Chronic Ovine Model of Right Ventricular Failure and Functional Tricuspid Regurgitation
Summary
Right ventricular failure and functional tricuspid regurgitation are associated with left-sided heart disease and pulmonary hypertension, which contribute significantly to morbidity and mortality in patients. Establishing a chronic ovine model to study right ventricular failure and functional tricuspid regurgitation will help in understanding their mechanisms, progression, and possible treatments.
Abstract
The pathophysiology of severe functional tricuspid regurgitation (FTR) associated with right ventricular dysfunction is poorly understood, leading to suboptimal clinical results. We set out to establish a chronic ovine model of FTR and right heart failure to investigate the mechanisms of FTR. Twenty adult male sheep (6-12 months old, 62 ± 7 kg) underwent a left thoracotomy and baseline echocardiography. A pulmonary artery band (PAB) was placed and cinched around the main pulmonary artery (PA) to at least double the systolic pulmonary artery pressure (SPAP), inducing right ventricular (RV) pressure overload and signs of RV dilatation. PAB acutely increased the SPAP from 21 ± 2 mmHg to 62 ± 2 mmHg. The animals were followed for 8 weeks, symptoms of heart failure were treated with diuretics, and surveillance echocardiography was used to assess for pleural and abdominal fluid collection. Three animals died during the follow-up period due to stroke, hemorrhage, and acute heart failure. After 2 months, a median sternotomy and epicardial echocardiography were performed. Of the surviving 17 animals, 3 developed mild tricuspid regurgitation, 3 developed moderate tricuspid regurgitation, and 11 developed severe tricuspid regurgitation. Eight weeks of pulmonary artery banding resulted in a stable chronic ovine model of right ventricular dysfunction and significant FTR. This large animal platform can be used to further investigate the structural and molecular basis of RV failure and functional tricuspid regurgitation.
Introduction
Right ventricular failure (RVF) is recognized as an important factor contributing to the morbidity and mortality of cardiac patients. The most common causes of RVF are left-sided heart disease and pulmonary hypertension1. During the progression of RVF, functional tricuspid regurgitation (FTR) may arise as a consequence of right ventricular (RV) dysfunction, annular dilatation, and subvalvular remodeling. Moderate to severe FTR is an independent predictor of mortality2,3, and it is estimated that 80%-90% of tricuspid regurgitation cases are functional in nature4. FTR itself may promote adverse ventricular remodeling by influencing either afterload or preload5. The tricuspid valve has historically been considered the forgotten valve6, and it was believed that the treatment of left-sided heart disease would resolve the associated RV pathology and FTR7. Recent data have shown this to be a faulty strategy, and current clinical guidelines advocate a much more aggressive approach to FTR4. However, the pathophysiology of severe FTR associated with right ventricular dysfunction is still poorly understood, leading to suboptimal clinical results8. The currently available large animal models of RVF are based on pressure, volume, or mixed overload. We have previously described a large animal model of RVF and TR but only in an acute setting9.
The current study focuses on a chronic ovine model of pulmonary artery banding (PAB) to increase RV afterload (pressure overload) and induce RV dysfunction and FTR. The afterload model is reliable and reproducible compared to pulmonary hypertension models, in which changes in microvasculature are less predictable and more likely10. The goal of the study was to develop a chronic large animal model of RVF and FTR that would most accurately mimic RV pressure overload in patients with left-sided heart disease and pulmonary hypertension. The establishment of such a model would permit in-depth studies on the pathophysiology of the ventricular and valvular remodeling associated with RV dysfunction and tricuspid insufficiency. The ovine model was chosen based on our prior work on the mitral valve and the published literature supporting the anatomical and physiological similarities between human and ovine hearts11,12,13.
For this study, 20 adult sheep (62 ± 7 kg) underwent a left thoracotomy and main pulmonary artery banding (PAB) to at least double the systolic pulmonary artery pressure (SPAP), thus inducing RV pressure overload. The animals were followed for 8 weeks, and the symptoms of heart failure were treated with diuretics when clinically apparent. Surveillance echocardiography was performed periodically to assess RV function and valvular competence. Following the completion of the experimental protocol for model development (8 weeks), the animals were taken back to the operating room for median sternotomy and the implantation of sonomicrometry crystals on the epicardial and intra-cardiac structures. This procedure was performed using cardiopulmonary bypass with the heart beating and with bicaval control. There were no problems in weaning the animals from the cardiopulmonary bypass or acquiring the sonomicrometry data in a stable steady-state hemodynamic environment without the need for inotropes for right heart support. We anticipate performing tricuspid ring annuloplasty and other right heart procedures in the near future using a right thoracotomy approach in both terminal and survival experiments. The current experience leads us to believe that it will be possible to wean the animals from the cardiopulmonary bypass without difficulty and that long-term survival is feasible. As such, we believe the model will permit the performance of clinically pertinent cardiac procedures. Below is a description of the steps (perioperative and operative) performed for carrying out the ovine experimental protocol.
