaminate any blood spills.
- Add 2 ml of defibrillated sheep’s blood to a microcentrifuge tube. Label this as “Control”. Repeat the process for the experimental tube labeled as “5 μM DZNep”.Add 6 μl of DZNep stock solution (see step 1.5) to the experimental tube labeled “5 μM DZNep” Gently mix the blood and drug by inverting it several times. Incubate the tube 10 min at RT.
- Using a pipette, add 2 ml of control and test blood to the top of correspondingly labeled feeders. Discard the pipette in the bleach solution. Cover the cage with a dark bag and breathe air on cage periodically to encourage females for feeding.
- Let the mosquitoes feed for approximately 30 min. Once feeding is completed, switch off the heating element, take the feeders out and strip the Parafilm. Soak the feeder in bleach solution for about 5 min and rinse well in deionized water.
- Remove the feeders and put cotton balls with sugar water on the cages for 48 hr. Place an egg dish containing water and filter paper in the cages overnight to facilitate egg laying. Label each egg dish with the respective test or control concentration.
- Remove the egg dish next day and examine the number and structure of eggs under a stereo microscope. Obtain images using the Q-colors5 software. Count the number of eggs. Analyze difference in the number of eggs obtained from test and control cages.
NOTE: An unpaired t-test can be used to determine if the number of eggs are significantly different (p ≤0.05).
3. Biochemical Assay of Mosquito Enzyme Inhibition by Drug
NOTE: This section describes an enzyme activity assay using DTNB as an indicator to determine the effect of DZNep on SAH hydrolase inhibition in adult male and female mosquitoes.
- Prepare a 0.1 M Na2HPO4 (dibasic sodium phosphate) solution by adding 14.19 g of Na2HPO4 to 1 L of dH2O. Next, prepare a 0.1 M NaH2PO4 (monobasic sodium phosphate) solution by adding 13.79 g of NaH2PO4 to 1 L of dH2O.
- Titrate the 0.1 M Na2HPO4 solution with the 0.1 M NaH2PO4 solution to pH 8.5.
NOTE: This will be the working stock solution of 0.1 M Na2HPO4 (pH 8.5). - Prepare a homogenization solution of 0.1 M Na2HPO4 (pH 8.5) by adding 0.3% Triton X-100.
NOTE: The homogenization solution can be stored at 4 oC. - Prepare a 1.6 mM SAH (MW = 384.4) solution by dissolving 6.15 mg of SAH in 10 ml of 0.1 M Na2HPO4 (pH 8.5).
- Prepare a 1.6 mM DTNB (MW = 396.4) solution by dissolving 6.3 mg of DTNB in 10 ml of 0.1 M Na2HPO4 (pH 8.5). Keep the fresh DTNB solution on ice until use.
- Prepare a 1 mM DZNep.HCl (MW = 298.73) solution by dissolving 0.3 mg of DZNep in 1ml dH2O. Next, dilute the 1 mM DZNep solution into a series of concentrations from 1,000 µM to 0.002 µM in dH2O.
- To measure the SAH hydrolase inhibition by DZNep, prepare a crude enzyme extract of mosquitoes: Homogenize 10 non-blood-fed adult mosquitoes in 1 ml of ice-cold 0.1 M NaH2PO4 (pH 8.5), containing 0.3% Triton X-100, using a glass tissue homogenizer. Transfer the homogenate to a 1.5ml microcentrifuge tube.
- Centrifuge the homogenate for 5 min at 10,000 x g at 4 °C. Transfer the supernatant to a clean 1.5 ml microcentrifuge tube. Use the supernatant as the enzyme source for the SAH biochemical assay.
- For the blank treatment (i.e., no DZNep, no enzyme), add 50 µl 1.6 mM SAH, 50 µl 1.6 mM DTNB, and 100 µl 0.1 M Na2HPO4 (pH 8.5) to the individual wells of a 96-well, flat bottom microplate.
- For the control (i.e., no DZNep), add 50 µl 1.6 mM SAH, 50 µl 1.6 mM DTNB, 50 µl 0.1 M Na2HPO4 (pH 8.5) and 50 µl enzyme (SAH hydrolase obtained from crude extract) to the individual wells.