Protocol
The protocol was approved by the Michigan State University Institutional Animal Care and Use Committee (IACUC) (Protocol 2020-035, approved on 7/27/2020). For this study, 20 adult male sheep weighing 62 ± 7 kg were used.
1. Preoperative steps
- Fast the animal 12 h prior to surgery (overnight).
- Place the animal in a sheep chair (Figure 1), and prepare for the right jugular vein cannulation using a long 11 Fr introducer sheath (sheath length = 10 cm).
- Shave with clippers along the IV placement site-the right anterior aspect of the neck around 10-15 cm lateral from the midline for the right jugular vein.
- Turn the animal's head to the left so that the right anterior and lateral aspects of the neck are exposed. Localize the jugular vein course. To facilitate this, compress the bottom of the neck to distend the vein.
- Clean with chlorhexidine and alcohol-based scrub, and anesthetize locally with 1% lidocaine.
- Canulate the jugular vein in the mid to upper third of the neck, as described.
- Cut the skin above the vein with a number 11 blade perpendicular to the vein.
- Cannulate with a 14 G angiocatheter; when in place (blood is coming out of the needle or blood is spotted), remove the needle, leave the catheter, pass the guidewire, remove the catheter, place the 11 Fr sheath, and secure it.
- Ensure the patency and proper placement of the cannula by withdrawing dark-red blood and performing a saline flush to confirm the flow and absence of swelling at the insertion site.
- Start the induction of propofol at 1.0-1.5 mg/kg intravenous (IV).
- Intubate with a number 9 endotracheal (ET) tube using a laryngoscope with a number 5 blade. For this, one person should secure the jaws and tongue, while the other person identifies the trachea, inserts the ET tube, and inflates the sealing cuff. Confirm proper placement by the bilateral breath sounds and condensation on the ET tube.
- Administer intravenously the analgesic buprenorphine at 0.01 mg/kg and use 240 mg of gentamicin and 1 g of cefazolin for antibiotic prophylaxis.
- Transfer the animal from the sheep chair onto a surgical table, and position it on its right side.
2. Surgery steps
- Ventilate at 15 mL/kg (12-18 breaths/min), with oxygen flow at 4 L/min and isoflurane at 2.5%-4.0%. Confirm proper anesthesia to ensure the subject is at the surgical level (stage 3) by checking the jaw tone and eye rotation.
- Lubricate both eyes by applying ophthalmic oinment and insert a gastric tube to ensure gas and food evacuation. Connect the electrocardiogram (EKG), pulse oximeter (SpO2), capnograph (ETCO2), and body temperature monitors. Attach the EKG limb leads (I, II, III) to the skin via alligator clips, the SpO2 sensor to the animal's cheek, and the ETCO2 tube to the endotracheal tube, and pass the temperature probe through the nostril into the nasopharynx.
- Prepare the operative field. Shave the left anterior thorax, clean with chlorhexidine and alcohol-based scrub, and cover with sterile drapes.
- Make a 10 cm long skin and subcutaneous incision at the level of the fourth intercostal space.
- Confirm the correct intercostal space by identifying the thoracic inlet and counting the intercostal spaces downward. Subsequently, continue the incision in the center and along the fourth intercostal space.
- Divide the intercostal muscles, open the chest cavity, and spread the ribs with a mini-thoracotomy Finochietto-style retractor. While performing the thoracotomy, take care not to injure the left internal mammary artery (LIMA) at the sternal border of the incision and the lung at the superior border.
- Perform baseline epicardial echocardiography to assess the biventricular function and valvular competence. The occurrence of non-standard views may be due to the mini-thoracotomy focused on the tricuspid valve (TV), right and left ventricular function, and pulmonary artery flow.
- Identify the LIMA at the sternal border of the incision, remove the adjacent tissues around it, and prepare to establish an arterial line for pressure monitoring.
- Place two 4-0 silk sutures around the artery, with one proximal and one distal to the cannulation site (used to secure the arterial catheter).