- For DZNep treatments, add 50 µl 1.6 mM SAH, 50 µl 1.6 mM DTNB, 50 µl enzyme and 50 µl selected DZNep concentration to individual wells. Prepare 4 replicates per treatment and control.
- Read the optical density (OD) of the SAH enzyme samples at 405 nm for 5 min at 20 sec intervals using a 96-well microplate reader.
- Subtract the blank OD from control and treatment OD obtained from each well. Calculate the percent remaining SAH hydrolase activity using the following equation: % Residual Activity = (Treatment OD/Control OD) x 100.
Representative Results
Figure 1 is a schematic representation of the assays; it describes the various steps of the procedure listed in this article. As the protocol is based on different life stages of mosquitoes, there is no particular sequence to be followed for the experiments detailed here. The user may choose to conduct one or more assays at the same time, depending upon sample availability.
Figure 2 demonstrates the plate set up for the immature mosquito development and survivorship assays. The number of test concentrations and replicates may be altered depending upon the user’s requirements and drug availability. For the immature mosquito assay, 1 mM stock solution of DZNep was added to water containing mosquitoes to achieve a final concentration of 0.5 µM and 5 µM along with a control pan with wells containing a drug-free population. The experiment demonstrated that more larvae and pupae survived in the control wells than in the DZNep-treated wells.
Figure 3 depicts the effect of DZNep on overall survivorship of malaria mosquitoes during the experiment. The results demonstrate that the epigenetic drug DZNep suppresses the growth and development and induces the mortality of immature mosquitoes. The majority of the mosquito exposed to 0.5 μM died by day 10 of the experiment, whereas a large number of the individuals exposed to 5 μM died by day 8. In contrast, the mortality of the mosquitoes in the control treatment (no drug) remained unaffected and several emerged as adults on day 8 of the experiment. An inverse relation was observed between drug concentration and mosquito body size (Figure 4).