- Use titanium clips with a clip applier to clip the LIMA distal to the planned cannulation site to prevent backflow bleeding during cannulation.
- Make a perpendicular incision that is half of the circumference of the catheter in the LIMA with a number 11 blade.
- Insert an 18 G angiocatheter, and attach it to the arterial line module. When a pressure of around 120/80mmHg is reached, secure the catheter in place using the two 4-0 silk sutures placed earlier.
- Perform pericardiotomy starting at the level of pulmonary artery sinuses and going 4-5 cm laterally along the main pulmonary artery (MPA), taking care not to injure the left phrenic nerve.
- Apply four to five retraction stitches to the opened pericardium to create a pericardial well, as this facilitates exposure and dissection between the pulmonary trunk and aorta.
- Dissect the MPA from ascending aorta (AA) around 2-3 cm from its origin using blunt right-angle forceps, starting at the level of the left atrial appendage and working toward the AA. To fully separate the MPA from the AA, use electrocautery or scissors to remove the connective tissue between the two structures.
- Pass an umbilical tape around the MPA with a blunt right-angle clamp. Establish an MPA pressure line by placing a 5-0 monofilament purse-string suture 1 cm distal from the MPA sinuses.
- Insert a 20 G angiocatheter, and connect it to a monitoring line. Ensure correct MPA and arterial line readings are achieved prior to cinching the umbilical tape; the arterial and pulmonary pressures may vary but should be comparable to human patient values.
- Hold both ends of the umbilical tape, and clip them together to reduce the lumen of the MPA.
- Progressively tighten the band with the successive application of a clip applier, with each clip placed below the previous clip until the systemic blood pressure begins to steadily decline (Figure 2). At this point, remove the last placed clip to stabilize the systemic blood pressure.
- When maximal cinching and stable hemodynamic conditions are achieved, secure the umbilical tape to the adventitia of the MPA using a 5-0 monofilament suture to avoid distal migration.
- Perform post-banding echocardiography to assess the biventricular function and valvular competence, as in step 2.7. Remove the MPA pressure line and arterial line, and ensure good hemostasis by checking for any bleeding coming from the area where the band and arterial lines were placed.
- Place a chest tube into the left thorax, with the entry site one intercostal space below the initial incision. Close the ribs with two Vicryl size 2 sutures, and close the wound with three-layer continuous sutures: Vicryl 2-0 for the muscle and subcutaneous tissues and Prolene 3-0 for the skin.
- When no signs of bleeding are seen, remove the chest tube before weaning the animal from the ventilator.
- Wean the animal from the ventilator, extubate, move it to a single cage, and closely follow it for at least 1 h. Leave the central IV line in place, and secure it using a loosely applied bandage around the neck.
NOTE: Postoperative intravenous analgesia was maintained with buprenorphine (0,05 mg/kg) and Flunixin (1.2 mg/kg) for 3 days post-surgery.
Representative Results
Following the completion of the experimental protocol for model development (nearly 8 weeks), the animals were taken back to the operating room for median sternotomy and the implantation of sonomicrometry crystals on the epicardial and intra-cardiac structures. This procedure was performed using cardiopulmonary bypass with the heart beating and with bicaval control, as described by our group in detail previously9. There were no problems in weaning the animals from the cardiopulmonary bypass or acquiring the sonomicrometry data in a stable steady-state hemodynamic environment.
Pulmonary artery banding acutely increased the SPAP from 21 ± 2 mmHg to 62 ± 9 mmHg (p = 0.001). Three animals died during the follow-up period due to stroke, hemorrhage, and acute heart failure. Of the surviving 17 animals, 3 developed mild TR, 3 developed moderate TR, and 11 developed severe TR. The mean TR grade (0-4; 0 = none or trace, 1 = mild, 2 = moderate, 3 = moderately severe, and 4 = severe) after the follow-up period increased from 0.8 ± 0.4 to 3.2 ± 1.2 (p = 0.0001). The presented data in Table 1 demonstrate signs of evolving right ventricular failure and the development of significant TR after 8 weeks of pulmonary banding, consistent with the echocardiographic examination of a representative animal shown in Figure 3.

Figure 1: Sheep chair. The sheep chair greatly facilitates animal imaging and the induction of anesthesia, as well as the placement of intravenous lines. It is customarily used in wool shearing, and animals are usually familiar with this position and remain quite docile for the necessary procedures. Please click here to view a larger version of this figure.