Figure 5 illustrates the effect of DZNep on mosquito fecundity. Adult female mosquitoes fed with blood containing DZNep exhibited a significant reduction in number of viable eggs. Figure 5A shows eggs obtained from control cage (no drug). Figure 5B shows the eggs obtained from test cage. A large number of eggs obtained from test cage were darker in color and lacked floats (air-filled expansions of exochorion) when compared with eggs obtained from normal blood-fed female mosquitoes (Figure 6). The absence of floats along with darker colored eggs indicates the potential role of epigenetics in exochorion formation.
Figure 7 demonstrates the effect of DZNep on SAH hydrolase activity in adult male and female malaria mosquitoes. A DZNep-dependent decrease in OD is observed for each drug treatment compared to the control treatment (no drug) indicating that DZNep inhibits SAH hydrolase activity.
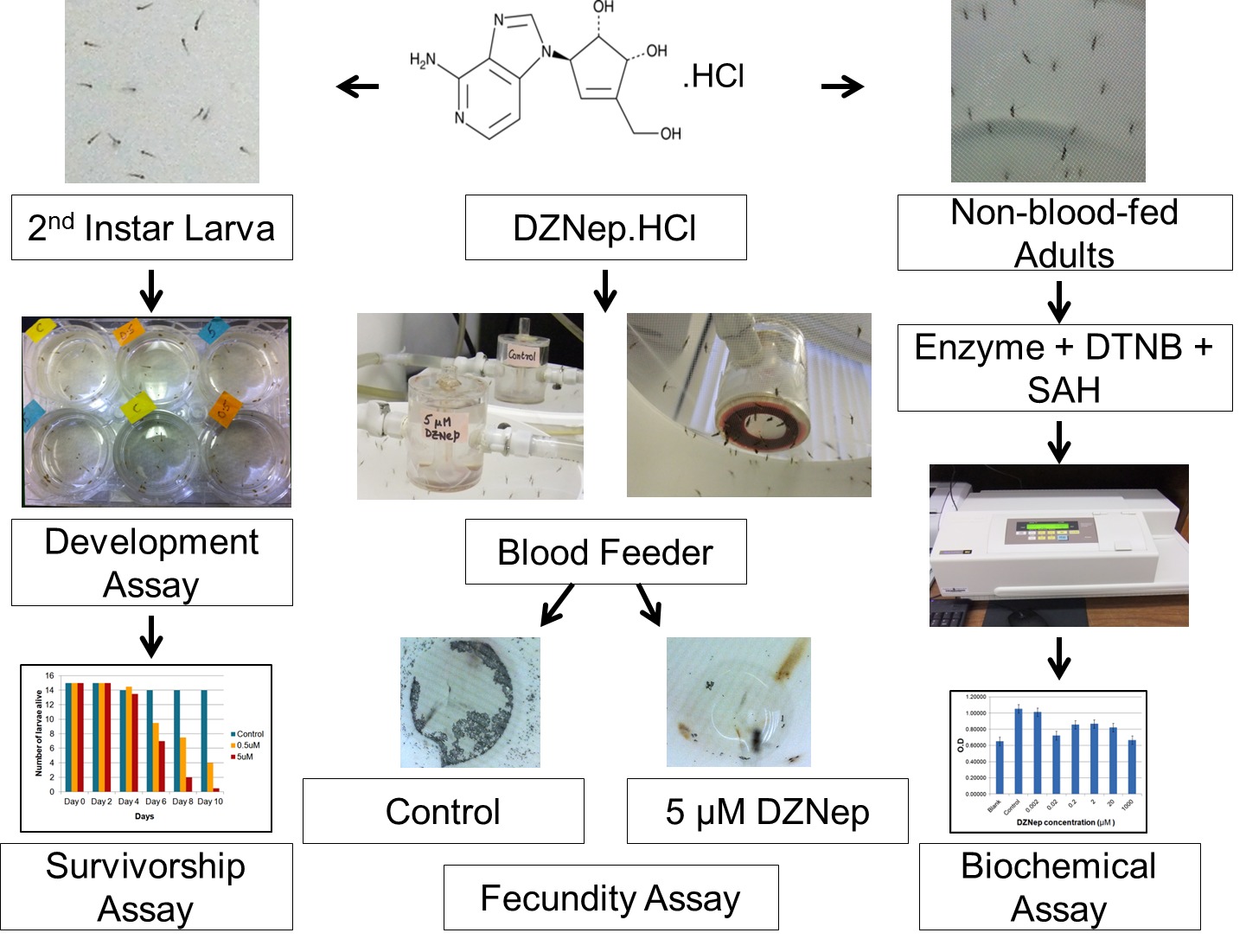
Figure 1: Schematic representation of assays using an epigenetic drug on malaria mosquitoes. The left part of the scheme shows the development and survivorship assays using immature mosquitoes. The central part demonstrates the fecundity assay via blood-feeding the drug to adult females. The right part depicts the biochemical assay of enzyme inhibition by the drug.

Figure 2: Plate depicting the immature mosquito development and survivorship assay. Label “C” marks control wells. Label “0.5” shows wells with 0.5 µM DZNep. Label “5” indicates wells with 5 µM DZNep.
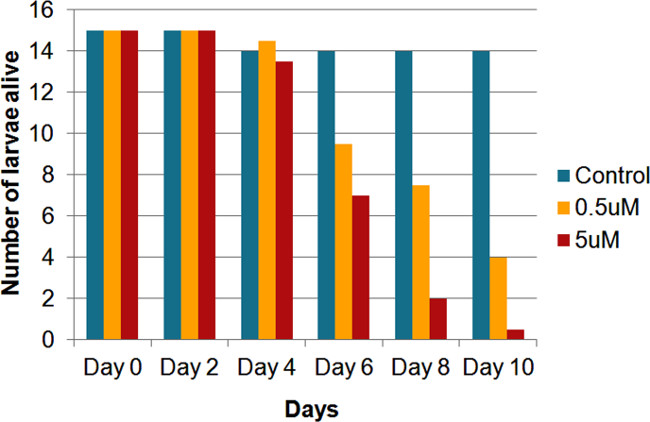
Figure 3: Effect of DZNep on survivorship of malaria mosquitoes. Concentration-dependent mortality is observed with immature mosquitoes.

Figure 4: Effect of DZNep on development of immature mosquitoes. (A) Larva from the control treatment (size = 5 mm). (B) Larva treated with 0.5 µM DZNep (size = 4 mm). (C) Larva treated with 5 µM DZNep (size = 3 mm). All larvae were measured on the same day of the experiment.

Figure 5: Effect of DZNep on mosquito fecundity. Egg dish from the control treatment (no drug) mosquitoes contains much greater number of eggs (A) compared with the egg dish of females treated with 5 µM DZNep (B).
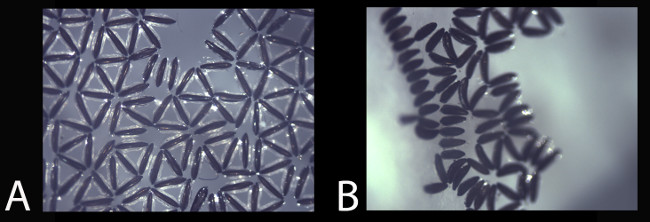
Figure 6: Effect of DZNep on egg structure in malaria mosquitoes seen under a microscope. (A) Eggs from the control treatment (no drug) mosquitoes display floats and rosette-like assemblies. (B) Eggs from the mosquitoes treated with 5 µM DZNep lack floats and rosette-like assemblies.
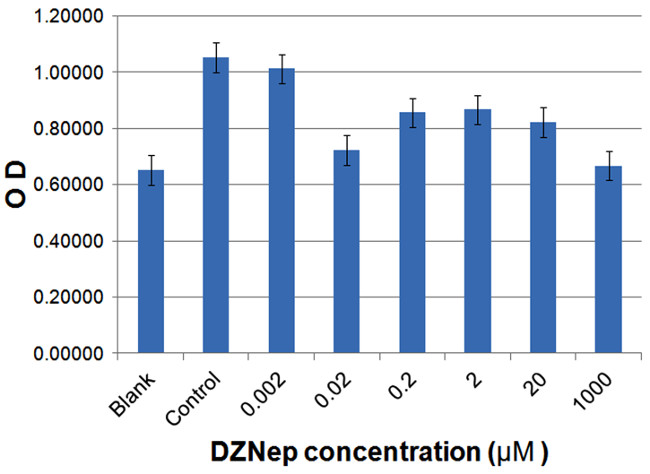
Figure 7: SAH hydrolase inhibition by DZNep in malaria mosquitoes. DZNep causes a decrease in OD compared with control treatment (no drug).
Discussion
There are several steps vital to the successful application of this protocol. For the larval assay, care should be taken to correctly label and replicate each test concentration. Randomizing test samples and adding the designated amount of drug to respective test wells is an important part of this experimental set up. Before adding 2nd instar mosquito larvae to a 96-well microplate, each larva can be put on a paper towel for 2-3 sec to remove excess water. To prevent desiccation, larvae must be immediately transferred to the microplate wells using a blunt pair of forceps. This ensures the volume of water in each microplate well and concentration of drug remains as intended. When transferring larvae for measuring their body length, it is recommended to put the larvae on ice for few minutes in order to keep them stationary. When adapting this protocol for a different compound, care should be taken to determine the amount of stock solution added to each well. In such cases, it might be better to dissolve the calculated amount of compound in water directly prior to adding the larvae in each microplate well.
For blood feeding assays, it is vital that mosquitoes are starved appropriately by removing sugar water at least 2 hr before of experiment. Failure to do so may result in incomplete feeding and little effect on the adult female egg production. Thorough mixing and uniform distribution of the drug in the blood is an important step as well, as DZNep takes a few minutes to dissolve. If the drug treated blood is fed immediately, DZNep does not get equally distributed in the mosquito system. It is strongly recommended that the defibrillated blood is fresh (i.e. less than a week since extraction date) for better results.