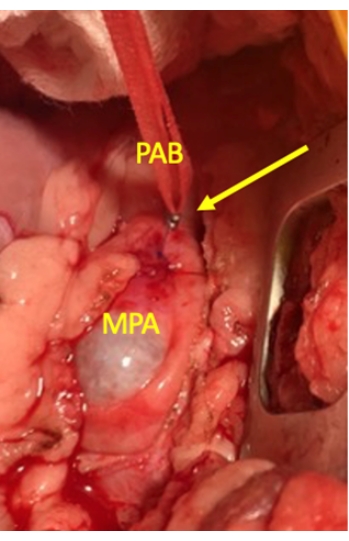
Figure 2: Intraoperative view of pulmonary artery banding. The photograph illustrates the pulmonary artery band formed by an umbilical tape passed around the main pulmonary artery, with surgical clips used to tighten and secure the band in place. The yellow arrow points to the clips applied to the umbilical cord. Abbreviations: MPA = main pulmonary artery; PAB = pulmonary artery band. Please click here to view a larger version of this figure.
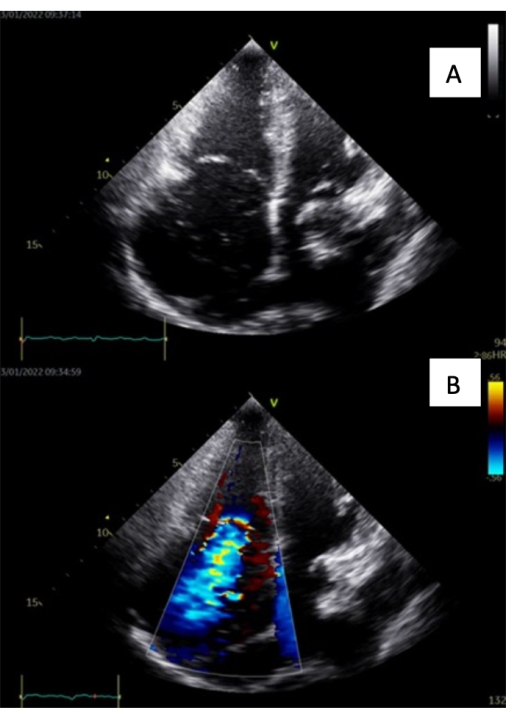
Figure 3: Intraoperative echocardiographic images at 8 weeks after PAB (A = four-chamber view, B = four-chamber view with a color doppler showing FTR). Please click here to view a larger version of this figure.
| Baseline | 8-weeks | |
| HR (b/min) | 107±15 | 88±11 |
| LVEF (%) | 62±3 | 58±4* |
| SPAP mmHg | 62±2 | 40±7* |
| RVFAC (%) | 50±14 | 38±7* |
| TAPSE | 1.2±0.1 | 0.8±0.1* |
| TR grade (0-4) | 0.4±0.5 | 3.2±1.2* |
| TV annulus (cm) | 2.4±0.2 | 3.1±0.2* |
Table 1: Echocardiographic and hemodynamic data. Abbreviations: HR = heart rate; LVEF = left ventricular ejection fraction; SPAP = systolic pulmonary artery pressure; RVFAC = right ventricle fractional area change; TAPSE = tricuspid annular plane systolic excursion; TR = tricuspid regurgitation (grade 0-4); TV = tricuspid valve. The data show mean ± SD; *p < 0.05 versus baseline by a paired t-test.
Discussion
In this model, 8 weeks of pulmonary artery banding resulted in a stable chronic ovine model of right ventricular dysfunction and, in most cases, significant FTR. The strengths of the presented chronic PAB model include the precise afterload adjustment during the procedure, although its influence on RV responses may differ. The model is suitable for evaluating varying degrees of RV failure or FTR, with the severity modulated by the degree of pulmonary artery constriction. Moreover, the application of fixed and stable resistance at the level of the main PA, unlike in pulmonary hypertension models, rules out the influence of changes in the pulmonary vascular bed on the afterload11. Ovine models of pulmonary hypertension with pulmonary artery embolization have not been demonstrated to predictably induce RVF14. However, it may be challenging to adequately tighten the band to achieve the desired degree (phenotype) of right heart failure15, not to mention the exact TR grade. This is reflected in the study, as similar peak PA pressure was achieved in all the animals (62 ± 9 mmHg), but it did not show any correlation with either TR or RHF. This may suggest biological variability in the remodeling responses of the strained RV to increased afterload. Nevertheless, in most cases, significant TR developed as a consequence of increased afterload and the subsequent changes related to RV remodeling and failure.