There are multiple steps critical to the success of a biochemical assay with a compound of interest. Concentrations of each stock solution used in this protocol have been optimized for DZNep. To adapt the protocol for a different compound, we suggest testing a range of different pH, SAH, DTNB concentrations and insect sample sizes to determine the most reliable results. Preparation of crude enzyme extract is the most time consuming step in this procedure. During homogenate preparation it is recommended to use not more than 10 adult mosquitoes, as lipids present in the supernatant may hinder absorbance reading. To avoid this issue, the top layer of lipids in the supernatant (using a single channel micropipette) can be removed after step 3.8. Supernatant must be transferred to a clean microfuge tube. Step 3.8 should be repeated to discard any remaining viscous lipids. The remaining clear supernatant must be pooled from all tubes and mixed gently. A multi-channel pipette can be used for increased efficiency to add SAH in the wells. DTNB is used as an indicator in the assay, and should be prepared last and kept on ice.
This protocol provides the user with simple steps to test the effects of a compound on different life stages of mosquitoes. However, there are some limitations to the use of this protocol. This technique depends primarily on the water solubility of DZNep. Immature larvae are exposed to the drug dissolved in their surrounding media. If the compound being tested is insoluble in aqueous media, it may render this protocol less efficient. For testing the fecundity, we subjected the adult females to drug dissolved in defibrillated sheep blood. This would not be possible if a lab rears mosquito colonies on mammalian blood directly (i.e., uses mice or guinea pigs for feeding mosquitoes).
The combination of different assays detailed in this article provides the user with a novel way of testing the effects of a water-soluble compound on insects. DZNep is mostly added to cell lines in vitro for cancer research; however, this protocol enables the user to examine any effects on various life stages of an insect in vivo. In addition, it also provides the researcher with step-by-step guidelines for toxicological assays. In the current literature, knowledge is limited with regard to the effects of epigenetic drugs on insects. Using DZNep as an example, we provide a procedure that may be utilized as a pre-requisite step towards exploring other epigenetic drugs or novel compounds as potential insect control agents. Although this protocol is developed for the malaria mosquito, it may be adapted to test the effect of DZNep, or any water-soluble compound of interest, in other mosquito or insect species.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We thank Victor Marquez for providing DZNep.HCl and Scotty Bolling for manufacturing the bloodfeeders. The Mopti and SUA2La strains of An. gambiae were obtained from the Malaria Research and Reference Reagent Resource Center (MR4). This work was supported by the Fralin Life Science Institute and the grant from National Institutes of Health 1R21AI094289 to Igor V. Sharakhov.
Materials
| Name of the Reagent/Equipment | Company | Catalgue Number | Comments |
| 96-well microplate | Fisher Scientific | 12565561 | |
| Cell culture plate | CytoOne | CC7682-7506 | |
| Centrifuge | Sorvall Fresco | 76003758 | A different centrifuge can be used |
| Colored tape rolls | Fisher | S68134 | |
| Dissection microscope | Olympus | SZ | |
| DTNB | Sigma Aldrich | D8130 | |
| DZNep.HCl | Sigma Aldrich | SMLO305 | |
| Egg dish cups | |||
| Filter papers | Fisher | 09-795E | |
| Glass feeders | Virginia Tech | ||
| Glass tissue homogenizer | |||
| Heating element | Fisher Scientific | NC0520091 | |
| Incubator | Percival scientific | I36VLC8 | A different incubator can be used |
| Microcentrifuge tube, 2 ml | Axygen | 22-283 | |
| Microcentrifuge tube, 1.5 ml | Axygen | MCT-150-C | |
| Micropipette | Eppendorf | 4910 000.069 | |
| Na2HPO4 | Fisher Scientific | M-3154 | |
| NaH2PO4 | Fisher Scientific | M-8643 | |
| pH meter | Mettler Toledo 7easy | S20 | |
| Plate reader | Spectramax | M2 | |
| SAH | Sigma Aldrich | A9384 | store at -20C |
| Triton X-100 | Sigma Aldrich | T8787 |
References
- Gatton, M. L., et al. The importance of mosquito behavioural adaptations to malaria control in Africa. Evolution; International Journal of Organic Evolution. 67, 1218-1230 (2013).