This ovine model was specifically designed to induce functional tricuspid regurgitation and differs from other models that are mainly focused on right heart dysfunction. The available models of TR are based mostly on structural damage to the TV and subvalvular apparatus16,17, thus meaning these are mostly volume overload models of RHF that do not represent the true nature of FTR. We have previously developed a model of tachycardia-induced cardiomyopathy18, which results in biventricular failure and functional mitral and tricuspid regurgitation. The current model permits the study and treatment of FTR in the case of isolated RV dysfunction. Recently, a model of gradual pulmonary artery banding with an inflatable band and a subcutaneous port has been introduced19, which may offer an extension of this technique. A catheter-based narrowing of the pulmonary artery has not yet been described, but such experimental techniques are surely on the horizon.
There are several critical steps while executing this protocol. Care must be taken while opening the fourth intercostal space so as not to injure the left internal mammary artery, which is used to establish an arterial line. The next critical step is freeing the MPA from the ascending aorta next to the left atrial appendage and passing an umbilical cord around the MPA. It is of utmost importance that, during pulmonary artery cinching, the tightness of the band is adjusted correctly, as over-tightening will result in early animal demise, while a band that is too loose will not induce an adequate degree of right heart failure and FTR. The band is progressively tightened with the successive application of a clip applier until the systemic blood pressure begins to steadily decline. It is crucial to be adept at removing the last clip expeditiously so as to avoid hemodynamic collapse and ventricular fibrillation. Emergency cardiac drugs should be on hand and easily available.
The model is limited by requiring open thoracotomy and direct surgical manipulation of the pulmonary artery, which represents a surgical risk and leads to the formation of adhesions that make subsequent surgeries more difficult. Furthermore, using the above-presented protocol, some animals experience a rapid evolution of heart failure and functional TR that is not compatible with 8 week survival. As such, an attrition rate of 15%-20% can be expected. The technique can be modified based on the scientific question at hand. In the current study, the goal of the experiment was to induce significant functional tricuspid regurgitation, and as such, aggressive pulmonary banding was used. However, the model may be modified to study the effects of various degrees of ventricular afterload (a surrogate for pulmonary hypertension) on right ventricular function and remodeling. In such scenarios, the pulmonary banding may be adjusted to achieve several different levels of pulmonary artery pressure to permit the study of the effects of different afterload levels. Additionally, the same model may be translated to rodents20 or be used in a graded fashion in sheep using an inflatable pulmonary band and subcutaneous injection port21.
The technique may be used in the future to study the mechanisms of functional tricuspid regurgitation with its associated right ventricular, annular, and subvalvular remodeling, as well as tissue changes. The model lends itself to the study of reverse remodeling as the pulmonary band is reversible through a repeat thoracotomy. Furthermore, this model has already been used to study right ventricular mechanical assist devices21, and it is anticipated that it will be harnessed more frequently as the field of right-sided mechanical support continues to evolve.
In conclusion, the presented large animal model of right heart failure and functional tricuspid valve regurgitation is reproducible and effective for producing FTR with a relatively low attrition rate. This large animal platform can be used to further investigate the structural and molecular basis of RV failure and functional tricuspid regurgitation. This model may also facilitate the evaluation of interventions targeting the failing RV and the TV apparatus.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The study was funded by an internal grant from the Meijer Heart and Vascular Institute at Spectrum Health.
Materials
| Anesthesia Machine Drager Narkomed MRI-2 | Drager | 4116091-001 | |
| angiocatheter | BD | BD382268 | 14GAx8.25cm |
| BD ChloraPrep Scrub Teal 26 ml applicator with a sterile solution | |||
| Blade #11 | Bard-Parker | 371111 | |
| Buprenorphine | HIKMA | ||
| cefazolin 1.0g | Hikma | 0143-9924-90 | |
| Diprivan 200mg/20ml | 63323-0269-29 | FRESENIUS KABI | |
| Electrosurgical generator Valleylab Force FX | Valleylab | CF5L44233A | |
| Gentamicin Sulfate 40 mg / mL | Fresenius | 406365 | |
| i-Stat Blood analyzer MN 300 | Abbott | ||
| Lidocaine HCl 1% | Pfizer | 243243 | |
| Open ligating clip appliers Horizon Medium | Teleflex | 237061 | |
| PERMAHAND Silk Suture | PERMA HAND | SA 63H | |
| Pinnacle Introducer sheath | Terrumo | RSS102 | sheath length 10cm |
| Prolene 3-0 | ETHICON | 8684H | |
| Titanium Clips Medium | Teleflex | 2200 | |
| Umbilical tape | Ethicon | EFA 1165 | |
| VICRYL 2 coated undyed 1X54" TP-1 | ETHICON | J 880T | |
| Vicryl 2-0 | ETHICON | J269H |
References
- Haddad, F., Hunt, S. A., Rosenthal, D. N., Murphy, D. J. Right ventricular function in cardiovascular disease, part I: Anatomy, physiology, aging, and functional Assessment of the right ventricle. Circulation. 117 (11), 1436-1448 (2008).