- Sternberg, E. D., Thomas, M. B. Local adaptation to temperature and the implications for vector-borne diseases. Trends in Parasitology. , (2014).
- Rocca, K. A., Gray, E. M., Costantini, C., Besansky, N. J. 2La chromosomal inversion enhances thermal tolerance of Anopheles gambiae larvae. Malaria Journal. 8, 147 (2009).
- Coluzzi, M., Sabatini, A., Petrarca, V., Di Deco, M. A. Chromosomal differentiation and adaptation to human environments in the Anopheles gambiae complex. Transactions of the Royal Society of Tropical Medicine and Hygiene. 73, 483-497 (1979).
- Donepudi, S., Mattison, R. J., Kihslinger, J. E., Godley, L. A. Update on Cancer Therapeutics. Science Direct. 2 (4), 157-206 (2007).
- Greer, E. L., Shi, Y. Histone methylation: a dynamic mark in health, disease and inheritance. Nature Reviews. Genetics. 13, 343-357 (2012).
- Kouzarides, T. Chromatin modifications and their function. Cell. 128, 693-705 (2007).
- Dawson, M. A., Kouzarides, T. Cancer epigenetics: from mechanism to therapy. Cell. 150, 12-27 (2012).
- Clough, E., Tedeschi, T., Hazelrigg, T. Epigenetic regulation of oogenesis and germ stem cell maintenance by the Drosophila histone methyltransferase Eggless/dSetDB1. Biologie du développement. 388, 181-191 (2014).
- Conrad, T., Akhtar, A. Dosage compensation in Drosophila melanogaster: epigenetic fine-tuning of chromosome-wide transcription. Nature Reviews. Genetics. 13, 123-134 (2011).
- Miranda, T. B., et al. DZNep is a global histone methylation inhibitor that reactivates developmental genes not silenced by DNA methylation. Molecular Cancer Therapeutics. 8, 1579-1588 (2009).
- Cui, B., et al. PRIMA-1, a Mutant p53 Reactivator, Restores the Sensitivity of TP53 Mutant-type Thyroid Cancer Cells to the Histone Methylation Inhibitor 3-Deazaneplanocin A (DZNep). J. Clin. Endocrinol. Metab. 99 (11), E962 (2014).
- Fujiwara, T., et al. 3-Deazaneplanocin A (DZNep), an inhibitor of S-adenosyl-methionine-dependent methyltransferase, promotes erythroid differentiation. The Journal of Biological Chemistry. , (2014).
- Li, Z., et al. The polycomb group protein EZH2 is a novel therapeutic target in tongue cancer. Oncotarget. 4, 2532-2549 (2013).
- Nakagawa, S., et al. Epigenetic therapy with the histone methyltransferase EZH2 inhibitor 3-deazaneplanocin A inhibits the growth of cholangiocarcinoma cells. Oncology Reports. 31, 983-988 (2014).
- Crea, F., et al. Pharmacologic disruption of Polycomb Repressive Complex 2 inhibits tumorigenicity and tumor progression in prostate cancer. Molecular Cancer. 10, 40 (2011).
- Vella, S., et al. EZH2 down-regulation exacerbates lipid accumulation and inflammation in in vitro and in vivo NAFLD. International Journal of Molecular Sciences. 14, 24154-24168 (2013).
- Tan, J., et al. Pharmacologic disruption of Polycomb-repressive complex 2-mediated gene repression selectively induces apoptosis in cancer cells. Genes & Development. 21, 1050-1063 (2007).
- Chiang, P. K., Cantoni, G. L. Perturbation of biochemical transmethylations by 3-deazaadenosine in vivo. Biochemical Pharmacology. 28, 1897-1902 (1979).
- Lu, Y. X., Denlinger, D. L., Xu, W. H. Polycomb repressive complex 2 (PRC2) protein ESC regulates insect developmental timing by mediating H3K27me3 and activating prothoracicotropic hormone gene expression. The Journal of Biological Chemistry. 288, 23554-23564 (2013).