- Taramasso, M., et al. The growing clinical importance of secondary tricuspid regurgitation. Journal of the American College of Cardiology. 59 (8), 703-710 (2012).
- Mangieri, A., et al. Mechanism and implications of the tricuspid regurgitation: From the pathophysiology to the current and future therapeutic options. Circulation: Cardiovascular Interventions. 10 (7), 005043 (2017).
- Otto, C. M., et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive summary: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 143 (5), 35-71 (2021).
- Vonk-Noordegraaf, A., et al. Right heart adaptation to pulmonary arterial hypertension: physiology and pathobiology. Journal of the American College of Cardiology. 62, 22-33 (2013).
- Yoganathan, A., et al. Tricuspid valve diseases: Interventions on the forgotten heart valve. Journal of Cardiac Surgery. 36 (1), 219-228 (2021).
- Vachiéry, J. L., et al. Pulmonary hypertension due to left heart diseases. Journal of the American College of Cardiology. 62, 100-108 (2013).
- Chin, K. M., Coghlan, G. Characterizing the right ventricle: Advancing our knowledge. American Journal of Cardiology. 110, 3-8 (2012).
- Malinowski, M., et al. Large animal model of acute right ventricular failure with functional tricuspid regurgitation. International Journal of Cardiology. 264, 124-129 (2018).
- Borgdorff, M. A., Dickinson, M. G., Berger, R. M., Bartelds, B. Right ventricular failure due to chronic pressure load: What have we learned in animal models since the NIH working group statement. Heart Failure Review. 20 (4), 475-491 (2015).
- Andersen, A., et al. Animal models of right heart failure. Cardiovascular Diagnosis and Therapy. 10 (5), 1561-1579 (2020).
- Dixon, J. A., Spinale, F. G. Large animal models of heart failure: A critical link in the translation of basic science to clinical practice. Circulation: Heart Failure. 2 (3), 262-271 (2009).
- Miyagi, C., et al. Large animal models of heart failure with preserved ejection fraction. Heart Failure Review. 27 (2), 595-608 (2022).
- Sato, H., et al. Large animal model of chronic pulmonary hypertension. American Society for Artificial Internal Organs Journal. 54 (4), 396-400 (2008).
- Bogaard, H. J., et al. Chronic pulmonary artery pressure elevation is insufficient to explain right heart failure. Circulation. 120 (20), 1951-1960 (2009).
- Xie, X. J., et al. Tricuspid leaflet resection in an open beating heart for the creation of a canine tricuspid regurgitation model. Interactive Cardiovascular and Thoracic Surgery. 22 (2), 149-154 (2016).
- Hoppe, H., et al. Percutaneous technique for creation of tricuspid regurgitation in an ovine model. Journal of Vascular and Interventional Radiology. 18, 133-136 (2007).
- Malinowski, M., et al. Large animal model of functional tricuspid regurgitation in pacing induced end-stage heart failure. Interactive Cardiovascular and Thoracic Surgery. 24 (6), 905-910 (2017).
- Ukita, R., et al. A large animal model for pulmonary hypertension and right ventricular failure: Left pulmonary artery ligation and progressive main pulmonary artery banding in sheep. Journal of Visualized Experiments. (173), e62694 (2021).
- Dufva, M. J., et al. Pulmonary arterial banding in mice may be a suitable model for studies on ventricular mechanics in pediatric pulmonary arterial hypertension. Journal of Cardiovascular Magnetic Resonance. 23 (1), 66 (2021).
- Verbelen, T., et al. Mechanical support of the pressure overloaded right ventricle: An acute feasibility study comparing low and high flow support. American Journal of Physiology-Heart and Circulatory Physiology. 309 (4), 615-624 (2015).

